- Review article
- Abstract
- Introduction
- Link between Type 2 Diabetes and Chronic Kidney Disease
- Prevention of Chronic Kidney Disease
- Patients
- Mineralocorticoid Receptor Antagonists
- Calcium Channel Antagonists
- Glucose Lowering Therapies
- Endothelin Antagonists
- Potential Future Therapeutic Options
- Control and Renal Function Preservation
- Evaluation of Clinical Outcomes
- Special Considerations and Challenges
- Conclusion
- References
Non-streptococcal Bacteria as the Leading Cause of Infection-related Glomerulonephritis in Adults
Laura Sofía Triviño Cuellar, Miguel Ángel Gutierrez Mejía, Ezequiel Jose Guevara Romero, Javier Aldair Ramírez Lovos, Valeria Di Stefano Perez, Karen E Reales Hernandez, María Isabel Murillo Pineda, Alejandra María Aleman Reyes, Andrea Gabriela Galecio Chao, and Maria Isabel Gomez*
1Fundación Universitaria Juan N. Corpas, Colombia
2Universidad de El Salvador, El Salvador
3Universidad de Ciencias Médicas de Centroamerica (UCIMED), Costa Rica
4Universidad Metropolitana Ciencias de la Salud, Colombia.
5Universidad católica de Honduras, Honduras
6Universidad de Guayaquil, Ecuador
7Universidad del Valle, México
Submission:September 21, 2023;Published:October 06, 2023
*Corresponding author: Maria Isabel Gomez, Department of Medicine, Universidad del Valle de, Mexico
How to cite this article: Laura Sofía Triviño C, Miguel Ángel Gutierrez M, Ezequiel Jose Guevara R, Javier Aldair Ramírez L, Valeria Di Stefano P, et al. Natera R, et al. Non-streptococcal Bacteria as the Leading Cause of Infection-related Glomerulonephritis in Adults. JOJ Urology & Nephrology, 2023; 8(3): 555736 DOI:10.19080/JOJUN.2023.08.555976.
Abstract
Infection-related glomerulonephritis (IRGN) is a kidney disorder characterized by inflammation of the glomeruli, the kidney's filtering units, triggered by infections elsewhere in the body. Recent data reveal a shift in the United States, with non-streptococcal bacteria emerging as significant causes of IRGN in adults, replacing the historical dominance of streptococcal infections. This condition arises when the body's immune response to an infection deposes immune complexes in the kidney's glomeruli, resulting in inflammation and damage. Factors like the type and virulence of the bacteria, host immune response, and genetic predisposition contribute to its pathogenesis. Non-streptococcal bacterial infections, including staphylococcal and Escherichia coli (E. coli), are now prominent contributors to adult IRGN. This shift in epidemiology may vary over time and by region. Patients with non-streptococcal bacterial IRGN commonly present with acute kidney injury symptoms, such as swelling, hypertension, hematuria, and proteinuria. Diagnosis involves clinical evaluation, laboratory tests, and kidney biopsy to reveal characteristic glomerular changes. Management focuses on treating the underlying infection with targeted antibiotics, controlling inflammation, and providing supportive care. Severe cases may require dialysis until kidney function recovers. This comprehensive narrative review aims to provide an up-to-date perspective on non-streptococcal bacteria as the leading cause of IRGN in adults. It covers epidemiology, etiology, clinical presentation, diagnosis, and treatment. The prognosis varies based on factors such as the underlying infection and promptness of intervention. This evolving landscape underscores the importance of early diagnosis and tailored management to enhance outcomes in changing IRGN epidemiology among adults. Staying vigilant in diagnosing and managing IRGN remains crucial as it presents new challenges and opportunities for improved patient care.
Keywords: Infection-related glomerulonephritis; Glomeruli; Non-streptococcal bacteria; Immune complexes; Glomerulonephritis; Nephritic syndrome
Abbreviations: IRGN: Infection-related glomerulonephritis; MRSA: Methicillin-resistant Staphylococcus aureus; MSS: Methicillin-sensitive Staphylococcus aureus; GN: Glomerulonephritis; SLE: Systemic Lupus Erythematosus; HSP: Henoch-Schönlein Purpura; MCD - Minimal Change Disease; FSGS: Focal Segmental Glomerulosclerosis; DDD: Dense Deposit Disease: GFR - Glomerular filtration rate; ACE inhibitors - Angiotensin-converting enzyme inhibitors; ARB: Angiotensin receptor blockers; NSAIDs - Non-steroidal anti-inflammatory drugs; GFR: Glomerular filtration rate; C3: Complement component 3; C4: Complement component 4; PSGN: Post-streptococcal glomerulonephritis; IgAN: IgA Nephropathy; APIGN - Acute Postinfectious Glomerulonephritis; SAGN: Staphylococcal-associated glomerulonephritis; LM: Light microscopy; IF: Immunofluorescence; EM: Electron microscopy
Introduction
Infection-related glomerulonephritis (IRGN) is a kidney disorder characterized by inflammation of the glomeruli, the filtering units of the kidneys, resulting from an infection elsewhere in the body. In adults, non-streptococcal bacteria have emerged as a significant cause of IRGN. This condition occurs when the immune system's response to the infection leads to the deposition of immune complexes in the kidney's glomeruli, causing inflammation and damage. In the United States, the epidemiology of non-streptococcal bacteria as the leading cause of IRGN in adults has steadily evolved. While streptococcal infections historically dominated the landscape of IRGN, recent data suggest a shift towards non-streptococcal bacterial infections, including staphylococcal and Escherichia coli (E. coli) infections, as significant contributors to this condition [1-3]. The exact prevalence may vary over time and by geographic region. The etiology of non-streptococcal bacterial IRGN lies in the body's immune response to bacterial antigens. In these infections, bacterial antigens or their immune complexes may circulate in the bloodstream and get trapped in the glomeruli. This triggers an inflammatory response, causing damage to the glomerular filtration barrier. Factors like the type and virulence of the bacteria host immune response and genetic predisposition play a role in the pathogenesis of this condition [2,4].
Patients with non-streptococcal bacterial IRGN typically present with signs and symptoms of acute kidney injury, such as swelling (edema), hypertension, hematuria (blood in the urine), and proteinuria (excess protein in the urine). They may also have symptoms related to the underlying infection, including fever, chills, and localized signs of infection. Diagnosing IRGN involves a combination of clinical evaluation, laboratory tests, and kidney biopsy. Urinalysis often reveals hematuria and proteinuria, while blood tests may show elevated serum creatinine levels, indicating kidney dysfunction. A definitive diagnosis is made through a kidney biopsy, demonstrating characteristic glomerular changes, including immune complex deposition [1,5,6]. Management of non-streptococcal bacterial IRGN focuses on treating the underlying infection and alleviating kidney inflammation. Antibiotics targeted at the specific causative bacteria are crucial for infection control. Additionally, supportive care may be necessary, including blood pressure management and diuretics to reduce edema. In severe cases where kidney function is significantly impaired, dialysis may be required until kidney function recovers [5]. This narrative review aims to provide a comprehensive overview of non-streptococcal bacteria as the leading cause of infection-related glomerulonephritis in adults. This review synthesizes current knowledge on the epidemiology, etiology/pathogenesis, clinical presentation, diagnosis, and treatment of this condition to offer a comprehensive and up-to-date perspective.
Epidemiology
The incidence of IRGN in children and adults in industrialized and developing countries has decreased over the past 5 decades. This trend is likely due to improvements in living conditions, early and effective use of antibiotics, and the increasing practice of water fluoridation, which attenuates the expression of Streptococcus pyogenes virulence factors. Based on a review of 11 population-based studies published between 1988 and 2000, Carapetis et al.8 estimated the incidence of IRGN in adults at 2 and 0.3 cases per 100,000 person-years in developing and developed countries, respectively, compared with 24.3 and 6 cases per 100,000 person-years in children [7].
The global burden of IRGN in adults was 68,000 per year. The true incidence is likely much higher as epidemiologic studies included only symptomatic patients, and it is known that subclinical IRGN is at least four times more common than clinically evident disease. Haas reported incidental ultrastructural evidence of largely healed or resolved IRGN in 10.5% of renal biopsies (including 22% of biopsies with diabetic nephropathy with no other immune-mediated glomerular disease). The renal biopsy incidence of adult IRGN in developed countries ranges from 0.6 to 4.6%,11-13 which is lower than that of adult IgA nephropathy, focal segmental glomerulosclerosis, membranous glomerulopathy, and membranoproliferative glomerulonephritis [7]. Furthermore, an increasing number of adult cases have been observed in alcoholics, diabetics, and intravenous drug abusers [8].
Etiology and Pathogenesis
Historically, post-infectious glomerulonephritis has been characterized as arising from streptococcal infections, especially after episodes of pharyngitis or skin infections, being more prevalent in the pediatric population [9]. However, the etiological landscape of the disease has evolved, especially in adults and the contexts of developed countries. While the Streptococcus and Staphylococcus genera remain the primary identified pathogens, there has been a particular surge in Staphylococcus, especially MRSA (Methicillin-resistant Staphylococcus aureus). Its emergence has led to this pathogen becoming as prevalent as Streptococcus in the etiology of glomerulonephritis in certain regions [10,11]. Furthermore, the etiological spectrum has diversified even more in recent years. The involvement of other bacteria, both Gram-positive and Gram-negative, in the disease has been evidenced. Examples of these microorganisms include Propionibacterium acnes, Neisseria gonorrhoeae, and Escherichia coli, among others, expanding the range of potential infectious causes associated [12]. An overview of most common bacteria involved in IRGN is depicted in Table 1. It's crucial to highlight that in the adult population, there are times when the underlying infection does not show evident clinical symptoms. In these cases, the infection is often discovered only after the diagnosis of glomerulonephritis, underscoring the importance of considering it in the differential diagnosis, even in the absence of apparent infectious symptoms [11].
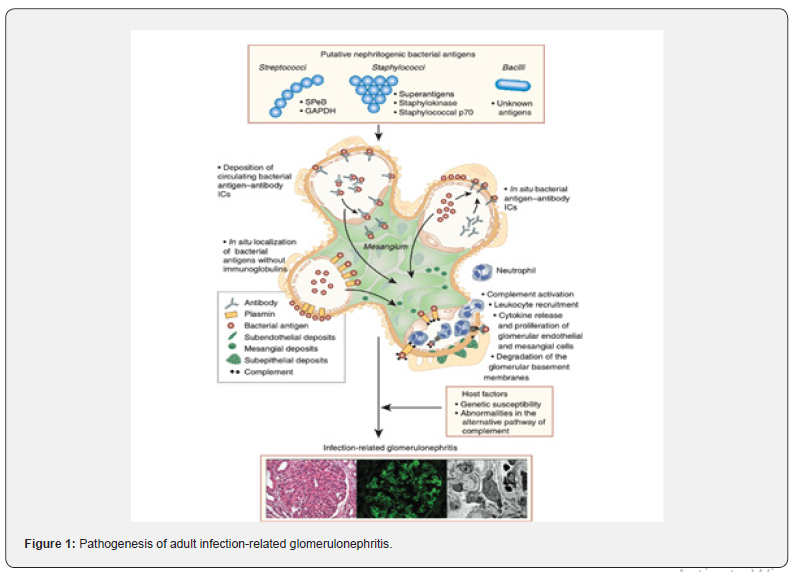
Infection-associated glomerulonephritis (GN) is believed to result from glomerular injuries mediated by the immune system following systemic infections. Although the exact mechanisms are not precise, it is suggested that bacterial proteins contribute to the formation of immune complexes in circulation that later deposit in the glomerulus, causing inflammation and capillary damage. This accumulation can activate complement pathways, release chemotactic factors, and produce other harmful immune mediators. Various mechanisms are proposed for forming these immune complexes in the kidney, such as molecular mimicry between bacterial antigens and glomerular proteins. An illustration of the pathogenesis of infection-related glomerulonephritis in adults can be found in Figure 1.
Fewer studies have been conducted on staphylococcal-associated glomerulonephritis (SAGN). It is suggested that staphylococcal enterotoxins act as superantigens that activate large subsets of T cells, causing a cytokine storm. It has also been proposed that SAGN might be related to the over-activation of neutrophils and the formation of neutrophil extracellular traps. However, not all patients with infections develop GN, indicating that factors like the host-pathogen interaction and individualized immune response are crucial. Lastly, certain genetic variations in the HLA region might influence susceptibility to infections and SAGN [12].
Clinical Features
Most often occurring cases had been reported in Caucasians, Asians, Hispanics, and Native Americans. In the United States, men are more likely than women to develop type 2 diabetes; nevertheless, drinking is the leading risk factor in Europe. Contrary to infantile streptococcal infections, non-streptococcal infections are more prevalent in adults. Among the most common clinical features, non-streptococcal infections can manifest hematuria, proteinuria, edema, hypertension, and impaired renal function, all symptoms of acute nephritic syndrome. Hypertension is prevalent in 60–84% of patients, and the risk rises with age. Due to water and sodium retention, the edema can be present in two-thirds of adult patients. From 1 to >3 g of protein per day, proteinuria might potentially result in nephrotic syndrome. The patients may have leukocyturia, microhematuria, or extensive hematuria.
When a patient first presents, their blood creatinine level is raised. However, it might be more significant in older patients than younger individuals and higher in those with a history of diabetic glomerulosclerosis. The low C3 and C4 complement can be restored to normal within two months of the presentation [13].
Diagnosis
No pathognomonic clinical or pathological manifestations exist for infection-associated glomerulonephritis [14]. However, in one study, at least 3 of the following were used as inclusion criteria: 1) clinical or laboratory evidence of infection preceding the onset of glomerulonephritis (GN); 2) depressed serum complement; 3) endocapillary proliferative and exudative GN on light microscopy (LM); 4) C3-dominant or codominant glomerular staining on immunofluorescence (IF); and 5) ‘‘hump-shaped’’ subepithelial deposits on electron microscopy (EM). Among these, 37.2% met 5/5 criteria, 40.7% 4/5 criteria, and 22.1% 3/5 criteria [15]. Unlike post-streptococcal glomerulonephritis (PSGN), where there is no need for biopsy unless some specific indications are present [16]. In most cases of non-streptococcal postinfectious glomerulonephritis (NSGN), biopsy is necessary to confirm the diagnosis and exclude other forms of GN [1], especially in cases of atypical postinfectious GN, defined by 1) persistent hematuria and proteinuria, with or without a history of preceding infection; 2) renal biopsy showing features of postinfectious GN 3) abnormalities of the alternative pathway of complement. This is because it may be associated with abnormalities in the alternative complement pathway [17]. A biopsy can be deferred in those with a clear history of previous or concurrent bacterial infection and low C3 complement level with spontaneous resolution of signs and symptoms [14].
Differential diagnosis
The differential diagnosis of Non-Streptococcal PSGN involves considering various other kidney diseases and conditions that share clinical features, histopathological findings, and laboratory abnormalities. This complex diagnostic process requires a comprehensive evaluation, often relying on clinical presentation, medical history, serological tests, imaging studies, and renal biopsy results [18-22].
Acute Postinfectious Glomerulonephritis (APIGN)
Distinguishing between Non-Streptococcal PSGN and APIGN, commonly caused by Streptococcus infections, is crucial due to their similar clinical presentations. A thorough review of the patient's medical history and microbiological data is essential in making this distinction [23].
IgA Nephropathy (IgAN)
IgAN is a common form of glomerulonephritis characterized by the deposition of IgA antibodies in the glomeruli. It can share clinical features with PSGN, such as hematuria and proteinuria, making renal biopsy and immunofluorescence analysis vital for differentiation [24,25].
Lupus Nephritis
Systemic Lupus Erythematosus (SLE) can present with renal involvement, known as lupus nephritis. It may mimic PSGN clinically, but a detailed autoimmune panel and serological markers specific to SLE help distinguish the two conditions [25].
Henoch-Schönlein Purpura (HSP) Nephritis
HSP is a systemic vasculitis associated with kidney IgA deposition. Clinically, it can resemble PSGN, especially in children. A skin biopsy and clinical history may aid in the diagnosis [26].
Membranoproliferative Glomerulonephritis (MPGN)
MPGN can manifest as hematuria, proteinuria, and hypertension, overlapping with PSGN. Renal biopsy with electron microscopy is crucial in establishing the diagnosis of MPGN [27].
IgM Nephropathy
IgM nephropathy is characterized by IgM deposition in the glomeruli. It can share clinical features with PSGN, necessitating renal biopsy and immunofluorescence studies for differentiation [21,28].
C3 Glomerulopathy
Disorders within the C3 glomerulopathy spectrum, including Dense Deposit Disease (DDD) and C3 Glomerulonephritis, can present similar clinical and histopathological features to PSGN. Specialized complement pathway tests are crucial for differentiation [29].
Postinfectious Glomerulonephritis with Streptococcus
Although PSGN primarily arises from non-Streptococcal infections, distinguishing it from Streptococcal PSGN is essential. Streptococcal serology and microbiological data can help differentiate the two [27,29].
Minimal Change Disease (MCD)
MCD is a nephrotic syndrome characterized by proteinuria and normal renal histology under light microscopy. It can sometimes present with hematuria, potentially mimicking PSGN. Renal biopsy with electron microscopy helps distinguish the two conditions [28].
Focal Segmental Glomerulosclerosis (FSGS)
FSGS is a cause of nephrotic syndrome and can occasionally present with hematuria, leading to diagnostic challenges. A renal biopsy is necessary to differentiate between PSGN and FSGS [22-24].
Infection-Associated Glomerulonephritis
Various viral and bacterial infections can lead to glomerulonephritis. Distinguishing between these infectious causes and PSGN requires a detailed clinical evaluation and serological testing to identify the causative pathogen [25,29].
As a result, the differential diagnosis of Non-Streptococcal Postinfectious Glomerulonephritis involves considering a wide range of kidney diseases and conditions that share clinical features, histopathological findings, and laboratory abnormalities. A multidisciplinary approach, including clinical evaluation, serological tests, imaging studies, and renal biopsy, is essential for accurately diagnosing and differentiating Non-Streptococcal PSGN from these various entities, ensuring appropriate management and treatment strategies tailored to the specific underlying condition.
Treatment
Most cases of Non-Streptococcal postinfectious glomerulonephritis (NSGN) have proven to be self-limiting, and its management revolves around controlling fluid overload and stabilizing changes in blood pressure. Treatment does not reverse the disease but prevents further progression to permanent damage and chronic kidney disease [30]. Supportive care is treated with loop diuretics, as well as dietary salt restriction (<2g/day) and fluid restriction (<2 L/day). Antihypertensive therapy is indicated for patients with persistent elevated proteinuria and hypertension refractory to supportive management [31]. These include drugs that modulate the Renin-Angiotensin-Aldosterone system, such as Angiotensin-converting enzyme inhibitors (ACE inhibitors) or Angiotensin receptor blockers (ARB). ACE inhibitors and ARBs should be avoided in patients with proven rapidly progressive acute renal failure, given the risk of exacerbating its advancement and potential hyperkalemia [30,31]. Antibiotic therapy is oriented at detecting the causal agent and eradicating such infections. Most adult patients present with active infection, and the antibiotic should be selected according to bacterial sensitivity [31].
Immunosuppressive therapy, such as corticosteroids, azathioprine, cyclophosphamide, and rituximab, have been demonstrated in various studies to be minimally effective in treating NSGN in adults given most patients present in an active state of infection and/or have adjacent immunosuppressive comorbidities such as type II Diabetes Mellitus [31]. Although the pathogenesis of the disease involves the immune system as a mediator and partial causative agent, the potential risks outweigh the limited benefit as a probable therapy. They should only be considered in cases refractory to other measures [31,33]. Vulnerable patients, such as the elderly or those with indications for dialysis, a severe decline in glomerular filtration rate (GFR), acute renal failure, or hypertensive crisis, should be hospitalized to monitor blood pressure, electrolyte and metabolic panel, urinalysis, and complement levels [31-33].
Prognosis & Outcomes
Both the prognosis and outcomes of Non-Streptococcal Postinfectious Glomerulonephritis (PSGN) can vary depending on several factors, including the underlying causative infection, the promptness of diagnosis and treatment, and individual patient characteristics. This condition is relatively rare, and limited research explicitly addresses its prognosis. However, drawing from general principles of glomerulonephritis and related conditions, we can provide insights into its potential course and outcomes. Non-Streptococcal PSGN encompasses a heterogeneous group of glomerular diseases triggered by various infections, such as staphylococcal, viral, or parasitic. The prognosis can vary widely based on the type and severity of the underlying infection [34]. In some cases, especially when the underlying infection is adequately treated, Non-Streptococcal PSGN can resolve spontaneously, leading to complete recovery without long-term renal impairment [35].
Some patients may experience persistent proteinuria even after resolution of the acute phase, which could indicate ongoing renal involvement. This may require long-term follow-up and monitoring [36]. In more severe cases or when diagnosis and treatment are delayed, Non-Streptococcal PSGN can progress to chronic kidney disease. Long-term inflammation and scarring of the glomeruli can impair renal function [36,37]. Renal biopsy plays a crucial role in assessing prognosis. The extent of glomerular and interstitial damage and the presence of specific histopathological findings can help predict the likelihood of recovery versus progression [38].
Certain risk factors, such as hypertension, older age, and severe initial kidney dysfunction, may be associated with a poorer prognosis in glomerulonephritis. However, the specific impact on Non-Streptococcal PSGN is not well-documented [36,39]. If indicated, the response to treatment, including antibiotics for the underlying infection and immunosuppressive therapy, can significantly influence the outcome. Prompt and appropriate treatment is essential for improving the prognosis [40]. While Non-Streptococcal PSGN is generally considered a one-time event, there have been rare reports of recurrence, particularly in cases associated with certain viral infections. Continuous monitoring may be necessary in such instances [40,41].
Overall, the prognosis and outcomes of Non-Streptococcal PSGN are highly variable and depend on multiple factors, including the type and severity of the underlying infection, the timeliness of treatment, and the presence of risk factors. While some patients may experience complete resolution, others may face persistent proteinuria or even progress to chronic kidney disease. Regular follow-up monitoring and appropriate management of the underlying infection and glomerulonephritis are crucial to optimize patient outcomes [42]. It is important to note that specific studies or clinical guidelines addressing Non-Streptococcal PSGN may be limited, and the information provided here is based on general principles of glomerulonephritis and kidney disease management. Therefore, individual patient prognoses should be determined through careful clinical evaluation considering the unique circumstances of each case.
Conclusion
Infection-related glomerulonephritis (IRGN) is a kidney disorder characterized by glomerular inflammation resulting from infections originating elsewhere in the body. While traditionally associated with streptococcal infections, recent data indicate a significant shift towards non-streptococcal bacterial infections, including staphylococcal and Escherichia coli (E. coli) infections, emerging as substantial contributors to IRGN among adults in the United States. The prevalence of this shift may exhibit temporal and geographical variations, highlighting the evolving landscape of IRGN epidemiology. The management of non-streptococcal bacterial IRGN focuses on two key aspects: addressing the underlying infection with targeted antibiotics and managing kidney inflammation. Additionally, patients may require supportive care, including meticulous blood pressure control and managing complications associated with kidney dysfunction. Timely diagnosis and prompt intervention are pivotal in enhancing patient outcomes in this evolving IRGN landscape among adults. This evolving epidemiological trend underscores the importance of staying vigilant in diagnosing and managing IRGN as it continues to present challenges and opportunities for improved patient care.
References
- Parikh SV, Nadasdy T (2020) Epidemiology, pathogenesis, treatment and outcomes of infection-associated glomerulonephritis. Nat Rev Nephrol 16(1): 32-50.
- Somers MJG (2019) Infection-Related Glomerulonephritis. Pediatr Clin North Am 66(1): 59-72.
- Shimohata H, Kobayashi M, Koyama A (2022) Staphylococcus aureus Infection-Related Glomerulonephritis with Dominant IgA Deposition. Int J Mol Sci 23(13): 7482.
- Oda T, Yoshizawa N (2021) Factors Affecting the Progression of Infection-Related Glomerulonephritis to Chronic Kidney Disease. Int J Mol Sci 22(2): 905.
- Lamba P, Nam KH, Contractor J, Kim A (2020) Nephritic Syndrome. Prim Care 47(4): 615-629.
- Zappa M, Salviani C, Gaggiotti M, Alberici F, Scolari F (2020) Infection-related glomerulonephritis: the new face of an old disease]. G Ital Nefrol 37(6): 2020-vol6.
- Nasr SH, Radhakrishnan J, D’Agati VD (2013) Bacterial infection–related glomerulonephritis in adults Kidney Int 83(5)792-803.
- YaoKo Wen (2009) The Spectrum of Adult Postinfectious Glomerulonephritis in the New Millennium, Renal Failure 31:8, 676-682.
- Wen YK (2009) The spectrum of adult postinfectious glomerulonephritis in the new millennium. Ren Fail 31(8): 676-682.
- Wen YK (2010) Clinicopathological study of infection-associated glomerulonephritis in adults. Int Urol Nephrol Jun 42(2): 477-485.
- Nasr SH, Radhakrishnan J, D Agati VD (2013) Bacterial infection-related glomerulonephritis in adults. Kidney International Nature Publishing Group 83(5): 792-803.
- Satoskar AA, Parikh SV, Nadasdy T (2020) Epidemiology, pathogenesis, treatment and outcomes of infection-associated glomerulonephritis. Nat Rev Nephrol 16(1): 32-50.
- Radhakrishnan J, D’Agati VD (2013) Bacterial infection-related glomerulonephritis in adults. Vol. 83, Kidney International. Nature Publishing Group 83(5): 792-803.
- D’Agati VD (2013) Bacterial infection–related glomerulonephritis in adults. Kidney Int 83(5): 792-803.
- Nasr SH, Markowitz GS, Stoke MB, Said SM, Valeri AM, et al. (2008) Acute postinfectious glomerulonephritis in the modern era: experience with 86 adults and review of the literature. Medicine (Baltimore) 87: 21-32.
- Rawla P, Padala SA, Ludhwani D (2023) Poststreptococcal Glomerulonephritis (2022) In: StatPearls. Treasure Island (FL): StatPearls Publishing.
- Sethi S, Fervenza FC, Zhang Y, Zand L, Meyer NC, et al. (2013) Atypical postinfectious glomerulonephritis is associated with abnormalities in the alternative pathway of complement. Kidney Int 83(2): 293-299.
- Chadban SJ, Atkins RC (2005) Glomerulonephritis. Lancet 365(9473): 1797-806.
- Elizabeth A K Hunt, Somers MJG (2019) Infection-Related Glomerulonephritis. Pediatr Clin North Am 66(1): 59-72.
- Anders HJ, Kitching AR, Leung N, Romagnani P (2023) Glomerulonephritis: immunopathogenesis and immunotherapy. Nat Rev Immunol 23(7): 453-471.
- Couser WG (2016) Pathogenesis and treatment of glomerulonephritis-an update. J Bras Nefrol 38(1): 107-122.
- Sethi S, Fervenza FC (2019) Standardized classification and reporting of glomerulonephritis. Nephrol Dial Transplant 34(2): 193-199.
- Keskinyan VS, Lattanza B, Reid Adam J (2023) Glomerulonephritis. Pediatric Rev 44(9): 498-512.
- Vinen CS, Oliveira DB (2003) Acute glomerulonephritis. Postgrad Med J 79(930): 206-213, quiz 212-213.
- Boudoirs C, Copin MC, Wacrenier S, Piccoli GB, Croue A, et al. (2022) Histopathological prognostic factors in ANCA-associated glomerulonephritis. Autoimmun Rev 21(9): 103139.
- Moroni G, Ponticelli C (2014) Rapidly progressive crescentic glomerulonephritis: Early treatment is a must. Autoimmun Rev 13(7): 723-729.
- Wasserstein AG (1997) Membranous glomerulonephritis. J Am Soc Nephrol 8(4): 664-674.
- Mookerje BK, Lohr JW, Jenis EH, Heffner HM (2001) Glomerulonephritis for the generalist. J Med 32(3-4): 115-134.
- Srivastava RN (1999) Acute glomerulonephritis. Indian J Pediatr 66(2): 199-205.
- Kazi, Ahmad M., and Muhammad F (2023) Hashmi "Glomerulonephritis." National Library of Medicine NIH.
- Nasr HS, Vivette D, Jai R (2013) Bacterial Infection Related Glomerulonephritis in Adults, International Society of Nephrology 83(3): 792-803.
- Vecihi B (2021) "Glomerulonephritis Associated with Nonstreptococcal Infection Treatment Management."
- Jadoul, Michel, Winkelmayer, Wolfgang C, Eknoyan, et al. (2021) KDIGO Clinical Practice Guideline for the Management of Glomerular Diseases, Kidney International Org 100(4).
- Schratzberger G, Mayer G (2006) Primäre Glomerulonephritiden [Primary glomerulonephritis]. Wien Klin Wochenschr 118(9-10 Suppl): 57-66, quiz 67.
- Floege J, Amann K (2016) Primary glomerulonephritides Lancet 387(10032): 2036-2048.
- Towers C, MM Lang (2001) Identifying poststreptococcal glomerulonephritis. Nurse Pract 26(8): 34, 37-42, 44-7, quiz 48-9.
- Zamd M, Hachim K, Soulami K, Ezzahidy M, Souiri M, et al. (2012) Glomérulonéphrites aiguës postinfectieuses [Acute postinfectious glomerulonephritis]. Nephrol Ther 8(4): 247-258.
- Mayer U, Bräsen JH, Pape L (2019) Die Rasch Progessive Glomerulonephritis im Kindesalter [Rapid Progressive Glomerulonephritis in Children]. Klin Padiatr 231(1): 4-13.
- Gaggiotti M, Alberici F, Scolari F, Zappa M, Salviani C (2020) [Infection-related glomerulonephritis: the new face of an old disease]. G Ital Nefrol 37(6):2020-vol6.
- Weisse ME, Sainato RJ (2019) Poststreptococcal Glomerulonephritis and Antibiotics: A Fresh Look at Old Data. Clin Pediatr (Phila) 58(1): 10-12.
- Watson S, Cailhier JF, Hughes J, Savill J (2006) Apoptosis and glomerulonephritis. Curr Dir Autoimmune 9: 188-204.
- Barsoum RS (2010) Glomerulonephritis in disadvantaged populations. Clin Nephrol 74 Suppl 1: S44-50.






























