Renal Tubular Acidosis: Recognition and Treatment Strategies
Pushan Aggarwal1, Elsa Carolina Bonilla la Rosa2, Ronald Mauricio Blanco Montecino3, Jhon Navarro Gonzalez4, Jose Luis Hernandez Aparicio3, Peggie Crisalida Mendoza Robles5, Felix Ricardo Bonilla Bonilla3, Patricia Maza Hill6, Rossy I Valecillos Paez7, Patricia V Visbal L8, Isaac Alberto Gomez Hernandez3, Maria Belen Roman9, Stephany Valko9 and Maria Isabel Gomez10*
1Kasturba Medical College Manipal, India
2Universidad Peruana Cayetano Heredia, Peru
3Universidad de El Salvador, El Salvador
4Universidad del Zulia, Venezuela
5Universidad San Martin de Porres, Peru
6Universidad de Carabobo, Venezuela.
7Universidad Central de Venezuela, Venezuela
8Universidad de los Andes, Venezuela
9Pontificia Universidad Catolica del Ecuador, Ecuador
10Universidad del Valle, México
Submission:July 13, 2023;Published: July 20, 2023
*Corresponding author: Maria Isabel Gomez, Department of Medicine, Universidad del Valle de, Mexico
How to cite this article: Pushan A, Elsa Carolina Bonilla la R, Ronald Mauricio Blanco M, Jhon Navarro G, Jose Luis Hernandez A, et al. Renal Tubular Acidosis: Recognition and Treatment Strategies. JOJ Urology & Nephrology, 2023; 8(1): 555732. DOI: 10.19080/JOJUN.2023.08.555732
Abstract
Renal tubular acidosis (RTA) is a group of kidney disorders characterized by impaired renal acidification, leading to disturbances in acid-base balance. This review article provides a comprehensive overview of RTA, including its epidemiology, pathophysiology, clinical presentation, and diagnosis. Three main types of RTA are discussed: distal RTA (Type 1), proximal RTA (Type 2), and hyperkalemic RTA (Type 4). Each type’s underlying causes and mechanisms are explored, highlighting the importance of accurate diagnosis for appropriate management. Diagnostic approaches involving clinical evaluation, laboratory tests, and specialized renal function studies are described in detail. The treatment strategies for each type of RTA are outlined, emphasizing the importance of correcting the acid-base imbalance and managing associated electrolyte abnormalities. Lifestyle modifications and pharmacological interventions are discussed, including alkali supplementation and potassium regulation. Finally, the need for further research and prospective studies to enhance our understanding of RTA and improve therapeutic interventions is emphasized. Advancements in diagnosis and treatment approaches have the potential to maximize patient outcomes and improve the quality of life for individuals affected by RTA.
Keywords: Renal tubular acidosis; RTA; Acid-base balance; Distal RTA; Proximal RTA; Hyperkalemic RTA; Epidemiology; Pathophysiology; Clinical presentation; Diagnosis; Treatment strategies; Alkali supplementation; Potassium regulation; Electrolyte abnormalities; Metabolic acidosis
Abbreviations: RTA: Renal Tubular Acidosis, GFR: Glomerular Filtration Rate, ACE: Angiotensin-Converting Enzyme, ARBs: Angiotensin Receptor Blockers, NH4Cl: Ammonium Chloride, HCO3-: Bicarbonate, KCL: Potassium Chloride, CKD: Chronic Kidney Disease, ENaC: Epithelial Sodium Channel, DASH: Dietary Approaches to Stop Hypertension, SNHL: Sensorineural Hearing Loss, CT: Computed Tomography, ATR: Acidification Test, DASH: Dietary Approaches to Stop Hypertension, SCZ: Zirconium Cyclosilicate, RAAS: Renin-Angiotensin-Aldosterone System, K+: Potassium, NaHCO3: Sodium Bicarbonate, K-citrate: Potassium Citrate, ACE inhibitors: Angiotensin-Converting Enzyme Inhibitors, ARBs: Angiotensin Receptor Blockers, DASH: Dietary Approaches to Stop Hypertension, CKD: Chronic Kidney Disease, K+: Potassium, SZC: Zirconium Cyclosilicate, NH4+: Ammonium, H+: Hydrogen, TRC101: Veverimer
Introduction
Renal tubular acidosis (RTA) is a group of kidney disorders characterized by impaired renal acidification, leading to the inability of the kidneys to regulate acid-base balance effectively [1]. There are three main types of RTA. Type 1, or distal RTA, is the most common form associated with impaired hydrogen ion secretion in the distal tubules of the kidneys. It often presents with metabolic acidosis and can be either inherited or acquired. Type 2, or proximal RTA, is characterized by impaired bicarbonate reabsorption in the proximal tubules. It can be inherited or acquired and is usually associated with generalized dysfunction of the proximal tubules. Type 4, or hyperkalemic RTA, is primarily caused by aldosterone deficiency or resistance, impairing potassium excretion [1,2]. The epidemiology of RTA varies depending on the type and underlying etiology. Type 1 RTA is the most prevalent, with estimated frequencies ranging from 1 to 4 cases per 10,000 population. Type 2 RTA is less common, and its prevalence is not well-established. Type 4 RTA is rare and often associated with diabetes mellitus and chronic kidney disease [3,4]. Various factors, including geographical location, population demographics, and the condition’s underlying causes, influence the prevalence and incidence of RTA. Accurate estimates are challenging due to the heterogeneity of RTA and the potential for underdiagnosis [2,4]. The clinical presentation of RTA can vary depending on the specific type and severity of the condition. Common symptoms include chronic metabolic acidosis, which may manifest as fatigue, weakness, growth retardation in children, and muscle pain. Other features may include electrolyte imbalances, such as hypokalemia or hyperkalemia, urinary abnormalities, and nephrocalcinosis [4].
The diagnosis involves a combination of clinical evaluation, laboratory tests, and specialized renal function studies. Initial screening typically includes measurement of serum electrolytes, arterial blood gas analysis, and urinary pH. Confirmatory tests may involve a urine acidification test, a bicarbonate-loading test, or genetic testing, depending on the suspected type of RTA [5]. Additionally, imaging studies like ultrasound or CT scans may be performed to assess for nephrocalcinosis or other kidney abnormalities. The treatment of RTA aims to correct the acidbase imbalance and manage associated electrolyte abnormalities. This often involves oral alkali supplementation with bicarbonate or citrate preparations to correct metabolic acidosis. In Type 1 RTA, thiazide diuretics may be used to enhance renal bicarbonate reabsorption [1,5]. Management of Type 2 and Type 4 RTA focuses on addressing the underlying causes and optimizing treatment for associated conditions, such as addressing aldosterone deficiency or resistance in Type 4 RTA [2,4]. This narrative review article aims to provide a comprehensive overview of renal tubular acidosis, a commonly misdiagnosed condition. It describes the different types of RTA and outlines the treatment strategies for managing this condition.
RTA Physiopathology
RTA is a condition characterized by impaired renal acidification, causing the kidneys to be unable to excrete hydrogen ions or reabsorb bicarbonate effectively [6]. This leads to a disruption of acid-base homeostasis in the body. There are three main types of RTA: distal RTA (Type 1), proximal RTA (Type 2), and hyperkalemic RTA (Type 4). Each type has distinct causes and physiopathology. Type 1 distal RTA is primarily caused by a defect in the distal tubular cells’ ability to secrete hydrogen ions [6,7]. This defect can be acquired, as in autoimmune diseases like Sjögren’s syndrome or systemic lupus erythematosus, or it can be inherited due to mutations in genes involved in the acidification process. The impaired hydrogen ion secretion reduces urine acidification and urinary acid excretion [8]. This leads to a buildup of acid in the blood and subsequently decreases blood bicarbonate levels, causing metabolic acidosis. Type 2 proximal RTA is characterized by impaired bicarbonate reabsorption in the proximal tubules. It can be inherited as an autosomal recessive disorder known as Fanconi syndrome or acquired due to various causes such as multiple myeloma, drugs (e.g., acetazolamide, ifosfamide), or heavy metal exposure [8,9]. The defect in bicarbonate reabsorption leads to increased bicarbonate loss in the urine and decreased bicarbonate levels in the blood, resulting in metabolic acidosis. Type 4 hyperkalemic RTA is primarily associated with impaired ammonium ion excretion and decreased urinary acidification in the distal tubules. It is often seen in individuals with reduced aldosterone activity or resistance to aldosterone, such as those with Addison’s disease or taking medications like ACE inhibitors or angiotensin receptor blockers. Aldosterone is crucial in potassium excretion and acid-base regulation [6,9]. In Type 4 RTA, the reduced aldosterone activity leads to impaired ammonium ion excretion, reducing urinary acidification and increasing blood potassium levels. The elevated potassium levels can also inhibit the secretion of hydrogen ions, contributing to metabolic acidosis.
The underlying physiopathology of all types of RTA ultimately leads to metabolic acidosis. Metabolic acidosis stimulates respiratory compensation, resulting in hyperventilation and decreased arterial carbon dioxide levels [10]. This compensatory response helps to maintain acid-base balance by attempting to decrease the concentration of hydrogen ions in the blood. However, if the underlying cause of RTA is not addressed, the compensatory mechanisms may not fully correct the acid-base imbalance. In summary, the different types of renal tubular acidosis have distinct causes and physiopathology [6,8]. Type 1 distal RTA is characterized by impaired hydrogen ion secretion, Type 2 proximal RTA by impaired bicarbonate reabsorption, and Type 4 hyperkalemic RTA by impaired ammonium ion excretion and decreased urinary acidification. These defects lead to metabolic acidosis and trigger compensatory mechanisms to maintain acid-base balance [11]. Identifying the underlying cause of RTA is crucial to guide appropriate treatment and prevent longterm complications.
RTA Clinical Presentation
The primary change caused by Renal Tubular Acidosis is a dyselectrolytemia that is partial to some particular ions. A change in these ions is the first sign of this condition. Subsequent effects of these electrolyte anomalies cause Renal Tubular Acidosi (RTA) pathognomonic symptoms. The primary dyselectrolytemia caused by Type-1 and Type-2 RTA is- low Potassium and bicarbonate levels in the serum. Potassium is essential for the normal functioning of nerves and muscles, especially cardiac muscle. Hence, hypokalemia after Type-1 and 2 RTA can cause extreme lethargy and weakness due to impaired muscular conduction. Such effects compounded on the heart muscle can cause dysrhythmias and irregular heartbeats, often with coupled conduction anomalies. When afflicting skeletal muscles in large groups, it can even result in paralysis. Smooth muscle paralysis can even lead to death if essential muscles such as respiration are affected [12,13].
The significant signs and symptoms of Type 4 RTA are elevated Potassium and low bicarbonate levels in the serum. The subsequent effects are primarily associated with altered levels of Potassium in the blood. Some frequently seen symptoms of Type 4 RTA are in common with those of Type-1 and 2 RTA, such as fatigue and weakness of skeletal muscles. Additionally, some studies have noted weight changes and moderate to severe abdominal pain as a common occurrence [14]. Additionally, inherited forms of RTA, such as the recessive form, can be associated with other clinical manifestations, such as hyperchloremic metabolic acidosis, nephrocalcinosis, dehydration, poor growth, and bilateral SNHL in some cases [15,16]. The dominant form often presents more in line with renal pathology, with common initial findings being nephrocalcinosis, persistent hypokalemia, and repeated episodes of hypovolemia [17].
RTA Diagnosis
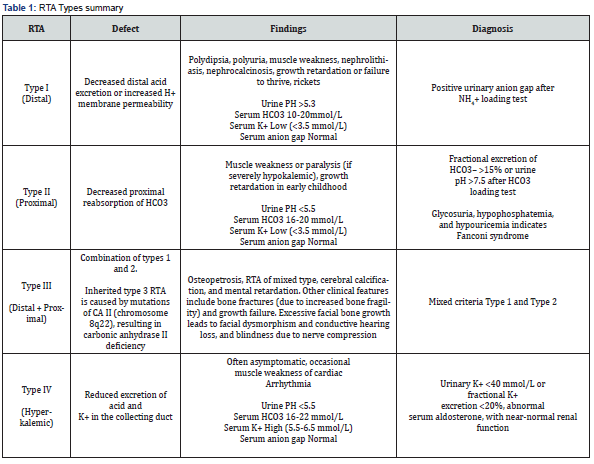
When a patient presents with a clinical picture resembling renal tubular acidosis (RTA), it is appropriate to follow a stepwise approach [18]. First, the clinician should determine the patient’s plasma anion gap. With a normal value that ranges between 8 and 16 mEq/L, it represents the unaccounted anions in the body. A high anion gap suggests clinical conditions with increased anions, like diabetic or starvation ketoacidosis, lactic acidosis, uremia, and some drug toxicities [18,19]. Hypokalemic hyperchloremic metabolic acidosis with a normal anion gap and a normal glomerular filtration rate suggest either RTA or gastrointestinal bicarbonate losses, such as diarrhea, pancreatic fistula, chronic laxative use or nasojejunal suctioning [20]. After determining the presence of metabolic acidosis with a normal plasma anion gap, it is essential to measure potassium; K+ disturbance is also a common finding on RTA and may guide evaluation and treatment. Hypokalemia is characteristic of distal and proximal RTA, while hyperkalemia indicates hyperkalemic RTA [21]. At the same time, measuring the urine anion gap to differentiate renal from extrarenal causes [18]. In metabolic acidosis due to extrarenal causes, the kidney responds by increasing NH4+ excretion. This response is not seen in renal tubular acidosis, where an increased NH4+ elimination is absent. Thus, it presents a normal Urine anion Gap. With a normal value of 30- 50 mEq/L, the urinary anion gap is the difference between the sum of urinary Na+ and K+ and Cl- [18]. Therefore, urinary Cl− indirectly measures NH4+ secretion [21].
The distinction between hyperkalemic RTA (Type 4) and distal and proximal RTA (Type 1 and 2) is relatively straightforward. Hyperkalemic RTA is readily diagnosed by hyperkalemia and serum aldosterone measurements in near-normal GFR [21]. Following identifying a renal cause of metabolic acidosis, the third step would be determining the urine pH, allowing to determine the appropriate distal urinary acidification. A urine pH >5.3 in a patient with metabolic acidosis indicates distal RTA [18]. However, urinary pH alone is a poor diagnostic tool. In various circumstances, pH can lead to a false diagnosis. Both intravascular volume depletion and urinary tract infection by urea-cleaving organisms can raise urinary pH while simultaneously causing nonanion gap metabolic acidosis, mimicking the appearance of ATR [21]. In patients without systemic acidosis, this can be induced by administering ammonium or calcium chloride, depending on liver function [18]. Administration of an acid load in the form of ammonium chloride (NH4Cl) should acidify the urine in a normally functioning kidney to buffer the pH of the blood. Urinary pH will not decrease as expected in patients with distal RTA and impaired urinary H + and NH4+ secretion [22]. Although the ammonium chloride test is the gold standard for distal RTA, the furosemide with fludrocortisone test is a more accessible, functional, and tolerable (but less specific) option and can be used for screening. This combination increases Na+ reabsorption by ENaC, causing a negatively charged tubular lumen and an increased H+ secretion by alpha-intercalated cells. In those patients who can reduce their urinary pH below 5.3, distal RTA is excluded, but confirmation with ammonium or calcium chloride may be needed in those patients who cannot [20].
The fourth step is the bicarbonate loading test, which allows the accurate diagnosis of proximal RTA [23]. The principle is that in patients with acidemia, low plasma HCO3-concentration, and intact proximal tubule function, HCO3- administration should not alter urinary HCO3- levels because HCO3 - is avidly reabsorbed from urine. In patients with proximal ATR, urinary HCO3 - concentration will increase after HCO3-infusion due to impaired reabsorption [24]. In metabolic acidosis and fractional excretion, >15% is diagnostic of proximal RTA [18,23]. ATR III, This rare mixed syndrome has diagnostic findings characteristic of both distal and proximal RTA. RTA type 3 manifests with debilitating congenital syndromes in children and is primarily associated with carbonic anhydrase II deficiency [25]. Additionally, mutation tests can be performed to identify alterations in transporters and their regulators, allowing insight into the pathogenesis of RTA, longterm outcome prognosis, and extrarenal phenotype; furthermore, it provides a ground for prenatal counseling [18] (Table 1).
Treatment: RTA Type I
Distal renal tubular acidosis should be managed with lifestyle modifications, including keeping a good weight, diet, and pharmacotherapy. Focusing on the diet can help reduce acid intake; a diet based on vegetables contains a lower acid load. Compared to acid-producing food such as animal protein, this latter should be reduced in patients with metabolic acidosis. They need to increase the alkali-containing foods such as fruits and vegetables. Decreasing animal protein intake also raises total CO2 serum concentrations [26]. Modifications in the diet, including citrus fruits and beverages, in addition to reducing Na, oxalate, fructose, and animal protein source, with normal calcium intake, can also help patients with RTA type 1 and kidney stones. Diet is an essential factor that is related to acid-base status. Previous studies have shown that a high acid-loading diet increases the risk and development of chronic renal disease. The central part of the diet is to eat more fruits and vegetables and reduce animal protein. However, an inappropriate diet may lead to protein malnutrition, which must be avoided [27]. Type 1 RTA features are hyperchloremic metabolic acidosis and hypokalemia. The urine pH can be more than 5.5. Pharmacotherapy is fundamental to correcting metabolic acidosis and requires oral bicarbonate replacement at 1-2 meq/kg per day by sodium bicarbonate [28,29]. Citrate salts correct hypocitraturia and prevent nephrolithiasis. This organic anion’s mechanism of action helps correct metabolic acidosis and decrease urine calcium excretion. As a result, it prevents recurrent kidney stone formation. Patients with symptomatic hypokalemia should be treated with KCL or K-citrate; this management will prevent decreasing serum K levels [26,30].
HCO3- Bicarbonate, K+ potassium, KCl: potassium chloride, Na+ sodium, NaHCO3- Sodium bicarbonate, NH4+ ammonium, RTA: Renal Tubular Acidosis.
Treatment: RTA Type II
The management of RTA type II involves a combination between lifestyle modifications and pharmacological strategies. Nutritional management is crucial for renal dysfunctions. Diets like DASH, Mediterranean, plant-based, deficient protein (0.3 g/kg/day), or vegan (0.7 g/kg/day) help reduce phosphorus, sodium, and uremic toxin reuptake. Maintaining nitrogen balance while correcting protein intake, including lipids and enough energy (30-35 Kcal/kg/day), is important [31]. However, strict low-protein diets are challenging for patients, making the Mediterranean (primarily plant-based) a more feasible long-term option [32]. Moreover, increasing fiber intake from fruits and vegetables, particularly in vegan diets, helps control metabolic acidosis by producing alkalizing acids. It also relates to lower saline load, regulating aldosterone, angiotensin II, and endothelin [31]. Consuming orange juice (0.5 or 1 L/day) rich in citrates helps alkalize urine, but glucose should be controlled. Mineral water intake (1.4 L/day) raises urinary pH, similar to potassium citrate supplementation. Finally, vitamin K1 supplementation is beneficial due to its association with reduced mortality in renal and cardiovascular diseases. Food sources like kimchi, spinach, and onion are rich in vitamin K [31].
Besides diet and lifestyle modifications, pharmacotherapy is vital for treating RTA type II. Long-term alkaline therapy is necessary for proximal renal tubular acidosis, especially for autosomal dominant and recessive type II renal tubular acidosis. High doses of alkaline medication like K-citrate (10-15 mmol/kg/ day) can be complicated. Deficiency in bone mineral density due to impaired phosphate reabsorption and vitamin D conversion requires supplementation of vitamin D and phosphate (1-3 g/day) to prevent skeletal abnormalities. Hydrochlorothiazide might aid HCO3-reabsorption and improves alkali supplementation, but close monitoring and supplementation of K+ are essential to prevent depletion caused by diuretics [32].
Treatment: RTA Type IV
Treatment for RTA type IV involves both non-pharmacological and pharmacological strategies. In patients with hyperkalemic RTA, dietary restriction of K+ has previously been the standard of care; however, new data suggest that increased intake of alkaliproducing fruits and vegetables (which are often high in K+) and limiting intake of acid-producing foods may correct acidosis. Lower serum potassium is achieved with dietary modification, withdrawal or modification of offending medications, and potassium binders. The treatment initially aims to normalize serum K+ concentration. This may improve metabolic acidosis by increasing urinary NH3 responsible for buffering secreted H+ and enhancing HCO3-generation in the proximal tubule by improving glutamine metabolism. Additionally, any non-essential medications affecting renal K+ excretion, aldosterone synthesis, or activity should be discontinued. ACE inhibitors and ARBs reduce urinary K+ excretion by inhibiting aldosterone secretion in the adrenal gland and may cause hyperkalemia in patients with preexisting conditions that cause impaired K+ excretion. However, ACE inhibitors and ARBs are usually continued in patients with CKD because of their cardiovascular and reno-protective benefits.
Pharmacotherapy of type 4 RTA aims to correct acidemia to prevent the maladaptive effects on bone health and the progression of CKD. This can be attained with alkali replacement, typically sodium bicarbonate. The Kidney Disease Improving Global Outcomes guidelines suggest the administration of oral HCO3-therapy to maintain serum HCO3-in the normal range or less than 22 mmol/L for patients with CKD. NaHCO3 administration will correct metabolic acidosis and minimize hyperkalemia risk. Mineralocorticoid replacement with fludrocortisone aims to correct hypoaldosteronism or overcome resistance to its actions. A lower dose of fludrocortisone, 0.1 mg per day, effectively manages hyperkalemia associated with aldosterone deficiency. The challenge in many cases is that patients with type 4 RTA tend to have multiple comorbidities, including CKD and diabetes, associated with hypervolemia. Therefore, administering additional sodium as the bicarbonate salt risks exacerbating volume retention and hypertension. Loop diuretics and NaHCO3 therapy may be beneficial in patients with type 4 RTA; they may reduce serum K+ and help control volume overload by increasing Na+ delivery and flow rates to the cortical collecting duct while lowering blood pressure, exceptionally when fludrocortisone is not tolerated. Concurrent administration of loop diuretics may help ameliorate salt and volume retention and potentiate the action of mineralocorticoids by promoting distal sodium delivery.
For patients with hyperkalemic type 4 RTA, there are newer K+-binding agents available that can be used to treat hyperkalemia and improve acidosis. Patiromer is a polymeric cation exchange resin that binds K+ ions in exchange for calcium ions in the colon. In contrast, zirconium cyclosilicate (SCZ) is a non-polymeric, selective K+ binder that entraps K+ and NH4+ ions in exchange for H+ and Na+ ions throughout the gastrointestinal tract. Both Patiromer and SZC are effective in patients receiving RAAS inhibitor therapy, allowing the continuation of these therapeutic agents in hyperkalemia. Veverimer (TRC101) is a treatment for metabolic acidosis currently in clinical development. Veverimer selectively binds H+ in the gastrointestinal tract, thereby increasing serum HCO3-concentrations and correcting acidosis. In patients with CKD and metabolic acidosis, Veverimer significantly increases serum HCO3-concentrations within 24-72 h of administration and can have continuous efficacy in the next 2 weeks and sometimes even up to 12 weeks. Although studies in patients with RTA are needed to confirm the efficacy and safety of Veverimer in RTA, this drug represents an emerging alternative to alkali therapy.
Conclusion
Renal tubular acidosis is a complex disorder characterized by impaired renal acidification, resulting in metabolic acidosis. This review article has shed light on RTA’s treatment strategies, which aim to correct the underlying acid-base imbalance, manage electrolyte abnormalities, and prevent complications. The mainstay of therapy involves alkali supplementation to correct metabolic acidosis. Additionally, potassium supplementation may be necessary in cases of hypokalemia, while measures to prevent renal stone formation, such as increased fluid intake and dietary modifications, are crucial. Although current treatment strategies have shown efficacy in managing RTA, it is essential to note that further large-scale prospective studies are necessary to enhance understanding of this condition and develop better therapeutic approaches. These studies should explore novel treatment modalities, investigate long-term outcomes, and address the gaps in our knowledge regarding the optimal management of RTA. While significant progress has been made in the diagnosis and management of RTA, there remains a need for further research to advance knowledge and refine therapeutic strategies. Only comprehensive investigations and well-designed prospective studies can result in improved therapeutic interventions that maximize patient outcomes and quality of life for individuals affected by RTA.
References
- Palmer BF, Kelepouris E, Clegg DJ (2021) Renal Tubular Acidosis and Management Strategies: A Narrative Review. Adv Ther 38(2): 949- 968.
- Giglio S, Montini G, Trepiccione F, Gambaro G, Emma F (2021) Distal renal tubular acidosis: a systematic approach from diagnosis to treatment. J Nephrol 34(6): 2073- 2083.
- Buckalew VM (1968) Familial renal tubular acidosis. Ann Intern Med 69(6):1329- 1330.
- Honore PM, Jacobs R, Hendrickx I, De Waele E, Van Gorp V, et al. (2015) Renal tubular acidosis with hyperchloremic acidosis: harmless with a sting? Crit Care 19(1): 305.
- Golding PL (1975) Renal tubular acidosis in chronic liver disease. Postgrad Med J 51(598): 550-556.
- Daniel Batlle, Haque SK (2012) Genetic causes and mechanisms of distal renal tubular acidosis. Nephrol Dial Transplant 27(10): 3691-3704.
- Kraut JA, Madias NE (2010) Metabolic acidosis: pathophysiology, diagnosis, and management. Nat Rev Nephrol 6(5): 274-285.
- Syed Haque K, Gema Ariceta, Batlle DC (2012) Proximal renal tubular acidosis: a not-so-rare disorder of multiple etiologies. Nephrol Dial Transplant 27(12): 4273-4287.
- Karet FE (2009) Mechanisms in hyperkalemic renal tubular acidosis. J Am Soc Nephrol 20(2):251-254.
- Bruce LJ, Wrong O, Toye AM, Young MT, Ogle G (2000) Band 3 mutations, renal tubular acidosis and South-East Asian ovalocytosis in Malaysia and Papua New Guinea: loss of up to 95% band 3 transport in red cells. Biochem J 350 Pt 1(Pt 1): 41-51.
- Feld LG, Langford DJ (2016) Renal tubular acidosis. In: Avner ED, Harmon WE, Niaudet P, eds. Pediatric Nephrology. 7th edn. Springer-Verlag 891-926.
- Ghimire A, Li D, Amin L (2023) Ibuprofen-Induced Renal Tubular Acidosis: Case Report on a Not-So-Basic Clinical Conundrum. Can J Kidney Health Dis 10: 20543581231183813.
- Mhlana N, Korf M, Davids MR, Chothia MY (2023) Distal renal tubular acidosis in a patient with Hashimoto's thyroiditis: a case report. Biochem Med (Zagreb) 33(2): 020802.
- Adrenal insufficiency & Addison’s disease. National Institute of Diabetes and Digestive and Kidney Diseases. Accessed (2020).
- Batlle D, Ghanekar H, Jain S, Mitra A (2001) Hereditary distal renal tubular acidosis: new understandings. Annu Rev Med 52: 471-484.
- Palazzo V, Provenzano A, Becherucci F, Sansavini G, Mazzinghi B, et al. (2017) The genetic and clinical spectrum of a large cohort of patients with distal renal tubular acidosis. Kidney Int 91(5):1243-1255.
- Gómez-Conde S, García-Castaño A, Aguirre M, Herrero M, Gondra L, et al. (2021) Molecular aspects and long-term outcome of patients with primary distal renal tubular acidosis. Pediatr Nephrol 36(10): 3133- 3142.
- Bagga A, Sinha A (2020) Renal Tubular Acidosis. Indian J Pediatr 87(9): 733-744.
- Joseph Laakman, Matthew Krasowski (2021) Frequency and Clinical Significance of Negative Anion Gap Values in an Academic Medical Center Population. American Journal of Clinical Pathology 156.
- Magni G, Unwin RJ, Moochhala SH (2021) Renal tubular acidosis (RTA) and kidney stones: Diagnosis and management. Arch Esp Urol 74(1): 123-128.
- Yaxley J, Pirrone C (2016) Review of the Diagnostic Evaluation of Renal Tubular Acidosis. The Ochsner Journal 16(4): 525- 530.
- Bagga A, Sinha A (2007) Evaluación de la acidosis tubular renal. Indio J Pediatr julio 74 (7): 679-686
- Alexander RT, Bitzan M (2019) Renal Tubular Acidosis. Pediatr Clin North Am 66(1): 135-157.
- Smulders YM, Frissen PH, Slaats EH, Silberbusch J (1996) Acidosis tubular renal: fisiopatología y diagnó Arch Intern Med 12-26 de agosto 156(15): 1629-1636.
- Goswami RP, Mondal S, Karmakar PS, Ghosh A (2012) Acidosis tubular renal tipo 3. Indio J Nephrol 22(6): 466-468.
- Siener R (2018) Dietary Treatment of Metabolic Acidosis in Chronic Kidney Disease 10(4): 512.
- Cupisti A, Dalessandro C, Gesualdo L, Cosola C, Gallieni M, et al. (2017) Non-Traditional Aspects of Renal Diets: Focus on Fiber, Alkali and Vitamin K1 Intake. Nutrients 9(5): 444.
- Torreggiani M, Fois A, Lippi F, Attini R, Longhitano E, et al. (2022) Plant-based diets for CKD patients: fascinating, trendy, but feasible? A green nephrology perspective. Clinical kidney journal 16(4): 647- 661.
- Kashoor I, Batlle D (2019) Proximal renal tubular acidosis with and without Fanconi syndrome. Kidney research and clinical practice 38(3): 267-281.
- Adomako E, Maalouf N (2023) Type 4 renal tubular acidosis and uric acid nephrolithiasis: Two faces of the same coin? Current Opinion in Nephrology and Hypertension 32 (2): 145-152.
- Manoocher Soleimani, Asghar Rastegar (2016) Pathophysiology of Renal Tubular Acidosis: Core Curriculum 2016, American Journal of Kidney Diseases 68(3): 488- 498.
- Mustaqeem R, Arif A (2022) Renal Tubular Acidosis. In: StatPearls. Treasure Island (FL): StatPearls.






























