Echocardiographic anomalies in a chronic hemodialysis population in the center of Senegal
Sarr Ibrahima Lyra1*, Sakho Binta2, Ndiaye Ousmane3, Dieye Mamadou3 and Diop Ndeye Diama2
1Department of Nephrology, Military hospital at Ouakam, Senegal
2Department of Nephrology, Henrich Lubke hospital, Senegal
3Department of Cardiology, Henrich Lubke hospital, Senegal
Submission: April 06, 2024;Published: April 23, 2024
*Corresponding author: Ibrahima Lyra Sarr, Nephrologist, Military hospital at Ouakam, Dakar, Senegal, Email: militaire474@gmail.com
How to cite this article: Sarr Ibrahima Lyra*, Sakho Binta, Ndiaye Ousmane, Dieye Mamadou and Diop Ndeye Diama. Echocardiographic anomalies in a chronic hemodialysis population in the center of Senegal. JOJ Urology & Nephrology, 2024; 8(5): 555750. DOI:10.19080/JOJUN.2024.08.555750.
Abstract
Introduction: The aim of this study was to determine echocardiographic anomalies in chronic hemodialysis patients and to identify factors associated with their occurrence.
Patients and methods: This was an observational, cross-sectional, descriptive, and analytical study carried out in the nephrology and hemodialysis department of the Heinrich Lübke regional hospital in Diourbel from September 1 to October 18, 2023. Echocardiographic data as well as other sociodemographic data, history, dialysis parameters and biological results were collected.
Results: Thirty-three patients (20 women, mean age 49.64 ± 12.62 years) were included, with a mean duration of hemodialysis of 37.8 ± 29.3 months. Echocardiographic anomalies were dominated by left ventricular diastolic dysfunction in 90.9 % of patients and left ventricular hypertrophy (LVH) in 66.7 %. Pulmonary arterial hypertension (PAH) was noted in 60.6 % of patients, and valvular calcifications in 45.5 %. Kinetic disturbances (12.1 %) and systolic dysfunction (9.1 %) were the least common anomalies. Phosphatemia (p = 0.04) and phosphocalcium product (p = 0.04) were significantly higher in patients with valvular calcifications. Diameters of the inferior vena cava (p < 0.001) left atrium (p < 0.03), tele diastolic (p < 0.03) and tele systolic left ventricle (p < 0.003) were significantly higher in patients with PAH.
Conclusion: Echocardiographic anomalies are frequent and may be a source of morbimortality in chronic hemodialysis patients. It is therefore essential to screen these patients regularly for echocardiographic anomalies.
Keywords: Transthoracic echocardiography; Chronic hemodialysis patients; Cardiovascular complications; Diourbel; Adaptive mechanism
Abbreviations: LVH: Left Ventricular Hypertrophy; PAH: Pulmonary Arterial Hypertension; TTE: Transthoracic Echocardiography; AVF: Arteriovenous Fistula; AKI: Acute Kidney Injury; CKD: Chronic Kidney Disease; ADPKC: Autosomal Dominant Polycystic Kidney Disease; LV : Left ventricle; RA : Right auricle; RV : Right ventricle
Introduction
Chronic hemodialysis is associated with major cardiovascular complications, which are a significant cause of morbidity and mortality. Indeed, they are the leading cause of death [1,2] and a frequent reason for hospitalization [3] in chronic hemodialysis patients. These complications often appear well before the dialysis stage, and worsen the longer the patient is on dialysis, despite the spectacular therapeutic and technical advances of recent years. The high incidence of heart disease in chronic hemodialysis patients is due to several risk factors, which may be interrelated. These include arterial hypertension, anaemia, chronic inflammation, mineral and bone disorders, homocysteinemia and many others [4] that generate structural and/or functional cardiac abnormalities. Effective management of these risk factors could reduce cardiovascular morbidity and mortality. Cardiac abnormalities in chronic hemodialysis patients can affect all tunics of the heart. They are dominated by left ventricular hypertrophy (LVH) and left ventricular systolic or diastolic dysfunction, which, even if asymptomatic, are responsible for high mortality.
They are diagnosed by transthoracic echocardiography (TTE), a non-invasive and accessible test, which should be performed regularly in these patients to detect and treat these anomalies early. In Senegal, studies conducted by Cissé et al [5] Aw et al [6] in 2012 and 2020 respectively in Dakar and Ziguinchor on cardiovascular complications in chronic hemodialysis patients, had found LVH to be the main echocardiographic abnormality, with a prevalence of 71.05 % and 41.66 % respectively. The present study follows the same approach, evaluating echocardiographic anomalies in a chronic hemodialysis population and identifying the factors associated with their presence in a semi-urban area in the center of Senegal.
Patients and methods
This was an observational, cross-sectional, descriptive, and analytical study carried out in the nephrology and hemodialysis department of the Heinrich Lübke regional hospital in Diourbel from September 1 to October 18, 2023. All patients over 18 years of age, hemodialysis patients for more than 3 months and willing to participate were included in the study. Patients with an unstable clinical condition during the study period and those undergoing transient hemodialysis at the center were not included. All included patients had a TTE performed after the hemodialysis session by the same cardiologist. TTE was performed using an ultrasound scanner equipped with a pulsed and continuous color Doppler. Measurements were taken in the dorsal and left lateral decubitus positions. Echocardiographic data as well as other sociodemographic data, history and comorbidities, dialysis parameters and biological results were collected.
Left ventricular diastolic dysfunction is defined by an E’ wave < 8 cm/s septal or < 10 cm/s lateral. A distinction was then made between grade I (E/A < 0.8), grade II (E/A < 1.5) and grade III (E/A > 2). LVH was defined as left ventricular mass indexed to body surface area greater than 131 g/m2 in men and 100 g/m2 in women. Pulmonary arterial hypertension (PAH) was defined by a systolic pulmonary arterial pressure (SPAP) greater than 25 mm Hg. It was mild if PAPS [25-35[ mm Hg, moderate for PAPS between [35-45[ mm Hg and severe if PAPS > 45 mm Hg. Left ventricular systolic dysfunction was defined by a systolic ejection fraction < 55 %. Statistical analysis was performed using EPI-Info software. Quantitative variables were expressed as mean ± standard deviation and qualitative variables as percentage. Student’s t test and chi 2 test were used for comparison of quantitative and qualitative variables respectively. A p-value < 0.05 is considered significant.
Results
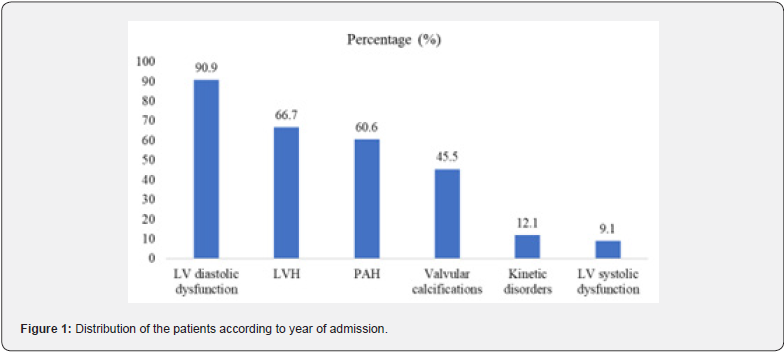
Thirty-three (33) patients were included with a mean age of 49.64 ± 12.62 years. There were twenty (20) women and thirteen (13) men, giving a sex ratio of 0.65. The mean duration of hemodialysis was 37.8 ± 29.3 months. Twenty-nine patients (87.6 %) were hypertensive, and five patients (15.2 %) were diabetic. Hypertensive nephropathy, noted in 15 patients (45.5 %), was the most frequent cause of end-stage renal failure, followed by nephropathy of undetermined etiology in 12 patients (36.4 %). All patients received 12 hours of dialysis per week, divided into three 4-hour sessions. Nineteen (19) patients (57.6 %) had an arteriovenous fistula (AVF) as their vascular access, of which 14 (73.7 %) were proximal AVFs. Mean inter-dialytic weight gain (IDWG) was 1.89 ± 1.05 kg and mean weekly Kt/v 1.67 ± 0.24. Biologically, mean hemoglobin was 7.40 ± 1.49 g/dl. Mean calcemia was 86.28 ± 8.66 mg/L. Other demographic, clinical and biological parameters of our population are shown in Table 1.
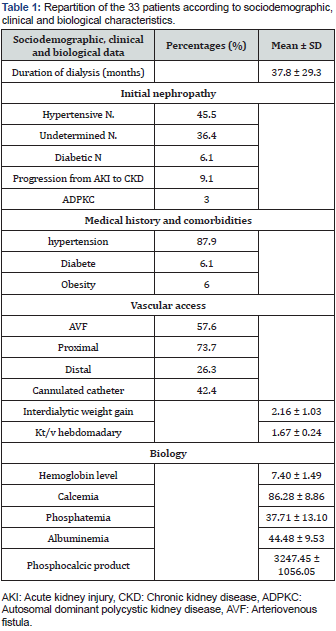
All patients had at least one anomaly on TTE. Echocardiographic anomalies were dominated by left ventricular diastolic dysfunction in 30 patients (90.9 %), with a predominance of type I diastolic dysfunction in 18 patients (60 %). Type II diastolic dysfunction was noted in 11 patients (36.7 %) and type III diastolic dysfunction in 1 patient (3.3 %). LVH was noted in twenty-two patients (66.7 %). It was concentric in eleven patients (50 %) and excentric in the remaining eleven. PAH was observed in twenty patients (60.6 %), of whom ten (50 %) had severe PAH. It was moderate in 5 patients (25 %) and mild in 5 patients (25 %). Valvular calcifications were observed in 15 patients (45.5 %), predominantly in the aortic region (60 %). In 40 % of cases, they were located on both the aortic and mitral valves at the same time. Kinetic disorders in 4 patients (12.1 %) and left ventricular systolic dysfunction in 3 patients (9.1 %) were the least common anomalies.
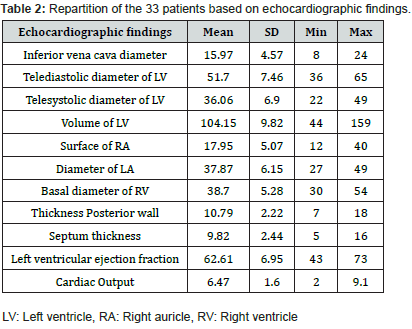
No patient had pericarditis. We found no factors associated with diastolic dysfunction or LVH. However, Phosphatemia (p = 0.04) and phosphocalcium product (p = 0.04) were significantly higher in patients with valvular calcifications. The diameters of the inferior vena cava (p < 0.001) left atrium (p < 0.03), telediastolic (p < 0.03) and tele systolic left ventricle (p < 0.003) were significantly higher in patients with PAH. The data and the various echocardiographic anomalies are shown in Table 2 and Figure 1 respectively.
Discussion
The mean age of patients was 49.64 ± 12.62 years, with a female predominance (60.6 %). Our results are like those reported by Cissé et al. and Maria et al. in 2022, with mean ages of 52 and 51.42 years respectively [5,7]. The young age of our patients may be explained by the ignorance of renal disease in rural areas, but also and above all using traditional medicine and therefore the important use of phytotherapy in rural areas.
We noted a high prevalence of echocardiographic anomalies. This can be explained on the one hand by the direct impact of chronic kidney disease on cardiac tissue, but also by the comorbidities that accompany this pathology.
In our study, left ventricular diastolic dysfunction (90.9 %) and LVH (66.7 %) were the most common echocardiographic anomalies. These results are in accordance with the literature. Indeed, LVH is always cited among the main anomalies observed on echocardiography of chronic hemodialysis patients [6-8]. It is strongly associated with left ventricular diastolic dysfunction [9]. These anomalies are the breeding ground for many cardiovascular pathologies such as heart failure, ischemic heart disease, rhythm disorders and even sudden death. The latter are often fatal, making cardiovascular pathologies the main cause of mortality in these patients [1,2]. The frequency of LVH in chronic hemodialysis patients can be explained by several risk factors specific to this population, such as hypertension, arteriovenous fistula, anemia, and fluid retention [10,11]. This is an adaptive mechanism of cardiac tissue to volume and pressure overload. Initially beneficial, it can later become maladaptive and deleterious due to cell death secondary to continuous ventricular overload, resulting in reduced capillary density and the development of myocardial fibrosis [12,13].
However, we did not find an association between LVH and diastolic dysfunction on statistical analysis (p = 0.25). This may be explained by the small size of our sample. PAH was noted in 60.6 % of patients. These results contradict those reported by Maria et al [7] and Aw et al [6], who noted a lower prevalence of 31 % and 20 % respectively. The pathophysiological mechanism of PAH in chronic hemodialysis patients is poorly elucidated. The prevalence of valvular calcifications in our study was 45.5 %. This is higher than those of Maria et al [7] and Anziani et al [8], who reported prevalences of 28.73 % and 14 % respectively in their studies. These calcifications are associated with the mineral and bone disorders of chronic kidney disease. Phosphatemia and phosphocalcium product were significantly higher in patients with valvular calcifications. In chronic renal disease in general, and in hemodialysis patients in particular, they represent a major complication in terms of mortality risk. The main limitation of our study was the small sample size. However, it has the particularity of being a cross-sectional study with data collected prospectively, unlike other publications on the subject where data is often collected retrospectively based on old echocardiography reports. In addition, the same cardiologist performed TTEs for all included patients.
Conclusion
In this study, we found a high frequency of cardiac anomalies in chronic hemodialysis patients in our setting. These anomalies are dominated by left ventricular diastolic dysfunction followed by LVH, which can be the cause of significant morbidity and mortality. Their regular detection by means of a periodic TTE scan enables early and appropriate management.
References
- Coulibaly M, Samaké M, Fofana AS, Coulibaly SB, Yattara H, et al. (2020) Déterminants de la mortalité chez les hémodialysés de l’Hôpital Mali Gavardo de Sébénikoro (Bamako). Health Sci Dis 21(6).
- Foley RN, Parfrey PS, Sarnak MJ (1998) Epidemiology of cardiovascular disease in chronic renal disease. J Am Soc Nephrol 9(12 Suppl): S16-S23.
- Adeniyi M, Kassam H, Agaba EI, Sun Y, Servilla KS, et al. (2009) Hospitalizations in patients treated sequentially by chronic hemodialysis and continuous peritoneal dialysis. Adv Perit Dial Conf Perit Dial 25: 72-75.
- Zoccali C, Benedetto FA, Mallamaci F, Tripepi G, Giacone G, et al. (2004) Left ventricular mass monitoring in the follow-up of dialysis patients: prognostic value of left ventricular hypertrophy progression. Kidney Int 65(4): 1492-1498.
- Cardiologie Pratique (2010) The hemodialysis patient: the role of the cardiologist.
- Moustapha CM, Tall LA, Maria F, Khodia F, Moustapha F, et al. (2016) Evaluation des complications cardiaques chez les hémodialysés chroniques de Dakar. Pan Afr Med J 23: 43.
- Faye M, Lemrabott AT, Ba B, Faye M, Boudal FE, et al. (2022) Anomalies électriques et échocardiographiques chez les patients hémodialysés Chroniques à Health Sci Dis 23(6).
- Ezziani M, Najdi A, Mikou S, Elhassani A, Akrichi MA, et al. (2014) Anomalies échocardiographiques chez l’hémodialysé chronique: prévalence et facteurs de risque. Pan Afr Med J 18: 216.
- Allibou S, Ouhammou W, Chettati M, Laouad I, Fadili W (2021) La dysfonction diastolique du ventricule gauche chez les hémodialysés chroniques: prévalence et facteurs de risque. Rev Med Int 42(2): A435.
- London G, Marchais S (1997) Conséquences cardiovasculaires de l'insuffisant rénal chronique. Encycl Méd chir - Néphrologie-urologie.
- Barberato SH, Pecoilts-Filho R (2010) Altérations échocardiographiques chez les patients atteints d'insuffisance rénale chronique subissant une hé Arq Bras Cardiol 94(1): 140-146.
- Londres GM, Pannier B, Guerin AP, Jacques B, Sylvain JM, et al. (2001) Altérations de l'hypertrophie ventriculaire gauche et survie des patients hémodialysés : suivi d'une étude interventionnelle. J Am Soc Néphrol 12(12): 2759-2767.
- BA AM, Kane Y, Manga S, Dieng A, BA B, et al. (2021) Évaluation des complications cardiovasculaires au centre d’hémodialyse de l’hôpital régional de Ziguinchor au sud du Séné J Marocain Sci Méd 23(1): 25-28.






























