Treatment of Acute Spondylolysis in Elite athletes. Literature Review and Presentation of a New Percutaneous Grafting Technique
Joris Robberecht*, Nick Stevens and Jan SYS
Department of Orthopaedic surgery, AZ St-Blasius, Kroonveldlaan 50, Dendermonde, Belgium
Submission: October 25, 2016; Published: November 07, 2016
*Corresponding author: Joris Robberech, Department of Orthopaedic surgery, Reetsesteenweg, Aartselaar, Belgium, Email;jorisrobberecht@gmail.com
How to cite this article: Joris R, Nick S, Jan SYS. Treatment of Acute Spondylolysis in Elite athletes. Literature Review and Presentation of a New 006 Percutaneous Grafting Technique. Open Access J Neurol Neurosurg. 2016; 1(4): 555568. DOI: 10.19080/OAJNN.2016.01.555568
Abstract
Introduction:The initial treatment of acute spondylolysis in young elite athletes is conservative. Excellent clinical results are seen when there is osseous healing of the defect. When there is no osseous healing, repetitive and maximal loading of the lumbar spine often remain painful. Osseous healing is more likely when lesions are diagnosed and treated early. When no signs of healing are present at computed tomography (CT-scan) after 4 months of conservative treatment and when pain persists, percutaneous surgical treatment can be considered in elite athletes. Methods:We present a new percutaneous bone grafting technique for young elite athletes with acute spondylolysis. Results:Osseous healing was achieved and the patient was able to resume competitive sport activities within 6 months after surgery. Conclusion:This technique can improve fracture biology without muscle damage and without affecting the normal mobility of the spine, eventually leading to osseous healing. Keywords:Spondylolysis; Treatment; Percutaneous; Grafting; Athletes
Introduction and Literature Review
Spondylolysis is a defect of the pars interarticularis, eventually causing low back pain. It mostly occurs as a stress-fracture, but can be congenital as well. The incidence in the adult Caucasian population is around 6%. It’s more common in males then in females [1]. Young athletes are most vulnerable, especially in sports requiring repetitive hyperextension and rotational loading: gymnasts, football players, weightlifters, wrestlers and dancers [2]. It mostly affects L5 (71-95%). Secondary spondylolisthesis occurs in 75% of cases [3,4]. For acute spondylolysis in young athletes conservative treatment is the preferred initial treatment. When diagnosed early the majority of athletes will resume their sporting activities after 3-6 months [5]. The longer the symptoms are present, the more likely that conservative treatment will be insufficient [6]. Unilateral active lesions have a higher tendency to heal then bilateral lesions [7]. Conservative treatment consists of lumbosacral orthosis, activity modification, and rehabilitation [6]. There is no consensus on the value and duration of bracing, the type of brace used, and the duration of sports restriction. Athletes should be pain free before they return to sport [5].
Morita et al. [8] classified spondylolysis defects on radiographs into early, progressive and terminal stages. The early stage was characterized by focal bony absorption or a hairline defect. In the progressive stage the defect was wide, and small fragments were present. Sclerotic changes indicated the terminal stage of development. With conservative treatment of 346 pars defects in 185 patients he found healing in 73.0% of the early, 38.5% of the progressive and none of the terminal defects [8]. Sys et al. [7] classified spondylolysis as unilateral, bilateral and pseudo-bilateral acutespondylolysis. In pseudo-bilateral spondylolysis, only the fracture with increased uptake on bone scintigraphy is considered recent or “active”. The fracture on the other side is considered a pseudarthrosis, and is an older or “scintigraphically inactive” lesion. The term “acute” spondylolysis is related to the appearance of symptoms rather than to the age of the lesion. In their study they found that bonyhealing is more likely to occur in unilateral active spondylolysis [7]. Consequently, acute spondylolysis must be diagnosed as early as possible and treated appropriately in order to maximize the chance for osseous healing and success of conservative treatment. Surgery is reserved for those athletes in whom conservative treatment fails and pain persists.
Non-union or delayed union is the result of insufficient stability and/or inappropriate biology (osteogenesis, osteoinduction, and osteoconduction). Treating spondylolysis surgically can act on two causative factors in the development of pseudarthrosis. Biology can be improved by introducing bone graft or osteoinductive proteins such as recombinant human bone morphogenic protein (rhBMP-2) [9]. When autologous bone grafts are used, osteogenesis as well as osteoinduction and osteoconduction are optimized. Stability at the fracture site can be improved stability with an internal fixation device. Improvement of biology by adding bone graft without internal fixation was first published by Kimura [10]. In order to improve stability at the fracture site, the patient had to remain recumbent for 2 months followed by immobilization in a plaster cast till union of the spondylolysis occurred (ca. 6 months) [10]. Dai et al. [11] also used bone grafting without internal fixation in 20 patients. The patients were allowed out of bed from the 10th postoperative day, wearing a plaster cast without inclusion of the thigh for 3-4 months. Restricted activities were recommended for 6 months. 10 patients returned to former occupation and sports with no restrictions. Nine suffered occasional pain and were able to resume sports activities but were restricted from strenuous sports. One had less complaint than preoperatively, but pain was still experienced as problematic, restricting sports, work, and recreational activities. All had bony healing except one. This study was not limited to young athletes and chronic lesions were included [11].
Internal fixation can be performed in two ways: segmental fusion and direct repair of the pars interarticularis. In segmental fusion the mobility of the affected level is sacrificed, erector spinae muscles are partially denervated and devascularized and there is an increased risk for degeneration of the adjacent lumbar segment [12]. In direct repair of the pars interarticularis normal bony anatomy and biomechanics are preserved, but denervation and devascularisation of the erector spinae muscles do occur depending on the approach and the extent of dissection. Buck described a technique for direct repair in 1970, using a compression screw through the pars defect and introduction of bone graft [13]. Nicol & Scott [14] used bone graft and fixation with a cerclage wire around the adjacent transverse and spinous processes in order to stabilize the pars defect [14]. More recent techniques make use of pedicle screws and hooks that hitch on under the caudal lamina. Drazin et al. [15] reviewed 9 studies, reporting 84 young athletes with a direct pars repair showing good results. Seventy-one athletes (84%) returned to their preoperative sports activities, 7 switched to a less incriminating sport, and 6 completely stopped practicing sports [15].
In order to maximize the chance for sports resumption at a high level, alteration of normal anatomy and normal biomechanics should be avoided as much as possible. Direct pars repair is generally preferred to segmental fusion in young athletes with spondylolysis, when no or minimal spondylolisthesis (3mm) and a normal intervertebral disc are present [16]. Still, the expected theoretical benefits (preservation of lumbar spine motion, avoidance of adjacent segment degeneration) could not be proven after a mean follow-up of 14.8 years [17], suggesting that even in a primary repair the normal biomechanics and lumbar muscle function are altered. Back muscle degeneration after surgery is undesirable, especially for sports players. In young athletes it is especially important to avoid surgery-related back muscle injury as much as possible [9].
Performing lumbar spine surgery with traditional surgical approach causes harm to the erector spinae muscles, tendons, innervation and vascularization leading to tissue necrosis, scar formation and loss of muscle function [12,18]. Attempting to limit these adverse events and to achieve faster recovery and shorter hospital stay, minimally invasive techniques were developed. The modified Buck technique consists of percutaneous placement of cannulated screws with the aid of an image intensifier or intraoperative CT-scan. This percutaneous technique does not allow the insertion of bone graft [9]. This modified Buck technique can be performed through a stab incision with minimal muscle splitting, but does not improve biology at the fracture site. Amoretti et al. [16] found good results in 10 patients with this technique, without any complications [16]. Care must be taken not to harm the facet joint or neurovascular structures when placing the screws. Correcting angular and translational deformities is limited in this technique [5].
Any other technique with hooks, screw, are cables requires a larger incision for the placement of pedicle screws and some split of the erector spinae muscle for placement of the hook, rod, or cable and application of bone graft.
In accordance with Kimura [10] & Dai et al. [11], we propose a new technique to improve the fracture biology by autologous bone graft and to improve the stability by external immobilization [10,11]. In contrast to the above named studies, we restrict this procedure to young elite athletes with acute spondylolysis, not responding to 4 months of conservative treatment and with a lesion located high in the pars interarticularis. The procedure is performed in a percutaneous way. External support is provided by a modified Boston brace instead of a plaster cast.
Surgical Technique
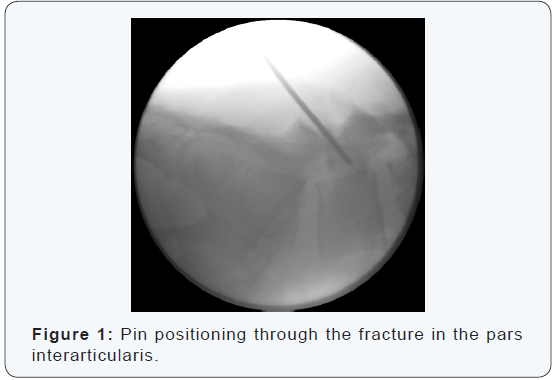
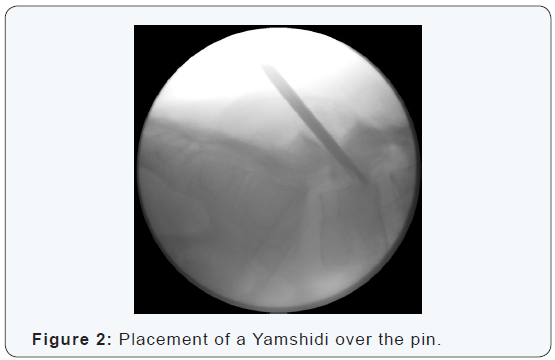
The procedure is performed under general anesthesia with the patient in a prone position. Localization and angulation of the pars interarticularis are determined with fluoroscopy in antero-posterior and lateral view (Figure 1). Preferably, 2 C-arms are used simultaneously. A stab incision is made at the affected site. Blunt dissection is performed and the lumbar fascia is opened allowing the introduction of a 2.0 mm kirschner wire. A hand-driven wire is placed from inferior to superior, perforating the area of nonunion but without perforating the pedicle or the upper end plate of the vertebra. Placement and positioning of the pin are controlled with antero-posterior and lateral fluoroscopic images (Figure 2). When the correct position is confirmed a Yamshidi needle (8 gauge, length 6 inch) is placed over the pin (Figure 3).

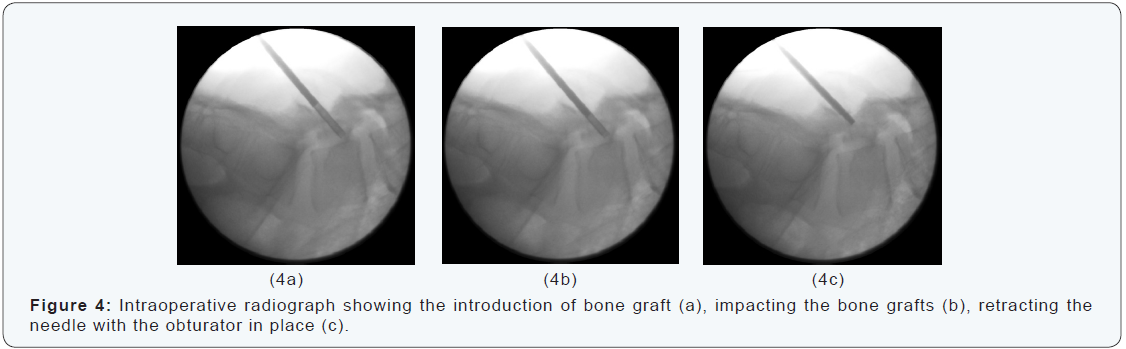
A cone of autogenous bone graft is harvested from the iliac crest with another Yamshidi needle (Figure 4). This can be performed through the same stab incision when the spondylolysis is at L5. The cone of bone graft is introduced and impacted in the lesion through the first needle. While retracting the needle the cone of bone grafts is kept in place by using an obturator (Figure 5). The incision is closed with a non-resorbable suture. Patients remain in bed for 12 hours and are discharged the day after surgery. They wear a modified Boston brace for 8 weeks postoperatively. Recreational swimming and cycling are allowed without the brace. A control CT-scan is performed after 6 weeks and after 12 weeks. The field of view was limited to the L5 vertebra in order to limit the cumulated exposure to X-rays.
Illustrative case
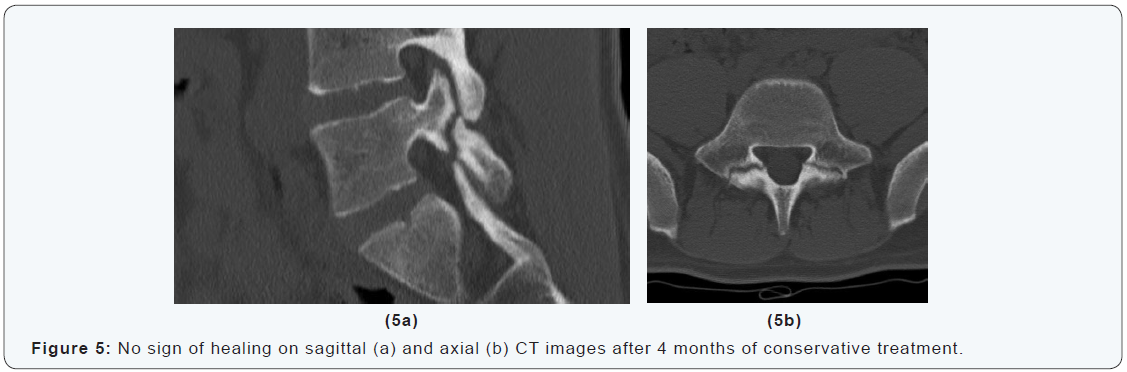
A 16-years-old goalkeeper presented with bilateral spondylolysis at L5. Conservative treatment was initiated. Sports were abandoned. A modified Boston brace was worn. After four months, CT-scan showed no signs of healing (Figure 6). Percutaneous autologous bone grafting was performed. A Boston brace was prescribed for another 3 months postoperatively. CTscan at 6 weeks and 3 months showed progressive healing. At 6 months the patient was pain free, and maximal sport activities were resumed (Figure 6 & 7). The aim of this technique is to improve fracture biology without muscle damage and without affecting the normal mechanics of the spine. In the case presented a good results was achieved. Osseous healing was obtained and the patient was able to resume competitive sports activities within 6 months after surgery.
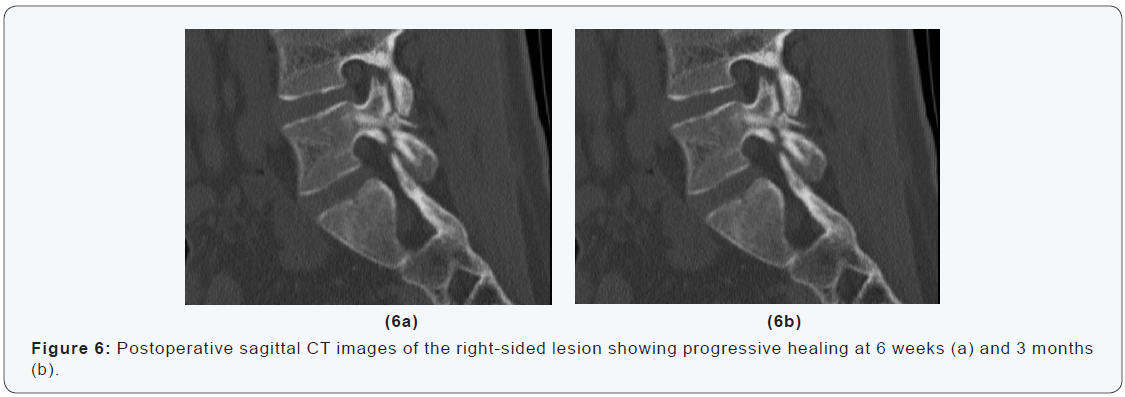
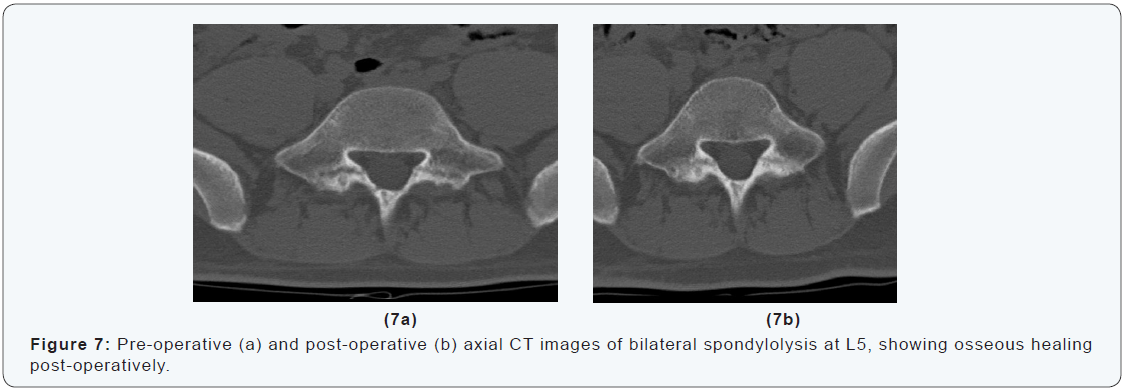
Discussion
The initial treatment of acute spondylolysis in young elite athletes is conservative. Acceptable clinical results can often be seen even when there is no osseous healing of the defect, depending on the solidity of the fibrous tissue at the pseudarthrosis [19,20]. In spondylolysis, maximal and repetitive loading of the lumbar spine should be avoided, as this often elicits pain [7]. In the long term the deficient pars interarticularis may cause increased shear stress on the intervertebral disc, eventually leading to accelerated disc degeneration. Szypryt et al. [21] found a higher prevalence of intervertebral disc degeneration and Spondylolysis [21]. In a high demand population, pain and/or degeneration are expected in the long course when a bilateral pseudarthrosis of the pars defect exists. Clinical follow-up and consecutive CT-scan allow differentiation between bony healing and delayed union or non-union. When a comprehensive conservative treatment of 4 months fails and pain persists, and CT-scan shows no signs of healing or evolution towards pseudarthrosis, a percutaneous surgical intervention can be considered in those athletes who definitely want to resume high-level sports activities.
The location of the lesion at the pars articularis is a determining factor for this technique. Only fractures located in the upper half of the pars are accessible for percutaneous perforation and grafting In the present literature, the location of the lesion has never been taken into account. In none of the case series describing surgical treatment in spondylolysis, authors have taken into account the level of sports activity. Only in the conservatively treated series of Sys et al. [7] the average time per week dedicated to sports was 10.9 hours, which gives some indication about the level of sports [7]. As spondylolysis mostly occurs in adolescents, we cannot distinguish between professional and non-professional athletes. We reserve the presented technique for adolescents performing sports daily, aiming for a professional career. Although athletes have a high probability for sports resumption after direct surgical repair [15], iatrogenic injury to the erector spinae is inevitable, even when a muscle split technique is used. These minimally invasive techniques are performed with the aid of tubular retractors which are brought in through small incisions but are distracted inside the erector spinae muscle. An incision is needed and there is some muscle damage, so these techniques are not strictly percutaneous [11].
Damage to the paraspinal musculature after posterior spinal surgery has been shown to correlate with increased postoperative back pain and an overall poorer outcome [22,23]. In elite athletes, we want to avoid any muscle damage and prefer a technique which is strictly percutaneous and can be performed through a stab incision.
The mostly used percutaneous technique for a direct pars repair is the lag screw method of Buck, which has recently been refined by Wilson [9]. This percutaneous technique is less invasive than minimal invasive techniques using a muscle split approach, but does not allow the insertion of bone graft through the same incision. Moreover, it cannot be used in patients with small laminae and the presence of a screw through the fracture site hampers the judgement of bone formation on CT-scan. Introducing this new percutaneous technique extends the indication for surgery in elite athletes. It aims at increasing the chance for ultimate osseous healing and fast competitive sports resumption at the pre-injury level. We hereby assume that osseous healing of the defect is the only guarantee for pain free and maximal sport resumption in the short term and for prevention of complications in the long term. Consequently, bony fusion must be the ultimate goal of treatment in acute spondylolysis in athletes.
Conclusion
Percutaneous autologous bone grafting can be used for young elite athletes with acute spondylolysis located high in the pars interarticuaris, when no signs of healing are present at CTscan after 4 months of conservative treatment and when pain persists. This technique can improve fracture biology without muscle damage and without affecting the normal mobility of the lumbar spine. We’ve performed this technique of percutaneous bone grafting in 2 athletes. Both showed osseous healing within 3 months after the intervention. Both could resume sporting activities and reach their pre-injury level within 6 months.
References
- Sakai T, Sairyo K, Suzue N, Kosaka H, Yasui N (2010) Incidence and etiology of lumbar spondylolysis: review of the literature. J Orthop Sci 15(3): 281-288.
- Syrmou E, P P Tsitsopoulos, D Marinopoulos, C Tsonidis, I Anagnostopoulos, et al. (2010) Spondylolysis: a review and reappraisal. Hippokratia 14(1): 17-21.
- Toueg CW, Mac-Thiong JM, Grimard G, Parent S, Poitras B, et al. (2010) Prevalence of spondylolisthesis in a population of gymnasts. Stud Health Technol Inform 158: 132-137.
- Gurd DP (2011) Back pain in the young athlete. Sports Med Arthrosc 19(1): 7-16.
- Bouras T, Korovessis P (2014) Management of spondylolysis and low-grade spondylolisthesis in fine athletes. A comprehensive review. Eur J Orthop Surg Traumatol 25(1): S167-S175.
- Blanda J, Bethem D, Moats W, Lew M (1993) Defects of pars interarticularis in athletes: a protocol for nonoperative treatment. J Spinal Disord 6(5): 406-411.
- Sys J, Michielsen J, Bracke P, Martens M, Verstreken J (2001) Nonoperative treatment of active spondylolysis in elite athletes with normal X-ray findings: literature review and results of conservative treatment. Eur Spine J 10(6): 498-504.
- Morita T, Ikata T, Katoh S, Miyake R (1995) Lumbar spondylolysis in children and adolescents. J Bone Joint Surg Br, 77(4): 620-625.
- Wilson L, Altaf F, Tyler P (2016) Percutaneous pars interarticularis screw fixation: a technical note. Eur Spine J 25(6): 1651-1654.
- Kimura M (1968) [My method of filing the lesion with spongy bone in spondylolysis and spondylolistesis]. Seikei Geka 19(4): 285-296.
- Dai LY, Jia LS, Yuan W, Ni B, Zhu HB et al. (2001) Direct repair of defect in lumbar spondylolysis and mild isthmic spondylolisthesis by bone grafting, with or without facet joint fusion. Eur Spine J 10(1): 78-83.
- Hu ZJ, Fang XQ, Fan SW (2014) Iatrogenic injury to the erector spinae during posterior lumbar spine surgery: underlying anatomical considerations, preventable root causes, and surgical tips and tricks. Eur J Orthop Surg Traumatol 24(2): 127-135.
- Buck JE (1970) Direct repair of the defect in spondylolisthesis. Preliminary report. J Bone Joint Surg Br 52(3): 432-437.
- Nicol RO, Scott JH (1986) Lytic spondylolysis. Repair by wiring. Spine (Phila Pa 1976) 11(10): 1027-1030.
- Drazin D, Shirzadi A, Jeswani S, Ching H, Rosner J, et al. (2011) Direct surgical repair of spondylolysis in athletes: indications, techniques, and outcomes. Neurosurg Focus 31(5): E9.
- Amoretti N, Huwart L, Hauger O, Browaeys P, Marcy PY, Nouri Y, et al. (2012) Computed tomography- and fluoroscopy-guided percutaneous screw fixation of low-grade isthmic spondylolisthesis in adults: a new technique. Eur Radiol 22(12): 2841-2847.
- Schlenzka D, Remes V, Helenius I, Lamberg T, Tervahartiala P, et al. (2006) Direct repair for treatment of symptomatic spondylolysis and low-grade isthmic spondylolisthesis in young patients: no benefit in comparison to segmental fusion after a mean follow-up of 14.8 years. Eur Spine J 15(10): 1437-1447.
- Mayer TG, Vanharanta H, Gatchel RJ, Mooney V, Barnes D, et al. (1989) Comparison of CT scan muscle measurements and isokinetic trunk strength in postoperative patients. Spine (Phila Pa 1976) 14(1): 33-36.
- Standaert CJ, Herring SA (1980) Spondylolysis: a critical review. Br J Sports Med 34(6): 415-422.
- Micheli LJ, Hall JE, Miller ME (1980) Use of modified Boston brace for back injuries in athletes. Am J Sports Med 8(5): 351-356.
- Szypryt EP, Twining P, Mulholland RC, Worthington BS. (1989) The prevalence of disc degeneration associated with neural arch defects of the lumbar spine assessed by magnetic resonance imaging. Spine (Phila Pa 1976) 14(9): 977-981.
- Styf JR, Willen J (1998) The effects of external compression by three different retractors on pressure in the erector spine muscles during and after posterior lumbar spine surgery in humans. Spine (Phila Pa 1976) 23(3): 354-358.
- Sihvonen T, Herno A, Paljärvi L, Airaksinen O, Partanen J, et al. (1993) Local denervation atrophy of paraspinal muscles in postoperative failed back syndrome. Spine (Phila Pa 1976) 18(5): 575-581.







