Asparagus Saponins: Chemical Characterization, Bioavailability and Intervention in Human Health
Amel Hamdi1,2*, Ana Jiménez-Araujo1, Rocío Rodríguez-Arcos1, Sara Jaramillo-Carmona1, Mokhtar Lachaal2 Najoua Karray Bouraoui2, Rafael Guillén-Bejarano1
1Phytochemicals and Food Quality Group, Instituto de la Grasa (CSIC), Spain
2Unité de Physiologie et de Biochimie de la réponse des plantes aux contraintes abiotiques, Tunisie
Submission: June 19, 2018;Published: July 10, 2018
*Corresponding author: Rafael Guillén-Bejarano, Phytochemicals and Food Quality Group, Instituto de la Grasa (CSIC), 41013 Seville, Spain, Tel: 954611550; Fax: 954616790; Email: rguillen@ig.csic.es
How to cite this article: Hamdi A, Jiménez-Araujo A, Rodríguez-Arcos R, Jaramillo-Carmona S, Lachaal M, et al. Asparagus Saponins: Chemical 002 Characterization, Bioavailability and Intervention in Human Health. Nutri Food Sci Int J. 2018; 7(1): 555704. DOI:10.19080/NFSIJ.2018.07.555704.
Abstract
In the last years, there has been increasing interest in the research on saponins from food sources. Many epidemiological studies have pointed out their versatile health benefits. Since saponins are directly associated with human dietary ingredients and health, there is a need to evaluate their structure and function relationship. Asparagus spears are one of the main dietary sources of steroidal saponins.Most recent researches have focused on the health aspects of asparagus saponins for humans. Many asparagus saponins have cytotoxic and antitumor activities together with cholesterol lowering effect. This review highlights the structural features of saponins in asparagus, their beneficial roles in human health, and as well as their bioavailability and role on diet.
Keywords: Saponins; Asparagus; Biological activity; Bioavailability; Human health; Hypocholesterolemic; Monodesmosidic; Bioactive fiber; Sterols
Introduction
Asparagus is a perennial and monocotyledonous member of the Liliaceae family. It is grown for its edible stems (spears) which are consumed as a seasonal vegetable and are highly appreciated for their delicious taste, low energy content, and nutritional quality.
Now-a-days, there is an increasing scientific interest in studying the health benefits of asparagus not only for their nutritional properties, but also for their richness in bioactive compounds such as phenols, flavonoids, saponins, bioactive fiber, and sterols [1]. Saponins play an important role on the organoleptic and functional properties of asparagus. Some studies have shown their hypocholesterolemic effect in both experimental animals and humans and are considered as potential nutritional supplements in the control of dyslipidemias and obesity [2,3]. Steroidal saponins from different varieties of asparagus showed cytotoxic and anti-tumor activities in different human cell lines [4,5]. However, the saponins present in edible plants of the genus Asparagus are not only responsible for the biological activity but can also contribute to the asparagus flavor; recently, saponins have been identified as the molecules responsible for the bitter taste of white asparagus[6,7].
Chemical Structure and Classification
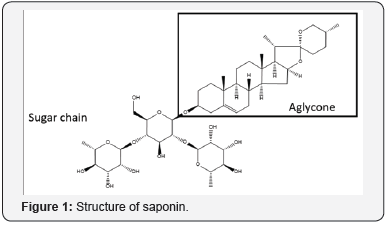
Saponins are glycosylated compounds composed of two main parts: a water-soluble glycidic chain and a liposoluble structure (Figure 1). The non-sugar and sugar components are called aglycone and glycone, respectively. The aglycone portion is composed by a triterpenoid or steroidal backbone. L-arabinose, D- xylose, D-glucose, D-glucuronic acid, D-galactose, L-rhamnose and D-fructose are among the sugars constituents of saponins. The sugar moiety is linked to the aglycone through a covalent linkage at one or two glycosylation sites; in the first case the saponins are called monodesmosidic and in the later bidesmosidic. In the monodesmosidic saponins the oligosaccharide chain is generally attached at the C3 position, while bidesmosidic saponins have an additional sugar moiety at the C26 or C28 position. The aglycone may contain one or more unsaturated C-C bonds, and depending on their nature saponins are classified in two main groups (Figure 2):
1.- Steroidal saponins, whose aglycone is steroidal, most of which are present in monocolyledonous angiosperms. The aglycone can be of the spirostanoid (Figure 2A) or furostanoid types (Figure 2B).
2.- Triterpene saponins, whose aglycone is terpene (Figure 2C), most of which are present in dicotyledonous angiosperms.

Saponins from the genus Asparagus belong to the group of steroidal glycosides [8]. In A. officinalis L., and most of the green and white commercial hybrids, basically derived from this specie, the main and almost only saponin is protodioscin (C51H84O22), which is a glycoside derivative of diosgenin of the furostanoid type [9,11]. Beside commercial hybrids, in some markets, it is found the so-called “triguero” asparagus which grows either wild or cultivated from species other than A. officinalis. We found that “triguero” asparagus from Huétor-Tájar, a place from Granada in Spain, has a distinct saponin profile compared to commercial hybrids [10,12]. The results revealed that, although commercial hybrids contain protodioscin as the major saponin, “triguero” from Huétor- Tájar presents a more complex profile. This consists on a combination of protodioscin and at least 12 different new saponins derived from a furostanol-type steroidal genin with a single bond between C5 and C6 of the B ring. The same kind of saponins have been found in different wild species while only protodioscin was found in wild A.officinallis[10,12-13]. The chemical structure of all of these saponins was classified by their retention time, molecular weight and fragmentation pathway, and as well as the co-injection with authentic reference saponins previously purified and identified from Huétor- Tájar “triguero” [10,12]. The fragmentation pathway has been studied through the mass spectra obtained in negative (100V-) and positive (50V+) modes (Table 1).
Changes in the structure of saponins can cause functional alterations. Mimaki and co-workers [14,15] have systematically examined the cytotoxic activities of the steroidal saponins mainly isolated from the Liliaceae plants, against HL-60 human promyelocytic leukemia cells and found several structures-activity relationships. Some steroidal saponins evaluated in the assay system showed considerable cytotoxic activities, which were almost as potent as that of etoposide used as a positive control. The activities were found to be sensitive to the monosaccharides constituting the sugar moieties and their sequences, as well as to the structures of the aglycones. In that study, they revealed that in the diosgenylα- l-rhamnopyranosyl-(1 → 2)-β-d- glucopyranoside derivative, which is cytotoxic, the diglycoside exists in a vertically oriented conformation against the steroid plane.However,in the diosgenylα-l-rhamnopyranosyl-(1→4)-β-d-glucopyranoside derivative, which presents a conformation with the diglycoside and the steroid in the same plane, the activity was not relevant. According to our previous studies [10,12,13], the different saponins identified from wild and cultivated asparagus spears (Table 1), have different substituents in the sapogenin as well as differences in the composition, linkage and number of sugar chains. This significant structural variation suggests that they might also have different bioactivities.

Saponins in the Diet
Saponins are present in more than 100 plant families and in some marine sources [16]. As previously, mentioned, steroidal saponins are distributed in monocotyledonous plants (Agavaceae, Dioscoreaceae, Liliaceae, Asparagaceae) and triterpenes are mainly found in dicotyledons (Leguminosae, Araliaceae, Caryophyllaceae). Most of the saponins present in the diet are of the triterpene type; in fact, the main sources are the legumes (soya, chickpeas, beans, beans, lentils, etc). In addition to legumes, pumpkin, licorice, beetroot, spinach and tea are also sources of triterpenic saponins. On the other hand, steroidal saponins are found in some foods such as asparagus, tomato, yucca, garlic or leek. In the case of cultivated white and green asparagus (A. officinalis), it has been reported that the main saponin they contain is protodioscin at concentrations of 1.4 to 5mg/100g fresh weight [11] and 0.024 -2.5mg/100g fresh weight [9], depending on the variety and the part of the shoot.
Although A. officinalis L. is the only cultivated asparagus nowadays, other wild species are also edible and traditionally consumed such as A. Pseudoscaber Grecescu, A. matitimus (L.) Mill., A. brachiphyllus Turcz., A. prostratus Dumort and A. officinalis L. [13]. In addition, both in Spain and in other countries of the Mediterranean area, three climbing plants of wild origin have been traditionally consumed under the name of "asparagus" (Bryonia dioica Jacq., Tamus communis L. and Humulus lupulus L.), due to their great resemblance to asparagus in terms of its phenotypic and organoleptic characteristics [17]. In the case of T. communis, its saponins are of the steroidal type derived from diosgenin, as in the case of most asparagus. In contrast, B. dioica and H. lupulus contain triterpene saponins. As they occur in the other wild asparagus, the concentrations of saponins in these three edible climbing plants are high [18] and vary depending on the part of the plant that is ingested [19].
It is important to mention that saponins can also be administered as food additives in order to improve the stability of the food itself or to give concrete effect at the functional level. These additives come from other foods that contain saponins naturally. Examples are Mohave yucca (Yucca schidigera Roezl Fla) and quillaja (Quillajasaponaria Mol Fla), which have been classified as food additives in the United States under section 172.50 (Natural Flavoring Substances and Natural Substances Used in Conjuction with Flavors) [20].
Bioavailability of Saponins
Scientific studies on the absorption, metabolism and pharmacokinetics of saponins have shown that saponins are not practically absorbed after oral administration, because they are mainly eliminated by faeces[4]. It is known that saponins have a low intestinal absorption mainly due to unfavorable physicochemical characteristics. In the understanding of absorption of saponins, it is important to remark that sapogenins have shown improved chemical properties compared to their precursor saponin, that enhance their permeability, such as a lower molecular weight, higher lipophilicity or lower molecular flexibility [21,22].
However, the aqueous solubility in the intestinal media is worst for sapogenins due to the lack of a hydrophilic sugar chain, which limits their bioaccessibility and therefore their bioavailability [22,23]. Liu et al. [24] effectively showed that the solubility of saponins significantly decreased for compounds with fewer or no sugar moieties attached. As example, glycyrrhetic acid, the sapogenin from glycyrrhizin of licorice was found in plasma after oral digestion of this saponin, whereas glycyrrhizin was barely detectable [25]. Kamo et al. [26] also described a higher bioavailability of soyasapogenols, the sapogenin of soyasaponins from soya in rats, compared to the bioavailability of soyasaponins. Additionally, these authors also found that the bioavailability was different depending on the type of saponin and sapogenin. Furthermore, due to the great variability on the chemical structure of saponins and sapogenins, the reported values of bioavailability are diverse. Studies on the bioavailability of steroidal saponins, such as those found in asparagus, have shown that saponins, such as dioscin and protodioscin, have a very low absorption, reaching values of bioavailability of 0.2%. However, they were found to have a very high average life time (120hours) [27,28]. Similarly, He et al. [29] observed that the absorption of methylprotodioscin was greater than dioscin. These effects are similar to those described for triterpene saponins [30]. Additionally, considering the reviews performed by Gao et al. [22] about the bioavailability of popular saponins in different animals, some of the observed values were as low as 0.1% for saponins from ginseng (ginsenosides) in rats, or 3% in dogs, and as high as 90% for the sapogenin from licorice (glycyrrhetic acid), also in rats. Moreover, due to the poor small intestinal absorption, saponins reach the colonic tract, suggesting that the microbiota is the main responsible for their hydrolysis and production of sapogenins[31].
It is important to remark that the events that take place during digestion are highly related to the bioactivities of saponins. This is because most studies have suggested that saponins are poorly absorbed, therefore their residence time within the gastrointestinal lumen causes important bioactive events. Additionally, some studies have shown interesting inhibitory activities of saponins of digestive enzymes such as lipase, amylase or glucosidase enzymes, which are effects related to their final bioactivities [32].
The Important Biological Activities of Asparagus Saponins
Hypocholesterolemic action
One of the main functions that have been attributed to asparagus saponins for years is its hypolipidaemic effect because diets rich in saponins have been shown to lower cholesterol levels, improving the lipid profile [3,5]. Moreover, it has been shown that the presence of steroidal saponins in the asparagus is able to improve the lipid profile by decreasing the levels of total cholesterol, LDL and triglycerides [2,33-34]. These authors justify the hypolipidaemic effect by decreasing the absorption and synthesis of cholesterol.
Cytotoxic and antitumor action
Numerous studies have shown the cytotoxic and antitumor properties of steroidal saponins present in asparagus edible part [4,35-36]. We have shown [4] that steroidal saponins from edible spears of “triguero” asparagus from Huétor-Tájar, when in contact with human colon cancer HCT-116 cells block ERK, AKT, and p70S6 (mTOR) signaling pathways, arrest the cell cycle at G0/G1 phase by interfering the expression of cyclins D, E, and A, and induce cell death through the apoptotic pathway via caspase-3, leading to PARP-1 cleavage and DNA fragmentation.
In addition, the cytotoxic capacity of saponins depends on their structural characteristics. Indeed, some authors [16,37] have shown that the structure of sugars in steroidal saponins plays a very important role in the cytotoxicity against certain tumors. Other studies have shown that the type of aglycone also influences the cytotoxic effect [38-40].
Antifungal action
Asparagus steroidal saponins have also antifungal activity; however, it seems that only spirostanol derivatives are active while furostanol derivatives are inactive [41]. The crude saponin fraction obtained from the bottom cut of A. officinalis L . h as s pecific a ctivity t o c ertain f ungi s uch as Candida, Cryptococcus, Trichophyton, Microsporum and Epidermophyton[42]. The mechanism of antifungal action of saponins is not well understood but it is believed that they complex with sterols in the cell membrane, leading to pore formation and consequent loss of membrane integrity.
Conclusion
Asparagus saponins are characterized by their structure containing a steroidal aglycone and one or more sugar units. Consumer’s demand for natural products, coupled with their physicochemical (surfactant) properties and biological activity (anticancer and anticholesterol activities), has led to the emergence of asparagus saponins as commercially significant compounds with expanding applications in food, cosmetics, and pharmaceutical sectors. The full realization of their commercial potential requires development of commercially feasible processes that can address processing challenges posed by their complex nature, including their stability. Information on the composition (qualitative and quantitative) and properties of the saponins present in asparagus, and the effects of processing on their composition and properties are key elements for successful process design. The abundance of saponins in asparagus spears and their presence in significant quantities in their processing and cultivation by-products result in a wide range of natural materials that can be exploited for saponin commercial production.
Acknowledgement
This work was supported by the Tunisian Ministry of Scientific Research and Technology and by the Ministerio de Ciencia e Innovación of Spain (AGL2017-82428-R).
References
- Lee JW, Lee JH, Yo IH, Gorinstein S, Bae JH, et al. (2014) Bioactive compounds, antioxidant and binding activities and spear yield of Asparagus officinalis L. Plant Foods for Human Nutrition 69: 175-181.
- García MD, De la Puerta R, Sáenz MT, Marquez-Martín A, Fernández- Arche MA (2012) Hypocholesterolemic and hepatoprotective effects of triguero asparagus from Andalusian in rats fed a high cholesterol diet. Evidence-Based Complementary and Alternative Medicine, pp. 6.
- Vázquez-Castilla S, De la Puerta R, García Giménez MD, Fernández- Arche MA, Guillén-Bejarano R (2013) Bioactive Constituents from “Triguero” Asparagus Improve the Plasma Lipid Profile and Liver Antioxidant Status in Hypercholesterolemic Rats. Int J Mol Sci 14(11): 21227-21239.
- Jaramillo S, Muriana FJG, Guillen R, Jiménez-Araujo A, Rodríguez-Arcos R, et al. (2016) Saponins from edible spears of wild asparagus inhibit AKT, p70S6K, and ERK signalling, and induce apoptosis through G0/ G1 cell cycle arrest in human colon cancer HCT-116 cells. Journal of Functional Foods 26: 1-10.
- Ji Y, Ji C, Yue L, Xu H (2012) Saponins isolated from Asparagus induce apoptosis in human hepatoma cell line HepG2 through a mitochondrialmediated pathway. Curr Oncol 19(Suppl 2): eS1-eS9.
- Dawid C, Hofmann T (2012) Identification of sensory-active phytochemicals in asparagus (Asparagus officinalis L.) J Agric Food Chem 60(48): 11877-11888.
- Dawid C, Hofmann T (2012 b) Structural and sensory characterization of bitter tasting steroidal saponins from asparagus spears (Asparagus officinalis L.) J Agric Food Chem 60 (48): 11889-11900.
- Scwarzbach A, Scheiner M, Knorr D (2006) Effect of cultivars and deep freeze storage on saponin content of white asparagus spears (Asparagus officinalis L.). European Food Research and Technology 222: 32-39.
- Wang M, Tadmor Y, Wu QL, Chin CK, Garrison SA, et al. (2003) Quantification of protodioscin and rutin in asparagus shoots by LC/MS and HPLC methods. J Agric Food Chem 51(21): 6132- 6136.
- Vázquez-Castilla S, Jaramillo-Carmona S, Fuentes-Alventosa JM, Jiménez-Araujo A, Rodríguez-Arcos R, et al. (2013) Saponin profile of green asparagus genotypes. J Agric Food Chem 61(46): 11098-11108.
- Lee W, Yoo K, Patil B (2010) Development of a rapid HPLC-UV method for simultaneous quantification of protodioscin and rutin in white and green asparagus spears. J Food Sci 75(9): C703-C709.
- Vázquez-Castilla S, Jaramillo-Carmona S, Fuentes-Alventosa JM, Jiménez-Araujo A, Rodrı́guez-Arcos R, et al. (2013) Optimization of a method for the profiling and quantification of saponins in different green asparagus genotypes. J Agric Food Chem 61(26): 6250-6258.
- Jaramillo-Carmona S, Rodriguez-Arcos R, Jiménez-Araujo A, López S, Gil J, et al. (2017) Saponin Profile of Wild Asparagus Species. J Food Sci 82(3): 638-664.
- Mimaki Y, Yokosuka A, Kuroda M, Sashida Y (2001) Cytotoxic Activities and Structure-Cytotoxic Relationships of Steroidal Saponins. Biol Pharm Bull 24(11): 1286-1289.
- Hernández JC, Leon F, Brouard I, Torres F, Rubio S, et al. (2008) Synthesis of novel spirostanic saponins and their cytotoxic activity. Bioorg Med Chem 16(4): 2063- 2076.
- Sparg SG, Light ME, Van Staden J (2004) Biological activities and distribution of plant saponins. J Ethnopharmacol 94(2-3): 219-243.
- Hadjichambis AC, Paraskeva-Hadjichambi D, Della A, Giusti ME, De Pasquale C, et al. (2008) Wild and semi-domesticated food plant consumption in seven circum-Mediterranean areas. Int J Food Sci Nutr 59(5): 383-414.
- Hadad C, Moradi Z (2005) The amounts and distribution of diosgenin and saponin and their carbohydrate moiety of Tamus communis L. Journal of Agricultural Science and Technology 12(2): 55-66.
- García-Herrera P, Cortés M, Cámara Montaña C, Tardío J, Olmedilla- Alonso B (2014) Carotenoid content of wild edible Young shoots traditionally consumed in Spain (Asparagus acutifolius L., Humulus lupulus L., Bryonia dioica Jacq. and Tamus communis L.) J Sci Food Agric 94(9):1914-1916.
- Natural Flavoring Substances and Natural Substances Used in Conjuction with Flavors: US FDA (2003)
- Ramasamy S, Chin SP, Sukumaran SD, Buckle MJC, Kiew LV, et al. (2015) In silico and in vitro analysis of bacoside A aglycones and its derivatives as the constituents responsible for the cognitive effects of Bacopa monnieri. PLoS ONE 10(5): e0126565.
- Gao S, Basu S, Yang Z, Deb A, Hu M (2012) Bioavailability challenges associated with development of saponins as therapeutic and chemopreventive agents. Curr Drug Targets 13(14): 1885-1899.
- Furtado N, Pirson L, Edelberg H, Miranda LM, Loira-Pastoriza C, et al. (2017) Pentacyclic triterpene bioavailability: An overview of in vitro and in vivo studies. Molecules 22(3): 400.
- Liu H, Yang J, Du F, Gao X, Ma X, et al. (2009) Absorption and disposition of ginsenosides after oral administration of Panax notoginseng extract to rats. Drug Metab Dispos 37(12): 2290-2298.
- Wang HY, Qi LW, Wang CZ, Li P (2011) Bioactivity enhancement of herbal supplements by intestinal microbiota focusing on ginsenosides. Am J Chin Med 39(6): 1103-1115.
- Kamo S, Suzuki S, Sato T (2014) Comparison of bioavailability (I) between soyasaponins and soyasapogenols, and (II) between group A and B soyasaponins. Nutrition 30(5): 596-601.
- Li K, Tang Y, Fawcett JP, Gu J, Zhong D (2005) Characterization of the pharmacokinetics of dioscin in rat. Steroids 70(8): 525-530.
- Ren H, Chen JP, Tan BY, Wang DM, Lin SH, et al. (2008) The disposition and pharmacokinetics of Dioscorea nipponica Makino extracts in rats. African Journal of Biotechnology 7(22): 4015-4018.
- He X, Qiao A, Wang X, Liu B, Jiang M, et al. (2006) Structural identification of methyl protodioscin metabolites in rats´ urine and their antiproliferative activities against human tumor cell lines. Steroids 71(9): 828-833.
- Liang MJ, Zhang WD, Zhang C, Liu RH, Shen YH, et al. (2007) Quantitative determination of the anticancer agent tubeimoside I in rat plasma by liquid chromatography coupled with mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 845(1): 84-89.
- Dong WW, Xuan FL, Zhong FL, Jiang J, Wu S, et al. (2017) Comparative analysis of the rats’ gut microbiota composition in animals with different ginsenosides metabolizing activity. J Agric Food Chem 65(2): 327-337.
- Ercan P, El SN (2016) Inhibitory effects of chickpea and Tribulus terrestris on lipase, α-amylase and α-glucosidase. Food Chem 205: 163-169.
- Zhu X, Zhang W, Zhao J, Wang J, Qu W (2010) Hypolipidaemic and hepatoprotective effects of ethanolic and aqueous extracts from Asparagus officinalis L. by-products in mice fed a high-fat diet. J Sci Food Agric 90(7): 1129-1135.
- Zhu X, Zhang W, Zhao J, Wang J, Qu W, et al. (2011) Hypolipidaemic Effect of n-Butanol Extract from Asparagus officinalis L. in Mice fed a High-fat Diet. Phytotherapy Research 25(8): 1119-1124.
- Liu W, Huang XF, Qi Q, Dai QS, Yang L, et al. (2009) Asparanin A induces G2/M cell cycle arrest and apoptosis in human hepatocellular carcinoma HepG2 cells. Biochemical and Biophysical Research Communications 381(4): 700-705.
- Shao Y, Chin CK, Ho CT, Ma W, Garrison SA, et al. (1996) Anti-tumor activity of the crude saponins obtained from asparagus. Cancer Lett 104(1): 31-36.






























