Coffee Diterpenes: before Harvesting the Bean to your Cup
Fábio Junior Moreira Novaes*
Universidade Federal do Rio de Janeiro, Brazil
Submission: May 25, 2018;Published: July 17, 2018
*Corresponding author: Novaes FJM, Universidade Federal do Rio de Janeiro, Instituto de Química, Laboratório de Análise de Aromas, Avenida Athos da Silveira Ramos, 149, Bloco A, Sala 626, Rio deJaneiro, RJ 21941-895, Brazil; Tel: +55 21 3938-7121; Email: fabiojmnovaes@yahoo.com.br
How to cite this article: Novaes FJM, Coffee Diterpenes: before Harvesting the Bean to your Cup. Nutri Food Sci Int J. 2018; 7(2): 555706. DOI:10.19080/NFSIJ.2018.07.555706.
- Mini Review
- Abstract
- Introduction
- Biosynthesis and Concentration of Cafestol and Kahweol (C&K) in Coffee
- Degradation of C&K due to green coffee bean storage and its roasting process
- C&K extraction in coffee beverage preparation and their absorption, metabolism and excretion
- Conclusion
- Acknowledgement
- References
Abstract
Cafestol and kahweol are molecules that exist only in coffee beans, even though they are also available for each consumer of the coffee beverage. Endogenous coffee enzymes and the absence of these molecules in other matrices confer them the title of taxonomic markers of plants of the genus Coffee. The trajectory of these molecules initiates with their biosynthesis and accumulation in the coffee fruit, passes through transformations during the storage and roasting of beans, reaches their transference to the beverage and ends when the human organism absorbs and metabolizes them. The present work deals with the chemical transformations imposed on these molecules during their trajectory in the coffee commercial cycle, as well as aspects related to human health.
Keywords: Coffee diterpenes; Human consumption; Metabolism; Biological activities; Storage; Roasting degradation
- Mini Review
- Abstract
- Introduction
- Biosynthesis and Concentration of Cafestol and Kahweol (C&K) in Coffee
- Degradation of C&K due to green coffee bean storage and its roasting process
- C&K extraction in coffee beverage preparation and their absorption, metabolism and excretion
- Conclusion
- Acknowledgement
- References
Introduction
Coffee is one of the most consumed beverages in contemporary society. About 80% of the world's adult population appreciates the beverage, which is usually marketed in the form of blends between Coffea arabica L. and C. canephora P. [1]. Due to the great popular appreciation, coffee has been the object of studies within the Food Sciences, some directly related to its chemical composition [2-4].
In the last three decades, studies related to the diterpenic composition of coffee bean gained attention. Two main factors contributed to this interest. The first was related to the representative presence of cafestol and kahweol (C&K) derivates (Figure 1) in the beans (≤2.5% w/wof its dry mass) and in its lipid fraction (≤20 % w/w) [5,6], and the second their anti-carcinogenic, antioxidant, anti-inflammatory and hypercholesterolemic activities [7-13].
- Mini Review
- Abstract
- Introduction
- Biosynthesis and Concentration of Cafestol and Kahweol (C&K) in Coffee
- Degradation of C&K due to green coffee bean storage and its roasting process
- C&K extraction in coffee beverage preparation and their absorption, metabolism and excretion
- Conclusion
- Acknowledgement
- References
Biosynthesis and Concentration of Cafestol and Kahweol (C&K) in Coffee
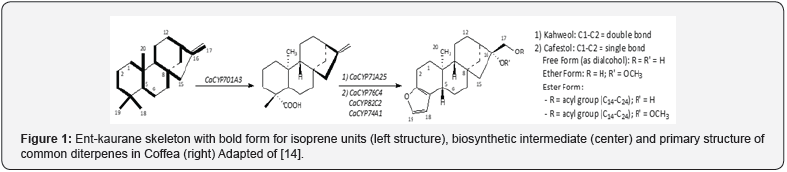
The coffee tree biogenetically modifies the ent-kaurane skeleton to produce C&K and distributes them throughout the plant as dialcohols (free form), ethers or esters (Figure 1) [14]. They are found in all the main tissues of the shrub (roots, branches, leaves and fruits), but mainly in coffee fruits, where their amount is dependent on the fruit stage of maturity [14]. Cafestol is the most common coffee diterpene in not only the major commercial species, C. arabica and C. robusta, but also in wild coffee [15]. The Arabica coffee beans have kahweol:cafestol proportions between 1:2 and 2:1, and this ratio is a coffee quality parameter for the beverage [2]. Kahweol may be absent in robusta coffee, or present in very low concentrations (≤0.01% w/w) [16]. Kahweol differs from cafestol by an unsaturation present in the ring A of the diterpene nucleus, between carbons 1 and 2 (Figure 1). The Robusta coffee has also ethers derived from C&K on carbon 16 (Figure 1) [5]. The presence and the ratio between Kahweol and 16-O-methyl-cafestol, when applied in a polynomial equation, can discriminate between different blend proportions of Arabica and Robusta beans in mixtures [17].
Commercial and wild coffee species have the esterified diterpenes in a proportion greater (250-fold) than their respective alcohols (Figure 1) [5-6]. However, according to SCOPUS database, there are few articles dealing with these compounds: only 16papers compared to 362 for the free form of the dialcohol [18]. This is probably due to the dispersion of C&K into 24 esters of different chain lengths and degrees of unsaturation (Figure 1), and the absence of effective techniques to obtain them individually [3,19-21]. For this reason, the research is concentrated on the free form of C&K after hydrolysis of their respective esters, simplifying their determination in coffee matrices and increasing the bulk of these diterpenes for further studies.
- Mini Review
- Abstract
- Introduction
- Biosynthesis and Concentration of Cafestol and Kahweol (C&K) in Coffee
- Degradation of C&K due to green coffee bean storage and its roasting process
- C&K extraction in coffee beverage preparation and their absorption, metabolism and excretion
- Conclusion
- Acknowledgement
- References
Degradation of C&K due to green coffee bean storage and its roasting process
When the green (or raw) coffee beans are stored, there is an increase in the content of both free form of C&K and fatty acids. Endogenous lipases in the beans hydrolyze the ester forms of these diterpenes (Figure 1) and the acylglycerol derivatives [22,23]. When the green bean is submitted to the roasting conditions (180-265 °C / 2-21min), C&K lose water generating several dehydrocafestol and dehydrokahweol, respectively, as well as the ether and aldehyde derivatives [22,24]. The amounts of both compounds increase proportionally with raising roasting temperatures or time of exposition. The cafestol/dehydrocafestol ratio is a good indicator of the coffee roasting degree [25]. Values below 15 for this ratio signal dark roasted coffee and values between 15 and 25 indicate medium roasting. Values above 25 describe a light-roasted coffee [25,26].
- Mini Review
- Abstract
- Introduction
- Biosynthesis and Concentration of Cafestol and Kahweol (C&K) in Coffee
- Degradation of C&K due to green coffee bean storage and its roasting process
- C&K extraction in coffee beverage preparation and their absorption, metabolism and excretion
- Conclusion
- Acknowledgement
- References
C&K extraction in coffee beverage preparation and their absorption, metabolism and excretion
The C&K extraction from roasted and grounded beans happens during the coffee beverage preparation. However, the final C&K concentration in the beverage is dependent on the kind of extraction procedure employed. The boiled coffee beverage - Scandinavian-type - reaches up to 1.766mg L-1 of C&K, whereas in beverages prepared with paper filter, C&K are widely retained, and their concentration reaches only 2.4mg L-1 after the percolation of these compounds through the paper [27,28].
When ingested by humans, about 70% of the total coffee diterpenes are absorbed in the intestinal tract, whereas 24% are degraded in the stomach and only 1% is excreted in the urine after conjugation with glucuronic acid and sulfate salts [15,29]. Results in rats indicate the presence of C&K esters in the body for 5 to 10 days after its administration, whereas the hydrolyzed forms persist for less than 48h [30]. These findings show that the hydrocarbon chain of the esterified coffee diterpenes increases their lipossolubility and the residence time in the tissues, which, therefore, increases their active period in the organism. Therefore, the investigations should be directed to the esterified forms, unless these esters are hydrolyzed in the human organism, releasing the free form.
- Mini Review
- Abstract
- Introduction
- Biosynthesis and Concentration of Cafestol and Kahweol (C&K) in Coffee
- Degradation of C&K due to green coffee bean storage and its roasting process
- C&K extraction in coffee beverage preparation and their absorption, metabolism and excretion
- Conclusion
- Acknowledgement
- References
Conclusion
The present work discussed the trajectory of coffee diterpenes - cafestol and kahweol - on the molecular level intheir environment/food/man relationship. It initiates with C&K biosynthesis in the plant, followed by their concentration in the coffee fruit, until their absorption in and elimination from the human organism. This multidisciplinary approach opens perspectives for several studies on these molecules and their derivatives along the coffee commercial path that ends with its consumption.
- Mini Review
- Abstract
- Introduction
- Biosynthesis and Concentration of Cafestol and Kahweol (C&K) in Coffee
- Degradation of C&K due to green coffee bean storage and its roasting process
- C&K extraction in coffee beverage preparation and their absorption, metabolism and excretion
- Conclusion
- Acknowledgement
- References
Acknowledgment
The author thanks CAPES, CNPq and FAPERJ agencies for funding and the editorial staff of NFRIJ for the invitation to write this article. The author also thanks Dr. Claudia Moraes de Rezende and Dr. Ademario Iris da Silva Junior for their kind scientific and English revision of the manuscript.
- Mini Review
- Abstract
- Introduction
- Biosynthesis and Concentration of Cafestol and Kahweol (C&K) in Coffee
- Degradation of C&K due to green coffee bean storage and its roasting process
- C&K extraction in coffee beverage preparation and their absorption, metabolism and excretion
- Conclusion
- Acknowledgement
- References
References
- Siridevi V, Giridhar P, Ravishankar GA (2011) Evaluation of roasting and brewing effect on antinutritional diterpenes - cafestol and kahweol in coffee. Glob JMed Res 11(5): 1-7.
- Novaes FJM, Oigman SS, Souza ROMA, Rezende CM, Aquino Neto FR (2015) New approaches on the analyses of thermolabile coffee diterpenes by gas chromatography and its relationship with cup quality. Talanta 139: 159-166.
- Novaes, FJM, Kulsing C, Bizzo, HR, Aquino Neto FR, Rezende CM, et al. (2018) Analysis of underivatised low volatility compounds by comprehensive two-dimensional gaschromatography with a short primary column. J Chromatogr A 1536: 75-81.
- Rosa JS, Freitas-Silva O, Rouws, JRC, Moreira IGS, Novaes FJM, et al. (2016) Mass spectrometry screening of Arabica coffee roasting: A nontarget and non-volatile approach by EASI-MS and ESI-MS. Food Res Int 89(2): 967-975.
- Kölling-Speer I, Strohscneider S, Speer K (1999) Determination of Free Diterpenes in Green and Roasted Coffees. J High Resol Chromatogr 22(1): 43-46.
- Kurzrock T, Speer K (2001) Diterpenes and Diterpene Esters in Coffee. Food Rev Int 17(4): 433-450.
- Urgert R, Essed N, Vand der Weg G, Kosmeijer-Schuil TG, Katan MB (1997) Separate effects of the coffee diterpenes cafestol and kahweol on serum lipids and liver aminotransferases. Am J Clin Nutr 65(2): 519-524.
- Lee KJ, Cho JH, Jeong HG (2007) Hepatoprotective and antioxidant effects of the coffee diterpenes kahweol and cafestol on carbon tetrachloride-induced liver damage in mice. Food Chem Toxicol 45(11): 2118-2125.
- Wang S, Yoon YC, Sung MJ, Park JH (2012) Antiangiogenic properties of cafestol, a coffee diterpene, in human umbilical vein endothelial cells. Biochem. Biophys Res Communic 421(3): 567-571.
- Cárdenas C, Quesada AR, Medina MA (2014) Insights on the antitumor effects of kahweol on human breast cancer: Decreased survival and increased production of reactive oxygen species and cytotoxicity. Biochem. Biophys Res Communic 447(3): 452-458.
- Cárdenas C, Quesada AR, Medina MA (2015) Chapter 70 - Kahweol, a coffee diterpene with anti-inflammatory properties. In: Preedy V (Ed), Coffee in Health and Disease Prevention (1st Edn), Academic Press, 32 Jamestown Road, London NW1 7BY, UK, pp. 627-633.
- Sakai E, Tsukuba T (2015) Chapter 96 - Coffee and bone metabolism: kahweol and osteoclastonegesis. In: Preedy V (Ed), Coffee in Health and Disease Prevention (1st Edn), Academic Press, 32 Jamestown Road, London NW1 7BY, UK, pp. 869-875.
- Guzzo LS, Romero TRL, Queiroz-Junior CM, Caliari MV, Azevedo AO, et al. (2015) Involvement of endogenous opioid peptides in the peripheral antinociceptive effect induced by the coffee specific diterpene kahweol. Pharmacol Rep 67(5): 1010-1015.
- Ivamoto ST, Sakuray LM, Ferreira LP, Kitzberger CSG, Scholz MBS, et al. (2017) Diterpenes biochemical profile and transcriptional analysis of cytochrome P450s genes in leaves, roots, flowers, and during Coffea arabica L. fruit development. Plant Physiol Biochem 111: 340-347.
- Roos B, Van der Weg G, Urgert R, Van de Bovenkamp P, Charrier A, et al. (1997) Levels of Cafestol Kahweol, and Related Diterpenoids in Wild Species of the Coffee Plant Coffea. J Agric Food Chem 45(8): 3065-3069.
- Mori ALB, Kalschne DL, Ferrão MAG, Fonseca AFA, Ferrão RG, et al. (2016) Diterpenes in Coffea canephora. J Food Compos Anal 52: 52-57.
- Pacetti D, Boselli E, Balzano M, Frega NG (2012) Authentication of Italian Espresso coffee blends through the GC peak ratio between kahweol and 16-O-methylcafestol. Food Chem 135(3): 1569-1574.
- www.scopus.com (2018).
- Pettit BC (1987) Identification of the diterpene esters in arabica and canephora coffees. J Agric Food Chem 35(4): 549-551.
- Speer K, Buchmann S, Kölling-Speer I (2012) TLC screening for the detection of Robusta admixtures to Arabica coffee. CAMAG Bibliography Service: Planar Chromatogr 109: 2-4.
- Moeenfard M, Erny GL, Alves A (2016) Variability of some diterpene esters in coffee beverages as influenced by brewing procedures. J Food Sci Technol 53(11): 3916-3927.
- Speer K, Kölling-Speer I (2006) The lipid fraction of the coffee bean. Braz J Plant Physiol 18(1): 201-216.
- Toci AT, Neto VJMF, Torres AG, Farah A (2013) Changes in triacylglycerols and free fatty acids composition during storage of roasted coffee. LWT - Food Sci Technol 50(2): 581-590.
- Guerrero G, Suárez M, Moreno G (2004) Chemosystematic study of diterpenoids in green coffee beans. 20th International Scientific Colloquium on Coffee, India.
- Kölling-Speer I, Kurt A, Nguyen T, Speer K (1997) Cafestol and dehydrocafestol in roasted coffee. 17th International Scientific Colloquiumon Coffee, Kenya.
- Dias RCE, Faria-Machado AF, Mercadante AZ, Bragagnolo N, Benassi MT (2014) Roasting process affects the profile of diterpenes in coffee. Eur Food Res Technol 239(6): 961-970.
- Erny GL, Moeenfard M, Alves A (2015) Liquid chromatography with diode array detection combined with spectral deconvolution for the analysis of some diterpene esters in arabica coffee brew. J Sep Sci 38(4): 612-620.
- Rendón MY, Scholz MBS, Bragagnolo N (2018) Physical characteristics of the paper filter and low cafestol content filter coffee brews. Food Res Int 108: 280-285.
- Van Cruchten STJ, de Waart DR, Kunne C, Hooiveld GJEJ, Boekschoten MV, et al. (2010) Abosrption, distribution, and biliary excretion of cafestol, a potent cholesterol-elevating compound in unfiltered coffees in mice. Drug Metab Dispos 38(4): 635-640.
- Huber WW, Teitel CH, Coles BF, King RS, Wiese FW, et al. (2004) Potential chemoprotective effects of the coffee components kahweol and cafestol palmitates via modification of hepatic N-acetyltransferase and glutathione S-transferase activities. Environ Mol Mutagen 44(4) 265-276.






























