Lower Psoas Muscle Area is associated with increased mortality after endovascular aneurysm repair in older adults.
Jiuan Ting1, Kien Chan2, Eileen Zhang3, Warren Raymond4, Joy Lu5, Chi Ho Ricky Kwok6, Shirley Jansen7 and Charles Inderjeeth8*
1Jiuan Ting, Bachelor of Medicine and Surgery (University of Western Australia); FRACP. No institutional affiliations
2Kien Chan, Bachelor of Medicine and Surgery, FRACP. Department of Geriatric Medicine, Sir Charles Gairdner Hospital, Perth, Australia
3Eileen Zhang, Bachelor of Medicine and Surgery (University of Western Australia), MMed (critical care). No institutional affiliations
4Warren Raymond, Bachelor of Science, University of Western Australia, Australia
5Joy Lu, Bachelor of Medicine and Surgery, RANZCR. Sir Charles Gairdner Osborne Park Health Care Group.
6Chi Ho Ricky Kwok, MBChB FRACS(Vasc). Dept of Vascular and Endovascular Surgery, Sir Charles Gairdner Hospital, WA
7Shirley Jansen MBChB FRACS FRCS PhD. Professor Vascular Surgery, Curtin Medical School, Curtin University, Perth WA. Head of Dept of Vascular and Endovascular Surgery, Sir Charles Gairdner Hospital, Perth WA. Director Heart and Vascular Research Institute, Harry Perkins Medical Research Institute, Perth WA. Clin Prof University of Western Australia
8Charles Inderjeeth. Consultant Physician (Geriatrician & Rheumatologist), NHMRC-NICS DVA Fellow. Clinical Professor (University of Western Australia). Clinical Dean (Curtin University). Clinical Epidemiologist. Director of Clinical Training, Research and Programs (Osborne Park Hospital Program). Linear Clinical Research
Submission: October 09, 2023; Published: November 02, 2023
*Corresponding author: Charles Inderjeeth, Sir Charles Gairdner Osborne Park Hospital Group, Hospital Avenue, Nedlands, WA 6009, Australia Email: Charles.Inderjeeth@health.wa.gov.au
How to cite this article: Jiuan Ting, Kien Chan, Eileen Zhang, Warren Raymond, Joy Lu, Chi Ho Ricky Kwok, Shirley Jansen and Charles Inderjeeth*. Lower Psoas Muscle Area is associated with increased mortality after endovascular aneurysm repair in older adults.. OAJ Gerontol & Geriatric Med. 2023; 7(5): 555721. DOI: 10.19080/OAJGGM.2023.07.555721
Abstract
Lower Psoas Muscle Area is associated with increased mortality after endovascular aneurysm repair in older adults.
Introduction: Psoas muscle area (PMA) is an easily measurable surrogate of sarcopenia, which may be associated with mortality and other adverse post-operative outcomes. This study aimed to further explore the relationship between PMA and outcomes post elective infrarenal aortic aneurysm (AAA) endovascular repair of (EVAR) in older adults.
Methods: A retrospective observational study of people aged over 65 years undergoing EVAR for asymptomatic AAA over 5 years. Patients with connective tissue disease, mycotic or inflammatory AAA, emergency or non-elective cases were excluded. The primary outcome was 5-year mortality. Secondary outcomes were 30-day readmissions, post-operative complications, and length of stay.
Results: Ninety-seven patients (mean age 77.5 years, 78% male) were assessed. Adjustment for age, sex and body mass index also showed that patients in the lowest PMA tertile had an increased 5-year mortality (HR 2.76, 95%CI 1.08, 7.03; P=0.034). Adjustment for frailty, measured by modified frailty index, showed only a minimal attenuation of the association (HR 2.30, 95%CI 0.99, 5.34; p=0.054). Being in the lowest PMA tertile did not increase LOS, 30-day readmissions, complications, or influence discharge destination.
Conclusion: The lowest tertile of PMA, as a surrogate marker for sarcopenia, was associated with increased mortality in patients undergoing EVAR for asymptomatic infrarenal AAA, which was only marginally attenuated by frailty adjustment. Although sarcopenia is a complex phenomenon and the literature surrounding it is still evolving, PMA may form one component of risk assessment in patients undergoing EVAR.
Keywords: Frailty; Psoas Muscle Area; Sarcopaenia; Vascular Surgery
Keywords: PMA: Psoas Muscle Area; AAA: Abdominal Aortic Aneurysm; EVAR: Endovascular Aneurysm Repair; LOS: Length of Stay; PMA: Psoas Muscle Area; CT: Computed Tomography; GEKO: Governance Evidence Knowledge Outcomes; SPSS: Statistical Package for the Social Sciences
Introduction
The evolution of abdominal aortic aneurysm (AAA) repair from open to endovascular aneurysm repair (EVAR) has great relevance in older adults, who often have more comorbid conditions, are frailer and have higher levels of sarcopenia. EVAR, where technically feasible and where the maximum aortic diameter has reached threshold (>5.5cm in men and >5.2cm in women) is minimally invasive and involves the deployment of a stent graft inside the aneurysm via percutaneous femoral access, and has several advantages including avoidance of general anesthesia, reduced length of stay (LOS) and reduced postoperative mortality [1].
Sarcopenia and frailty are distinct but overlapping conditions. Frailty is “a biologic syndrome of decreased reserve and resistance to stressors, resulting from cumulative declines across multiple physiologic systems, and causing vulnerability to adverse outcomes” [2]. There have been multiple screening tools developed in an attempt to assess frailty, which usually follow either a phenotypic or cumulative deficits model [3]. Despite the differing tools used to measure frailty, higher frailty scores are associated with increased mortality, post-operative complications, and length of stay in surgical patients [4], higher mortality following open AAA repair and carotid endarterectomy [5] and vascular surgery [6], and LOS and discharge destination in vascular ward patients [7].
Sarcopenia, on the other hand, is defined as “a progressive and generalised skeletal muscle disorder that is associated with an increased likelihood of adverse outcomes including falls, fractures, physical disability and mortality” and “confirmed by the presence of low muscle quantity or quality” [8,9]. Several parameters have been used to define muscle quantity, including psoas muscle area (PMA). A decline in PMA seems to be representative of decreased lean body muscle mass and sarcopenia, at least in haemodialysis patients. However, the European guidelines suggest more research is required to formalise whether PMA is a reasonable surrogate for skeletal muscle mass [10].
There is emerging evidence to suggest that PMA is associated with mortality in older vascular surgical patients following endovascular aortic surgery, and to a lesser extent post-operative complications. Some, but not all studies [11,12], have shown that a reduction in PMA correlated with higher mortality following EVAR, open AAA repair or other vascular intervention [13-21]; but none of the studies to date have included frailty scoring in conjunction with PMA in this setting.
In the AAA population, widespread use of computer tomography (CT) imaging enables easy measurement of PMA as a representation of sarcopenia. PMA could therefore be an easy to obtain predictive tool for outcomes following EVAR, and to determine whether the procedure is in the patient’s favour or not. Our study further explored the relationship between PMA with post-operative outcomes and mortality in older adults undergoing asymptomatic infrarenal EVAR, seeking to assess whether similar correlations between PMA and mortality were found in the local surgical population in Western Australia. We chose to include frailty to further clarify this issue.
Methods
A retrospective observational study of patients 65 years and older who had undergone an elective EVAR for asymptomatic infrarenal AAA, in a tertiary metropolitan centre in Western Australia, from 1st January 2013 to 31st December 2017. Patients were excluded if the cause of the AAA was connective tissue disease, mycosis, or inflammation, and if EVAR was performed as an emergency or non-elective procedure for symptomatic AAA.
Retrospective data was collected from chart review and electronic medical records. Baseline patient characteristics included age, sex, current smoking status, Charlson comorbidity index, the presence of coronary artery disease (history of previous myocardial infarction, coronary artery bypass graft surgery or percutaneous coronary intervention), cerebrovascular disease, chronic kidney disease, diabetes, hypertension, peripheral vascular disease (intermittent claudication, critical limb ischemia, revascularisation or lower limb arteries), as well as, maximum antero-posterior AAA diameter, BMI, and preoperative haemoglobin levels. A modified frailty index (following a cumulative deficits model of frailty) was also calculated for each patient based on information found in medical records. This provided a score out of 11, with a score of 0 indicating an absence of frailty, and a score of 11 indicating the highest level of frailty [22].
PMA was measured at the mid L4 vertebral body on preoperative computed tomography (CT) scans. Where possible, 3mm slice thickness was chosen for measurement, however if this was not possible, 5mm or 1mm slice thickness was chosen. 3mm slice thickness was the most common and thus was the preferred slice thickness for consistency. A radiologist assisted with initial training of one researcher. Following this, the researcher proceeded to calculate the PMA for the rest of the patients. If there were any uncertainties regarding the psoas muscle calculation, the primary researcher verified the measurement with the radiologist.
The primary outcome was survival (calculated as days from operation). Follow up date selected was 31/10/2020. Study end date, the date of final data collection, was 23/02/2021. Determining the survival was based on hospital death records. This was also checked by means of seeing whether the patient had attended subsequent appointments or had subsequent admissions in patients without hospital record of death. This was essential as in Western Australian hospital records as patient death is not always recorded unless the patient has died during a subsequent public hospital admission, or the hospital is formally informed. Patients who could not be confirmed to be alive (or dead) as of the 31/10/2020 were censored in the survival analysis at the last known date that they were alive.
Secondary outcomes were 30-day readmissions related to EVAR, discharge destination (home, residential care, rehabilitation), post-operative complications, and length of stay (LOS) measured in days. This project was approved under the GEKO (Governance, Evidence, Knowledge, Outcomes database used to register information about quality improvement studies and clinical governance) system of WA Health, for projects deemed low- to negligible-risk to patients (GEKO#: 27118).
Statistical Analysis
Results were presented as a measure of central tendency, i.e., median with inter-quartile range, mean with standard deviation or as frequency and proportion. Differences between groups assessed with an independent t-test, non-parametric Mann-Whitney U test, and Fisher’s exact test, where appropriate. Univariate correlations were quantified by Spearman’s (rho) correlation coefficients (Rs). Linear and logistic regression analysis determined the crosssectional association between exposure (PMA) and outcomes. Cox proportional hazards regression analysis determined the uniand multi-variate association between the lowest tertile of psoas size for either sex with mortality. Multivariate models included further adjustment for age, body mass metrics, sex, and other comorbidities associated with mortality.
Statistical significance was set at α=0.05 or 5% level. The statistical analysis was performed on Statistical Package for the Social Sciences (SPSS) Version 22.0 (IBM Corp.; Armonk, NY, USA).
Results
Of the 122 patients identified as having undergone an EVAR procedure, complete data was available for 97 (79.5%). Five patients were excluded as they did not meet inclusion criteria (3 due to age, 2 due to missing information about the type of operation performed). Eighteen patients were excluded as imaging was not available to ascertain PMA data. Although these patients had similar demographics and clinical characteristics to those who were included, the exception was a lower complication rate in the excluded patients, although this failed to reach statistical significance (44% vs. 22%, p=0.06).
The study sample had a median age of 78 years, male preponderance (78.4%), and 79% were ex- or current smokers. Participants had a high prevalence of cardiovascular disease (85%, n=82/97), including coronary disease, cerebrovascular disease, hypertension, and peripheral vascular disease. Complications occurred in 44% of patients - the majority of which were categorized as ‘other’ (that is, not a fall, delirium, venous thromboembolism, bleeding, or infection). Most common was type 1 and 2 endoleak (n=8, with one requiring surgical intervention) and ischaemic complications (n=6 -testicular infarct, 3 non- ST elevation myocardial infarct, ischaemic lower limb, splenic infarct). Twenty-six (27%) patients had died by end of follow up. Twenty-six patients were unable to be confirmed to be alive at the time of follow up (31/10/2020). Median follow-up was 3.7 years with IQR 2.51 to 5.3 years (Range: 0.01 - 8.84 years) (Table 1).
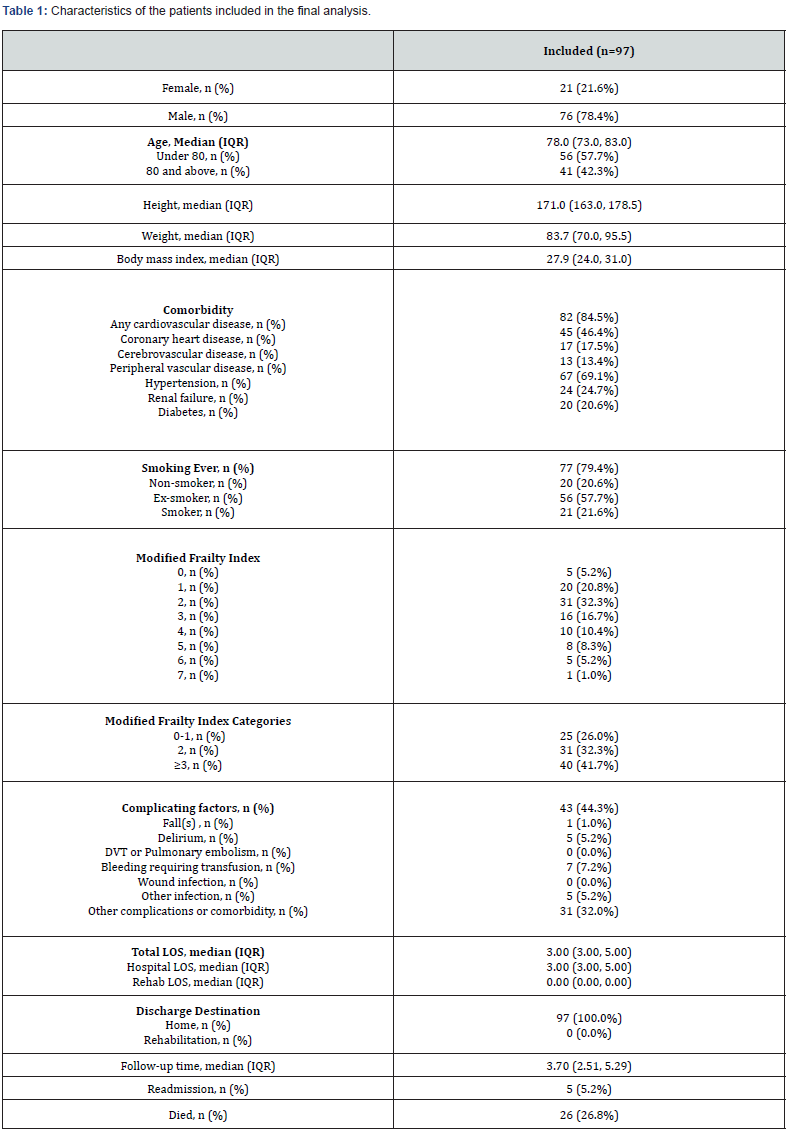
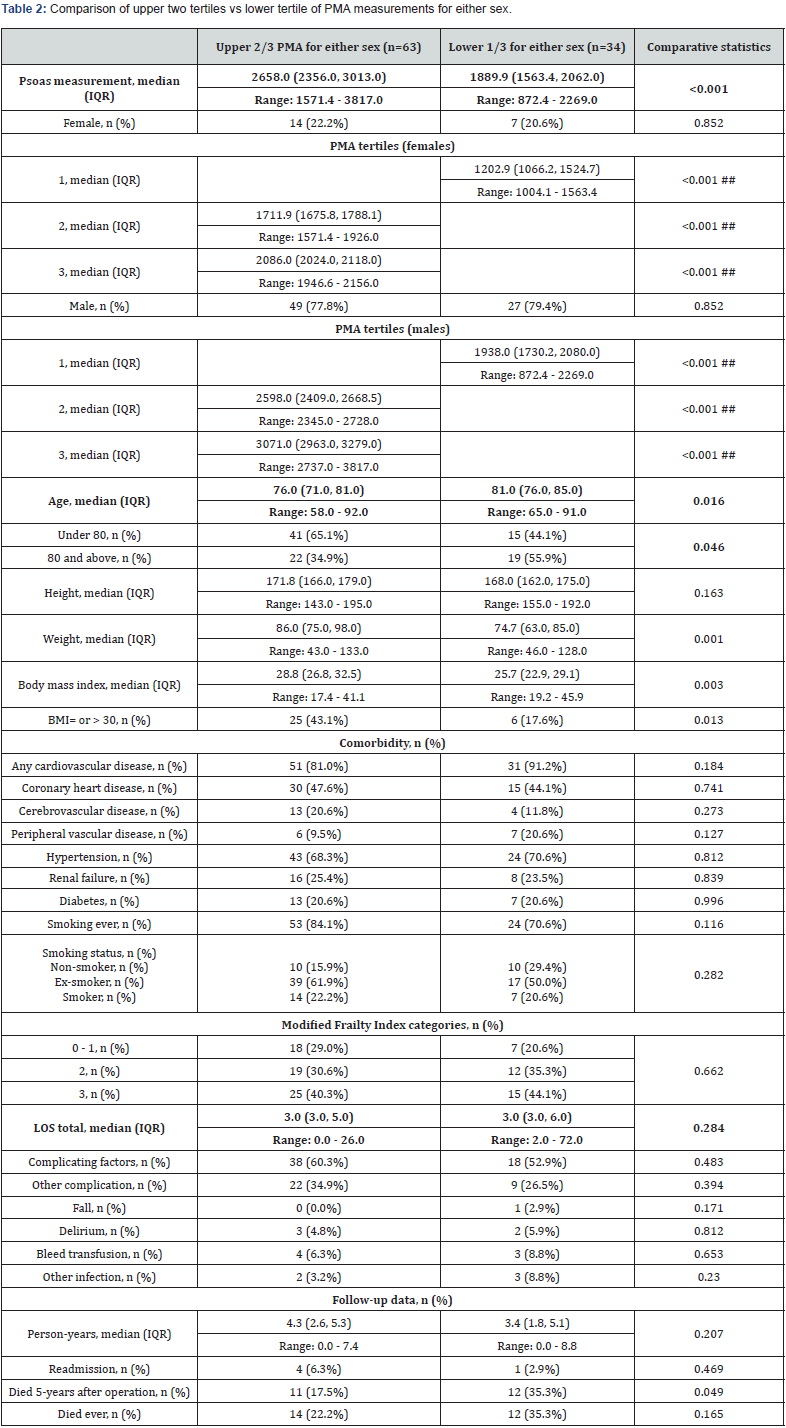
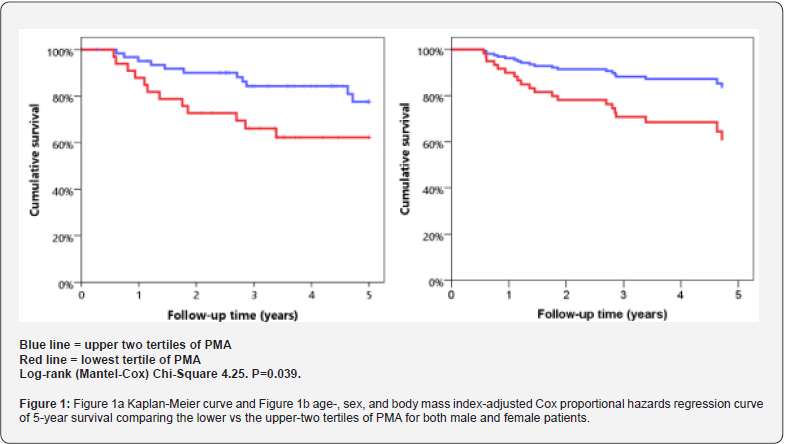
PMA exhibited pronounced age- and gender-specific differences. Female patients and those over >80 years of age had lower PMA. However, median PMA was similar for females patients regardless of age group. PMA was also shown to vary significantly by body mass index, with a higher BMI significantly associated with a higher than median PMA for sex and age. PMA was divided into tertiles, with comparison made between the lower tertile and upper 2 tertiles. Those in the lower tertile (PMA less than 1938 mm2 in males and less than 1202 mm2 in females) tended to be older, with a lower weight and BMI. Degree of frailty as per modified frailty index and comorbidities including cardiovascular disease, hypertension, renal failure, diabetes, and smoking status were similar in both groups. There was no significant difference in complications, readmissions, or LOS between the two groups, and no significant difference in length of follow-up. There was however a significant difference in crude 5-year mortality following operation, with 35% (12) of those in the lowest tertile having died after 5 years compared with 17.5% (11) of those in the upper two tertiles (p=0.049), (Table 2). The lowest tertile of PMA for either sex was associated with 5-year mortality after elective EVAR surgery for asymptomatic infrarenal AAA, as demonstrated by Kaplan Meier survival curve. This was maintained after adjustment for age, sex and BMI as per (Figure 1).
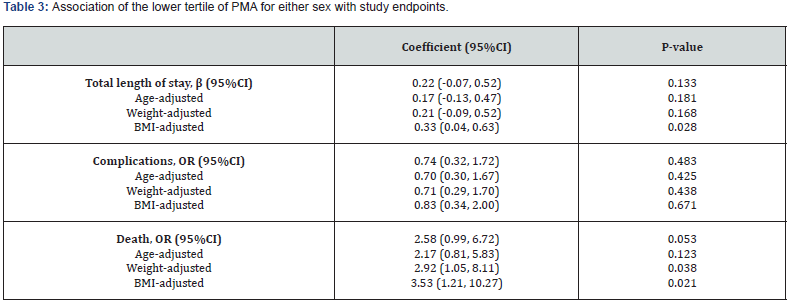
There was a significant association also noted between the lower tertile of PMA for either sex, when adjusted for BMI, for increased risk of death (OR 3.53, 95%CI 1.21 -10.27, p=0.021) and increased LOS (β 0.33, 95%CI 0.04-0.63, p=0.028). However, there was no association between lower tertile PMA for complications in either gender (Table 3).
Sensitivity Analysis
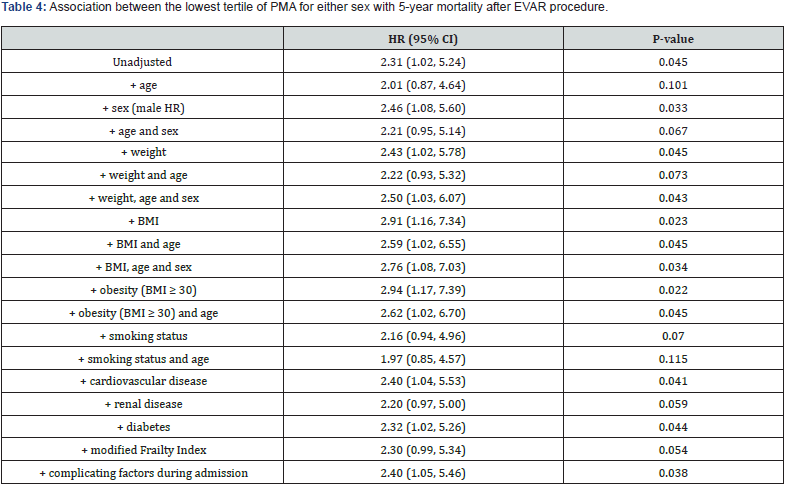
The lowest tertile of PMA for either sex after EVAR was associated with increased risk of 5-year mortality in the unadjusted model (HR 2.31, 95%CI 1.02, 5.24; P=0.045), which remained after adjustment for BMI (HR 2.91, 95%CI 1.16, 7.34; P=0.023) and after adjustment for age, sex, and BMI (HR 2.50, 95%CI 1.03, 6.07; P=0.043). The association between the lowest tertiles of PMA for either sex was attenuated by age-adjustment alone, but the association was strengthened after the inclusion of sex and body mass metrics. Lowest tertile PMA associated with an increased risk of 5-year mortality after adjustment for prevalent diabetes, cardiovascular disease and complications during admission. Modified Frailty Index minimally attenuated the association between lowest tertile of PMA and risk of 5-year mortality after EVAR procedure (Table 4).
Discussion
This study reported a two-to-three-fold increase in 5-year mortality in the lowest tertile of PMA, which was strengthened after adjusting for age, sex, and BMI, in elderly patients undergoing elective EVAR for asymptomatic infrarenal AAA, in a single tertiary centre in Western Australia. This association was also strengthened after adjusting for cardiovascular disease, diabetes, and obesity, but was marginally attenuated with adjustment for frailty. The association between mortality and PMA has been observed in several studies before, showing a similar association between low PMA and higher mortality in the diverse EVAR population. Most studies are single centre, retrospective and heterogenous in nature. Drudi et al (open repair of AAA and EVAR) showed decreased 5-year mortality by 14% for a 1cm2 increase in PMA [14]. Lindstrom et al. (elective and emergency EVAR for infrarenal and more complex AAA) showed a lean PMA (calculated by Hounsfield units multiplied by total PMA) was independently associated with 26% reduced mortality per standard deviation increase of lean PMA, and more strongly associated with mortality than total PMA at L3 (22%) [18] respectively. Newton et al and Oliveira et al, (elective EVAR), also showed an association between the lowest tertile of PMA with increased mortality, with OR of 3.9 [15] and HR of 4.1 respectively [19]. Similarly, Huber et al showed the lowest quartile of PMA was associated with reduced median survival time (65 months vs 91 months) (25). Karkkainen et al (fenestrated-branched EVAR) found reduced lean PMA had inverse correlations with major adverse events and mortality, and further showed in a prospective cohort similar correlation with mid-term survival and quality of life [16,17].
In opposition, Chowdury et al, (vascular patients admitted to vascular unit having had CT abdomen) did not show a link between PMA and mortality and readmission [11]. This opposing result may be due to the heterogeneity of the patient population examined - roughly 52% did not undergo EVAR, lower duration of follow up (12-24 months), and BMI not being accounted for as potential confounder. Indrakusuma et al (asymptomatic infrarenal AAA) did not find an association between PMA and survival in either EVAR/open repair or conservative management groups [12]. However, the surgical cohort was generally younger than other studies and again BMI was not accounted for as a potential confounding factor.
The effect size of our study was smaller than in most other studies. This may be explained by our calculation of PMA by dimension, rather than lean PMA. Lindstrom et al. indicated that a stronger association is found with lean PMA rather than total PMA [18]. Frailty was not assessed in previous studies and differences in outcomes may be explained by differences in frailty. Furthermore, our study was specific to elective infrarenal AAA, whereas other studies had a significantly more heterogenous population, ranging from vascular surgery patients admitted who had CT abdomen, to patients undergoing EVAR for aortic aneurysmal disease in general.
The association between lowest tertile of PMA in either sex with 5-year mortality was attenuated by age-adjustment alone. This may be due to a narrowing of the differences in PMA with increasing age for either sex, or a lack of sufficient study power. The actual number of deaths in each arm was small and the older cohort and expected life expectancy make interpretation difficult. Additionally, “old age mortality advantage”, a phenomenon describing relative improved mortality in those 70-79 years, as compared to younger ages and other countries, may also explain the attenuation [23]. This result is also reflected in other studies [15,16]. Similarly, smoking status (seen in Karkkainen et al.) and the presence of underlying renal disease appeared to attenuate the association of PMA and mortality, again suggesting that PMA may be less useful in predicting mortality outcome in smokers or those with underlying renal disease [16].
The novelty of our study was the simultaneous measurement of frailty which minimally attenuated the association between 5-year mortality and the lowest tertile of PMA suggesting that despite adjusting for frailty, the association between PMA and mortality remains. The impact of frailty, as mentioned previously, has not been formally assessed previously in any other study of PMA and outcomes in vascular patients. This reinforces the message that although there is intuitively a link between PMA / sarcopenia and frailty, the concepts are not interchangeable. It may imply that sarcopenia as assessed by PMA may be a better marker of mortality risk than measures of frailty. Our study did not show statistically significant associations between PMA and postoperative complications, discharge destination and readmissions within 30 days. This may relate to many of the complications being technical or procedure related rather than due to lack of physiological reserve. The only studies looking at PMA and have published complication rates show overall surgical complication rates of 21.9% [18] and ‘post-operative complication’ of 27% [15]. Although this is much lower than our study, it is difficult to compare these as the former study references both open and endovascular aneurysm repair and the latter study does not describe what they mean by ‘post-operative complications’. The lack of association with complications would more likely be reinforced by the difference noted in complication rate in those patients who were excluded from the study due to lack of PMA data
There was a statistically significant association found between PMA and LOS after adjusting for BMI, however it is unlikely to be clinically significant at 8 hours or approximately one third of a day. Chowdury et al. did not show correlation with LOS or discharge destination, similar to this study [11]. However, this does contrast with Karkkainen et al [16]. The increased complexity of the operation (fenestrated-branched EVAR for thoracic and pararenal aneurysms) may account for this, as well as the larger sample size (n=504).
There is no clear consensus as to which value of PMA leads to increased mortality and poorer outcomes (8). In this study, the lowest tertile of PMA for men was less than 19.4 cm2 and for women, less than 12cm2. Other studies that have examined total psoas muscle area have a cut off for the lowest tertile of PMA at 13cm2 and 24cm2, both measured at L4, which is the level we chose [14,15]. We also showed that PMA to be associated with age, sex and BMI as did all previous studies mentioned. The limitations to our study include: the retrospective nature of the study at a single metropolitan centre; the small sample size which contributes to lower statistical power; and missing data as a potential source of bias. Furthermore, European consensus guidelines on sarcopenia have been recently updated to place more emphasis on muscle strength (potentially measured by grip strength or chair stand test), with poor muscle performance (potentially measured by low gait speed) defining severe sarcopenia [8]. These new measures should be included in future studies [24-30].
Conclusions
Lower PMA is associated with 2.76 times increased mortality in patients undergoing EVAR for asymptomatic infrarenal AAA. However, it did not predict shorter term outcomes. Despite adjusting for frailty, this association with PMA and mortality remains, which has not previously been assessed in other studies. Low PMA would be considered as less than 19.4cm2 in men and less than 12cm2 in women in this study, however further studies are required to determine what PMA would be best representative of sarcopenia and significantly worse mortality. As patients undergoing EVAR routinely have pre-operative CT scanning, PMA is an easy way to assess sarcopenia and to determine risk for operative management of these patients adding additional valuable information to risk stratification and indication for treatment in this group.
References
- Moll FL, Powell JT, Fraedrich G, Verzini F, Haulon S, et al. (2011) Management of abdominal aortic aneurysms clinical practice guidelines of the European society for vascular surgery. European Journal of Vascular & Endovascular Surgery 41(Suppl 1): S1-S58.
- Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, et al. (2001) Frailty in older adults: evidence for a phenotype. Journals of Gerontology Series A-Biological Sciences & Medical Sciences 56(3): M146-M156.
- Buta BJ, Walston JD, Godino JG, Park M, Kalyani RR, et al. (2016) Frailty assessment instruments: Systematic characterization of the uses and contexts of highly cited instruments. Ageing Research Reviews 26: 53-61.
- Lin HS, Watts JN, Peel NM, Hubbard RE (2016) Frailty and post-operative outcomes in older surgical patients: a systematic review. BMC Geriatrics 16(1): 157.
- Ehlert BA, Najafian A, Orion KC, Malas MB, Black JH, et al. (2016) Validation of a modified Frailty Index to predict mortality in vascular surgery patients. Journal of Vascular Surgery 63(6): 1595-1601.e2.
- Kraiss LW, Al-Dulaimi R, Presson AP, Arya S, Lee GK, et al. (2016) A Vascular Quality Initiative-Based Frailty Instrument Predicts 9-Month Postoperative Mortality. Journal of Vascular Surgery 64(2): 551-552.
- McRae PJ, Walker PJ, Peel NM, Hobson D, Parsonson F, et al. (2016) Frailty and Geriatric Syndromes in Vascular Surgical Ward Patients. Annals of Vascular Surgery 35: 9-18.
- Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyere O, et al. (2019) Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing 48(1): 16-31.
- Janssen I (2006) Influence of sarcopenia on the development of physical disability: the Cardiovascular Health Study. Journal of the American Geriatrics Society 54(1): 56-62.
- Morrell GR, Ikizler TA, Chen X, Heilbrun ME, Wei G, et al. (2016) Psoas Muscle Cross-sectional Area as a Measure of Whole-body Lean Muscle Mass in Maintenance Hemodialysis Patients. Journal of Renal Nutrition 26(4): 258-264.
- Chowdhury MM, Ambler GK, Al Zuhir N, Walker A, Atkins ER, et al. (2018) Morphometric Assessment as a Predictor of Outcome in Older Vascular Surgery Patients. Annals of Vascular Surgery 47: 90-97.
- Indrakusuma R, Zijlmans JL, Jalalzadeh H, Planken RN, Balm R, et al. (2018) Psoas Muscle Area as a Prognostic Factor for Survival in Patients with an Asymptomatic Infrarenal Abdominal Aortic Aneurysm: A Retrospective Cohort Study. European Journal of Vascular & Endovascular Surgery 55(1): 83-91.
- Lee JS, He K, Harbaugh CM, Schaubel DE, Sonnenday CJ, et al. (2011) Frailty, core muscle size, and mortality in patients undergoing open abdominal aortic aneurysm repair. Journal of Vascular Surgery 53(4): 912-917.
- Drudi LM, Phung K, Ades M, Zuckerman J, Mullie L, et al. (2016) Psoas Muscle Area Predicts All-Cause Mortality After Endovascular and Open Aortic Aneurysm Repair. European Journal of Vascular & Endovascular Surgery 52(6): 764-769.
- Newton DH, Kim C, Lee N, Wolfe L, Pfeifer J, et al. (2018) Sarcopenia predicts poor long-term survival in patients undergoing endovascular aortic aneurysm repair. Journal of Vascular Surgery 67(2): 453-459.
- Karkkainen JM, Oderich GS, Tenorio ER, Pather K, Oksala N, et al. (2021) Psoas muscle area and attenuation are highly predictive of complications and mortality after complex endovascular aortic repair. J Vasc Surg 67(2): 453-459.
- Karkkainen JM, Tenorio ER, Oksala N, Macedo TA, Sen I, et al. (2020) Pre-operative Psoas Muscle Size Combined with Radiodensity Predicts Mid-Term Survival and Quality of Life After Fenestrated-Branched Endovascular Aortic Repair. Eur J Vasc Endovasc Surg 59(1): 31-39.
- Lindstrom I, Khan N, Vanttinen T, Peltokangas M, Sillanpaa N, et al. (2019) Psoas Muscle Area and Quality Are Independent Predictors of Survival in Patients Treated for Abdominal Aortic Aneurysms. Ann Vasc Surg 56: 183-193 e3.
- Oliveira VC, Oliveira P, Moreira M, Correia M, Lima P, et al. (2021) Impact of Total Psoas Area and Lean Muscular Area on Mortality after Endovascular Aortic Aneurysm Repair. Ann Vasc Surg 72: 479-487.
- Huber TC, Keefe N, Patrie J, Tracci MC, Sheeran D, et al. (2019) Predictors of All-Cause Mortality after Endovascular Aneurysm Repair: Assessing the Role of Psoas Muscle Cross-Sectional Area. J Vasc Interv Radiol 30(12): 1972-1979.
- Lindstrom I, Protto S, Khan N, Sillanpaa N, Hernesniemi J, et al. (2020) Developing sarcopenia predicts long-term mortality after elective endovascular aortic aneurysm repair. J Vasc Surg 71(4): 1169-1178 e5.
- Farhat JS, Velanovich V, Falvo AJ, H Mathilda Horst, Andrew Swartz, et al. (2012) Are the frail destined to fail? Frailty index as predictor of surgical morbidity and mortality in the elderly. J Trauma Acute Care Surg 72(6): 1526-1530.
- Palloni A, Yonker JA (2016) Is the us old-age mortality advantage vanishing? Population & Development Review 42(3): 465-489.
- Masuda T, Shirabe K, Ikegami T, Harimoto N, Yoshizumi T, et al. (2014) Sarcopenia is a prognostic factor in living donor liver transplantation. Liver Transplantation 20(4): 401-407.
- Onesti JK, Wright GP, Kenning SE, Tierney MT, Davis AT, et al. (2016) Sarcopenia and survival in patients undergoing pancreatic resection. Pancreatology 16(2): 284-289.
- Saji M, Lim DS, Ragosta M, LaPar DJ, Downs E, et al. (2016) Usefulness of Psoas Muscle Area to Predict Mortality in Patients Undergoing Transcatheter Aortic Valve Replacement. American Journal of Cardiology 118(2): 251-257.
- Garg L, Agrawal S, Pew T, Hanzel GS, Abbas AE, et al. (2017) Psoas Muscle Area as a Predictor of Outcomes in Transcatheter Aortic Valve Implantation. American Journal of Cardiology 119(3): 457-460.
- Kleczynski P, Tokarek T, Dziewierz A, Sorysz D, Bagienski M, et al. (2018) Usefulness of Psoas Muscle Area and Volume and Frailty Scoring to Predict Outcomes After Transcatheter Aortic Valve Implantation. American Journal of Cardiology. 122(1): 135-140.
- Heberton GA, Nassif M, Bierhals A, Novak E, LaRue SJ, et al. (2016) Usefulness of Psoas Muscle Area Determined by Computed Tomography to Predict Mortality or Prolonged Length of Hospital Stay in Patients Undergoing Left Ventricular Assist Device Implantation. American Journal of Cardiology 118(9): 1363-1367.
- Zuckerman J, Ades M, Mullie L, Trnkus A, Morin JF, et al. (2017) Psoas Muscle Area and Length of Stay in Older Adults Undergoing Cardiac Operations. Annals of Thoracic Surgery 103(5): 1498-1504.






























