Computer Simulation in Pharmacokinetic and Pharmacodynamic Studies
Durgawati Patel1 and Puspendra Kumar2*
1Department of Chemistry, Banasthali University, India
2KIET School of Pharmacy, KIET Group of Institutions, India
Submission: August 21, 2017; Published: September 22, 2017
*Corresponding author: Puspendra Kumar, KIET Group of Institutions, India, Email: puspendrapatel9@gmail.com
How to cite this article: Durgawati Patel, Puspendra Kumar. Computer Simulation in Pharmacokinetic and Pharmacodynamic Studies. Mod Appl Bioequiv Availab. 2017; 2(2): 555582 DOI: 10.19080/MABB.2017.02.555582
Abstract
Computer simulation in the field of Pharmacokinetic and Pharmacodynamics or in silica model is need of the hour in the biomedical field. In silica studies helps the research with ease and effectiveness. The current review summarises the various computer simulation models for different drugs with their outcomes. The current field is still under developed and needs more number of researches to achieve the goals.
Keywords: Computer simulation; Pharmacokinetic; Pharmacodynamics; Monte carlo simulation; In-silico studies
Mini Review
Current scenario is based on rapid development of technologies and computer simulation is an integral part in the field of pharmacokinetic and pharmacodynamics studies. It helps to rapid development of dosage forms with cheaper price and by using less manpower [1]. Medical field is still slow in accepting the computer simulation models. Simulation can play a major role for selection of studies to be performed; clinical trial simulation covers many disciplines i.e. pharmacokinetics, pre clinical pharmacologist, statistician, computer programmer etc. So, all experts can discuss and precede the appropriate research [2]. Understanding of the aim and objectives of the work is very essential for all experts and it's a tedious job; and the main reason behind the less development of the simulation technique in the field of medical field [3].
Computer simulation methods are based on availability of literature and studies regarding pharmacokinetic and pharmacodynamics parameters of the selected drugs [4]. Success of computer simulations methods are depended on quality of data inputs available [5]. Previous studies are taken as a reference to predict the simulation; and computer simulations demonstrate the pharmacokinetic parameters (i.e. half lives) of different drugs [6]. Computer simulations can give atomic details which are not accessible from experiments and help to elucidate the mechanism of the passive permeation process at a molecular level [7].
Current Development in Computer Simulation
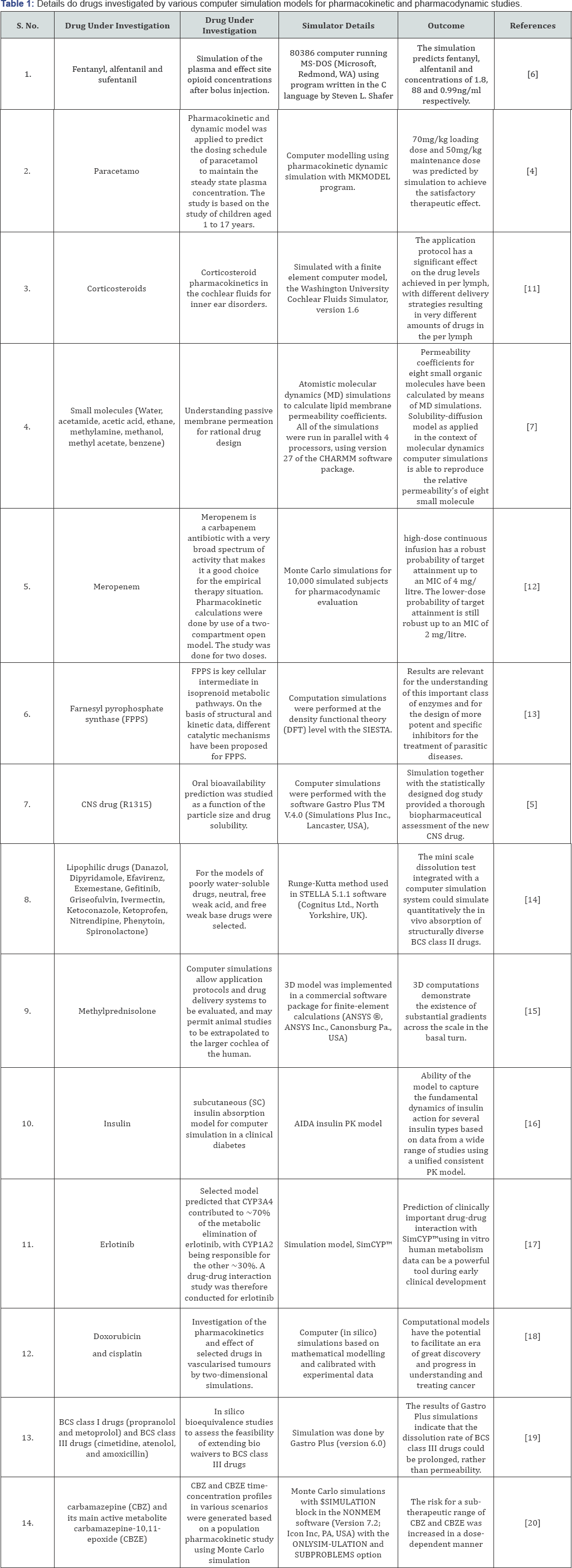
All the medicines should undergo to a development and assessment processes before launching to the market. Safety of the patient is main concern for everyone especially in the case of drugs; so scrutiny and rigorous testing of the product before commercialization is highly essential. Some products fall into the potential harmful substances and repeated preclinical and clinical studies are harmful for animals and humans; so computer simulation can fill the gap and gives better result. Currently computer simulation models are promoted to overcome all these issues, but still this area is under developed and requires more development (Table 1) [8-10].
Conclusion
More number of research and experts are required in the field of in silico computer simulation field to improve the product trials and replacement of animals and human during clinical trials. Subject specific model studies are recommended in which living models can be easily replaced by in silico models. The major challenge in the field of in silico pharmacokinetic and Pharmacodynamic studies is the harmony of understanding between the pharmacokinetics and computer programmer.
References
- Bonate PL (2000) Clinical trial simulation in drug development. Pharmaceutical research 17(3): 252-256.
- Bonate PL (2001) A brief introduction to Monte Carlo simulation. Clinical pharmacokinetics 40(1): 15-22.
- Johnson SC (1998) The role of simulation in the management of research: what can the pharmaceutical industry learn from the aerospace industry? Therapeutic Innovation & Regulatory Science.
- Anderson B, Holford N (1997) Rectal paracetamol dosing regimens: determination by computer simulation. Pediatric Anesthesia 7(6): 451-455.
- Kuentz M, Nick S, Parrott N, Rothlisberger D (2006) A strategy for preclinical formulation development using GastroPlus™ as pharmacokinetic simulation tool and a statistical screening design applied to a dog study. European journal of pharmaceutical sciences 27(1): 91-99.
- Shafer SL, Varvel JR (1991) Pharmacokinetics, pharmacodynamics, and rational opioid selection. Anesthesiology 74(1): 53-63.
- Bemporad D, Essex JW, Luttmann C (2004) Permeation of small molecules through a lipid bilayer: a computer simulation study. The Journal of Physical Chemistry B 108(15): 4875-4884.
- Viceconti M, Henney A, Morley-Fletcher E (2016) In silico clinical trials: how computer simulation will transform the biomedical industry. International Journal of Clinical Trials 3(2): 37-46.
- Fuchs A, Csajka C, homa Y, Buclin T, Widmer N (2013) Benchmarking therapeutic drug monitoring software: a review of available computer tools. Clinical pharmacokinetics 52(1): 9-22.
- Chabaud S, Girard P, Nony P, Boissel JP (2002) Clinical trial simulation using therapeutic effect modeling: application to ivabradine efficacy in patients with angina pectoris. Journal of pharmacokinetics and pharmacodynamics 29(4): 339-363.
- Plontke SK, Salt AN (2003) Quantitative interpretation of corticosteroid pharmacokinetics in inner fluids using computer simulations. Hearing research 182(1-2): 34-42.
- Krueger WA, Bulitta J, Kinzig-Schippers M, Landersdorfer C, Holzgrabe U, et al. (2005) Evaluation by Monte Carlo simulation of the pharmacokinetics of two doses of meropenem administered intermittently or as a continuous infusion in healthy volunteers. Antimicrobial agents and chemotherapy 49(5): 1881-1889.
- Sanchez VM, Crespo A, Gutkind JS, Turjanski AG (2006) Investigation of the catalytic mechanism of farnesyl pyrophosphate synthase by computer simulation. The Journal of Physical Chemistry B 110(36): 18052-18057.
- Takano R, Sugano K, Higashida A, Hayashi Y, Machida M, et al. (2006) Oral absorption of poorly water-soluble drugs: computer simulation of fraction absorbed in humans from a mini scale dissolution test. Pharmaceutical research 23(6): 1144-1156.
- Plontke SK, Siedow N, Wegener R, Zenner HP, Salt AN (2007) Cochlear pharmacokinetics with local inner ear drug delivery using a three-dimensional finite-element computer model. Audiology and Neurotology 12(1): 37-48.
- Wong J, Chase JG, Hann CE, Shaw GM, Lotz TF, et al. (2008) A subcutaneous insulin pharmacokinetic model for computer simulation in a diabetes decision support role: validation and simulation. Journal of diabetes science and technology 2(4): 672-680.
- Rakhit A, Pantze MP, Fettner, Jones HM, Charoin JE, et al. (2008) The effects of CYP3A4 inhibition on erlotinib pharmacokinetics: computer- based simulation (SimCYP) predicts in vivo metabolic inhibition. European journal of clinical pharmacology 64(1): 31-41.
- Sinek JP, Sanga S, Zheng X, Frieboes HB, Ferrari M, et al. (2009) Predicting drug pharmacokinetics and effect in vascular zed tumors using computer simulation. Journal of mathematical biology 58(4-5): 485-510.
- Tsume Y, Amidon GL (2010) The biowaiver extension for BCS class III drugs: the effect of dissolution rate on the bioequivalence of BCS class III immediate-release drugs predicted by computer simulation. Molecular pharmaceutics 7(4): 1235-1243.
- Ding JJ, Zhang YJ, Jiao Z, Wang Y (2012) The effect of poor compliance on the pharmacokinetics of carbamazepine and its epoxide metabolite using Monte Carlo simulation. Acta Pharmacological Sinica 33(11): 1431-1440.






























