The Altered Hormonal Homeostasis with Aging, Neuronal Dysfunction and Cognitive Decline
Vinod Nikhra*
Senior Chief Medical Officer and Consultant, Department of Medicine, Hindu Rao Hospital and NDMC Medical College, India
Submission: February 26, 2018; Published: March 19, 2018
*Corresponding author: Vinod Nikhra, Department of Medicine, Hindu Rao Hospital and NDMC Medical College, New Delhi, India, Email: drvinodnikhra@gmail.com
How to cite this article: Vinod Nikhra. The Altered Hormonal Homeostasis with Aging, Neuronal Dysfunction and Cognitive Decline. OAJ Gerontol & Geriatric Med. 2018; 3(4): 555619. DOI: 10.19080/OAJGGM.2018.03.555619
Abstract
Overview-Neurohormonal Aging: The human aging brings about various changes in hormonal homeostasis which include alterations in hormonal secretion, feedback loops and sensitivity of receptors and tissues. These changes have fallouts on cognitive functions through the receptors in brain areas, hormonal effects on neurotransmitters (NTM) and increased oxidative stress, and neuronal degeneration. The hormonal alterations occur across lifespan and play a role in cognitive dysfunction in form of mild cognitive impairment (MCI) and Alzheimer's disease (AD), and affect the health and activity of daily living (ADL) and quality of life (QOL) during later years.
The Endocrinological Alterations: Most of the hormones decrease with aging, like estrogen (in women), testosterone (in men), growth hormone and melatonin. The thyroid hormones may also decrease and subclinical hypothyroidism is common in older adults. Some hormones tend to remain at physiological levels as in younger adults, like cortisol and insulin except in disease conditions. But, the endocrine function may suffer with age because the hormonal receptors may become less sensitive. Whereas, some hormones increase in absolute or a relative ratio and include FSH, LH, norepinephrine, epinephrine, leptin and parathormone.
Pathophysiology of Neuronal Impact: Among the hormones, estrogen, progesterone and testosterone act directly on neurones in the brain, facilitate neurotransmission, enhance cerebral vasodilatation and blood flow, and protect neurones from neurotoxins and free radicals. Progesterone stimulates the GABA receptors and has an overall calming effect on the brain. The thyroid hormones (TH) regulate brain glucose utilization, neuronal metabolic activity and cerebral blood flow. The alterations in TH manifest as cognitive decline and increased risk for AD. With insulin deficiency, in the type 1 diabetes (T1DM) there occurs a slower information processing and cognitive decline. Whereas, in type 2 diabetes (T2DM), the associated deranged metabolic function and insulin resistance, leads to memory and cognitive dysfunctions, and neurodegenerative disorders. The decline in the overall GH level with age is called somatopause, and manifests as sleep disturbance and cognitive dysfunction. Adrenal hormones, DHEA and Cortisol also play an important role in aging related cognitive decline.
Applying Research - Hormone Replacement Strategies: The HRT may improve cognitive changes and consists of either combined hormone treatment having estrogen plus a progestin (HT) or estrogen alone (ET). The thyroxine (L-T4) treatment administered both sub- chronically and chronically, has been demonstrated to improve cognitive function, possibly mediated by an enhancement of cholinergic activity. But, as documented in clinical practice, the L-T4 treatment may not always completely restore normal cognitive functioning in patients with hypothyroidism. The use of thyroid hormone should be clinically relevant. Higher levels of thyroxine can potentiate oxidative stress and damage neurons, and are associated with accelerated cognitive decline. The replacement therapy for androgens is ridden with controversies, and the studies document that testosterone replacement therapy in a hypogonadal men does not have a positive effect on cognitive functions.
Conclusion-Finding the Solutions: The aim of hormone therapy is to suitably replenish the hormonal deficiencies that come with aging and to take care of their fallouts in the aging man or woman to maintain cognitive health. The hormonal replacement (HR) for a failing hormone is a grossly simple concept. But, then it will affect the altered feedback loops, attenuated receptors, and atrophied neurones. In all, the recent research provides various possibilities, but also puts various limitations and restraints on the therapeutic choices.
Keywords: Neurological Aging; Hormonal Homeostasis; Estrogen; Progesterone; Menopause; Testosterone; Andropause; DHEA; Thyroid Hormone; Cortisol; Calcium Homeostasis; Growth Hormone; Somatopause; Cognitive Decline; Alzheimer's Disease; Hormonal Replacement Therapy; ET; HRT
Abbreviations: ADL: activities of daily living; QOL: quality of life; AD: Alzheimer's disease; MCI: Mild Cognitive Impairment; NTMs: Neurotransmitters; ERs: Estrogen Receptors; HPG: Hypothalamus-Pituitary-Gonadal; BDNF: Brain-Derived Neurotrophic Factor; MAP: Mitogen- Activated Protein; pAkt: Phosphorylated Akt; CAlsr: CA1 Stratum Radiatum; dlPFC: Dorsolateral Prefrontal Cortex; WHI: Women's Health Initiative; WHIMS: WHI Memory Study; CEE: Conjugated Equine Estrogen; MPA: Medroxy Progesterone Acetate; HRT: Hormone Replacement Therapy; HT: Hormone Therapy; PADAM: Partial Androgen Deficiency in Aging Male; THs: Thyroid Hormones; Ach: Acetylcholine; T1DM: Type 1 Diabetes Mellitus; WMH: White Matter Hyperintensity; T2DM: Type 2 Diabetes Mellitus; GH: Growth Hormone; HR: Hormonal Replacement
The Neurohormonal Aging
The aging and underlying pathophysiological alterations are the gradual but heterogeneous. Simultaneously, there are declines of physiological functions, often accompanied by cognitive decline. These alterations are orchestrated through various molecular mechanisms, which complexly interact to alter the homeostasis. The endocrine system is involved in all of the integrative aspects of life, including cognitive faculties. The endocrine functions undergo major changes during aging, and include alterations in hormonal networks and signaling, and concomitant hormonal deficits/excess, augmented by decreased sensitivity of receptors and tissues to their action [1]. These hormonal alterations occur across lifespan, and affect the health, activities of daily living (ADL) and quality of life (QOL) during later years.
The demographic studies suggest that there is significant increase in the prevalence of Alzheimer's disease (AD) in older adults, of which 68% are female and 32% are male [2]. Further, because women have a longer life expectancy than men, the absolute number of women with AD exceeds that of men. In fact, the women are 1.5 times more likely to develop AD than age- matched men [3]. Traditionally, the aging process, including the development of physical frailty and a gradual loss in cognitive function during later years of life, has been considered to be physiological and unavoidable. In recent years, however, it has become evident scientifically and we can reject the grim stereotype of aging as an unalterable process of physical and cognitive decline, and optimistically can look forward to healthy and successful aging with good quality of life [4].
Essential Hormonal Alterations
With aging, the levels of various hormones undergo alterations [5]. Most of the hormones decrease with aging, like estrogen (in women), testosterone (in men), growth hormone and melatonin. The gonadal steroid hormones include androgen, estrogen, and progesterone, which decrease with aging. These hormones transduce their effects via hormone-specific receptors which are localized throughout the brain. The androgen, estrogen, progesterone, and the glucocorticoid receptors have been identified in brain regions associated with learning and memory areas such as the hippocampus [6]. In women, the acute decline in estrogen levels leads to menopause. In men, testosterone levels decrease gradually. Apart from, reproductive functions, estrogen, progesterone and testosterone play a critical role in modulation of cognition with aging and protect from loss of memory, cognition and progression of dementia [7].
The sex hormones estrogen, testosterone, and progesterone decline with age, while hormones regulating the HPA axis, such as corticosteroids have been shown to increase with age. The gonadal steroid hormones appear to act in concert with each other. The testosterone can be aromatized to estrogen, thus the loss of testosterone may also result in a reduction in estrogen. Further, the progesterone receptor contains an imperfect estrogen-binding site, thus loss of estrogen can potentially impact actions initiated through the progesterone receptor. The role of gonadal steroid hormones in cognition has been studied using both animal models and clinical trials [8]. Mood swings, memory loss, Mild Cognitive Impairment (MCI) and dementia are common symptoms and signs indicating that estrogen, progesterone and testosterone levels are declining [9].
The thyroid hormones decrease and subclinical hypothyroidism is common in older adults. There is evidence from animal models and observational human studies that thyroid hormone has an important influence on cognitive function, not only in childhood and adolescence but also in younger adults and the elderly [10]. The clinical undiagnosed hypothyroidism may present as difficulty in concentration, short-term memory loss and brain fog. The decreased levels of growth hormone lead to decreased muscle mass and strength, and affect cognitive functions; whereas the decreased melatonin levels play a role in the loss of normal sleep-wake patterns and affect the biological clock [11].
Some hormones tend to remain at physiological levels as in younger adults, like cortisol and insulin except in disease conditions. But, even when hormone levels do not change, endocrine function suffers with age because the hormonal receptors and tissues may become less sensitive [12]. This way an imbalance results especially with regard to Neurotransmitters (NTMs) like dopamine and serotonin, and hormones like cortisol, epinephrine and norepinephrine. Because of increased resistance and decreased production, the same is true for insulin, availability of which becomes chronically low in the brain in older adults and affects the cognitive functions and appetite regulation [13]. Cognitive performance is dependent on adequate glucose supply to the brain. Insulin, which regulates systemic glucose metabolism, has recently been shown both to regulate hippocampal metabolism and to be a mandatory component of hippocampally-mediated cognitive performance.
In addition, the adiposity with age increases serum triglycerides, which affects various bioactive molecules and hormones including leptin. The obese individuals appear to be more responsive to stress. Stress activates the sympathetic nervous system and the adipose-tissue cytokine leptin has been shown to stimulate SNS activity in animals. There is evidence that individuals with greater adiposity and/or higher plasma leptin would be more stress-responsive [14]. Apart from leptin, some hormones increase in absolute or a relative ratio and include FSH, LH, norepinephrine, epinephrine and parathormone.
Physiological Vs. Abnormal Homeostasis
The brain is part of a larger integrated biological system, which relies on signaling and neurotransmission throughout the body. There are balanced interactions between organs that release hormones and the nervous system which acts through neurotransmission in purview of the integrated biological system. Certain areas of brain are concerned with various cognitive functions. Whereas the hippocampus is concerned with short term memory, the temporal lobe is concerned with memory, emotions, hearing and language. The amygdola also shares emotions and social behaviour, whereas the frontal lobe is important for decision making, problem solving and planning [15].
Among the hormones, testosterone is Crucial for maintaining mental sharpness and clarity. The testosterone, along with estrogen and progesterone act directly on neurones in the brain, facilitating neurotransmission, and protect neurones from neurotoxins and free radicals, and enhance cerebral vasodilatation and blood flow. Estrogen facilitates higher cognitive functions by exerting effects on brain regions such as the prefrontal cortex and hippocampus. It induces spinogenesis and synaptogenesis in these two brain regions and also initiates a complex set of signal transduction pathways via Estrogen Receptors (ERs). The estrogen effects are mediated by activation of ER α and ER β, which in turn act on nuclear DNA, which leads to gene expression and protein synthesis, and enhance the NTM, associated functions by NTM associated proteins, spines and neurotrophins [16]. Progesterone stimulates the GABA receptors in brain, the feel-good, calming NTM, and has an overall calming effect on the CNS. A decreased progesterone level or the altered receptors lead to altered cognitive function [17].
The Pathophysiology Of Endocrinological Alterations
Estrogen
The HPG Axis: Estrogen plays an important role in the neurobiology of aging, and both endocrine and neural senescence overlap and is intertwined in complex feedback loops. The brain controls estrogen release through the Hypothalamus-Pituitary- Gonadal (HPG) axis. The GnRH neurons in the hypothalamus release RH, a decapeptide, which acts on the pituitary gland, to release the gonadotropins LH and FSH, which act on their receptors on the ovary to regulate production of sex steroid hormones, which are released into the circulation to exert feedback actions on the hypothalamus and pituitary gland. But, the brain also responds to estrogen through ERa and ERp receptors, which are distributed throughout the brain [18]. The neuroendocrine function is initiated in the hypothalamus and the circuitry responding to estrogen includes neocortex, hippocampus and brainstem (Figure 1). With the drop in circulating estrogens at menopause and the brain being a target organ for estrogen, women are prone for neurological changes, the cognitive dysfunctions and risk for depression, at menopause and in post-menopausal period [19].
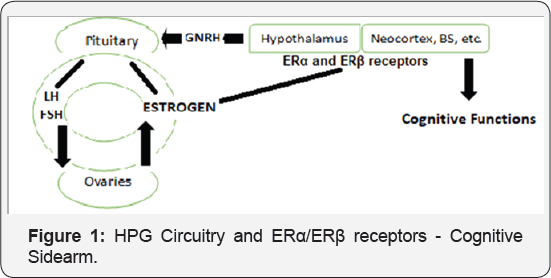
Estrogen and Aging: The major estrogen from ovaries is 17-β-estradiol. The process of reproductive aging in women has several unique features and occurs in stages spread over few years. In fact, the menopause transition represents a complex interplay of actions at all levels of the hypothalamic- pituitary-ovarian axis [20]. Another aspect is the impact of falling concentration of the reproductive hormones on brain function and behaviour during the menopause transition. For the majority of women, the reproductive aging is not associated with either depressive symptoms or the syndrome of significant cognitive decline.
But, in a minority of women, progress through the menopause transition and during the post-menopausal period, symptoms of depression, decreased concentration and memory decline are common [21]. There appears a probability that the declining ovarian estrogen secretion exacerbates the age-related decrement in episodic memory [22,23]. Several longitudinal and community-based studies have also documented an association between the menopause transition and an increased risk for depression [24]. Some recent longitudinal studies that followed women with no past history of depression demonstrated an increased risk of depressions during the menopause transition [25] .
The Neurological Mechanisms
a) The Estrogen Receptors (ERs): The brain undergoes many structural and functional changes during aging, some of which are regulated by estrogens which act mainly through the intracellular estrogen receptors, ERa and ERp. The expression of these receptors depends upon several factors including their own ligand estrogen, and others such as growth hormone and thyroid hormone. The levels of these factors also decrease during aging and affect cognitive functions. Another important factor, the nucleotide polymorphisms of ERa has been documented with an increased risk of cognitive dysfunction and dementia [26] .
b) Behavioural Alterations and Cognitive Decline: The age-related changes in estradiol concentrations affect function in hypothalamus and cortex, thalamus, amygdala, brainstem, cerebellum, and most other brain regions through ERs. Though, the GnRH cell numbers do not change with aging, the inputs to GnRH neurons from NMDA (N-methyl-D-aspartate) receptors though glutamate and other neurotransmitters, which regulate GnRH gene expression and biosynthesis, cause age-associated changes in GnRH output [27,28]. These changes in NMDARs may also contribute to, other dysfunctions including alterations in sexual behaviour and/or libido, and decline in cognitive function [29]. In other brain regions such as hippocampus and frontal cortex, the loss or change in estradiol signaling may be manifested by morphological and behavioural changes.
c) Loss of Neuroprotection: The efficacy of neurological function and preventing neurodegeneration depends on the binding of estradiol and other estrogenic ligands to membrane- associated, mitochondrial, and nuclear estrogen receptors in hippocampal and cortical neurons [30,31]. Estrogen-activated cellular signaling cascade promotes enhanced mitochondrial function, leading to increased calcium load tolerance, enhanced electron transport chain efficiency, and promotion of antioxidant defence mechanisms pivotal to sustaining calcium homeostasis and the estradiol-induced cascade that leads to neurotrophic and neuroprotective benefit [32]. Thus, the postmenopausal fall in estrogen levels have effects on the neuronal aging and cognitive performance. Consistent with these alterations, incidence of depression, cognitive dysfunction and dementia, including AD, has been reported in observational studies.
d) The miRNA levels: It is thought that menopause associated rapid age-related decline of circulating 17 β-estradiol levels alters the miRNA levels in an age- and brain region- dependent manner. This results in differential gene expression involving genes that are important for memory and stress regulation, such as Brain-Derived Neurotrophic Factor (BDNF), glucocorticoid receptor, and SIRT-1, and consequently alter neuronal function [33].
The Research Studies
a) The Animal Studies: The studies in rodents and NHP have provided evidence that estrogen favourably modifies synaptic circuitry in hypothalamus, hippocampus and neocortex and is neuroprotective [34,35]. The hippocampus is a forebrain structure and consists of the subiculum, CA1, CA2, CA3 and dentate gyrus regions. The studies in female rodents provide evidence that the synaptic effects of estrogen in hippocampus and prefrontal cortex influence cognitive aging through synaptic circuitry of CA1 in the hippocampus which is NMDAR dependent. These hippocampal circuits sub-serve memory functions. Effects of estradiol on GABAergic and cholinergic systems augment the glutamatergic impact on hippocampal function. In addition, estradiol rapidly stimulates signaling cascades such as the Mitogen-Activated Protein (MAP) kinase family and the phosphotidylinositol 3-kinase (PI3K), pathway leading to the phosphorylation of Akt, a key signaling molecule. Furthermore, Phosphorylated Akt (pAkt) is present in CA1 dendrites, spines, and synapses and is increased by the presence of estradiol [36-38].
Further, the synaptic effects and behavioural impact of estrogen in CA1 differ in young and aged female rats. There is an age-related attenuation of the beneficial cognitive effects of estrogen modulations in rats [39]. This is related to age and duration of loss of estrogen, after which ET is less effective. The activated signaling molecules such as pAkt, which is present in CA1 dendrites and spines play a critical role in estradiol-induced synaptic alterations. In addition, synaptic pAkt thought to be activated by ERα is also decreased dramatically in aged CA1 axospinous synapses as is ERβ suggesting that several key players in the local synaptic response to estradiol are compromised with age in female rats [40,41].
In young and aged rhesus monkeys, as compared to female rats, estrogen increases axospinous synapses in CA1. The studies using an NHP model have revealed the cognitive and neurobiological effects of ET in the context of aging [42]. The studies in rhesus monkeys used a regimen of cyclic exposure to estradiol in an attempt to replicate a pulsatile peak. Initially, the total number of spines in CA1 Stratum Radiatum (CA1sr) of both young and aged monkeys, showed an increase of 35%. Also, long-term cyclical ET enhances cognitive performance and spine density in the Dorsolateral Prefrontal Cortex (dlPFC) of aged rhesus monkeys. The dlPFC is an area in the prefrontal cortex of the brain involved in executive functions. It is one of the most recently evolved parts of the human brain and undergoes a prolonged period of maturation which lasts until adulthood and is highly vulnerable in aging and AD.
b) Observation and Clinical Studies: Numerous studies have documented beneficial effects of HT/ET regimens on cognition [43]. The trials demonstrated the beneficial effects of short-term (i.e., 2-12 weeks) ET on measures of verbal learning and memory [44]. There were beneficial effects of HT/ET on several domains of cognitive function, like verbal memory, vigilance, reasoning, and motor speed, in symptomatic perimenopausal women and in women in whom hypogonadism was recently induced by medical or surgical interventions.
The Women's Health Initiative (WHI) studies published in 2003 failed to collaborate these findings. Simultaneously, the WHI Memory Study (WHIMS) concluded that this HT regimen did not improve cognitive function but increased the risk for probable dementia in postmenopausal women aged 65 or older [45,46]. Other related WHIMS studies with ET also failed to protect against cognitive decline, although this regimen did not carry the same risk for increased incidence of dementia [47]. But, there were two notable points, these studies used the pharmaceutical formulations which different from the natural hormones estradiol and progesterone, and the HRT was initiated in women 65 years or older, which may be too late to expect the neurological benefits of ET or combined HT [48,49].
Both observational- and clinic-based studies have suggested that combined HT and unopposed ET in hypogonadal women improve cognition, lessen the risk for the development of dementia, and possibly, improve the severity and course of dementia [50]. Estrogen seems to enhance neuronal function, improve neuronal resilience, and serve as a neuro protective agent. In addition to the potential beneficial effects of estrogen on episodic memory, there is evidence to suggest that HT reduced the risks of both MCI and dementia including AD. The therapeutic response to estradiol was observed in both major and minor depression as well as in women with and without hot flushes in perimenopausal women. In the observational studies, the early initiation and adherence to HT/ET treatment were related to the ongoing relief of the affective, cognitive, and behavioral symptoms.
The impact of declining reproductive function on sexual function is variably expressed in women. Many of postmenopausal symptoms reported in women, such as vasomotor symptoms, vaginal dryness, etc. are directly attributable to low circulating estradiol. These may have correlation with loss of libido and sexual dysfunction in postmenopausal period. However, estradiol fails to restore sexual functioning. Some recent RCTs reported improvements in libido after testosterone therapy in hypogonadal women [51].
Hormone Therapy for the Cognitive Decline: The HRT may improve cognitive changes and consists of either combined hormone treatment having estrogen plus a progestin (HT) or estrogen alone (ET). The neuro-selective estrogen receptor modulator (NeuroSERM) and phyto-selective estrogen receptor modulator (PhytoSERM) molecules appear to be safe and efficacious estrogen alternatives for preserving neurological function and preventing neurodegenerative disease. The research indicates that a low dose estradiol exerts neuroprotection in the perimenopausal period in acute, continuous or intermittent mode [52].
Whereas, a high estradiol dose is ineffective at inducing neuroprotection regardless of pattern of dose, and in fact, exacerbate neurodegeneration. The mechanism by which high estradiol exacerbates neurodegeneration is likely through dysregulation of calcium homeostasis. Thus, estrogen-induced calcium signaling pathways both promote neuronal function and could exacerbate neuronal apoptosis in neurodegenerative states. The epidemiological studies indicate that women receiving ET at the time of menopause, in a prevention mode well before age-associated degeneration is prevalent, have a lower risk of developing AD than women who have never received ET or HT.
The retrospective studies have investigated the issue of ET or HT formulation as a determinant of the adverse outcomes of treatment trials. The results of a frequently prescribed combined estrogen/progestin formulation, Conjugated Equine Estrogen (CEE) plus Medroxy Progesterone Acetate (MPA) - Depo-Provera or PremPro - have documented that MPA is neither neuroprotective nor synergistic with estradiol. In fact, MPA antagonized estradiol-induced neuroprotection [53]. Moreover, MPA exacerbated glutamate induced excito-toxic neuronal death [54]. Results of the WHIMS trial in which the hormone therapy group (CEE plus MPA) documented a twofold risk of developing dementia strongly suggest that the addition of MPA has deleterious outcomes for the brain [55].
The evidence from various studies endorse that in the hypogonadal states, the Hormone Therapy (HT) or Hormone Replacement Therapy (HRT) is likely to prevent and reverse age- related alterations in the relevant neural circuitry. The HRT is protective against cognitive decline and AD, has no detrimental effect on cognitive function and has shown benefits on memory, attention and reasoning [56]. The observational data also suggest that HRT may reduce the risk of cognitive decline and dementia. In a population cohort of older women, lifetime HRT supplementation was associated with improved global cognition and attenuated decline over a 3-year interval [57]. A 2011 meta-analysis study concluded that the overall data from epidemiologic studies, observational studies and clinical trials of HRT, indicate that the age-related hormone decline plays an important role in the pathogenesis of cognitive decline and risk for Alzheimer's disease and there was a decreased risk of dementia in HRT users [58].
As per the latest research on HRT, last updated on Oct 12, 2017 with over 713 News and research items available on the subject online - 'Hormone Replacement Therapy News Widget', the postmenopausal estrogen-based hormone therapy lasting longer than ten years was associated with a decreased risk of AD in a large study [59]. Finally, the HT has very limited effect when administered to postmenopausal women with AD, as an adjuvant with anti-cholinesterasic treatment, HT was only efficient when administered before the onset of dementia and most therapeutic trials conducted to date have been unable to conclude that HT could significantly prevent the development of AD or decrease its severity [60,61].
The identification of the hormonal determinants of cognitive and neurodegenerative disorders in the older adults and the characterization of sub-groups of 'hormone-responsive' subjects potentially at risk may open up the possibility of offering tailor- made therapies based on hormones favouring natural steroids. The natural formulations (micronized progesterone, transdermal 17-β-estradiol) are preferable because of their fewer side effects, especially when given at the peri-menopause and have potentially greater impact on neuropsychiatric disorders [62]. For the postmenopausal hormone treatment, there is prognostic importance of the following parameters: women's age, age at start of hormone use, duration of therapy, dosage, route of administration, and the exact type and combination of estrogen and progestogen.
Testosterone
Testosterone and Cognitive Function: The testosterone secretion and serum levels gradually decline with age after attaining a peak in early twenties (Figure 2). Also, the advancing age is the most significant risk factor for the onset of general cognitive decline [63]. But, the decline is variable among individuals. Though, the underlying neuro- pathogenic mechanisms for cognitive decline with age are not fully understood, there is a mounting evidence, that it may also be related to the declining testosterone in men, like estrogen in women. Testosterone, an androgen, is a precursor of certain neurotransmitters. The testosterone appears to be neuroprotective by mediating neuronal and vascular aging in neuronal cells of the hippocampus and other areas which are involved in cognitive function [64]. The results from cell culture and animal studies support testosterone as neuroprotective hormone [65].
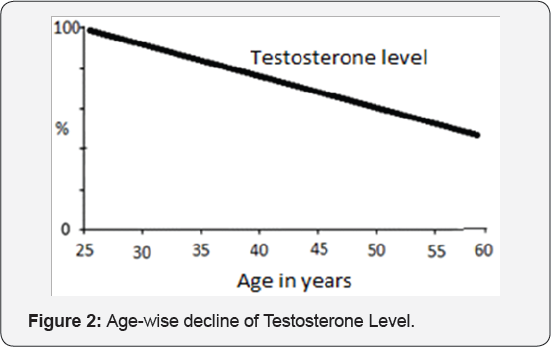
With aging, both cognitive functions and testosterone levels decline. Further, a recent meta-analysis, in 2016, documented that most men with AD and other cognitive disorders have low testosterone. Also, the testosterone levels are lower in men with AD compared with controls. In fact, the low testosterone levels may precede onset of AD by many years. Further, there is a documented evidence to prove that testosterone therapy in older men may improve cognitive functions [66].
Mechanism of neuroprotective action: A recent animal study evaluated a possible mechanism by which testosterone may inhibit cognitive decline is via the influence on neuronal and vascular aging in hippocampal cells. In the mouse model with hypogonadism and cognitive impairment, the treatment with testosterone decreased senescent changes in hippocampal vascular endothelial cells and inhibited cognitive decline. At the sub cellular level, key enzyme, SIRT1, an NAD+-dependent acetylase, was induced by testosterone therapy and led to decreased oxidative stress-induced endothelial senescence [67].
In a cross-sectional study, the OPTIMA trial, the testosterone levels were significantly lower in the older cohort, aged~80 years with AD than in controls. Also, a younger cohort with AD aged~66 years; the testosterone levels were significantly lower than the controls without AD [68]. The studies have investigated the probability of lower testosterone levels as an independent risk factor for AD. In the large and well-designed Baltimore Longitudinal Study of Aging, it was documented that the free testosterone serum levels at 2, 5, and 10 years before diagnosis of AD were documented to be reduced [69]. In another study, using post-mortem data, documented that in preclinical male AD cases, testosterone brain levels were lower than in controls [70]. These studies indicate that testosterone may play a part in early, preclinical phases of AD, suggesting a causative role. Evidence supporting this is the high densities of androgen receptors in hippocampus and nearby regions [71]. Further, these regions also exhibit the earliest functional declines in AD. As suggested in another study testosterone may modulate neuronal damage caused by oxidative stress and it probably decreases neuronal apoptosis, a key step in both AD and age-related cognitive decline [72].
Testosterone Deficiency States: The studies show the testosterone replacement therapy in hypogonadal men does not have a positive effect on cognition. Certain other factors also appear to influence the neuroprotective effect of testosterone and include sex-hormone-binding globulin, thyroid hormone, gonadotrophins and estrogen levels. An imbalance or deficiency of these factors may affect testosterone's neuroprotective actions and lead to cognitive decline. Evidence of the relationship between androgen deficiency and male depression also comes from studies that have assessed depression in hypogonadal subjects, the association between low testosterone levels and male depressive illness, and the antidepressant action of androgen replacement. The etiology of depressive symptoms of Partial Androgen Deficiency in Aging Male (PADAM) is multifactorial, and results from the interaction of the biological and psychosocial changes that take place during the mid-life transition [73].
Recently, a preliminary study of testosterone therapy in older men with low levels of the hormone and clinical conditions related to low testosterone, found that restoring levels to those of healthy young men improved sexual function. The treatment had a smaller effect on other aspects of health, such as the ability to walk or the sense of vitality [74]. The positive effect on cognitive functions was not documented. The PADAM is responsible for a variety of behavioural symptoms, such as weakness, decreased libido and erectile dysfunction, lower psychological vitality, depressive mood, anxiety, insomnia, difficulty in concentrating, and memory impairment. The psychological and behavioural aspects of PADAM may overlap with signs and symptoms of major depression [75].
Testosterone Therapy for Cognitive Dysfunction: Testosterone is an important modulator of cerebral functions. It appears to activate the cortical network, the ventral processing stream, during spatial cognition tasks, and the addition of testosterone improves spatial cognition in younger and older hypogonadal men. In addition, reduced testosterone is associated with depressive disorders. In the hypogonadal men, testosterone supplementation enhances vigor and energy and many aspects of mood and cognition [76].
Androgens play a neuroactive role during the aging process when it affects hippocampal spine synapse density, suggesting a role for androgen in the modulation of cognitive function and development of neurodegenerative disease. The sex steroids modulate brain function and the ability of the brain to process, store and retrieve sensory information [77]. The maintenance of neural elements in brain systems that support memory, such as synapse formation in prefrontal cortex and hippocampus, are critical for cognitive health in aging. There is a biological basis for androgens as neuro-protectants or neuromodulators and the importance of androgens for memory. The androgen deprivation causes significant loss of synapses in the hippocampus in rodent and nonhuman primates, increases amyloid deposition in human and rodent models and causes changes in neurotransmission in prefrontal cortex in rodent models.
The research suggests that these changes modify age-related cognitive loss, particularly to memory in men. In addition, the conversion of testosterone to its androgen metabolites or to estradiol may play a special role in the preservation of memory in aging [78]. Androgen deficiency has been reported to cause changes in mood and cognitive function and the preliminary evidence suggests that testosterone loss may be a risk factor for cognitive decline and possibly for dementia. Conversely, the maintenance of higher testosterone levels may prove beneficial for cognitive and brain function in elderly men. Large-scale placebo-controlled intervention studies are required to resolve ambiguities in the literature [79].
Thyroid Hormone
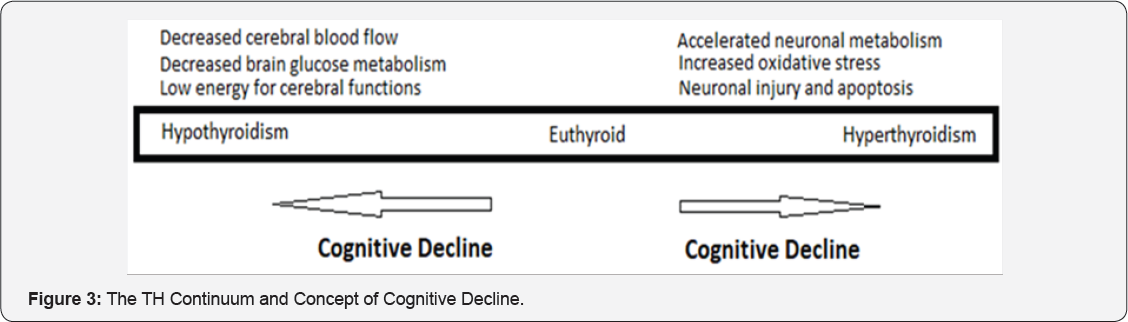
Thyroid Dysfunction and Cognitive Decline: The concentration of Thyroid Hormones (THs) declines with age. The cognitive decline is often concomitant with aging and the physiological changes in thyroid function might be causally related to changes in cognition during aging [80]. There is a continuum from low to increased concentrations of THs in which cognitive dysfunction can result (Figure 3). The clinical hypothyroidism has effects on cognition and mood, and there is evidence that subclinical hypothyroidism may be a predisposing factor for depression, cognitive impairment, and dementia. Subclinical hypothyroidism is common in the older adults, particularly in women. Further, the older adults are more vulnerable to the effects of subclinical hypothyroidism because of age-related changes to the hypothalamic-pituitary-thyroid axis [81].
The hyperthyroidism in older adults is also associated with decreased cognitive functioning, especially memory, visuospatial retention, attention, and reaction time. Thus, there is potentially increased risk of cognitive decline with thyroid dysfunction, and it is plausible to theorize that the thyroid status contributes to a risk, at least in part, to AD. In fact, hypothyroidism and hyperthyroidism have both been associated with cognitive impairment and dementia [82]. Even the mild variations of thyroid function can have significant consequences for cognitive function in the elderly, and the older adults with cognitive impairment should be assessed for hypothyroidism [83].
Mechanism of Cognitive Impairment in Hypothyroidism: The THs regulate cellular metabolic activity and neuronal development and function [84]. In fact, there exists interdependence between THs and Acetylcholine (Ach), nerve growth factor and hippocampal function. There occurs a decreased cerebral blood flow in mild hypothyroidism in regions mediating attention, motor speed, memory, and visuospatial processing. The THs regulate systemic glucose metabolism and may also be involved in regulation of brain glucose metabolism.
It may also decrease glucose metabolism in brain, thus, prevents the brain from adequately utilizing the energy needed for neurotransmission, memory, and other higher brain functions. Low brain uptake of glucose is commonly associated with deteriorating cognition and AD and can be present decades before clinical evidence of the disease occurs. Thus, brain hypo-metabolism is a precursor lesion increasing the risk of at least some forms of cognitive decline [85]. Further, the THs imbalance is often encountered in combination with metabolic disorders such as diabetes, and may cause additional metabolic dysregulation and hence worsening of disease states. THs may also modulate memory processes, at least in part by modulation of central insulin signaling and glucose metabolism.
Thyroid Replacement Therapy:
a) The results from animal studies: The L-T4 treatment administered both sub-chronically and chronically, significantly enhances the ability of rats to learn a spatial memory task, compared with controls. Moreover, both short-term and longterm L-T4 treatment reduced the cognitive-impairing effects of scopolamine. Improvements in performance were shown to occur along with increased cholinergic activity in frontal cortex and hippocampus of treated animals. These findings demonstrate an augmentative effect of L-T4 upon cognitive function, possibly mediated by an enhancement of cholinergic activity. The results support a relationship between L-T4 and acetylcholine, and the possible mechanisms by which disorders of thyroid function may be associated with cognitive decline.
b) The Results from Clinical Studies: As documented by clinical research, the L-thyroxine treatment may not always completely restore normal cognitive functioning in patients with hypothyroidism [86]. The use of thyroid hormone should be clinically relevant. Higher levels of thyroxine can potentiate oxidative stress and damage neurons, and are associated with accelerated cognitive decline. Therefore treatment with thyroxine in those without thyroid disease is not recommended. In fact, the optimal therapeutic level in the older adults may be normally a bit on lower side because of the physiological slowdown of tissue metabolism.
Insulin
In the Type 1 Diabetes Mellitus (T1DM) patients, cerebral micro vascular damage is common. As life expectancy in patients with T1DM has increased because of better therapeutic modalities, living longer and the longer exposure to disease- related factors, contributes to cerebral micro vascular disease. Clinically relevant White Matter Hyperintensity (WMH) lesions are evident earlier among middle-aged patients with T1DM and lead to the slower information processing and cognitive decline [87]. Brain imaging in T1DM patients having cognitive dysfunctions, especially those with neuropathies, may delineate cerebral micro vascular lesions [88]. Signs of accelerated aging that are related to slower information processing are evident in the brains of middle-aged adults with T1DM. As the people with T1DM are now living longer than ever before and as the incidence of T1DM is increasing annually, the impact of the disease is seen more often and they increasingly present with accelerated brain aging and cognitive decline.
In Type 2 Diabetes Mellitus (T2DM) also, aging is associated with a deranged metabolic function, insulin resistance, increased incidence of neurodegenerative diseases, and memory or cognitive dysfunction [89]. The onset of type 2 diabetes in older adults appears to affect late-life cognition by reducing brain volume, as revealed by MRI studies in a population- based cohort without dementia. But, the T2DM onset after age 64 does not seem to impact brain pathology or cognitive functions, suggesting that the deleterious impact of diabetes on memory and other cognitive aspects develops over decades. The older adults with pre-diabetes and T2DM may suffer from an accelerated decline in brain volume and cognitive functions over as short as two years period, suggesting that other factors also play a role. Further, the aging brain is vulnerable to worsening blood sugar levels even before T2DM is diagnosable [90].
The researchers have documented that older adults having pre-diabetes and T2DM, may lose almost two and a half times more brain volume than the controls over two years. The reduction in size of the frontal lobe, associated with higher mental functions like decision-making, emotional control, and long term memory, has a significant impact on cognitive function and quality of life. The Sydney Memory and Ageing Study, compared MRI scans in older adults taken from the beginning and end of a two-year period. The participants were elderly aged between 70 and 90 years old (54% were male) and free from dementia. At the start of the study 41% had pre-diabetes and 13% had type 2 diabetes. The researchers found that a person's blood sugar status after two years can significantly predict their decline in brain volume [91].
The older adults with T2DM and hypertension may be found to have cortical or subcortical infarctions, WMH, and hippocampal and whole brain volume loss on MRI scanning, and cognitive decline with reduced performance in executive function during cognitive testing, and have a higher risk for AD. In addition, the older adults with T2DM show a two-fold increase in MCI risk as compared with those having no diabetes. The dysfunctional insulin signaling and altered glucose metabolism in the brain of diabetics appears to increase tau phosphorylation, increase insulin binding to insulin-degrading enzyme, decrease beta-amyloid clearance, and lead to neurodegeneration [92].
Growth Hormone
The Growth Hormone (GH) levels also decline with age and may contribute to the cognitive dysfunction typical of aging [93]. GH has a pulsatile pattern of release by the pituitary gland, and the normal pulsatile secretion may be lost before there develops actual GH deficiency. The decline in the overall GH level with age is called somatopause. The signs of somatopause in adults include decrease in lean body mass, decreased joint cartilage, decrease in cardiac endurance, delayed rate of wound healing and decreased sleep duration and quality. The recent clinical research suggests that somatropin or human GH is useful as an anti-aging hormone. The low GH has also been linked to chronic fatigue syndrome, fibromyalgia and osteoporosis.
The low GH states that may benefit from GH replacement therapy include obesity and plaque buildup leading to cardiovascular disease. Tesamorelin is a synthetic version of growth-hormone-releasing hormone, or GHRH. The drug stimulates the production of HGH. In a 20 week double blind study, the researchers found that supplemental GHRH had a positive effect on cognition and healthy brain function not only in healthy older adults but also in adults at increased risk of cognitive decline and dementia [94]. Other research works have also documented that GH supplementation in low GH states may be useful in treating cognitive decline [95].
Cortisol and Dhea
Cortisol is involved in balancing blood glucose levels, immune system responses, bone turnover rate, mood and thought, sleep and protein catabolism. Elevated cortisol is associated with anxiety, insulin resistance, obesity, osteoporosis, sex hormone imbalance, onset insomnia, accelerated aging and immune suppression and disrupts gastrointestinal microflora levels causing dysbiosis. Low cortisol levels, on the other hand, relate to CFS, depression, PMS, menopause, fibromyalgia, impotence in men, fertility and maintenance insomnia [96]. An increase in cortisol levels experienced during prolonged stress, will inhibit release of GH. Also there will be a reduction in the release of LH and testosterone. Cortisol also influences the activity of insulin and THs. Decreased sleep causes an increase in cortisol, a decrease in GH and testosterone. Also the release of melatonin will be affected and this further reduces the production of GH and quality of sleep. The sleep disturbances can exacerbate age- related changes in the brain through the endocrine system [97].
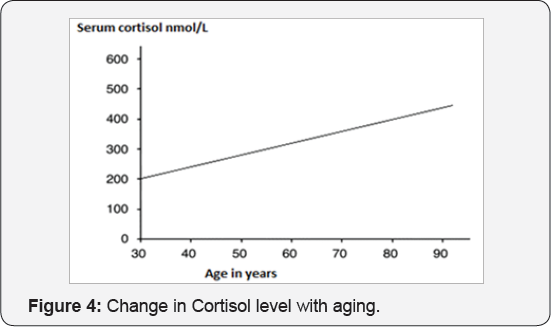
The cortisol levels increase with age (Figure 4). Adrenal hormones DHEA and Cortisol also play an important role in aging related cognitive decline. DHEA, the principle adrenal androgen, and its sulphonated ester DHEA-S, decrease with age [98]. In addition to its androgenic function, DHEA has been reported to have other functions such as improving carbohydrate metabolism, neurological function and general well-being. Low levels of DHEA-S are involved in the decline in immunity, chronic fatigue, nonspecific arthritis, insomnia, decreased libido, obesity, depression and osteoporosis. The hippocampal region of the brain show plasticity and resilience of brain cells to stress hormone action and aging. There is vulnerability of the aging hippocampus in relation to stress and AD [99]. Mechanisms of neuronal death or apoptosis in aging brain and AD include the role of endocrine-mediated altered calcium homeostasis [100].
Applying Recent Research For Action Plan
Research, Results and Concepts
The biological research including the animal studies and clinical research, and various related discoveries have led to wider understanding about hormonal homeostasis, hypothalamic- pituitary axis for hormonal control, the feedback loops which auto-adjust and auto-regulate and role and impact on brain and neurotransmitters, and cognitive functions [101]. The results have led to formulation of theories and concepts through which we can understand and act. The Hormonal Replacement (HR) for a failing hormone is a grossly simple concept. But, then it will affect the altered feedback loops, attenuated receptors, and atrophied neurones. In all, the recent research provides various possibilities, but also puts limitations on therapeutic choices.
HR: Controversies and Restrictions
As for the postmenopausal or perimenopausal women with the hypogonadal states, the HT is likely to prevent and reverse age-related cognitive alterations, and protect AD, but should not have any detrimental effect on other physiological functions [102]. It should also be free from serious side-effects and aftereffects. Further, the research indicates that a low dose estradiol exerts neuroprotection in acute, continuous or intermittent mode, but a high estradiol dose is ineffective at inducing neuroprotection regardless of pattern of dose and may have significant side-effects. Various clinical studies have provided mixed results, and have failed to endorse a unified therapeutic approach. Thus, the wonderful possibilities of HT have been put on back-foot, and lie vastly under-utilized.
The testosterone replacement therapy in PADAM in older men is also ridden with unclarified and mixed results from various clinical studies, hence the therapeutic controversies and confusion [103]. The DHEA treatment is equally controversial [104]. The androgen deficiency leads to mood disorders and cognitive dysfunction and predispose to dementia. A suitable replacement dose may help, but the maintenance of higher testosterone levels have been shown to be beneficial for cognitive and brain function in aging male. TH has a role is in regulating brain glucose metabolism and modulating hippocampal cognitive processes. The THs gradually declines with age and hypothyroidism is common. In fact, TH replacement is the most common in hormonal treatment in clinical practice. But, the L-T4 treatment may not always completely restore normal cognitive functioning in patients with hypothyroidism. On the other hand, higher levels of thyroxine can potentiate oxidative stress and damage neurons, and are associated with accelerated cognitive decline. Therefore, TH replacement should be regularly monitored.
Conclusion: Way Out of the Research Smog
Dissecting Data for Uniformity
With aging, various tissues and organs including the brain gradually fail. The latter is manifested as cognitive decline, MCI and AD. The cognitive decline with aging is part due to wear and tear processes in various neurones and faltering hormonal homeostasis including alterations in the concerned receptors and their impact on neuronal functioning. Over the year, animal studies as well as clinical trials and therapeutic practice have generated vast but diverse data. They provide a gamut of results from varying methodology with differing subjects. These data at times are lope-sided and contradictory. Many a time, the research deals with one aspect of the totality and fails to generate a wholesome inference. The conclusions drawn in various studies restricted and biased, and may preclude further research in the area. Thus, it is required that the research works are dissected out to find and develop relevancy for clinical applicability.
Finding Solutions and Final Words
The biological systems in an organism are regulated by brain through neurotransmitters and hormone releasing factors, influencing hormones secretion. The hormones communicate between organs and tissues by binding to specific receptors for physiological regulation. They also communicate with brain for physiologic and metabolic modulation by the feedback loops and through various receptors on different parts of the brain for behavioral modulation. The aim of hormone therapy, which aims to suitably replenish all the hormonal deficiencies which come with aging, is to prevent and treat their fallouts in the aging man or woman to maintain cognitive health. The hormone therapy needs to address to cognitive decline, and improve ADL and QOL.
References
- Diamanti Kandarakis E, Dattilo M, Macut D (2017) Mechanisms in Endocrinology: Aging and anti-aging, a Combo-Endocrinology overview. Eur J Endocrinol 176(6): R283-R308.
- Brookmeyer R, Gray S, Kawas C (1998) Projections of Alzheimer's disease in the United States and the public health impact of delaying disease onset. Am J Public Health 88(9): 1337-1342.
- Zandi PP, Carlson MC, Plassman BL (2002) Hormone replacement therapy and incidence of Alzheimer disease in older women: the Cache County Study. JAMA 288(17): 2123-2129.
- Steven W J Lamberts (2002) The Endocrinology of Aging and the Brain. JAMA Neurology 59(11): 1709-1711.
- Morley JE (2017) Effects of Aging on the Endocrine System, Merk Manuals.
- Lewis DK, Sohrabji F (2009) Hormonal Influences on Brain Aging and Age-Related Cognitive Decline. In Bizon J, Woods (Eds.) Animal Models of Human Cognitive Aging. Aging Medicine, Humana Press, New York, USA.
- Will MA, Randolph JF (2009) The influence of reproductive hormones on brain function in the menopausal transition. Minerva Ginecol 61(6): 469-481.
- Ulubaev A, Lee DM, Purandare N (2009) Activational effects of sex hormones on cognition in men. Clin Endocrinol 71(5): 607-623.
- Akishita M (2014) Sex hormones and cognitive function. Japanese Journal of Clinical Medicine 72(4): 657-661.
- Lazarus JH (2012) Thyroid hormones and cognitive function. J Exp Rev Endocrinol Metabol 7(4): 365-367.
- Beydoun MA, Beydoun HA, Kitner Triolo MH (2013) Thyroid Hormones Are Associated With Cognitive Function: Moderation by Sex, Race, and Depressive Symptoms. The Journal of Clinical Endocrinology & Metabolism 98(8): 3470-3481.
- Whalen RE (1980) Hormone receptors in the brain. Hormones and the Brain p. 3-10.
- Clark A, Jones LC, de Koning E (2001) Decreased Insulin Secretion in Type 2 Diabetes: A Problem of Cellular Mass or Function? Diabetes 50(1): S169-S171.
- Brydon L (2011) Adiposity, leptin and stress reactivity in humans. Biol Psychol 86(2): 114-120.
- Nugent, Pam MS (2016) Parts of the Brain and Cognitive Function. Psychology Dictionary.
- Hara Y, Waters EM, McEwen BS, Morrison JH (2015) Estrogen Effects on Cognitive and Synaptic Health over the Lifecourse. Physiological Reviews 95(3): 785-807.
- Callachan H, Cottrell GA, Hather NY (1987) Modulation of the GABAA receptor by progesterone metabolites. Proc R Soc Lond B Biol Sci 231(1264): 359-369.
- Morrison JH, Brinton RD, Schmidt PJ, Gore AC (2006) Estrogen, menopause, and the aging brain: How basic neuroscience can inform hormone therapy in women. J Neurosci 26(41): 10332-10348.
- Sherwin BB (2012) Estrogen and cognitive functioning in women: lessons we have learned. Behavioral Neuroscience 126(1): 123-127.
- Santoro N (2005) The menopausal transition. Am J Med 118: 8-13.
- Woods NF, Mitchell ES, Adams C (2000) Memory functioning among midlife women: observations from the Seattle Midlife Women's Health Study. Menopause 7(4): 257-265.
- Maki PM, Zonderman AB, Resnick SM (2001) Enhanced verbal memory in nondemented elderly women receiving hormone-replacement therapy. Am J Psychiatry 158(2): 227-233.
- Henderson VW, Guthrie JR, Dudley EC (2003) Estrogen exposures and memory at midlife: a population-based study of women. Neurology 60(8): 1369-1371.
- Schmidt PJ, Haq N, Rubinow DR (2004) A longitudinal evaluation of the relationship between reproductive status and mood in perimenopausal women. Am J Psychiatry 161(12): 2238-2244.
- Cohen LS, Soares CN, Vitonis AF (2006) Risk for new onset of depression during the menopausal transition: the Harvard study of moods and cycles. Arch Gen Psychiatry 63(4): 385-390.
- Freeman EW, Sammel MD, Lin H, Nelson DB (2006) Associations of hormones and menopausal status with depressed mood in women with no history of depression. Arch Gen Psychiatry 63(4): 375-382.
- Thakur MK, Sharma PK (2006) Aging of Brain: Role of Estrogen. Neurochemical Research 31(11): 1389-1398.
- Ryan J, Scali J, Carriere I (2008) Hormonal treatment, mild cognitive impairment and Alzheimer's disease. Int Psychogeriatr 20(1): 47-56.
- Yin W, Gore AC (2006) Neuroendocrine control of reproductive aging: roles of GnRH neurons. Reproduction 131(3): 403-414.
- Gore AC, Windsor-Engnell BM, Terasawa E (2004) Menopausal increases in pulsatile gonadotropin-releasing hormone (GnRH) release in a non-human primate (Macaca mulatta). Endocrinology 145(10): 4653-4659.
- Burke SN, Barnes CA (2006) Neural plasticity in the ageing brain. Nat Rev Neurosci 7(1): 30-40.
- Milner TA, Mc Ewen BS, Hayashi S (2001) Ultrastructural evidence that hippocampal alpha estrogen receptors are located at extra nuclear sites. J Comp Neurol 429(3): 355-371.
- Milner TA, Ayoola K, Drake CT (2005) Ultrastructural localization of estrogen receptor beta immunoreactivity in the rat hippocampal formation. J Comp Neurol 491(2): 81-95.
- Simpkins JW, Wang J, Wang X (2005) Mitochondria play a central role in estrogen-induced neuroprotection. Curr Drug Targets CNS Neurol Disord 4(1): 69-83.
- Rao YS, Mott NN, Wang Y, Chung WCJ (2013) MicroRNAs in the Aging Female Brain: A Putative Mechanism for Age-Specific Estrogen Effects. Endocrinology 154(8): 2795-2806.
- Lacreuse A (2006) Effects of ovarian hormones on cognitive function in nonhuman primates. Neuroscience 138(3): 859-867.
- Bryant DN, Sheldahl LC, Marriott LK (2006) Multiple pathways transmit neuroprotective effects of gonadal steroids. Endocrine 29(2): 199-207.
- Rudick CN, Woolley CS (2001) Estrogen regulates functional inhibition of hippocampal CA1 pyramidal cells in the adult female rat. J Neurosci 21(17): 6532-43.
- Tinkler GP, Tobin JR, Voytko ML (2004) Effects of two years of estrogen loss or replacement on nucleus basalis cholinergic neurons and cholinergic fibers to the dorsolateral prefrontal and inferior parietal cortex of monkeys. J Comp Neurol 469(4): 507-521.
- Znamensky V, Akama KT, Mc Ewen BS, Milner TA (2003) Estrogen levels regulate the subcellular distribution of phosphorylated Akt in hippocampal CA1 dendrites. J Neurosci 23(6): 2340-2347.
- Savonenko AV, Markowska AL (2003) The cognitive effects of ovariectomy and estrogen replacement are modulated by aging. Neuroscience 119(3): 821-830.
- Bailey M, Wang ACJ, Hao J (2011) Interactive Effects of Age and Estrogen on Cortical Neurons: Implications for Cognitive Aging. Neuroscience 191: 148-158.
- Yildirim M, Janssen WG, Lou WWY (2006) Effects of estrogen and aging on distribution of pAkt in CA1 of rat hippocampus. Soc Neurosci Abstr 32: 659-661.
- Janssen WG, Yildirim M, Lou WWY (2006) Estrogen receptor β is affected by estrogen and aging in CA1sr region of female rat hippocampus. Soc Neurosci Abstr 32: 659-617.
- Sherwin BB (2006) Estrogen and cognitive aging in women. Neuroscience 138(3): 1021-1026.
- Sherwin BB (2003) Estrogen and cognitive functioning in women. Endocr Rev 24(2): 133-151.
- Rapp SR, Espeland MA, Shumaker SA (2003) Effect of estrogen plus progestin on global cognitive function in postmenopausal women: the Women's Health Initiative Memory Study: a randomized controlled trial. JAMA 289(20): 2663-2672.
- Shumaker SA, Legault C, Rapp SR (2003) Estrogen plus progestin and the incidence of dementia and mild cognitive impairment in postmenopausal women: the Women's Health Initiative Memory Study: a randomized controlled trial. JAMA 289(20): 2651-2662.
- Espeland MA, Rapp SR, Shumaker SA (2004) Conjugated equine estrogens and global cognitive function in postmenopausal women: Women's Health Initiative Memory Study. JAMA 291(24): 2959-2968.
- Morrison JH, Brinton RD, Schmidt P, Gore AC (2006) Estrogen, Menopause, and the Aging Brain: How Basic Neuroscience Can Inform Hormone Therapy in Women. The Journal of Neuroscience 26(41): 10332-10348.
- Maki PM (2006) Hormone therapy and cognitive function: is there a critical period for benefit? Neuroscience 138(3): 1027-1030.
- Dubal DB (2002) Estrogen and neuroprotection: from clinical observations to molecular mechanisms. Dialogues Clin Neurosci 4(2): 149-161.
- Manson JE, Bassuk SS, Harman SM (2006) Postmenopausal hormone therapy: new questions and the case for new clinical trials. Menopause 13(1): 139-147.
- Simon J, Braunstein G, Nachtigall L (2005) Testosterone patch increases sexual activity and desire in surgically menopausal women with hypoactive sexual desire disorder. J Clin Endocrinol Metab 90(9): 5226-5233.
- Nilsen J, Deng J, Brinton RD (2005) Impact of clinically relevant progestins on the neural effects of estradiol and the signaling pathways involved. Drug News Perspect 18(9): 545-553.
- Nilsen J, Morales A, Brinton RD (2006) Medroxyprogesterone acetate exacerbates glutamate excitotoxicity. Gynecol Endocrinol 22(7): 355361.
- Shumaker SA, Legault C, Kuller L (2004) Conjugated equine estrogens and incidence of probable dementia and mild cognitive impairment in postmenopausal women: Women's Health Initiative Memory Study JAMA 291(24): 2947-2958.
- Maki P, Hogervorst E (2003) HRT and cognitive decline. Clinical Endocrinology & Metabolism 17(1): 105-22.
- Carlson MC, Zandi PP, Plassman BL (2001) Hormone replacement therapy and reduced cognitive decline in older women. The Cache County Study, Neurology. 57(12): 2210-2216.
- Janicki SC, Schupf N (2010) Hormonal Influences on Cognition and Risk for Alzheimer Disease. Curr Neurol Neurosci Rep 10(5): 359-366.
- (2017) Hormone replacement therapy (HRT).
- Hogervorst E, Yaffe K, Richards M, Huppert F (2002) Hormone replacement therapy for cognitive function in postmenopausal women. Cochrane Database of Systematic Reviews 2: CD003122.
- Rigaud AS, Andre G, Vellas B, Touchon J, Pere JJ (2003) No additional benefit of HRT on response to rivastigmine in menopausal women with AD. Neurology 60(1): 148-149.
- Pines A (2007) Postmenopausal hormone therapy: The way ahead. Maturitas 57(1): 3-5.
- Evans DA, Funkenstein HH, Albert MS (1989) Prevalence of Alzheimer's disease in a community population of older persons higher than previously reported. JAMA 262(18): 2552-2556.
- Pike CJ, Carroll JC, Rosario ER, Barron AM (2009) Protective actions of sex steroid hormones in Alzheimer's disease. Front Neuroendocrinol. 30(2): 239-258.
- Holland J, Bandelow S, Hogervorst E (2011) Testosterone levels and cognition in elderly men: a review. Maturitas 69(4): 322-337.
- Tan RS, Pu SJ (2003) A pilot study on the effects of testosterone in hypogonadal aging male patients with Alzheimer's disease. Aging Male 6(1): 13-17.
- Ota H, Akishita M, Akiyoshi T (2012) Testosterone deficiency accelerates neuronal and vascular aging of SAMP8 mice: protective role of eNos and SIRT1. PLoS ONE 7: e29598.
- Hogervorst E, Bandelow S, Combrinck M (2004) Low free T is an independent risk factor for Alzheimer's disease. Exp Gerontol 39(11- 12): 1633-1639.
- Moffat SD, Zonderman AB, Metter EJ (2004) Free T and risk of Alzheimer disease in older men. Neurology 62(2): 188-193.
- Rosario ER, Chang L, Head EH, Stanczyk FZ, Pike CJ (2009) Brain levels of sex steroid hormones in men and women during normal aging and in Alzheimer's disease. Neurobiol Aging 32: 604-613.
- Amore M, Scarlatti F, Quarta AL, Tagariello P (2009) Partial androgen deficiency, depression and testosterone treatment in aging men. Aging Clinical and Experimental Research 21(1): 1-8.
- Braak H, Braak E (1995) Staging of Alzheimer's disease-related neurofibrillary changes. Neurobiol Aging. 16(3): 271-278.
- Hammond J, Le Q, Goodyer C (2001) T mediated neuroprotection through the androgen receptor in human primary neurons. J Neurochem. 77(5): 1319-1326.
- Snyder PJ, Bhasin S, Cunningham GR (2016) For the Testosterone Trials Investigators. Effects of Testosterone Treatment in Older Men. N Engl J Med 374: 611-624.
- Amore M, Scarlatti F, Quarta AL, Tagariello P (2009) Partial androgen deficiency, depression and testosterone treatment in aging men. Aging Clinical and Experimental Research 21(1): 1-8.
- Onakomaiya MM, Henderson LP (2016) Mad men, women and steroid cocktails: a review of the impact of sex and other factors on anabolic androgenic steroids effects on affective behaviors. Psychopharmacology 233(4): 549-569.
- Genazzani AR, Pluchino N, Freschi L (2007) Androgens and the brain. Maturitas 57(1): 27-30.
- Aggarwal N, Razvi S (2013) Thyroid and Aging or the Aging Thyroid? An Evidence-Based Analysis of the Literature. Journal of Thyroid Research 481287.
- Janowsky JS (2006) The role of androgens in cognition and brain aging in men. Neuroscience 138(3): 1015-1020.
- Moffat SD (2006) Effects of Testosterone on Cognitive and Brain Aging in Elderly Men. Annals of the New York Academy of Sciences.
- Davis JD, Stern RA, Flashman LA (2003) Cognitive and neuropsychiatric aspects of subclinical hypothyroidism: Significance in the elderly Current Psychiatry Reports 5(5): 384-390.
- Smith JW, Evans T, Costall B, Smythe JW (2002) Thyroid hormones, brain function and cognition: a brief review. Neuroscience & Biobehavioral Reviews 26(1): 45-60.
- Hogervorst E, Huppert F, Matthews FE, Brayne C (2008) Thyroid function and cognitive decline in the MRC Cognitive Function and Ageing Study. Psychoneuroendocrinology 33(7): 1013-1022.
- Smith JW, Evans BT, Costall B, Smythe JW (2002) Thyroid hormones, brain function and cognition: a brief review. Neuroscience & Biobehavioral Reviews 26(1): 45-60.
- Jahagirdar V, Mc Nay EC (2012) Thyroid hormones role in regulating brain glucose metabolism and potentially modulating hippocampal cognitive processes. Metabolic Brain Disease 27(2): 101-111.
- Begin ME, Langlois MF, Lorrain D, Cunnane SC (2008) Thyroid Function and Cognition during Aging. Current Gerontology and Geriatrics Research 2008: 474868.
- Zhang J, Wang Y, Wang J (2014) White matter integrity disruptions associated with cognitive impairments in type 2 diabetic patients. Diabetes 63(11): 3596-3605.
- /a> Nunley KA, Ryan CM, Orchard TJ (2015) White matter hyper-intensities in middle-aged adults with childhood-onset type 1 diabetes. Neurology 84(20): 2062-2069.
- Roberts RO, Knopman DS, Przybelski SA (2014) Association of type 2 diabetes with brain atrophy and cognitive impairment. Neuol 82(13): 1132-1141.
- Samaras K (2012) Diabetes shrinks elderly brain. European Society of Endocrinology Science Daily
- Nyberg F, Hallberg M (2013) Growth hormone and cognitive function. Nature Reviews Endocrinology 9(6): 357-365.
- Baker LD, Barsness SM, Borson S (2012) Effects of Growth Hormone- Releasing Hormone on Cognitive Function in Adults with Mild Cognitive Impairment and Healthy Older Adults: Results of a Controlled Trial. Arch Neurol 1-10.
- Vitiello MV, Moe KE, Merriam GR (2006) Growth hormone releasing hormone improves the cognition of healthy older adults. Neurobiol Aging 27(2): 318-323.
- Comijs HC, Gerritsen L, Penninx B (2010) The Association between Serum Cortisol and Cognitive Decline in Older Persons. American J Geriatric Psychiatry 18(1): 42-50.
- Mander BA, Winer JR, Walker MP (2017) Sleep and human aging. Neuron 94(1): 19-36.
- Luppi C, Fioravanti M, Bertolini B (2009) Growth factors decrease in subjects with mild to moderate Alzheimer's disease (AD): potential correction with dehydroepiandrosterone-sulphate (DHEAS). Arch Gerontol Geriatr 49(1): 173-184.
- Lupien S, de Leon M, de Santi S (1998) Cortisol during human aging predict hippocampal atrophy and memory deficits. Nature Neuroscience 1(329): 69-73.
- Landfield PW, Thibault O, Mazzanti M (1992) Mechanisms of neuronal death in brain aging and alzheimer's disease: Role of endocrine-mediated calcium dyshomeostasis. Journal of Neurobiology 23(9): 1247-1260.
- Malenka RC, Nestler EJ, Hyman SE. 'Chapter 10: Neural and Neuroendocrine Control of the Internal Milieu'. In Sydor A, Brown RY. Molecular Neuropharmacology: A Foundation for Clinical Neuroscience. 2nd ed. 2009, New York: McGraw-Hill Medical. pp. 246, 248-259.
- Le Blanc ES, Janowsky J, Chan BKS, Nelson HD (2003) Hormone replacement therapy and cognition: Systematic review and metaanalysis. JAMA 285(11): 1489-1499.
- Tenover JL. Should there be hormone replacement therapy for aging men? Handbook of Andrology.
- Huppert FA, Van Niekerk JK (2001) Dehydroepiandrosterone (DHEA) supplementation for cognitive function. Cochrane Database Syst Rev 2: CD000304.






























