The Effect of Intravenous Iron Replacement Therapy on Calcium, Phosphorus, and Parathormone Levels in Advanced-Stage Chronic Kidney Disease
Pinarbaşli Akçam Ö1, Tunca O2*, Kazan S2 and Dizen Kazan E3
1 Department of Internal Medicine, Afyonkarahisar State Hospital, Turkey
2 Department of Nephrology and Medicine, Afyonkarashisar Health Sciences University, Turkey
3 Department of Internal Medicine, Afyonkarahisar Health Sciences University, Turkey
Submission:March 13, 2023;Published: March 23, 2023
*Corresponding author: Tunca O, Department of Nephrology and Medicine, Afyonkarahisar Health Sciences University, Turkey
How to cite this article: Pinarbaşli Akçam Ö, Tunca O, Kazan S and Dizen Kazan E. The Effect of Intravenous Iron Replacement Therapy on Calcium, Phosphorus, and Parathormone Levels in Advanced-Stage Chronic Kidney Disease. JOJ Urology & Nephrology, 2023; 8(1): 555728. DOI: 10.19080/JOJUN.2023.08.555728
Abstract
Objectives: One of the most important problems that will affect the quality of life and even the progression of patients with chronic kidney disease (CKD) is the development of anemia. Intravenous iron replacement therapy is more frequently preferred in patients with CKD because of the problems of tolerance of the drugs and the often unsatisfactory treatment response. Intravenous iron replacement can cause hypophosphatemia and can affect mineral and bone disorders seen in CKD patients. Our study was planned to examine the effects of intravenous iron preparations on calcium, phosphorus, and parathormone levels, which are frequently used in treatment.
Materials and Methods: Thirty-two patients with advanced stage 4-5d CKD followed by Afyon Health Sciences University, Department of Nephrology were included in the study. The patients were divided into 2 groups those who were treated with intravenous iron carboxymaltose and those who were treated with intravenous iron sucrose. The changes in a total of 4 measurements made for calcium, phosphorus, and parathormone before and after iron replacement were examined within the groups.
Results: Groups were similar in terms of gender, age, CKD etiology, and comorbidities, laboratory parameters at the beginning of the study, urea, creatinine, eGFR, sodium, potassium, AST, ALT, hemoglobin, thrombocyte, calcium, phosphorus, and parathormone levels. In terms of pre-treatment and post-treatment calcium values, it was found that the calcium levels in the iron carboxymaltose group showed statistically significant variability (p<0.001), while the calcium values in the iron sucrose group were found to be similar (p= 0.351). It was determined that there was no statistically significant change in phosphorus and parathormone levels in both groups at 3-month follow-ups.
Conclusion: It is supported by studies in the literature and our study that IV iron treatments may have effects on calcium, phosphorus, and PTH levels in CKD patients. Randomized, controlled studies with larger participation are needed to demonstrate this effect.
Keywords: Calcium; Chronic Kidney Disease; Parathormon; Parenteral iron replacement, Phosphorus; Stage 4-5D
Abbreviations: CKD: Chronic Kidney Disease; ADPKD: Autosomal Dominant Polycystic Kidney Disease; DM: Diabetes Mellitus; CHF: Congestive Heart Failure; HT: Hypertension; CAD: Coronary Artery Disease; AST: Aspartate Transaminase; ALT: Alanine Transaminase; eGFR: estimatet Glomerular Filtration Rate; PTH: Parathormone
Introduction
Chronic kidney disease (CKD) is an important health problem that has become widespread all over the world. Anemia management is an important component of reducing morbidity and improving the quality of life in CKD. The goal of treating anemia and iron deficiency in patients with advanced CKD is to improve their symptoms and quality of life and potentially reduce their cardiovascular risk. Studies have shown that there is a direct link between iron deficiency anemia and quality of life and mortality. Guidelines advocate the importance of treating iron deficiency anemia in patients with CKD undergoing dialysis [1].
Oral iron preparations are frequently used in the treatment of anemia in CKD patients; however, intravenous (IV) iron replacement therapy has started to be preferred more frequently due to the tolerance problem of drugs and the unsatisfactory treatment response most of the time, especially in patients with advanced-stage CKD. KDIGO CKD-Anemia guideline recommends IV iron therapy for CKD patients with anemia and transferrin saturation below 30 in the anemia, and oral iron preparations can be used for 3 months as an alternative to IV iron therapy [2].
Calcium, phosphorus, and PTH management in advanced CKD patients form the basis of the management of mineral and bone disorders in these patients. In the KDIGO mineral and bone disorders guideline, it is recommended that serum calcium, phosphorus, and PTH levels be measured at regular intervals in patients with advanced CKD, and this triad should be evaluated together and kept at the targeted values [3]. There are studies in the literature showing that IV iron replacement may cause hypophosphatemia [4,5]. Many theories have been put forward on the mechanism of hypophosphatemia development of IV iron preparations. Although the exact mechanism is still unknown, the variable effect of different iron preparations on the intracellular metabolism of FGF-23 (Fibroblast Growth Factor), increased fractional phosphorus excretion, and decreased active vitamin D production is emphasized [6,7]. The effects of IV iron preparations on serum calcium, phosphorus, and PTH in patients with advanced CKD suggest that these molecules may play a role in the management of mineral and bone disorders as well as their use in the treatment of anemia.
The aim of this study is to compare the effects of IV iron replacement as iron carboxymaltose and iron sucrose on serum calcium, phosphorus, and PTH levels in patients with advanced CKD and anemia.
Methods
Patients and Groups
Laboratory tests of patients diagnosed with end-stage renal disease and diagnosed with stage 4-5-5D (peritoneal and hemodialysis patients) in the Nephrology Clinic of Afyon Health Sciences University Medical Faculty Hospital were examined. Demographic data and laboratory values of all patients were recorded.
Serum calcium, phosphorus, and parathormone (PTH) levels measured during routine follow-ups before IV iron therapy was compared with serum calcium, phosphorus, and PTH levels at 1 week, 1 month, and 3 months after iron therapy.
Statistical Analysis
Categorical variables are presented as percentages and frequency. The chi-square test was used for the comparison of categorical variables between the iron sucrose and iron carboxymaltose groups, and Fisher's exact test was used when appropriate. The conformity of continuous variables to normal distribution was checked with the Shapiro-Wilk test, Kolmogorov-Smirnov test, and visual histograms. Normally distributed continuous variables are presented as mean±standard deviation, and non-normally distributed continuous variables are presented as median and minimum-maximum values. Independent sample t-test or Mann-Whitney U test was used according to the presence of normal distribution in the comparison of continuous variables between groups. The changes in a total of 4 measurements made for calcium, phosphorus, and parathormone before and after iron replacement within the groups were evaluated with the Repeated-Measures test. Wilcoxon Signed Rank test was used to determine which measure the meaning originated from in group comparisons that were found to be significant. A pairwise comparisons test was used to determine which measurement resulted from the parameters with significant changes within the groups. Type 1 error level was taken as 5% and p<0.05 values were expressed as statistically significant. Analyzes were performed using SPSS 26.0 (IBM Corp. 2019 IBM SPSS Statistics for Windows, version 26.0. Armonk, NY: IBM Corp.).
Results
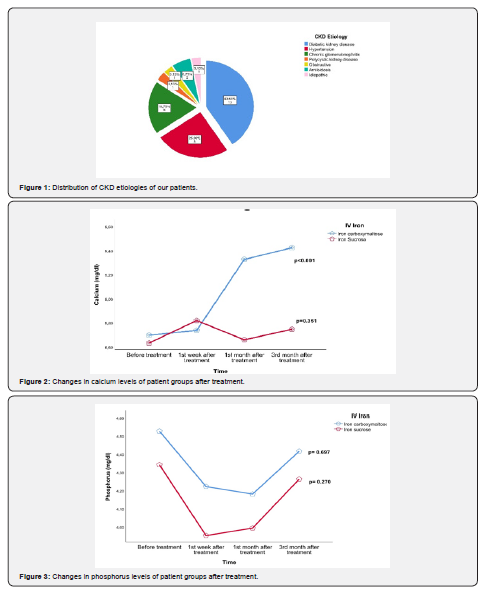
The study was conducted with 32 patients with advanced-stage kidney disease. 20 of the patients were women (62.5%) and 12 (37.5%) were men. The median age of the patients was found to be 63 years (min= 20 years, max=83 years). When the CKD etiologies of the patients were examined, it was found that diabetic nephropathy was the most common with 13 patients (40.6%), hypertensive nephropathy was the second with 8 patients (25%) and chronic glomerulonephritis was the third with 6 patients (18.8%) (Figure 1).
In terms of comorbidities, 14 (43.8%) of our patients had DM, 23 (71.9%) HT, 2 (6.3%) coronary artery disease, and 5 (15.6%) congestive heart failure. While 19 (59.4%) of our patients received iron sucrose treatment, 13 (40.6%) received iron carboxymaltose treatment.
It was found that the iron sucrose and iron carboxymaltose groups were similar to each other when compared in terms of gender, age, CKD etiology, and comorbidities.
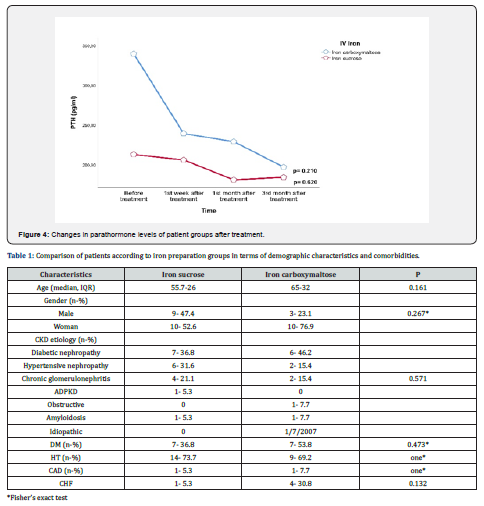
When calcium levels were examined before and after treatment, it was found that the calcium levels in the iron carboxymaltose group showed statistically significant variability (p<0.001), while the calcium values in the iron sucrose group were similar (p= 0.351) (Figure 2).
In the post-hoc analyzes for the iron carboxymaltose group, it was observed that the calcium level before the treatment and the calcium level in the first week of the treatment were similar. In addition, calcium levels were found to be similar in the 1st month of the treatment and the 3rd month of the treatment. However, all other measurements were found to be significantly different from each other.
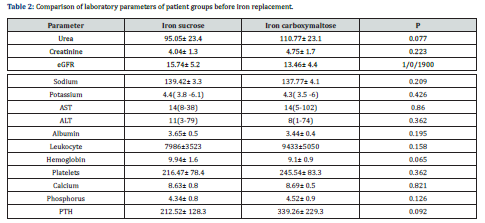
When the effects of iron replacement therapy on phosphorus were examined, it was found that there was no statistically significant change in phosphorus levels in both groups at 3-month follow-up (p= 0.270 for the iron sucrose group and p=0.697 for the iron carboxymaltose group) (Figure 3).
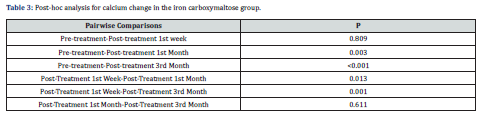
In the analyses performed for PTH, it was found that the parathormone values of both groups did not show significant variability (p= 0.620 for the iron sucrose group and p= 0.210 for the iron carboxymaltose group) (Figure 4) & (Table 1-3).
Discussion
In the etiology of chronic kidney disease, it is observed that DM and HT are the first two in our country, as in the whole world, and up to 65% of all causes of CKD are caused by these two diseases [8]. According to the results of our study, diabetic nephropathy was found to be the most common (40.6%), hypertensive nephropathy was the second (25%) and chronic glomerulonephritis was the third (18.8%) when looking at the etiologies of CKD patients.
Anemia is one of the leading causes of mortality and hospital admissions in CKD. The etiology of anemia in CKD lies mainly in the insufficiency of erythropoietin production. In order to obtain an adequate response to drugs that stimulate erythropoiesis, the adequacy of iron stores should be ensured, and the underlying iron deficiency anemia should be treated. Oral iron preparations have been replaced by IV iron therapy due to side effects and a lack of treatment for the treatment of iron-deficient anemia in people with CKD [9].
In our study, iron carboxy maltose and iron sucrose were included. There are numerous studies showing that asymptomatic and transient hypophosphatemia develops after IV iron treatment [5].
Many theories have been put forward on the mechanism of IV iron preparations for the development of hypophosphatemia. Although the exact mechanism is still unknown, the variable effect of different iron preparations on the intracellular metabolism of FGF-23 (Fibroblast Growth Factor 23), increased fractional phosphorus excretion, and decreased active vitamin D production is being focused on [6,7].
Some of the iron preparations have been compared in clinical trials; it has been suggested that the risk of hypophosphatemia may be lower compared to iron isomaltose than ferric carboxy maltose, but data from randomized trials comparing the two formulations directly are limited.
Myles Wolf et al. [10] examined the effects of iron isomaltose and ferric carboxy maltose on hypophosphatemia in two randomized clinical trials in which 554 patients, 123 were randomized to iron isomaltose and 122 to ferric carboxy maltose. 231 out of 245 people completed their trials. In this study, conducted in patients with iron deficiency anemia who cannot tolerate oral iron or do not respond to oral iron, iron isomaltose resulted in a lower incidence of hypophosphatemia for 35 days compared to ferric carboxy maltose. This study also provided mechanistic information about the role of FGF-23 in vitamin D metabolism in humans. The findings of this study show that ferric carboxy maltose activates this entire pathophysiological cascade by acutely increasing intact fibroblast growth factor 23 within 1 day. This was followed by increased urinary phosphate excretion and decreased 1,25-dihydroxycholecalciferol and ionized calcium, which were observed to accelerate secondary hyperparathyroidism, which maintains renal phosphate loss and hypophosphatemia even after intact FGF-23 returns to normal. Although the mechanism by which ferric carboxy maltose acutely elevates intact FGF-23 is unknown, it has been suggested that the carbohydrate carrier of iron in ferric carboxy maltose somehow prevents the cleavage of full-length fibroblast growth factor 23, which is normally upregulated in parallel with increased FGF-23 [10].
Eseoghene Ifie, Samson O Oyibo, Hareesh Joshi, and Olugbenro O Akintade explained the cause of hypophosphatemia developing after IV iron therapy as follows; Ferric carboxy maltose infusion therapy causes renal phosphate loss. It has been suggested that this iron compound increases serum levels of FGF-23 by inhibiting the degradation of FGF-23. FGF-23 is a peptide secreted by both osteoclasts and osteoblasts in response to elevated phosphate levels. Its function is to limit phosphate reabsorption in the renal tubules, maintaining phosphate homeostasis. Thus, the excess serum FGF-23 obtained causes hypophosphatemia by inappropriately reducing phosphate reabsorption in the proximal renal tubules, even in the presence of continued low serum phosphate levels. In addition, it has been reported that FGF-23 inhibits the production of calcitriol, which is necessary for phosphate absorption in the intestines [11]. Hypophosphatemia secondary to intravenous iron (particularly ferric carboxy maltose) has been reported, but this was generally considered to be both asymptomatic and transient. However, in this study, he pointed out that this side effect was not as asymptomatic and transient as previously thought. In another study, it was reported that 51% of patients receiving ferric carboxy maltose infusion developed hypophosphatemia, the severity of hypophosphatemia correlated with the dose administered, and the mean duration of hypophosphatemia was 6 months [12].
In this case reported by Eseoghene Ifie et al., low-dose and short-term (only 519 mg) intravenous iron was handled, and they showed that it caused both symptomatic and severe hypophosphatemia within a week [13].
In the multicenter study of Charytan et al. [14] with IV ferric carboxy maltose in the treatment of iron deficiency anemia in patients with CKD; The most common treatment-emergent adverse reactions in the ferric carboxy maltose group were nausea (4.4%), vomiting (2.9%), peripheral edema, arthralgia, dizziness, and hypertension (2.0% each), and edema (2.8%) in the iron sucrose or sodium ferric gluconate group. , peripheral edema, and hypertension (2.4% each). For subjects receiving iron sucrose or sodium ferric gluconate, the most common events were edema (4.0%), hypertension (3.2%), CHF, nausea, and fatigue. Hypophosphatemia was detected at a rate of 4.3% (8/186) after ferric carboxy maltose [14].
In our study, only stage 4-5-5d CKD patients were included and the effects of ferric carboxy maltose and iron sucrose preparations were examined. In our study, 32 patients were included, 19 (59.4%) of our patients received iron sucrose treatment, while 13 (40.6%) received iron carboxy maltose treatment. Patients in both groups were similar at the beginning of the study in terms of laboratory parameters and when compared in terms of gender, age, CKD etiology, and comorbidities. When the effects of iron replacement treatments on phosphorus were examined, it was found that the phosphorus level decreased at the end of the first week in both groups, but there was no statistically significant change in the 3-month follow-up.
In a meta-analysis including 19 randomized clinical trials in the literature published by Glaspy JA et al. in 2020, it was stated that all patients with hypophosphatemia associated with ferric carboxy maltose treatment were asymptomatic [15]. In our study, clinical findings such as fatigue, myalgia, rhabdomyolysis, and arrhythmia were not observed in any patient who developed hypophosphatemia.
We can summarize the pathophysiological mechanisms of IV iron-induced hypophosphatemia by looking at our own study as follows; Working through multiple feedback loops, PTH, 1,25- dihydroxycholecalciferol D [1,25(OH) 2 d] and FGF-23 regulate serum phosphate by modulating intestinal phosphate absorption, renal phosphate reabsorption, and bone metabolism. Malfunctioning of these feedback loops can cause various diseases. Primary increases in FGF23 cause hypophosphatemia and suppression of 1,25(OH)2D3 (1,25-dihydroxy-vitamin D3=calcitriol), leading to rickets or osteomalacia. Secondary increases in FGF-23 are one of the earliest indicators of decreased kidney function, and the magnitude of the FGF-23 elevation is strongly associated with and may contribute to an increased risk of future cardiovascular disease and death. Based on this study, the suppression of 1-25(OH) vitamin D due to the increase in FGF-23 may have played a role in the development of hypocalcemia in the iron carboxy maltose group.
The diagnosis of iron-induced hypophosphatemia requires a high level of alertness and suspicion and is based on the recognition of its temporal relationship with a new initiation of IV iron therapy [16]. In the patient group in our study, any side effects thought to be primarily related to IV iron were not reported by the patients.
Myles Wolf et al. [17] study and our study have similar features. However, FGF-23 and vitamin D levels were not measured in our study. In the study of Myles Wolf et al. [17] unlike ours, those who do not tolerate or respond insufficiently to oral iron; pregnant women and patients receiving dialysis treatment were excluded from the study. Patients in the USA, Canada, and Europe were included in the study between February 2016 and January 2017, and repeated measures of phosphate homeostasis in blood and urine were examined at baseline (week 0) and at weeks 1, 2, and 5 after baseline. The Ferumoxytol group maintained stable serum phosphate and urinary phosphate excretion throughout the trial. in the iron carboxymaltose group, serum phosphate gradually decreased from baseline to week 1 before reaching its lowest value at week 2 (mean difference between iron treatment groups in change from baseline to week 2, 7.3%; 95% CI 2.3–12.3; P = 0.004). Concomitantly, serum iFGF-23 approximately doubled at week 1 in the iron carboxymaltose group and then doubled again at week 2 and remained unchanged in the ferumoxymaltose group. Plasma cFGF-23 was significantly decreased in both groups. Compared with ferumoxymaltose, iron carboxymaltose caused significant decreases in serum 1,25-dihydroxycholecalciferol and calcium (mean difference between iron treatment groups in change from baseline to week 2, 0.37 mg/dl; 95% CI 0.25–0.50; P < 0.001 and There were significant increases in PTH, which peaked at week 5. Serum 25-hydroxyvitamin D was unchanged throughout the study in both groups, and when restricted to patients without CKD, all results were qualitatively similar [17].
This increase in PTH is explained as follows; There are studies showing that the production of C-terminal FGF-23 (cFGF23) in an inactive form in osteocytes increases in iron deficiency anemia, but the level of intact FGF-23 (iFGF-23), which is the biologically active form, does not change [6,17]. As a result of inhibition of differentiation of osteoblastic progenitor cells by iron replacement, production of FGF-23 from osteocytes, and disruption of the balance in decomposition into active/inactive form, cFGF-23 level decreases while iFGF-23 level increases. hypophosphatemia is observed [6,18,19]. In the study of Huang et al. with CKD patients, the iFGF-23/cFGF-23 ratio after DCM replacement was found to be 4.2±2.2 on day 2 and 3.0±1.8 on day 42. In addition, although not clearly explained, hypophosphatemia may develop as a result of the increase in tubular phosphorus excretion due to the increase in the amount of phosphorus transferred into the cell due to the rapidly increasing erythropoiesis after DCM replacement or due to the direct inhibition of NaPi2a and 2c cotransporters in the proximal tubule by IV iron therapy. In the study of Prats et al. conducted with 47 chronic kidney patients who were not on dialysis, it was shown that DCM treatment did not have a significant effect on 1,25-vitamin D and PTH levels [20]. This was explained by the suppression of 1,25-vit D production in the kidneys of iFGF23 in chronic kidney disease and its inhibitory effect on PTH secretion directly.
In our study, it was determined that the PTH values of both groups did not vary significantly in the analyzes performed for PTH in both groups. In terms of calcium values before and after treatment, it was found that calcium levels showed statistically significant variability in the iron carboxy maltose group, while calcium values were found to be similar in the iron sucrose group. In the analyzes performed for the iron carboxy maltose group, it was determined that the calcium level before the treatment and the calcium levels in the 1st week of the treatment and the calcium levels in the 1st month of the treatment and the 3rd month of the treatment were similar, while all other measurements were significantly different from each other.
One of the limitations of our study is that the FGF-23 level was not measured and a control group was not formed, in addition, the study period is short for long-term safety and efficacy evaluation. The amount of dietary phosphorus intake by patients during the study is unknown. In conclusion, ferric carboxy maltose and ferric sucrose are well-tolerated and fast-acting parenteral treatment options for the treatment of iron deficiency anemia in stage 4-5-5d chronic kidney disease patients. When electrolyte disorders, which are frequently asymptomatic and transient, are evaluated in terms of risk/benefit; It can be considered as an insignificant side effect that will not affect the treatment decision. There is a need for long-term studies with the participation of more patients on this subject.
Conclusion
It is supported by the studies in the literature and our study that IV iron treatments may have effects on calcium, phosphorus, and PTH levels in CKD patients. Randomized, controlled studies with larger participation are needed to demonstrate this effect.
References
- Zeidan A, Bhandari S (2017) Anemia İn Peritoneal Dialysis Patients; İron Repletion, Current and Future Therapies. Perit Dial Int 37(1): 6-13.
- Kidney Disease: Improving Global Outcomes (KDIGO) Anemia Work Group. KDIGO Clinical Practice Guideline for Anemia in Chronic Kidney Disease. Kidney inter 2: 279-335.
- Kidney Disease (2017) Improving Global Outcomes (KDIGO) CKD-MBD Update Work Group. KDIGO 2017 Clinical Practice Guideline Update for the Diagnosis, Evaluation, Prevention, and Treatment of Chronic Kidney Disease–Mineral and Bone Disorder (CKD-MBD). Kidney Int Suppl 7(1): 1-59.
- Emel Işıktaş Sayılar (2021) The İmpact Of Ferric Carboxymaltose Treatment on Serum Phosphorus Levels İn Chronic Kidney Disease, Ufuk Üniversitesi Tıp Fakültesi Dr. Rıdvan Ege Hastanesi, Nefroloji Bilim Dalı, Ankara, Turkey Cukurova Medical Journal 46(1): 266-272
- Schaefer B, Tobiasch M, Wagner S, Glodny B, Tilg H, et al. (2022) Hypophosphatemia after intravenous iron therapy: Comprehensive review of clinical findings and recommendations for management. Bone 154: 116202.
- Wolf M, Koch TA, Bregman DB (2013) Effects of İron Deficiency Anemia And İts Treatment on Fibroblast Growth Factor 23 and Phosphate Homeostasis İn Women. J Bone Miner Res 28: 1793-1803.
- Wolf M, White KE (2014) Coupling Fibroblast Growth Factor 23 Production and Cleavage: İron Deficiency, Rickets, and Kidney Disease. Curr Opin Nephrol Hypertens 23: 411-419.
- Seyahi N, Ateş K, Süleymanlar G (2017) Türkiye’de Renal Replasman Tedavilerinin Güncel Durumu: Türk Nefroloji Derneǧi Kayit Sistemi 2016 Yili Özet Raporu. Turkish Nephrol Dial Transplant J 26(2): 154-160.
- Agarwal R, Rizkala AR, Bastani B, Kaskas MO, Leehey DJ, et al. (2006) A Randomized Controlled Trial of Oral Versus İntravenous İron İn Chronic Kidney Disease. Am J Nephrol 26: 445-454.
- Wolf M, Rubin J, Achebe M, Econs MJ, Peacock M, et al. (2020) Effects of Iron Isomaltoside vs Ferric Carboxymaltose on Hypophosphatemia in Iron-Deficiency Anemia: Two Randomized Clinical Trials. JAMA 323(5): 432-443.
- Shimada T, Mizutani S, Muto T, Yoneya T, Hino R, Takeda S, Takeuchi Y, et al. (2001) Cloning and characterization of FGF23 as a causative factor of tumor-induced Osteomalacia. Proc Natl Acad Sci U S A. 98(11): 6500-6505.
- Hardy S, Vandemergel X (2015) Intravenous iron administration and hypophosphatemia in clinical practice. Int J Rheumatol 2015: 468675.
- Ifie E, Oyibo SO, Joshi H, Akintade O (2019) Symptomatic hypophosphatemia after intravenous iron therapy: an underrated adverse reaction. Endocrinol Diabetes Metab Case Rep 2019(1): 19-0065.
- Charytan C, Bernardo MV, Koch TA, Butcher A, Morris D, et al. (2013) Intravenous Ferric Carboxymaltose Versus Standard Medical Care İn The Treatment Of İron Deficiency Anemia İn Patients with Chronic Kidney Disease: A Randomized, Active controlled, Multi-Center Study. Nephrol Dial Transplant 28: 953-664.
- Tossani E, Cassano P, Fava M (2005) Depression and renal disease. Semin Dial 2: 73-81
- Glaspy JA, Wolf M, Strauss WE (2021) Intravenous Iron-Induced Hypophosphatemia: An Emerging Syndrome. Adv Ther 38(7): 3531-3549.
- Wolf M, Chertow GM, Macdougall IC, Kaper R, Krop J, et al. (2018) Randomized trial of intravenous iron-induced hypophosphatemia. JCI Insight 3(23): e124486.
- Prats M, Font R, García C, Cabré C, Jariod M, et al. (20013) Effect of ferric carboxymaltose on serum phosphate and C-terminal FGF23 levels in non-dialysis chronic kidney disease patients: post-hoc analysis of a prospective study. BMC Nephrol 14: 167.
- Schouten BJ, Hunt PJ, Livesey JH, Frampton CM, Soule SG (2009) FGF23 Elevation and Hypophosphatemia After İntravenous İron Polymaltose: A Prospective Study. J Clin Endocrinol Meta. 94: 2332-23327.
- Lazarus JM, Brenner BM, Chronic Renal Failure. Fauci AS, Braunwald E, Isselbacher KJ, Wilson JD, Martin JB, Kasper DL, Hauser SL, Longo DL (editors): Harrison's Principles of Internal Medicine. 14th The McGraw-Hill Companies, Inc USA. 1998; 1513-1520.






























