Event-Related Potential Elicited in Response to A Colour-Word Stroop Task in Young, Middle-Aged and Old Participants
Montserrat Zurrón*, Marta Ramos-Goicoa and Fernando Díaz
Department of Clinical Psychology and Psychobiology, IDIS, Universidade de Santiago de Compostela, Galicia, Spain
Submission: July 12, 2023; Published: July 24, 2023
*Corresponding author: Montserrat Zurrón, Department of Clinical Psychology and Psychobiology, Universidade de Santiago de Compostela, Galicia, Spain
How to cite this article: Zurron M, Ramos-Goicoa M, Diaz F. Event-Related Potential Elicited in Response to A Colour-Word Stroop Task in Young, Middle-Aged and Old Participants. OAJ Gerontol & Geriatric Med. 2023; 7(3): 555713. DOI: 10.19080/OAJGGM.2023.07.555713
Abstract
Electroencephalography (EEG) was used to record the brain electrical activity in healthy participants (Young, middle-aged, and old) who were carrying out a Stroop task, with the aim of studying the neurofunctional changes associated with processing stimuli and responses in healthy aging. The EEG data were subsequently processed to obtain the Event-Related Potentials (ERPs). Reaction Times (RT) and number of hits were also obtained. The RTs were longer in the old and middle-aged participants than in the young participants, and the middle-aged participants made more hits in response to incongruent stimuli than the old participants. The N2, P3b and LRP-S latencies and the time between the onset latency of LRP-R and the TR increased with age. Thus, the older and middle-aged participants processed the information more slowly that the young participants, and the slowing affected the evaluation and classification of the stimuli and the initiation and duration of the motor activity associated with the response. The frontal P3b amplitude was larger in middle-aged and old than in young participants, increasing towards posterior regions only in the young group. The central LRP-S and LRP-R amplitudes were larger in the old and middle-aged than in the young participants, which may indicate greater frontal recruitment for processing the stimuli and the responses from middle-age onwards.
Keywords: Event-Related Potentials; Stroop Task; Aging
Abbreviations: EEG: Electroencephalography; ERPs: Event-Related Potentials; LRP-S: Lateralized Readiness Potential related to Stimulus component; LRP-R: Lateralized Readiness Potential related to Response component; RT: Reaction Times; ICA: Independent Component Analysisc
Introduction
The increase in the proportion of elderly people in the population and the associated rise in the incidence of neurodegenerative diseases have led to increased interest in research into the cognitive changes that accompany healthy aging. The aim of such research is to determine whether these changes represent the initial deterioration characteristic of neurodegenerative disorders and thus to enable the design of more effective intervention strategies. Age-related cognitive changes can be objectively measured by evaluating how participants of different ages perform various executive control tasks, such as the Stroop task [1-4]. In the Stroop task, participants are shown colour patches (or colored strings of the letter X) and the names of colors in a colour that may be the same as or different from the word meaning (thus generating congruent and incongruent stimuli) and are asked to name (or indicate) the colour of the stimulus and to ignore its meaning [5].
The task generates the Stroop effect or Stroop interference, whereby the time taken to respond to the incongruent word is longer than the time taken to respond to the congruent or colored stimuli (patches or Xs), because of the interference in the automatic reading process and the conflict generated by the meaning of the word during the response. Performance of the task deteriorates between youth and old age, although the rate of decline varies depending on whether the speed of processing or precision of the response is measured. Thus, the time taken by participants to complete the task increases gradually from youth onwards, but the precision of the response remains stable for longer before decreasing in old age. In parallel to the changes in cognitive performance with aging, changes have been found to occur in the brain throughout life [6-9].
Recording Event Related Potentials (ERPs) is one way of studying the age-related changes in neural functioning associated with cognitive processes. ERPs represent neuroelectrical activity with a temporal resolution of milliseconds and can be used to monitor the temporal course of information processing in the brain, in relation to processing of stimuli and of responses. ERPs consist of the oscillations in the voltage that appear in relation to the presentation of stimuli or the emission of responses within certain temporal windows. These oscillations (waves) are denominated components and have been associated with cognitive processes. The values of the latency of the ERP components provide precise chronometric information about mental processes, and the voltage (amplitude) of the components provides information about the neural resources used and about their scalp distribution.
Previous studies investigating age-related effects on ERPs in the Stroop task have compared young and old participants [10- 13], young and middle-aged participants [14,15], and middleaged and old participants [16]. However, none of these studies compared all three groups of participants, which would provide more precise information about the age-related rate of change, i.e., whether the changes appear in middle age and are maintained or progress in older age, or whether they only appear in old age. Furthermore, although some of the findings of the studies are consistent, there are also some discrepancies, which require further investigation.
In relation to stimulus processing, the studies have generally revealed longer latencies of ERP components posterior to 200ms, including the N2 and P3b components. The latencies of these components have been associated with stimulus evaluation and categorization in working memory [17-19]. However, longer latencies of earlier components (P1, P150/N170 associated with perception processes) have not been observed in either old (over 65 years) or middle-aged participants, relative to young participants [10,11,13-15]. This finding is consistent with the findings of simple stimuli-discrimination tasks such as oddballtype tasks [20-22]. Overall, the findings support the hypothesis of age-related slowed information processing, although not the generalized slowing suggested by Salthouse [23], as not all stages of stimulus processing are affected. However, a study comparing old and middle-aged participants revealed that the latency of the components became longer from 150ms onwards, i.e., the slowing even appeared in perceptive processes in old relative to middle-aged participants [16]. Furthermore, Killikelly & Szücs [14] did not observe any differences between young and middleaged participants in the latencies of the components associated with response processing (LRP-S and LRP-R) in Stroop tasks. Falkenstein etal. [24] found that, in choice reaction time tasks, the longer RT in elderly than in young adults was associated with delays in the latency of motor ERP components. Therefore, the delays in the components associated with response processing the Stroop task may only appear in the older participants.
In relation to the amplitude of the components and their scalp distribution, the amplitude of the frontal P150 component has been found to be larger in old than in young participants [13]. P150 forms part of the P150 frontal-N170 occipital complex [25], which is involved in the perceptive processing of visual stimuli such as faces and words [25-27]. The amplitude of the fronto P3a component, which is associated with attention and orientation towards new stimuli [28-30], has been found to be larger in middle-aged than in young participants [14]. The amplitude of P3b has been observed to be smaller in parietal regions in old and middle-aged than in young participants, with a frontal pericraneal distribution in the first two groups and a parietal distribution in the young group [13,15]. These data on the P3b component are consistent with observations in older participants performing oddball-type tasks [20,31-33], and have been interpreted in terms of greater recruitment of frontal resources by old than by young adults [32]. By contrast, Killikelly & Szücs [14] did not observe any differences in the amplitude of the P3b component in centroparietal derivations between young and middle-aged participants in response to stimuli in the Stroop task. Finally, the central LRP-R has been observed to be larger in old than in middle-aged participants [16]; although in a comparison of young and middle-aged participants, Killikelly & Szücs [14] did not observe any differences in the amplitude of LRP-S and LRP-R between groups.
In the present study, we aimed to contribute to recording healthy patterns of neurocognitive functioning at different ages. For this purpose, we recorded the behavioural performance and obtained ERP data associated with the stimuli and responses to a Stroop task in three groups of participants: young, middle-aged, and old. The study aims were as follows:
i. To determine which components and therefore which processes measured in the Stroop task slow down with age (perception, evaluation, and categorization of stimuli, and/or response processing)
ii. To establish the age at which neurocognitive slowing first appears, i.e., in middle age or in old age (after 65 years)
iii. To determine whether age affects the amplitude of the ERP components and their scalp distribution, and, if so, d) at what age, i.e., middle, or old age.
Methods
Participants
The study participants (n=41) were divided into three groups: young (n= 15, aged between 19 and 21 years, mean of 20 years; 4 men), middle-aged (n= 16, aged between 52 and 64 years, mean of 57 years; 8 men) and old (n=10, aged between 66 and 81 years, mean of 72 years; 3 men). None of the participants had a history of clinical stroke, traumatic brain injury, motor-sensory defects or alcohol or drug abuse/dependence, and they were not diagnosed with any significant medical or psychiatric illnesses. All participants had normal audition and normal or correctedto- normal vision. Handedness was assessed using the Edinburgh inventory [34]. In the group of young participants, [14] were right-handed and one was ambidextrous; all of the middle-aged and old participants were right-handed.
All participants gave their written informed consent prior to participation in the study and voluntarily attended our laboratory for psychophysiological evaluation by the ERP technique. They also provided data on their educational, socioeconomic, medical, and personal backgrounds. The young participants were students attending the University of Santiago de Compostela (Galicia, Spain) and had received on average 13.8 years of education. The middle-aged participants received on average 12.4 years of education and the old participants, 10.3 years.
The three groups of participants did not differ significantly in years of education. The middle-aged and old participants also underwent psychological and neuropsychological assessment to ensure that they were not suffering from any neurocognitive disorders. A detailed description of the assessment is provided by Ramos-Goicoa, et al. [16]. The research project was approved by the Galician Clinical Research Ethics Committee (CEIC). The study was performed in accordance with the ethical standards established in the 1964 Declaration of Helsinki [35].
Task and Stimuli
Three different types of stimuli were presented: colored X-strings (“XXXX” displayed in red or blue), congruent colourword stimuli (e.g., the Spanish word rojo [red] displayed in red) and incongruent colour-word stimuli (e.g., the Spanish word azul [blue] displayed in red). Twenty X-string stimuli were displayed before the start of the experimental session, to familiarize the participants with the task, and the EEG activity of the participants was then recorded during presentation of the following stimuli: 70 colored X-string stimuli (35 displayed in red and 35 in blue in a pseudo-random order).
These non-word stimuli were presented first and separately from the other stimuli to ensure that they were only processed as chromatic stimuli and were not influenced by semantic expectations. 140 colour-word stimuli. 70 congruent colour-word stimuli (35 displayed in red and 35 in blue) and 70 incongruent colour-word stimuli were presented: the word Azul (blue) was displayed in red 18 times; the word rojo (red) was displayed in blue 18 times; the word Verde (green) was displayed in blue 17 times and in red 17 times. To prevent participants from using a read-only strategy, the stimuli (congruent and incongruent) were presented in a pseudo-random order.
Thus, no more than three words of the same type (congruent/ incongruent) were presented consecutively, and no more than two words were displayed in the same colour. The word stimuli were displayed as lowercase letters (font style Trebouchet MS, size 180) on a black background. The colored X-strings, of dimensions 12.5 x 3.3cm, were presented at a visual angle of 7.1º x 1.8º. The words azul (blue) and rojo (red) of dimensions 10.5 x 3.3cm, were displayed at a visual angle of 6º x 1.8º. The word verde (green), of dimensions 16 x 3.3cm, was displayed at a visual angle of 9.1º x 1.8º. The duration of presentation of each stimulus was 250ms, and the interstimulus interval was 2200, 2350 or 2500 milliseconds (varied at random).
The task involved pressing a red or blue button depending on the colour of the stimulus that appeared on the screen. The participants pressed each button with a different hand, with the wrists resting on a pad. Participants were instructed to respond to the colour of the stimulus as quickly and accurately as possible, without trying to correct any errors that they thought they had made, and to refrain from moving or talking during task execution. The position of the red and blue buttons (right or left) was counterbalanced across participants. Reaction Time (RT) and number of hits were obtained as measures of behavioural output.
Electroencephalographic (EEG) Recording
Participants were required to abstain from consuming drugs/ alcohol/caffeine and nicotine from about one hour prior to testing, and none of them reported fatigue caused by lack of sleep. None of the participants were familiar with the protocols used in the study. The participants were seated on a comfortable chair in a Faraday chamber with attenuated levels of light and noise, and they were asked to refrain from moving during the recording. Visual stimuli were presented on a 19″ flat screen monitor with a vertical refresh rate of 120 Hz. The distance between the monitor and the participant was one metre. The electroencephalogram was recorded via 49 ring electrodes placed in an elastic cap (Easycap, GmbH), according to the international 10-10 system. All electrodes were referenced to an electrode attached to the tip of the nose, and an electrode positioned at Fpz served as ground.
The horizontal electro-oculogram (EOG) was recorded at two electrodes placed at the outer canthi of both eyes, whereas the vertical EOG was recorded at two electrodes placed supra and infraorbitally on the right eye. The EEG was continuously digitized at a rate of 500 Hz (bandpass 0.01-100 Hz), and electrode impedance was maintained below 10 kΩ. Once the EEG signal was stored, ocular artefacts were corrected by Independent Component Analysis (ICA). The EEG was then segmented by extraction of -200 to 1400 ms epochs synchronized with each stimulus to yield the P150, N170, N2 and P3b components associated with stimulus processing. For each response hand, the EEG was segmented into -200 to 1400 ms epochs in synchrony with the stimulus and into -1000 to 300 ms epochs in synchrony with the response, to obtain the components associated with response processing, i.e., the Lateralized Readiness Potential related to Stimulus (LRP-S) and to Response (LRP-R) components. These epochs were then classified a posteriori as colored X-strings, congruent colour words and incongruent colour words. The signal was passed through a digital 0.1 to 30 Hz bandpass filter, and epochs were corrected to the mean voltage of the 200-ms pre-stimulus recording period (for direct waveforms and LRP-S) or from -1000 to -800 ms pre response recording period (LRP-R). EEG segments exceeding ±100 μV and those corresponding to incorrect responses were automatically excluded from the averages. The number of epochs included in the ERP averages was between 50 and 60, and there were no significant differences between the numbers of epochs for either type of stimulus or group. In order to obtain the LRP waveforms (LRP-S and LRP-R), the difference in contralateralipsilateral activation was calculated for C3 and C4 electrode pairs in each hemisphere, and the differences were then averaged. The method can be summarized by the following formula: [(C4 - C3) left hand movements + (C3 - C4) right hand movements]/2].
Identification of Components
For stimulus processing, the N170/P150 complex [25] was measured at the Oz (N170), AFz (P150) and Fz (P150) electrodes, with a latency of between 100 and 200 ms at Oz and of between 100 and 190 ms at frontal electrodes. N2 was measured at AFz, Fz and Cz, with a latency of 200 and 350 ms. Finally, the P3b component was recorded at AFz, Fz, Cz and Pz with a latency of between 300 and 550 ms. The P150/N170 complex was always identified when the polarity between frontal and occipital electrodes was reversed; N2 appeared as a negative wave prior to P3b, and P3b was always identified at the three mid-line electrodes (Fz, Cz and Pz). The peak amplitude of the components was measured relative to baseline, and the peak latency of each component was also determined.
These components were identified manually. In relation to response processing, the amplitude and the onset latency of the LRP-S and LRP-R components were measured using the ERPLAB measurement tool developed by Lopez-Calderón and Luck [36]. The amplitudes were measured as the mean voltage between two fixed latencies: 200 to 600 ms (LRP-S) and -300 to -25 ms (LRP-R). The onset latency was obtained using the Fractional Area Latency option and scored as the point at which 15% of the area (200 to 600 ms for LRP-S and-300 to -25 for LRP-R) was to the left of the point.
Data Analysis
The behavioural responses (RT and number of hits) and electrophysiological data (latencies and amplitudes of the ERP components) were analyzed by ANOVAs including three levels of the repeated measures factor stimulus (colored X-strings, congruent colour words and incongruent colour words) and three levels of the between-group factor age (young, middle-aged and old). A third repeated measurement factor, electrode, was added for the amplitudes and latencies of the P150, N2 and P3b components. When the ANOVAs revealed significant differences, pairwise comparison of means (with Bonferroni correction) was carried out to identify the source of the differences. Greenhouse- Geisser corrections were applied to the degrees of freedom in all cases in which the condition of sphericity was not met. In these cases, the original degrees of freedom are presented together with the corrected p and ε values. Differences were considered significant at p≤ 05, and only significant effects are reported. All statistical analyses were carried out with SPSS (version 19.0).
Results
Task Performance
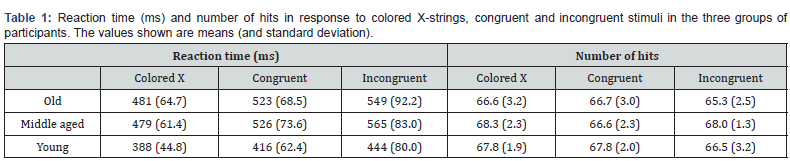
Age had a significant effect on the RTs (F (2,38) =12; p≤.001). The post hoc analysis revealed significantly longer RTs in the middle-aged and old participants than in the young participants (p≤.005 for both paired comparisons). Stimulus also had a significant effect on the RTs (F (2,76) =53; p≤.001 ε=.7); the post hoc analysis revealed significantly longer RTs in response too incongruent and to congruent stimuli than in response to the colored X-strings and significantly longer RTs in response to incongruent than to congruent stimuli (p≤.001 for three paired comparisons). The age x stimulus interaction had a significant effect on the number of hits (F (4,76) = 2.5; p≤.05). The paired comparisons revealed that the middle-aged participants made more hits than old participants in response to the incongruent stimuli (p≤.03); the middle-aged participants made significantly more hits in response to colored X-strings than in response to congruent stimuli (p≤.03). No other significant effects were observed in relation to these two dependent variables. The values of TR and hits are shown in Table 1.
Event Related Potentials
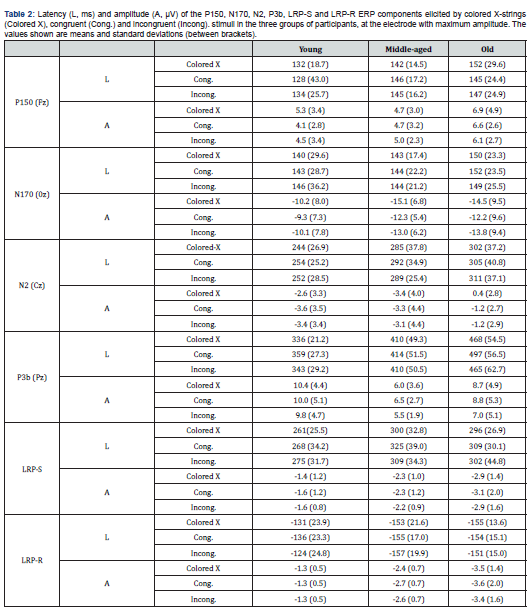
Grand average ERP waveforms for stimuli processing with P150, N170, N2, and P3b components are shown in Figure 1. Neither age nor stimulus significantly affected the P150 latency and amplitude. Stimulus had a significant effect on the N170 amplitude (F (2,50) =8.8; p≤.001), and the amplitude was significantly larger to colored X stimuli than to congruent stimuli (p≤.001). No other significant differences were observed. Age significantly influenced the N2 latency (F (2,21) =5.6; p≤.01). The post hoc analysis revealed that the latency was significantly longer in the old than in young participants (p≤.015). The stimulus factor also had a significant influence on the N2 latency (F (2,42) =7,5; p≤.002), and the N2was significantly longer to the incongruent and congruent stimuli than to the colored X stimuli (p≤.02 and p≤.007 respectively). Age (F (2,21) =4.1; p≤.03) and the Age x Electrode interaction (F (4,42) =2,7, p≤.05) significantly affected the N2 amplitude. The post-hoc analysis revealed that the N2 amplitude was significantly larger in the young than in the middle-aged participants (at AFz: p≤.02) and old participants (at AFz: p≤.003, and at Fz: p≤.02). Stimulus had a significant effect on the amplitude (F (2,42) =7.2; p≤.005), which was significantly larger to congruent than to colored X stimuli (p≤.007). Age had a significant influence on the latency of the P3b component (F (2,27) =21.5; p≤.001). The post hoc comparisons showed that at the latency was significantly longer in the old and middle-aged participants than in the young participants, and it was significantly longer in the old than in the middle-aged participants (p≤.01 for all paired comparisons). Stimulus had a significant effect (F (2,54) =7.2; p≤.005; ε=.7), and the latency was significantly longer too congruent than to colored X stimuli (p≤.002) and incongruent stimuli (p≤.001).
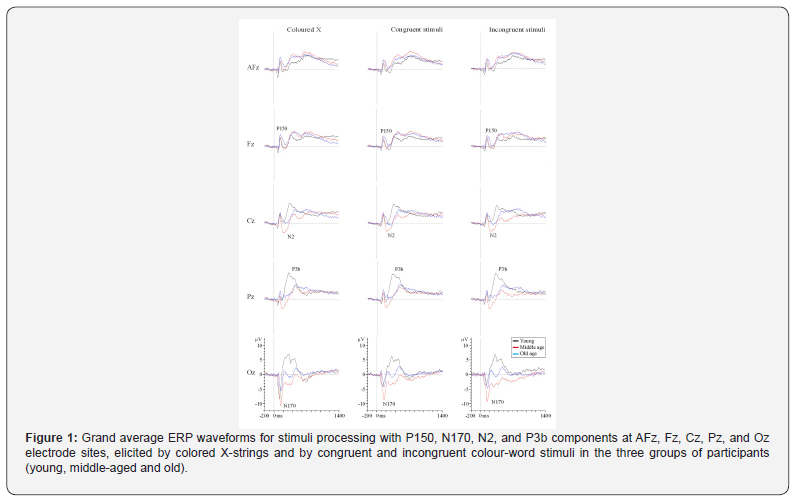
The electrode factor (F (3,81) =7.5; p≤.005; ε=.45) and the age x electrode interaction (F (6,81) =8.6; p≤.001) had significant effects on the amplitude of the P3b component. The post hoc analysis revealed that in the young participants, the amplitude was significantly larger at Pz, Cz and Fz than at AFz, significantly larger at Pz and Cz than at Fz, and significantly larger at Pz than at Cz (p≤.007 for all paired comparisons). In addition, at the AFz and Fz electrodes, the amplitude was significantly larger (p≤.02 for all paired comparisons) in the middle-aged (AFz: Mean=6.4 μV; Fz: Mean=6.3 μV) and old groups (AFz: Mean=6.6 μV; Fz: Mean=7.5 μV) than in the young group (AFz: Mean=1.7 μV; Fz: Mean=3.2 μV). The differences between groups in the scalp distribution of the P3bamplitude are shown in Figure 2.
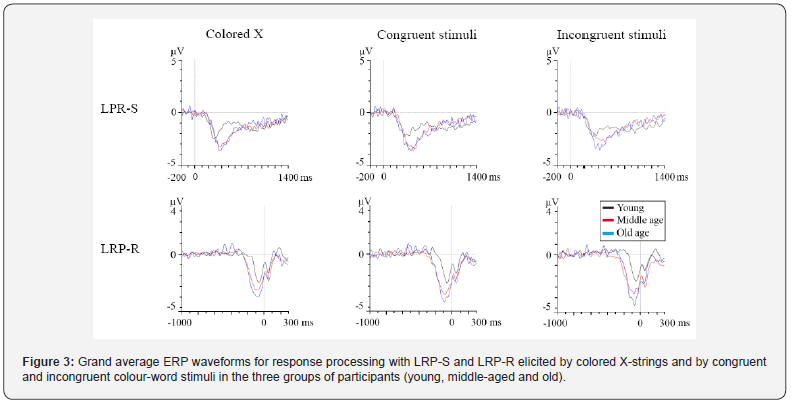
The stimulus factor also had a significant effect (F (2,54) =5.2; p≤.009), and the amplitude was significantly smaller to incongruent stimuli than to congruent(p≤.02) and colored X stimuli (p≤.03). No other interaction effects were observed. Grand average ERP waveforms for response processing with LRP-S and LRP-R components are shown in Figure 3. Age also had a significant impact on the onset latencies of the LRP-S (F (2,33) = 7.5; p≤.002) and LRP-R components (F (2,37) = 11.1; p≤.001). The LRP-S onset latency was significantly longer for the old participants (p≤.04) and the middle-aged participants (p≤.002) than for young participants. The time between the onset latency of LRP-R and the RT was significantly longer for the middleaged (p≤.001) and old participants (p ≤.003) than for the young participants. The stimulus factor was significant for LRP-S onset latency (F (2,66) = 4.4; p≤.02), the latency was significantly longer to congruent stimuli than to colored X strings (p≤.02). Finally, age also had a significant influence on the amplitudes of the LRP-S (F (2,32) = 3.8; p≤.03) and LRP-R (F (2,37) = 17.9; p≤.001) components. The post hoc analysis revealed that the amplitudes were significantly larger in the middle-aged (LRP-R: p≤.002) and old participants (LRP-S: p≤.03; LRP-R: p≤.001) than in the young participants. Latency and amplitude values of the P150, N170, N2, P3b, LRP-S and LRP-R ERP components are shown in Table 2.

Discussion
Task performance
The increase in RTs in participants older than 50 years old relative to young participants observed in this study is consistent with previous findings and with age-related slowed processing [1-4]. The well-known Stroop effect was observed (see [5] for a review): the RT was longer in response to incongruent stimuli than in response to congruent stimuli and colored-X-strings, due to the interference from the reading process and the conflict that incongruent meaning of the word generates in the task of responding to the colour. The RT was also longer in response to the congruent stimuli than in response to the colored X-strings, due to the interference from the reading process provoked by the random presentation of the congruent and incongruent stimuli in the same block, which prevented the participants from using a read-only strategy. In other words, no facilitation effect was observed in the processing of congruent stimuli (in which colour and meaning were consistent) relative to the colored stimuli. Furthermore, no interaction effect between the type of stimulus and age was observed in the RT. The Stroop effect or interference between the meaning of the colour word and the colour in which was written was therefore independent of age. By contrast, it has been found that interference in the Stroop task increases with age [1, 4, 5]. The lack of observation of any increase in the Stroop effect with age may be explained by the fact that the Stroop task used in this study, with only two response colour, was easier than Stroop tasks with 3-5 response colours, which are more commonly used. [37]. The three age groups did not differ in the number of hits in response to the colored X-strings and congruent stimuli, as also observed by Ratcliff et al. [38] in tasks such as numerosity discrimination, recognition memory and lexical decision tasks, which like our Stroop task, had only two types of response. According to Ratcliff and McKoon [39] the quality of the information on which decisions are based is not significantly worse in old than in young adults in this type of tasks. The results of the present study show that this concept can be applied to the processing of congruent stimuli and colored X-strings.
Regarding the incongruent stimuli, the middle-aged participants had more hits than old participants, which is consistent with the findings reported by Troyer et al. [2] and Uttl & Graf [3], as they found that in the incongruent condition of a Stroop task, the number of errors remained stable in young and middle-aged participants but was higher in old adults. This may indicate the successful use of strategies based on the accuracy of execution in the middle-aged group at the cost of slower processing, given the longer RT of middle-aged participants than of young participants. As also observed for the RTs, no facilitation effect was observed in relation to the number of hits, as we observed a lower number of hits in response to congruent stimuli than in response to the colored X-strings in the middle-aged group. In this respect, MacLeod [5] indicated that the facilitation effect is weak and highly dependent on the characteristics of stimulus presentation.
Event-Related Potentials
The findings regarding the latencies of the ER components are consistent with the behavioural results, as they demonstrate age-related slowing of the neural functioning associated with information processing that appears during middle age. In addition, examination of the ERPs shows that the processes that slow down with age are those related to the evaluation and categorization of the stimuli as well as with response processing, but not with perceptive processes. Therefore, the ERP recordings have enabled us to verify that the age-related slowing does not generally occur at all stages of information processing.
The frontal positive P150 waveform is considered, together with the N170 occipital waveform [25], which appears in response to words, as being specifically related to sub-lexical perceptual processes [27]. The findings of the present study indicate that there is no age-related delay in appearance of the P150/N170 complex and therefore the perceptive processes do not appear to undergo age-related slowing. This coincides with previous findings of studies with the colourword Stroop task, in comparisons of young and old participants [13], and middle-aged and young participants [14, 15]. They are also consistent with the findings of a Flanker task study [40] and with those obtained with oddball-type tasks [20, 21]. Considering the large number of consistent results, we believe that the longer P150 latency in old than in middle-aged adults observed by Ramos-Goicoa et al. [16] can be explained by the act that the study participants included healthy adults and adults with mild cognitive impairment.
The results of the effects of age on the latencies of the N2 and P3b components in the present study are consistent with those previously obtained with the Stroop task [13-16], with oddballtype tasks [21,31], and with Go- NoGo tasks [41], and they indicated that the evaluation and categorization of stimuli that are relevant to the task showed a gradual age-related slowing down.
Smulders & Miller [42] reported that the onset latency of LRP-S may indicate the start of response activation, and the onset latency of LRP-R can be considered an indicator of the duration of the response activation and peripheral motor processes. Thus, the present findings indicate that initiation of the neural activation of the response occurred later and the duration of the activation until emission of the response was longer in older and middleaged participants than in the young participants, i.e., the response processing was slower from middle age. These findings are consistent with previous findings in studies using other executive control tasks such as the Simon task [43], the Eriksen flanker task [40], and the -NoGo task [41], although Killikelly & Szücs [14] did not find any differences between young and middle-aged participants in the latency of these components.
In relation to the effect of the type of stimulus on the ERP latencies, we found that the latencies of N2, P3b and LRPS were shorter in response to the colored X-strings than in response to the congruent stimuli. These findings indicate that it is easier to evaluate and categorize the stimuli, as well as to activate the response to colored-X strings than the response to the congruent stimuli, because of the effect on the congruent stimuli of the interference from reading caused by presenting the stimuli together and randomly with the incongruent stimuli.
Paradoxically, we also found that the latency of P3b was shorter in response to incongruent stimuli than in response to congruent stimuli. Stroop task studies have consistently shown that the P3b latency does not differ between conditions in either young or old participants [12,13,15,44-51]. Future studies should therefore examine whether this discrepancy is due to the type of Stroop task (with only two response colours) used in the present study.
In regard to the amplitudes of the P150/N170, we did not observe any age-related effect due to the type of stimulus. The N170 amplitude was larger in response to colored X-string stimuli than in response to congruent stimuli, which is consistent with previous findings of a Stroop task study with young participants [52].
The N2 amplitude increases with the degree of cognitive control required for resolution of the task [17]; thus, the smaller amplitudes of the N2 component in the old and middle-aged participants than in the young participants may indicate a lower degree of cognitive control in the old and middle-aged than in the young participants. We also found that the N2 amplitude was larger in response to congruent stimuli than in response to the X-strings, which may indicate that the former generate greater cognitive control than the latter, which is consistent with the effect of interference from reading obtained in response to the congruent stimuli (longer RT in response to congruent stimuli than in response to X-strings).
Regarding the P3b amplitude, the scalp distribution depended on the age of the participants. Thus, the young participants showed the characteristic increase towards posterior locations [53]; by contrast, in the old and middle-aged participants the distribution of P3b was similar at anterior and posterior electrodes, as observed in previous studies with oddball-type tasks [32,54]. Mager et al. [15] and Zurrón et al. [13] observed a frontal distribution of P3b in older and middle-aged participants performing a Strrop task. These age-related differences in the distribution of the P3b amplitude led Van-Diteren et al. [32] to suggest greater recruitment of frontal resources. The larger P3b amplitude recorded at the frontopolar electrodes in older and middle-aged participants than in the young participants observed in the present study is consistent with this hypothesis. By contrast, we did not observe any age-related effect on the P3b parietal amplitude, as found by Killikelly & Szücs [14]. Zurrón et al. [13] found that the amplitude of the parietal P3b in a Stroop task was smaller in older than in young participants, coinciding with the findings of a study with oddball tasks carried out by Van-Diteren etal. [31,32], who observed that the P3b amplitude decreased at parietal electrodes between youth and old age. Thus, it appears that the greater frontal recruitment from middle age onwards may or may not be accompanied by a decrease in the parietal P3b amplitude. Regarding the effect of the type of stimulus on the P3b amplitude and therefore in agreement with the findings with samples of young and middle-aged participants [13- 15,48,51,52,55,56], we found that the P3b amplitude was smaller in response to incongruent stimuli than in response to congruent stimuli and colored X-strings, independently of age. This led us to consider that the P3b amplitude is sensitive to interference and to the conflict between incongruent words/colours [51,52]. By contrast, no differences were observed in the P3b amplitude in response to congruent and incongruent stimuli in a group of older participants [13], which was attributed to a possible floor effect in the older participants given the small P3b amplitude in this group of participants.
The amplitudes of both the LRP-S and the LRP-R components were larger in the old and middle-aged participants than in the young participants, which is consistent with the findings obtained by Ramos-Goicoa [16] with the Stroop task, Cespón et al. [43] with a Simon task and Wild- Wall et al. [40] with a Flanker task. By contrast, Killikelly & Szücs [14] did not find any differences between young and middleaged participants in the LRP-S and LRP-R amplitudes in a Stroop task, although the amplitudes were larger in the young participants than in a group of adolescents. The involvement of the motor cortex in generating these components [42] may indicate additional activation of the motor cortex in the processing of the correct response to stimuli in old and middleaged participants relative to young participants.
The larger amplitudes of the P3b, LRP-S and LRP-R components observed at the frontocentral electrodes in the old and middleaged participants than in the young participants may be evidence of greater recruitment of frontal neural resources. These findings are consistent with those of neuroimaging studies (fMRI and PET) showing greater activation of frontal regions in old than in young adults [57] and suggest that greater activation may also occur in middle aged adults.
In summary, the behavioural and ERP results indicate that the slowing of the neural processing of information that appears from middle-age onwards does not occur at all stages on the information processing and that it affects the evaluation and categorization of stimuli, as well the initiation and duration of the neural activity associated with processing of the motor response. In addition, the differences in the amplitude of the P3b, LRP-S and LRP-R components of the middle-aged and older participants relative to the young participants could be interpreted as being due to an age-related increase in the frontal neural resources for processing the stimuli and responses, The identification of neurofunctional changes from middle age onwards in healthy participants, as done in the present study, will contribute to discriminating these changes in the initial deterioration of pathological processes, i.e. at the early stages of illness, when treatment may be most effective.
Acknowledgments
The authors thank everyone who participated in this study. This study was financially supported by funds from the Spanish Government (Ministerio de Economia y Competitividad: PSI2014- 55316-C3-3-R) and from the Galician Government (Consellería de Cultura, Educacion e Ordinacion Universitaria; axudas para a consolidacion e Estruturacion de unidades de investigacion competitiva do Sistema Universitario de Galicia, GPC2014/047), by FEDER funds.
References
- Peña-Casanova J, Quiñones-Ubeda S, Gramunt-Fombuena N, Quintana M, Aguilar M, et al. (2009) Spanish Multicenter Normative Studies (NEURONORMA Project): Norms for the Stroop Color-Word Interference Test and the Tower of London-Drexel. Arch Clin Neuropsychol 24: 413-429.
- Troyer AK, Leach L, Strauss E (2006) Aging and Response Inhibition: normative Data for the Victoria Stroop Test. Aging Neuropsychol and Cognition 13: 20-35.
- Uttl B, Graf P (1997) Color-Word Stroop test performance across the adult life span. J Clin Exp Neuropsychol 19(3): 405-420.
- Van-der-Elst W, Van-Boxtel MPJ, Van-Breukelen GJP, Jolles J (2006) The Stroop color-word test Influence of age, sex, education; and normative data for a large sample across the adult age range. Assessment 13(1): 62-79.
- MacLeod CM (1991) Half a century of research on the Stroop effect: an integrative review. Psychol Bull 109(2): 163-203.
- Harada CN, Love MCN, Triebel KL (2013) Normal cognitive aging. Clin Geriatr Med 29(4): 737-752.
- Park D, Schwarz N (2002) Envejecimiento cognitivo. Médica Panamericana, Madrid.
- Raz N, Lindenberger U, Rodrigue KM, Kennedy KM, Head D, et al. (2005) Regional brain changes in aging healthy adults: general trends, individual differences and modifiers. Cereb Cortex 15(11): 1676-1689.
- Reuter-Lorenz PA, Park DC (2010) Human neuroscience and the aging mind: a new look at old problems. J Gerontol B Psychological Sciences Soc Sci. 65B (4): 405-415.
- Kray J, Eppinger B, Mecklinger A (2005) Age differences in attentional control: an event-related potential approach. Psychophysiology 42(4): 407-416.
- Eppinger B, Kray J, Mecklinger A, John O (2007) Age differences in task switching and response monitoring: evidence from ERPs. Biol Psychol 75(1): 52-67.
- West R, Alain C (2000) Age-related decline in inhibitory control contributes to the increased Stroop effect observed in older adults. Psychophysiology 37(2): 179-189.
- Zurrón M, Lindín M, Galdo-Alvarez S, Díaz F (2014) Age-related effects on event-related brain potentials in a congruence/incongruence judgment color-word Stroop task. Front Aging Neurosci 6(128): 1-8.
- Killikelly C, Szücs D (2013) Asymmetry in stimulus and response conflict processing across the adult life span: ERP and EMG evidence. Cortex 49(10): 2888-2903.
- Mager R, Bullinger AH, Brand S, Schmidlin M, Schärli H, et al. (2007) Age-related changes in cognitive conflict processing: an event-related potential study. Neurobiol Aging 28(12): 1925-1935.
- Ramos-Goicoa M, Galdo-Álvarez S, Díaz F, Zurrón M (2016) Effect of Normal Aging and of Mild Cognitive Impairment on Event-Related Potentials to a Stroop Color-Word Task. J Alzheimers Dis 52(4): 1487-1501.
- Folstein JR, Van-Petten C (2008) Influence of cognitive control and mismatches on the N2 component of the ERP: A Psychophysiology 45(1): 152-170.
- Hillyard SA, Kutas M (1983) Electrophysiology of cognitive processing. Annu Rev Psychol 34: 33-61.
- Kutas M, McCarthy G, Donchin E (1977) Augmenting mental chronometry: the P300 as a measure of stimulus evaluation time. Science 197(4305): 792-795.
- Amenedo E, Díaz F (1998) Aging-related changes in processing of non-target and target stimuli during an auditory oddball task. Biol Psychol 48(3): 235-267.
- Friedman D (2012) The components of aging. In: Luck SJ, Kappenman ES, editors. The Oxford handbook of event-related potential components, Oxford University Press, New York, pp. 513-536.
- Polich J (1996) Meta-analysis of P300 normative aging studies. Psychophysiology 33(4): 334-353.
- Salthouse TA (1996) The processing-speed theory of adult age differences in cognition. Psychol Rev 103(3): 403-428.
- Falkenstein M, Yordanova J, Kolev V (2006) Effects of aging on slowing of motor-response generation. Int J Psychophysiol 59(1): 22-29.
- Joyce C, Rossion B (2005) The face sensitive N170 and VPP components manifest the same brain processes: the effect of reference electrode site. Clin Neurophysiol 116(11): 2613-2631.
- Schendan HE, Ganis G, Kutas M (1998) Neuropsychological evidence for visual perceptual categorization of words and faces within 150 Psychophysiology 35(3): 240-251.
- Dien J (2009) The neurocognitive basis of reading single words as seen through early latency ERPs: a model of converging pathways. Biol Psychol 80(1): 10-22.
- Courchesne E, Hillyard SA, Galambos R (1975) Stimulus novelty, task relevance and the visual evoked potential in man. Electroencephalogr Clin Neurophysiol 39(2): 131-143.
- Friedman D, Cycowicz YM, Gaeta H (2001) The novelty P3: an event-related brain potential (ERP) sign of the brain's evaluation of novelty. Neurosci Biobehav Rev 25(4): 355-373.
- Squires NK, Squires KC, Hillyard SA (1975) Two varieties of long-latency positive waves evoked by unpredictable auditory stimuli in man. Electroencephalogr Clin Neurophysiol 38(4): 387-401.
- Van-Dinteren R, Arns M, Jongsma MLA, Kessels RPC (2014) P300 Development across the Lifespan: A Systematic Review and Meta-Analysis. PLoS One 9(2): e87347.
- Van-Dinteren R, Arns M, Jongsma MLA, Kessels RPC (2014) Combined frontal and parietal P300 amplitudes indicate compensated cognitive processing across the life span. Front Aging Neurosci 6(294): 1-9.
- Fabiani M, Friedman D (1995) Changes in brain activity patterns in aging: The novelty oddball. Psychophysiology 32(6): 579-594.
- Oldfield RC (1971) The assessment and analysis of handedness: The Edinburgh inventory. Neuropsychologia 9(1): 97-113.
- Lynöe N, Sandlund M, Dahlqvist G, Jacobsson L (1991) Informed consent: study of quality of information given to participants in a clinical trial. BMJ 303 (6803): 610-613.
- Lopez-Calderón J, Luck SJ (2014) ERPLAB: An open-source toolbox for the analysis of event-related potentials. Front Hum Neurosci 8: 213.
- Lezak MD, Howieson DB, Bigler ED, Tranel D (2012) Neuropsychological assessment. Oxford University Press, New York.
- Ratcliff R, Thapar A, McKoon G (2010) Individual differences, aging, and IQ in two-choice tasks. Cogn Psychol 60(3): 127-157.
- Ratcliff R, McKoon G (2008) The Diffusion Decision Model: Theory and Data for Two-Choice Decision Tasks. Neural Compute 20(4): 873-922.
- Wild-Wall N, Falkenstein M, Hohnsbein J (2008) Flanker interference in young and older participants as reflected in event-related potentials. Brain Res 1211: 72-84.
- Cid-Fernández S, Lindín M, Díaz, F (2014) Effects of Amnestic Mild Cognitive Impairment on N2and P3 Go/NoGo ERP Components. J Alzheimers Dis 38(2): 295-306.
- Smulders FTY, Miller JO (2012) Lateralized Readiness Potential. In: Luck S, Kappenman ES, editors. Oxford handbook of Event-Related Potential Components: Oxford University Press, New York, pp. 209-230.
- Cespón J, Galdo-Álvarez S, Díaz F (2013) Age-related changes in ERP correlates of visuospatial and motor processes. Psychophysiology 50(8): 743-757.
- Atkinson CM, Drysdale KA, Fulham WR (2003) Event-related potential to Stroop and reverse Stroop stimuli. Int J Psychophysio 47(1): 1-21.
- Duncan-Jonhson CC, Kopell BS (1980) The locus of interference in a Stroop task: When you read “blue”, do you see “red”?. Psychophysiology 17: 308-309.
- Duncan-Jonhson CC, Kopell BS (1981) The Stroop effect: brain potentials localize the source of interference. Science 214(4523): 938-940.
- Grapperon J, Vidal F, Leni P (1998) The contribution of cognitive evoked potentials to knowledge of mechanisms on the Stroop test. Neurophysiol Clin 28(3): 207-220.
- Ilan AB, Polich J (1999) P300 and response time from a manual Stroop Task. Clinical Neurophysiol 110(2): 367-373.
- Lavoie ME (1999) Toward a functional explanation of the locus of the Stroop inference: A psychophysiological study. Brain and Cognition 40(1): 167-170.
- Rosenfeld JP, Skogsberg KR (2006) P300-based Stroop study with low probability and target Stroop oddballs: the evidence still favours the response selection hypothesis. Int J Psychophysiol 60(3): 240-250.
- Zurrón M, Pouso M, Lindín M, Galdo S, Díaz F (2009) Event-Related Potentials with the Stroop colour-word task: Timing of semantic conflict. International Journal of Psychophysiology 72(3): 246-252.
- Zurrón M, Ramos-Goicoa M, and Díaz F (2013) Semantic Conflict Processing in the Color-Word Stroop and the Emotional Stroop: Event-related potential (ERP) correlates. J Psychophysiol 27(4): 149-164.
- Johnson R (1993) On the neural generators of the P300 component of the event-related potential. Psychophysiology 30(1): 90-97.
- Friedman D, Kazmerski VB, Fabiani MC (1997) An overview of age-related changes in the scalp distribution of P3b. Electroencephalography Clin Neurophysiology/Evoked Potentials Sections 104(6): 498-513.
- Houston RJ, Bauer LO, Hesselbrock VM (2004) Effects of borderline personality disorder features and a family history of alcohol or drug dependence on P300 in adolescents. Int J Psychophysiol 53(1): 57-70.
- Potter DD, Jory SH, Bassett MRA, Barrett K, Mychalkiw W (2002) Effect of mild head injury on event-related potential correlates of Stroop task performance. J Int Neuropsychol 8(6): 828-837.
- Park DC, Reuter-Lorenz P (2009) The Adaptive Brain: Aging and Neurocognitive Scaffolding. Annu Rev Psychol 60: 173-196.






























