Antibiotics Stewardship: Challenges and One Health Approach
Muhammad Kamran1*, Ayoun Tahir1, Amaila Qaiser2 and Muhammad Noman1
1Department of Biosciences, COMSATS University Islamabad, Pakistan
2Faculty of Rehabilitation & Allied Health Sciences, Riphah International University Islamabad, Pakistan
Submission: April 1, 2024; Published: April 24, 2024
*Corresponding author: Muhammad Kamran, Department of Biosciences, COMSATS University Islamabad, Pakistan. Email: kamranahmdani@gmail.com
How to cite this article: Muhammad Kamran*, Ayoun Tahir, Amaila Qaiser and Muhammad Noman. Antibiotics Stewardship: Challenges and One Health Approach. OAJ Gerontol & Geriatric Med. 2024; 8(1): 555729. DOI: 10.19080/OAJGGM.2024.08.555729
Abstract
Healthcare systems must prioritize antibiotic stewardship to combat bacterial infections caused by resistant organisms and reduce inappropriate antibiotic usage. Implementing effective antibiotic stewardship programs in ambulatory and outpatient care settings faces various obstacles, including a lack of specialized staff, inadequate documentation and tracking capabilities. Addressing global health issues such as zoonotic diseases and emerging infections requires an approach that prioritizes the interdependence of human, animal, and environmental health, known as the One Health approach. Comprehensive strategies that address underlying issues contributing to antibiotic stewardship challenges are necessary considering the emergence of antibiotic resistance. Factors such as antibiotic self-medication and structural challenges in healthcare systems contribute to this problem. Public health control measures, such as the fight against cat-transmitted sporotrichosis, have also been successful because of the One Health approach. Appropriate antimicrobial therapy and stewardship greatly assist in improving patient outcomes and clinical prognosis in the context of sepsis. When antibiotic stewardship programs are properly implemented in healthcare facilities, they have the potential to reduce antibiotic overuse and enhance patient safety.
Keywords: Antibiotics Resistance; Antibiotics Stewardship; One Health; Public Health; Rapid Diagnostics; Digital Health Solutions
Keywords: AMR: Antimicrobial Resistance; IDSA: Infectious Diseases Society of America; SCI/D: Spinal Cord Injury/Disorder; LMICs: Low-and-Middle-Income Countries; AMR: Addressing Antimicrobial Resistance
Introduction
Antibiotics Stewardship
Considering the current COVID-19 pandemic, the importance of antibiotic stewardship in healthcare has become vital. Rational use of antibiotics is crucial in effectively managing bacterial and fungal coinfections in COVID-19 patients [1]. To enhance antimicrobial stewardship, it is essential to have a comprehensive understanding of COVID-19 and antimicrobial resistance (AMR), including antibiotic use, as this virus has the potential to cause AMR [2]. The goal of antibiotic stewardship is to reduce the prevalence of antibiotic resistance by conserving antibiotics and optimizing their use [3]. Antimicrobial stewardship programs play a crucial role in providing reasonable empirical antibiotic recommendations within the context of the pandemic [4]. The significance of adhering to guidelines recommended by the Infectious Diseases Society of America (IDSA) is highlighted by interventions in adult oncology and hematopoietic stem cell transplant populations, demonstrating that antibiotic stewardship is not limited to specific patient populations [5]. Evidence-based strategies are needed to optimize antibiotic use, as evidenced by the implementation of antibiotic stewardship programs in various healthcare settings, such as ambulatory care and intensive care units [6,7]. Antibiotic stewardship is a multi-sector effort that begins in the healthcare system and extends to public education and awareness. Increasing public knowledge, sentiment, and behavior regarding antibiotic resistance is the initial step in establishing an effective antibiotic stewardship program [8,9].
To ensure accountability for antibiotic stewardship efforts in nursing homes, it is recommended to involve consultant pharmacists and nurses in these programs [10]. Antibiotic stewardship is equally important in pediatric care, with a focus on optimizing antibiotic use and its impact on antimicrobial resistance [11]. New competency frameworks for antibiotic stewardship in nursing education and practice emphasize the need for interdisciplinary collaboration to achieve optimal antibiotic use [12,13]. Addressing antibiotic resistance and prescribing antibiotics appropriately requires a multidisciplinary team consisting of clinicians, epidemiologists, nurses, and pharmacists to work together in implementing antibiotic stewardship programs [14,15]. The utilization of antibiotic stewardship teams and interventions did not have a negative impact on patient outcomes. Instead, it led to increased therapy that aligned with guidelines and decreased the total number of days of antibiotic therapy [16,17] (Figure 1).
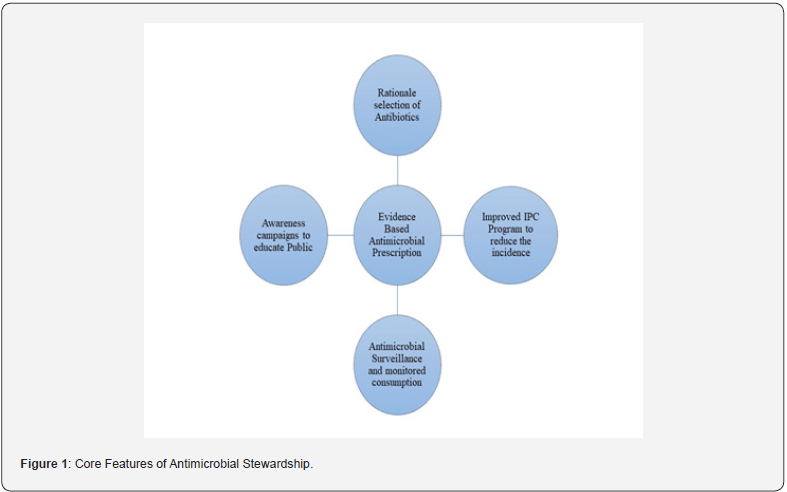
Challenges in Practicing Antibiotics Stewardship
Various healthcare settings present unique challenges to antibiotic stewardship. Antimicrobial stewardship programs face difficulties in outpatient care due to factors such as a lack of specialized staff, inaccurate records, and inadequate monitoring capabilities [18]. It is also challenging to reduce the administration of unnecessary medications in neonatal care, particularly to neonates [19]. A major obstacle to implementing antimicrobial stewardship programs in primary care settings is the detection of unnecessary antibiotic prescriptions [20].
Furthermore, small independent clinics find it difficult to allocate the necessary financial and technical resources to implement antibiotic stewardship initiatives [21]. The attitudes of primary care physicians towards outpatient antibiotic stewardship and antibiotic resistance reveal challenges in addressing antibiotic resistance and inappropriate prescribing [22]. The absence of dedicated time for stewardship, despite leadership commitment to antibiotic stewardship, is another obstacle faced by pharmacists [23]. Implementing effective antibiotic stewardship presents unique challenges in specialized care settings, such as those involving spinal cord injury/disorder (SCI/D) [24]. Despite the reported positive effects in reducing antibiotic use, hospitals in low- and middle-income countries face challenges when trying to implement antibiotic stewardship interventions [25].
Misuse of Antibiotics
One of the biggest obstacles to effective antibiotic stewardship is the overuse and misuse of antibiotics. Antibiotic resistance is becoming an increasingly serious problem due to the dramatic rise in antibiotic consumption worldwide [26]. Studies have shown that unprescribed use, storing antibiotics for later use, and not finishing courses are common patterns of antibiotic misuse [27]. The desire for a faster recovery with fewer side effects has also been associated with antibiotic overuse [28]. Effective stewardship is particularly challenging in ambulatory care settings due to a lack of specialized staff, inadequate documentation, and insufficient tracking capabilities, all of which pose problems for antimicrobial stewardship programs [18]. Antibiotic misuse is also common regardless of socioeconomic status or level of education [29,30]. Neonatal care faces issues with antibiotic administration, knowledge of antibiotic stewardship, and the availability of antibiotic education and resources [19,23].
Business practices contribute to the problem of antibiotic misuse as they are involved in the marketing and distribution systems [31]. Challenges in post-discharge antibiotic stewardship have been identified, including antibiotic overuse, prolonged use of antibiotics, and inefficient use of specific antibiotic treatments [32,33]. While antibiotic stewardship initiatives have proven effective in reducing antibiotic use, their implementation in low- and middle-income countries remains difficult. Systematic reviews and meta-analyses have shown that both developed, and less developed regions have high rates of antibiotic misuse among university students and the general population [30,34]. This suggests that the misuse of antibiotics is not limited to specific regions. Implementing effective stewardship practices is crucial because antibiotic misuse is a key factor in the rise of antibiotic resistance [35].
Campaigns for Public Awareness
One of the most pressing problems in healthcare today is the general public’s underestimation of the importance of antibiotic stewardship [36]. Prescribers’ actions can be impacted by patient demands and expectations, and most antimicrobials are prescribed in community settings. Community pharmacists continue to face obstacles in raising public awareness due to a lack of resources, including time, personnel, training, and technology, which prevents them from practicing antimicrobial stewardship effectively [37]. There may be a deficiency in public education and awareness initiatives if some members of the community pharmacy team are unaware of European Antibiotic Awareness Day [38].
Lack of understanding about the role of nurses in combating antibiotic resistance may be attributed, in part, to the fact that nursing prelicensure programs do not adequately teach students about stewardship interventions or the seriousness of antibiotic resistance [39]. Antimicrobial stewardship is an important topic, but interns and doctors in Nigeria don’t seem to understand it [40]. Medical students and residents lack awareness of the importance of antibiotic stewardship, and there is a need to improve the effectiveness of educational campaigns targeting this demographic on social media [41]. Stewardship activities can be hindered by a lack of education and by the culture of the hospital or unit, highlighting the necessity for increased educational initiatives to overcome these obstacles [42].
Discrepancies in Global Regulation
Antibiotic resistance is a growing concern worldwide due to increased antibiotic consumption. Therefore, it is essential to monitor antibiotic consumption globally to support policies aimed at reducing consumption and resistance while ensuring access to these essential medications [26]. However, issues such as unregulated over-the-counter sales, low-quality antibiotics, and limited access to necessary antibiotics continue to be major concerns, especially in low- and middle-income countries (LMICs) [43]. The fact that over half of all antibiotics are available over the counter exacerbates the problem, particularly in underdeveloped nations with poor drug control [44].
Optimizing the use of antimicrobials is crucial for preventing the emergence of resistant strains and protecting patients from the unnecessary harms of medication. This is particularly important in pediatric care, where antibiotic stewardship presents both challenges and opportunities, such as in the case of preterm infants [45]. Healthcare utilization disparities influence antibiotic use. To prevent underserved populations from experiencing reduced appropriate antibiotic use, it is important to consider the varying rates of antibiotic-treatable diseases and healthcare-seeking behaviors among different populations [46]. Exploring workarounds and their perceived impact on antibiotic stewardship in healthcare settings has revealed the complexities and barriers encountered in these efforts [47]. Challenges in antibiotic stewardship go beyond access alone. Targeted action is needed to reduce antibiotic overuse in vulnerable populations, such as individuals with spinal cord injuries or disorders (SCI/D), who face unique challenges in antibiotic stewardship [24].
The One Health Approach
The goal of the One Health strategy is to achieve optimal health outcomes for all living things by recognizing the interdependence of humans, animals, and plants with their shared environment [48]. This strategy operates at different levels, including local, subnational, national, regional, and global, with the objective of achieving optimal health through interdisciplinary collaboration [49]. The One Health strategy has received support from various groups, such as the WHO, Public Health England, and the American Medical Association [49] and has been incorporated into the plans of many nations and international organizations to address pressing issues like antibiotic resistance [50]. It has been recognized as a realistic, durable, and cost-effective approach to achieving high standards of health for all living things [21].
The One Health approach has been instrumental in understanding, preventing, and managing zoonotic diseases, both emerging and long-standing, by coordinating efforts between the public health and animal health sectors [51]. It has also been successfully utilized in collaboration with medical and veterinary institutions to study zoonotic diseases, demonstrating its effectiveness in addressing health issues across human, animal, and environmental domains [52]. Due to the interconnectedness of human, animal, and environmental health, a systems approach to One Health is necessary, highlighting the importance of considering the environment as an integral part of the One Health framework [53]. As a framework with guiding principles, the One Health approach facilitates cross-sectoral collaboration with various stakeholders to holistically control diseases, such as Q fever [54]. It has also been recognized as crucial in addressing global health concerns beyond zoonoses and has played a key role in responding to unprecedented pandemics like COVID-19 [55]. Many regions, including Kenya, have documented both the successes and failures of the One Health approach, with Kenya utilizing the approach to adopt and implement strategies for controlling and preventing zoonotic diseases [51].
Addressing antimicrobial resistance (AMR) and promoting responsible use of antibiotics requires a strong connection between antibiotic use in animals and humans within the One Health approach, as well as antibiotic stewardship. A collaborative, multisectoral strategy is needed to address antimicrobial resistance (AMR). This strategy incorporates antibiotic use in both animals and agriculture, in addition to human use [56]. This approach shows how the animal and human health sectors can work together more effectively. Antibiotic resistance can be better understood by taking a One Health approach, since research has shown that people, animals, and the environment are all interdependent [57]. The extensive use of antibiotics in animals, including protected antibiotics and crossover-use, highlights the importance of community-level interventions that promote responsible antibiotic use in animals through a One Health approach [58].
In addition, to promote antimicrobial stewardship, legislation, and advocacy, as well as to limit the dissemination cycle of antibiotic resistance, worldwide multidisciplinary and integrated approaches grounded in One Health principles are necessary [59]. An even more compelling argument in favor of a One Health strategy is the fact that antibiotic use in livestock raises the risk of antibiotic resistance and contaminates human food with antibiotic residues [60]. The goal of antibiotic stewardship is to ensure that antibiotics are prescribed sparingly and used appropriately by implementing evidence-supported interventions [61]. To standardize antibiotic use and prevent drug-resistant bacterial infections, stewardship guidelines have been developed by governments and health organizations worldwide [61]. Antibiotic stewardship and public health initiatives face obstacles, such as the improper prescription of antibiotics in ambulatory care clinics [62].
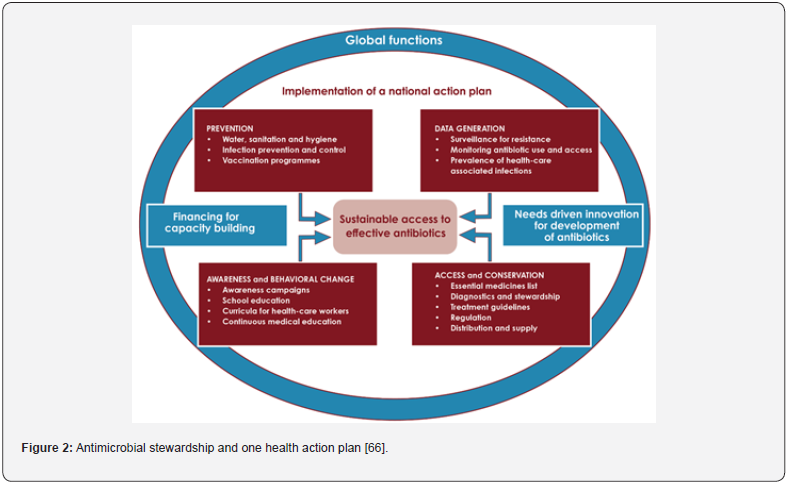
The urgency of antibiotic stewardship in maximizing antibiotic use is underscored by the fact that antibiotic resistance is significantly accelerated by inappropriate antibiotic use [63]. There needs to be action to combat the simultaneous pandemics of COVID-19 and antibiotic resistance since antibiotic use in treating the virus has compromised antibiotic stewardship [64]. ASPs are vital in encouraging responsible antibiotic use and have a significant impact on antibiotic use [65]. A team consisting of clinicians, epidemiologists, nurses, and pharmacists could be an effective way to encourage responsible antibiotic use [14]. To combat the rise of antibiotic-resistant microbes and encourage their responsible use, it is crucial to establish a connection between the One Health approach’s emphasis on animal and human antibiotic use and antibiotic stewardship. Antimicrobial resistance must be tackled through a collaborative, multisectoral strategy, as the One Health approach acknowledges the interdependence of human, animal, and environmental health. The promotion of responsible antibiotic use and improvement of antibiotic use are both greatly aided by antibiotic stewardship programs (Figure 2) [66].
Successful One Health Initiatives
Recognized as the most effective method for controlling diseases like rabies, the One Health approach emphasizes ecological concerns and large-scale oral vaccination campaigns. In 2020, Acharya and colleagues further supported this approach [21]. The importance of health system resilience and collaborative learning initiatives has also led to the recognition of the One Health concept as a critical framework for tackling global health challenges [67]. The One Health initiative has highlighted the importance of global partnerships in addressing public health issues, such as controlling antibiotic resistance through international collaborations [68]. A multidisciplinary approach is crucial in combating pandemics and public health threats. In Indonesia, the One Health concept has been successfully implemented to address the spread of zoonotic diseases [69].
One Health Approach & Technology Innovation
There is significant hope that rapid diagnostic testing can improve antibiotic prescribing behaviors and serve as an effective diagnostic antibiotic stewardship tool [70]. Diagnostic stewardship initiatives within healthcare systems can address infectious syndromes such as UTIs, GERDs, COVID-19, and bloodstream infections [71]. Antibiotic stewardship programs should involve the microbiology lab, and new diagnostic tools may assist doctors in prescribing the appropriate medication more quickly and accurately [72]. One-way diagnostic stewardship works is by reducing tests with a high likelihood of false positive results [33]. Regardless of illness severity, similar approaches should be considered for individuals with pathogen-negative sepsis due to the positive outcomes observed when antimicrobial stewardship programs and molecular rapid diagnostic testing are utilized together [73].
Digital health solutions have completely transformed the tracking of antibiotic usage and resistance patterns. Studies indicate that healthcare providers can make better decisions regarding antibiotic prescribing and stewardship through realtime data on antibiotic consumption and resistance provided by digital health solutions [5,74]. Telemedicine and mobile applications can be utilized to educate patients on the proper use of antibiotics and encourage their participation in stewardship initiatives [75,76]. Rapid diagnostics and digital health solutions have made substantial contributions to antibiotic stewardship efforts by improving bacterial infection identification, reducing unnecessary antibiotic prescriptions, and providing real-time data on antibiotic use and resistance patterns. These technological innovations have the potential to greatly enhance antibiotic stewardship practices and patient care.
Conclusion & Future Recommendations
The One Health approach and antibiotic stewardship play pivotal roles in addressing global health challenges, including antimicrobial resistance and zoonotic diseases. The One Health approach emphasizes the interconnectedness of human, animal, and environmental health, highlighting the need for collaborative, multisectoral strategies. Antibiotic stewardship focuses on promoting responsible use of antibiotics, reducing unnecessary prescriptions, and combating antimicrobial resistance. The success of these initiatives requires breaking down interdisciplinary barriers and fostering collaboration between healthcare professionals, veterinarians, environmental scientists, and other stakeholders. Future research should focus on evaluating the effectiveness of the One Health approach, developing specific evaluation methods and tools, and addressing the challenges associated with interdisciplinary collaboration. Additionally, efforts are needed to increase awareness and understanding of antibiotic stewardship programs among healthcare professionals and students. Technological innovations, such as rapid diagnostics and digital health solutions, should be further integrated into antibiotic stewardship efforts to improve the accuracy of bacterial infection identification, reduce unnecessary antibiotic prescriptions, and monitor antibiotic use and resistance patterns in real time.
The implementation of antibiotic stewardship programs in various healthcare settings, including emergency departments, urgent care centers, and long-term care facilities, has shown promising results. However, there is a need for continued efforts to overcome barriers and obstacles to antibiotic stewardship, such as the inappropriate use of antibiotics and the lack of awareness among healthcare professionals. In the future, collaborative learning initiatives, such as the Joint Initiative for Teaching and Learning on Global Health Challenges and One Health, can provide valuable opportunities for multidisciplinary education and research. These initiatives can foster impactful collaborations and support an interconnected/integrated approach to health. Moreover, the role of social media in guiding One Health initiatives and the potential of clinical One Health as a novel healthcare solution for underserved communities should be further explored.
References
- Nori P, Cowman K, Chen V, Bartash R, Szymczak W, et al. (2021) Bacterial and Fungal Coinfections in COVID-19 Patients Hospitalized During the New York City Pandemic Surge. Infect Control Hosp Epidemiol 42(1): 84-88.
- Adebisi YA, Alaran AJ, Okereke M, Oke GI, Amos OA, Olaoye O, et al. (2021) COVID-19 and Antimicrobial Resistance: A Review. Infect Dis Res Treat 31:14:11786337211033870.
- Aricò MO, Valletta E, Caselli D (2023) Appropriate Use of Antibiotic and Principles of Antimicrobial Stewardship in Children. Children 10(4): 740.
- Maryam S, Fatima R, Ashfaq M, Hassan N, Bibi A, et al. (2023) Epidemiological studies of COVID-19 disease: A story of SARS CoV-2 infection from origin to hospital.
- Pillinger KE, Bouchard J, Withers S, Mediwala KN, McGee E abasi U, et al. (2019) Inpatient Antibiotic Stewardship Interventions in the Adult Oncology and Hematopoietic Stem Cell Transplant Population: A Review of the Literature. Ann Pharmacother 54(6): 594-610.
- Keller SC, Cosgrove SE, Miller MA (2022) A Framework for Implementing Antibiotic Stewardship in Ambulatory Care: Lessons Learned from the Agency for Healthcare Research and Quality Safety Program for Improving Antibiotic Use. Antimicrob Steward Healthc Epidemiol 2(1): e109.
- Wunderink RG, Srinivasan A, Barie PS, Chastre J, Dela Cruz CS, et al. (2020) Antibiotic Stewardship in the Intensive Care Unit. An Official American Thoracic Society Workshop Report in Collaboration With the AACN, CHEST, CDC, and SCCM. Ann Am Thorac Soc 17(5): 531-540.
- Habib MB, Anayyat U, Usmani F, Jafri AR, Ramzan A, Safdar N, et al. (2023) Investigation of Food Borne Pathogens, Coliforms and Fecal Coliforms in Raw Meat Samples of Beef, Chicken and Fish. J Pharm Negat Results, pp. 4044-4054.
- Kamran M, Choudary MA, Amin H, Asghar S, Shahid A, et al. (2022) Antimicrobial Susceptibility Pattern of Pseudomonas aeruginosa isolated from Clinical and Environmental Sources in Punjab, Pakistan: Antimicrobial Susceptibility Pattern of Pseudomonas Aeruginosa. Pak Biomed J, pp. 34-38.
- Ashraf MS, Bergman S (2021) The Case for Consultant Pharmacists as Key Players in Nursing Home Antibiotic Stewardship Programs. J Am Med Dir Assoc 22(1): 6-8.
- Grammatico-Guillon L, Abdurrahim L, Shea K, Astagneau P, Pelton SI (2019) Scope of Antibiotic Stewardship Programs in Pediatrics. Clin Pediatr (Phila) 58(11-12):1291-1301.
- Courtenay M, McEwen J (2020) Applying an Antimicrobial Stewardship Competency Framework in Nurse Education and Practice. Nurs Stand 35(3): 41-46.
- Kamran M, Iqbal U, Saleem M, Khan AA, Haq MU, et al. (2022) Antibiogram and Prevalence of ESBL Genes in Escherichia Coli from Clinical Specimen. J Pharm Negat Results, pp. 1915-1924.
- Chang EY, Fatima S, Stevens ES (2018) Further Considerations Regarding Duration of Antibiotic Therapy for Sinusitis. Jama Intern Med 178(8): 1138-1139.
- Jackson BR, Chow N, Forsberg K, Litvintseva AP, Lockhart SR, et al. (2019) On the origins of a species: what might explain the rise of Candida auris? J Fungi 5(3): 58.
- Foolad F, Huang A, Nguyen CT, Colyer L, Lim M, et al. (2018) A Multicentre Stewardship Initiative to Decrease Excessive Duration of Antibiotic Therapy for the Treatment of Community-Acquired Pneumonia. J Antimicrob Chemother 73(5):1402-1407.
- Anwar I, Rafiq S, Ahmad K, Mubeen MU, Mudassar A, et al. (2022) Antimicrobial Susceptibility Pattern of E. Coli in Patients with Urinary Tract Infection at a Tertiary Care Hospital, Rawalpindi. Pak J Med Health Sci 16(06): 967-967.
- Drwiega EN, Griffith N, Herald F, Badowski ME (2023) How to Design and Implement an Outpatient Antimicrobial Stewardship Programme. Drugs Context 12: 2022-8-2.
- Hamidi M, Blatz MA (2022) A National Survey of Neonatal Nurses’ Knowledge, Beliefs, and Practices of Antibiotic Stewardship. Adv Neonatal Care 23(1): E22-E28.
- Singh-Franco D, Mohammed SH, Eckardt P, Goldman JL, Wolowich WR (2022) Identification of Inappropriate Antibiotic Orders During Implementation of a Multidisciplinary Antimicrobial Stewardship Program Within the Primary Care Setting. J Patient Saf 18(5): e889-e894.
- Acharya KR, Brankston G, Soucy, Cohen A, Hulth A, et al. (2021) Evaluation of an OPEN Stewardship Generated Feedback Intervention to Improve Antibiotic Prescribing Among Primary Care Veterinarians in Ontario, Canada and Israel: Protocol for Evaluating Usability and an Interrupted Time-Series Analysis. BMJ Open 11(1): e039760.
- Zetts RM, Stoesz A, Garcia AM, Doctor JN, Gerber JS, et al. (2020) Primary Care Physicians’ Attitudes and Perceptions Towards Antibiotic Resistance and Outpatient Antibiotic Stewardship in the USA: A Qualitative Study. BMJ Open 10(7): e034983.
- Appaneal HJ, Luther MK, Timbrook TT, LaPlante KL, Dosa D (2018) Facilitators and Barriers to Antibiotic Stewardship: A Qualitative Study of Pharmacists’ Perspectives. Hosp Pharm 54(4): 250-258.
- Skelton F, Suda KJ, Evans CT, Trautner BW (2018) Effective Antibiotic Stewardship in Spinal Cord Injury: Challenges and a Way Forward. J Spinal Cord Med 42(2): 251-254.
- Dijck CV, Vlieghe E, Cox J (2018) Antibiotic Stewardship Interventions in Hospitals in Low-and Middle-Income Countries: A Systematic Review. Bull World Health Organ 96(4): 266-280.
- Klein EY, Van Boeckel TP, Martínez E, Pant S, Gandra S, et al. (2018) Global Increase and Geographic Convergence in Antibiotic Consumption Between 2000 and 2015. Proc Natl Acad Sci USA 115(15): E3463-E3470.
- Jayawardhana Y, Premaratne A, Kalpani S, Jayasundara S, Jayawardhane G, et al. (2023) Investigating the Drivers for Antibiotic Use and Misuse Amongst Medical Undergraduates-perspectives From a Sri Lankan Medical School. Plos Glob Public Health 3(3): e0001740.
- Santana PT, Rosas SLB, Ribeiro BE, Marinho Y, de Souza HS (2022) Dysbiosis in inflammatory bowel disease: pathogenic role and potential therapeutic targets. Int J Mol Sci 23(7): 3464.
- Mallah N, Orsini N, Figueiras A, Takkouche B (2022) Income Level and Antibiotic Misuse: A Systematic Review and Dose-response Meta-Analysis. Eur J Health Econ 23(6): 1015-1035.
- Mallah N, Orsini N, Figueiras A, Takkouche B (2022) Education Level and Misuse of Antibiotics in the General Population: A Systematic Review and Dose-response Meta-Analysis. Antimicrob Resist Infect Control 11(1): 24.
- Kotwani A, Bhanot A, Singal GL, Gandra S (2022) Marketing and Distribution System Foster Misuse of Antibiotics in the Community: Insights from Drugs Wholesalers in India. Antibiotics (Basel) 11(1): 95.
- Soper NS, Appukutty AJ, Paje D, Petty LA, Flanders SA, et al. (2022) Antibiotic Overuse After Discharge from Medical Short-Stay Units. Infect Control Hosp Epidemiol 43(11): 1689-1692.
- Vaughn VM, Ratz D, Greene MT, Flanders SA, Gandhi T, et al. (2022) Antibiotic Stewardship Strategies and Their Association with Antibiotic Overuse After Hospital Discharge: An Analysis of the Reducing Overuse of Antibiotics at Discharge (Road) Home Framework. Clin Infect Dis 75(6): 1063-1072.
- Peng D, Wang X, Xu Y, Sun C, Zhou X (2018) Antibiotic Misuse Among University Students in Developed and Less Developed Regions of China: A Cross-Sectional Survey. Glob Health Action 11(1): 1496973.
- Alsuwayt B, Shiromwar S (2020) Understanding of the Antibiotic Misuse Among the General Public of Rafha City of Saudi Arabia. J Pharm Res Int 32(21): 123-130.
- Garau J, Bassetti M (2018) Role of Pharmacists in Antimicrobial Stewardship Programmes. Int J Clin Pharm 40(5): 948-952.
- Lee Y, Bradley N (2023) Antimicrobial Stewardship Practices in a Subset of Community Pharmacies Across the United States. Pharmacy (Basel) 11(1): 26.
- Tonna A, Weidmann AE, Sneddon J, Stewart D (2020) Views and Experiences of Community Pharmacy Team Members on Antimicrobial Stewardship Activities in Scotland: A Qualitative Study. Int J Clin Pharm 42(5): 1261-1269.
- Van Lonkhuyzen CL, Decker T, Carlson C (2023) The Christian Nurse’s Role in Antibiotic Stewardship. J Christ Nurs 40(2): E14-E18.
- Ayosanmi OS, Akafa TA, Adeniran O, Ayosanmi T, Omoregie DO, et al. (2023) Qualitative Assessment of Knowledge, Perception and Experience of Physicians About Antimicrobial Stewardship in Nigeria During COVID-19 Pandemic. Eur J Med Health Sci 5(1): 1570.
- Kalu IC, Mukhopadhyay S, Dukhovny D, Young RM, Guzman-Cottrill JA (2023) Knowledge, Attitudes, and Perceptions About Antibiotic Stewardship Programs Among Neonatology Trainees. Am J Perinatol 40(8): 893-897.
- Catanzaro M (2022) Antibiotic Stewardship for Nurses: Using E-Learning Modules to Bridge the Education Gap. Antimicrob Steward Healthc Epidemiol 2(1): e7.
- Pauwels I, Versporten A, Drapier N, Vlieghe E, Goossens H (2021) Hospital Antibiotic Prescribing Patterns in Adult Patients According to the WHO Access, Watch and Reserve Classification (AWaRe): Results from a Worldwide Point Prevalence Survey in 69 Countries. J Antimicrob Chemother 76(6): 1614-1624.
- Rani N, Kumar Ks, Saranya S (2022) Community Pharmacists’ Knowledge, Attitude, and Nonprescription Dispensing Practices of Antibiotics: An Explorative Study in a Selected City of South India. J Res Pharm Pract 11(2): 51-58.
- Mukhopadhyay S, Sengupta S, Puopolo KM (2018) Challenges and Opportunities for Antibiotic Stewardship Among Preterm Infants. Arch Dis Child - Fetal Neonatal Ed 104(3): F327-F332.
- Olesen SW, Kanjilal S, Kissler SM, Sun DS, Grad YH (2021) Effects of Racial/Ethnic Disparities in Healthcare Utilization on Antibiotic Use, United States, 2016/2018. Medrxiv 12(9): 21266965.
- Mula C, Human N, Middleton L (2019) An Exploration of Workarounds and Their Perceived Impact on Antibiotic Stewardship in the Adult Medical Wards of a Referral Hospital in Malawi: A Qualitative Study. BMC Health Serv Res 19(64).
- Lokossou V, Atama NC, Nzietchueng S, Koffi BY, Iwar V, et al. (2021) Operationalizing the ECOWAS Regional One Health Coordination Mechanism (2016-2019): Scoping Review on Progress, Challenges and Way Forward. One Health 13: 100291.
- Mackenzie JS, Jeggo M (2019) The One Health Approach-Why Is It So Important? Trop Med Infect Dis 4(2): 88.
- Collignon P, McEwen SA (2019) One Health-Its Importance in Helping to Better Control Antimicrobial Resistance. Trop Med Infect Dis 4(1): 22.
- Munyua P, Njenga MK, Osoro E, Onyango C, Bitek A, et al. (2019) Successes and Challenges of the One Health Approach in Kenya Over the Last Decade. BMC Public Health 19(Suppl 3): 465.
- Espí A, Cerro A del, Oleaga Á, Rodríguez-Pérez M, López C, et al. (2021) One Health Approach: An Overview of Q Fever in Livestock, Wildlife and Humans in Asturias (Northwestern Spain). Animals (Basel) 11(5): 1395.
- Essack SY (2018) Environment: The Neglected Component of the One Health Triad. Lancet Planet Health 2(6): e238-e239.
- Rahaman R, Milazzo A, Marshall H, Bi P (2019) Is a One Health Approach Utilized for Q Fever Control? A Comprehensive Literature Review. Int J Environ Res Public Health 16(5): 730.
- Villanueva-Cabezas JP, Rajkhowa A, Campbell A (2020) One Health Needs a Vision Beyond Zoonoses. Transbound Emerg Dis 67(6): 2271-2273.
- Torumkuney D, Kundu S, Vu GV, Nguyen HA, Pham HV, et al. (2022) Country Data on AMR in Vietnam in the Context of Community-Acquired Respiratory Tract Infections: Links Between Antibiotic Susceptibility, Local and International Antibiotic Prescribing Guidelines, Access to Medicines and Clinical Outcome. J Antimicrob Chemother 77(Suppl_1): i26-i34.
- Donner L, Staley ZR, Petali J, Sangster JL, Li X, et al. (2022) The Human Health Implications of Antibiotic Resistance in Environmental Isolates from Two Nebraska Watersheds. Microbiol Spectr 10(2): e0208221.
- Arnold JC, Day D, Hennessey M, Alarcon P, Gautham M, et al. (2021) If It Works in People, Why Not Animals?: A Qualitative Investigation of Antibiotic Use in Smallholder Livestock Settings in Rural West Bengal, India. Antibiotics (Basel) 10(12): 1433.
- Aslam B, Khurshid M, Arshad M, Muzammil S, Rasool M, et al. (2021) Antibiotic Resistance: One Health One World Outlook. Front Cell Infect Microbiol 11: 771510.
- Manyi-Loh CE, Mamphweli S, Meyer EL, Okoh AI (2018) Antibiotic Use in Agriculture and Its Consequential Resistance in Environmental Sources: Potential Public Health Implications. Molecules 23(4): 795.
- Gerber JS, Jackson MA, Zaoutis TE (2021) Antibiotic Stewardship in Pediatrics. Pediatrics 147(1): e2020040295.
- Ray M, Tallman GB, Bearden DT, Elman MR, McGregor JC (2019) Antibiotic Prescribing Without Documented Indication in Ambulatory Care Clinics: National Cross-Sectional Study. BMJ 367: l6461.
- Zhao X, Yu Z, Ding T (2020) Quorum-sensing regulation of antimicrobial resistance in bacteria. Microorganisms 8(3): 425.
- Mustapha A, Nikau J, Isa T (2020) COVID-19 and Antibiotic Resistance: Parallel Pandemics and Different Intercessions. Microbes Infect Dis 2(1): 15-24.
- Morgan JR, Barlam TF, Drainoni ML (2018) A Qualitative Study of the Real-World Experiences of Infectious Diseases Fellows Regarding Antibiotic Stewardship. Open Forum Infect Dis 5(9): ofy102.
- Cars O, Chandy SJ, Mpundu M, Peralta AQ, Zorzet A, et al. (2021) Resetting the agenda for antibiotic resistance through a health systems perspective. Lancet Glob Health 9(7): e1022-e1027.
- Yasobant S, Bruchhausen W, Saxena D, Falkenberg T (2019) One Health Collaboration for a Resilient Health System in India: Learnings from Global Initiatives. One Health 8: 100096.
- Cabrera-Pardo JR, Lood R, Udekwu KI, González-Rocha G, Munita JM, et al. (2019) A One Health - One World Initiative to Control Antibiotic Resistance: A Chile - Sweden Collaboration. One Health 8: 100100.
- Rachmawati F, Khariri K (2020) The Approach of One Health Concept in Addressing the Spread of Zoonotic Diseases in Indonesia. Int Conf Agromedicine Trop Dis 3(1): 43-50.
- Weragama K, Mudgil P, Whitehall J (2022) Diagnostic Stewardship-The Impact of Rapid Diagnostic Testing for Paediatric Respiratory Presentations in the Emergency Setting: A Systematic Review. Children 9(8): 1226.
- Coffey K, Morgan DJ, Claeys KC (2023) Diagnostic Stewardship: What Impacts Antibiotics Use? Curr Opin Infect Dis 36(4): 270-275.
- Palavecino E, Williamson J, Ohl C (2020) Collaborative Antimicrobial Stewardship. Infect Dis Clin North Am 34(1): 51-65.
- Lockhart SR, Etienne KA, Vallabhaneni S, Farooqi J, Chowdhary A, et al. (2017) Simultaneous Emergence of Multidrug-Resistant Candida auris on 3 Continents Confirmed by Whole-Genome Sequencing and Epidemiological Analyses. Clin Infect Dis 64(2): 134-140.
- Fowler VG, Jezek A, Spivak ES, Talkington K (2022) Urgent, Comprehensive Federal Action Needed to Stem Mortality and Medicare Costs Associated with Antimicrobial Resistance. Clin Infect Dis 74(6): 1107-1111.
- Aloni-Grinstein R, Rotem S (2021) COVID-19 Pandemic: A Lesson for Antibiotic and Antiseptic Stewardship. Am J Public Health Res 9(2): 48-51.
- Brinker DL, Foley KA, Zhou Y, Acevedo-Callejas M, Yuwei Li, et al. (2022) Use of a Smartphone Medication Reminder Application to Support Emerging Adult Adherence to Non-Antibiotic Treatment for Viral Upper Respiratory Tract Infection. J Prim Care Community Health 13: 21501319221129732.






























