Comparison of Proteinuria and Glycemic Parameters According to Dipping Status in Patients with Hypertension and Diabetic Kidney Disease
Tunca O1*, Kazan S1 and Dizen Kazan E2
1Department of Nephrology, Faculty of Medicine, Afyonkarahisar Health Sciences University, Turkey
2Department of Internal Medicine, Faculty of Medicine, Afyonkarahisar Health Sciences University, Turkey
Submission:February 14, 2022;Published:February 24, 2022
*Corresponding author:Tunca O, Department of Nephrology, Faculty of Medicine, Afyonkarahisar Health Sciences University, Mavi Hastane, F Blok, Diyaliz Ünitesi, Merkez/Afyonkarahisar/Turkey.
How to cite this article:Tunca O*, Kazan S and Dizen Kazan E. Comparison of Proteinuria and Glycemic Parameters According to Dipping Status in Patients with Hypertension and Diabetic Kidney Disease. JOJ Urology & Nephrology, 2023; 7(5): 555726. DOI: 10.19080/JOJUN.2023.07.555726
Abstract
Objective: In this study, we aimed to compare glycemic regulation parameters and proteinuria in patients with hypertension and diabetic kidney disease (DKD) in terms of 24-h ambulatory blood pressure monitoring (ABPM) groups.
Material-Methods: We retrospectively scanned all patients with DKD and who have ABPM for any reason. Patients were divided into three groups; dippers, non-dippers, and reverse-dippers. Groups were compared in terms of glycemic regulation parameters and proteinuria.
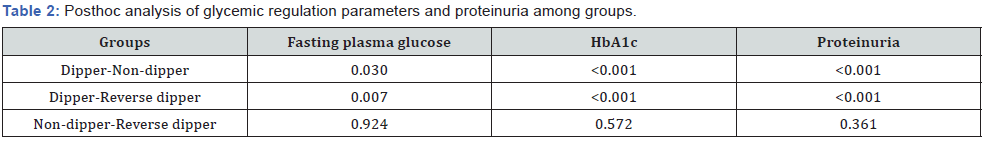
Results: The study included 126 patients with DKD and 24-h ABPM. The median age of the patients was 58 years. Non-dippers and reverse-dippers had higher fasting plasma glucose (FPG) (p= 0.030 and p= 0.007, respectively), HbA1c (both p<0.001), and proteinuria (both p<0.001) than dippers. Non-dippers and reverse-dippers were similar in terms of FPG, HbA1c, and proteinuria (p= 0.924, p= 0.572, and p= 361, respectively).
Conclusion: We found that non-dipping and reverse-dipping statuses are accompanied by poor glycemic regulation and higher proteinuria. Effective control of the shared pathophysiological processes that lead to this condition can improve both conditions.
Keywords: Diabetic kidney disease; Fasting plasma glucose; HbA1c; Proteinuria
Introduction
Diabetic Kidney Disease (DKD) is the leading cause of end-stage kidney disease [1]. The first sign of DKD is moderately increased albuminuria. It is recommended that patients with type 2 diabetes should be screened for DKD at regular intervals from the time of diagnosis [2]. Detection of microalbuminuria and proteinuria in spot urine is the most commonly used DKD screening method [3]. Hypertension is one of the most common comorbidities accompanying type 2 diabetes. Studies in the literature show that the prevalence of hypertension in patients with type 2 diabetes varies between 30-75% [4,5]. On the other hand, the prevalence of diabetes is higher in patients with hypertension compared to those without hypertension [6].
There are several modifiable and non-modifiable risk factors contributing to DKD. Whereas age, age of onset, sex, and genetics are non-modifiable risk factors, smoking, control of blood pressure, and glycemic control are the most known modifiable risk factors for DKD [7]. Hypertension and diabetes show a synergistic effect in the development of DKD [8]. This synergistic effect is the reason why hypertension guidelines set lower blood pressure targets for patients with diabetes. Effective control of hypertension is one of the most important modifiable causes in the course of DKD. In the ADVANCE (Action in Diabetes and Vascular disease: preterAx and diamicroN-MR Controlled Evaluation) study, the incidence of microalbuminuria and macroalbuminuria was reduced by 21% in the group taking angiotensin-converting enzyme inhibitors [9]. In diabetic and hypertensive patients, the incidence of abnormal dipping patterns increases in 24-hour ambulatory blood pressure monitoring (ABPM) [10]. Abnormal dipping patterns may also have an impact on the DKD stage and glycemic regulation.
In this study, we aimed to compare DKD stages and glycemic regulation parameters according to dipping status in patients with hypertension and DKD.
Methods
Patients and Groups
The files of all patients with type 2 diabetes mellitus and hypertension, who were followed up in our hospital’s nephrology outpatient clinic and were admitted between January 2015 and January 2022, were retrospectively scanned (n= 895). Patients with suspected primary glomerular disease (hypoalbuminemia, edema, >3.5 g/day proteinuria) and patients with urinary tract infection findings during urinalysis were excluded from the study. All DKD patients who had 24-hour ABPM for any reason between the specified dates were included in the study (n= 126). According to the amount of nighttime decrease in systolic blood pressure; the patients were divided into 3 groups dippers (who had a nighttime systolic blood pressure decline >10%), nondippers (who had a nighttime systolic blood pressure decline of 0-10%) and reverse dippers (who had a nighttime systolic blood pressure increase). The groups were compared in terms of glycemic regulation parameters and proteinuria.
Assessment of DKD
While evaluating albuminuria, the rates of albumin and creatinine excretion in the spot urine of the patients performed at 3 different times were recorded. The presence of DKD was determined by taking the average of these three measurements. Patients with albuminuria greater than 30 mg/g in the spot urine and who were using angiotensin-converting enzyme inhibitors or angiotensin receptor blockers due to albuminuria were considered DKD patients.
24-hours ABPM
A 24-hour ABPM was performed with an FDA (Food and Drug Administration) verified device (Cardioline monitor [Walk200b model] and Cardioline blood-pressure cuff). In the daytime; blood pressures were recorded every 30-min period (from 08:00 AM to 11:59 PM), and in the nighttime; blood pressures were recorded every 60-min period (from 00:00 PM to 7:59 AM). If the first measurement failed, the measurement was repeated once again and measurements were accepted only when at least 75% of the measurements were successful. Values outside of normal physiological ranges and spurious readings due to motion artifacts were automatically edited by the analysis software.
Statistical Analysis
Statistical analyzes were done with SPSS 26.0 (IBM Corporation 2019 IBM SPSS Statistics for Windows, version 26.0. Armonk, NY: IBM Corp) package program. Categorical variables were presented as percentages and frequencies. The conformity of continuous variables to normal distribution was checked with visual histograms, the Shapiro-Wilk test, and the Kolmogorov-Smirnov test. Normally distributed continuous variables were presented as mean±standard deviation, and nonnormally distributed continuous variables were presented as median (min-max). ANOVA test was used when comparing the normally distributed continuous variable between the groups. Continuous variables that were not normally distributed were compared between groups using the Kruskal-Wallis test. A pairwise comparisons test was used for post hoc analysis of the Kruskal-Wallis test and a Bonferroni correction was used for post hoc analysis of the ANOVA test. All presented p-values are bidirectional and p<0.05 is considered statistically significant.
Ethics Approval
Ethical committee approval was received from the Ethics Committee of Afyonkarahisar Health Sciences University (Date: Feb 03, 2023, Meeting No: 2023/2 Decision No: 66).
Results
This study was conducted with 126 DKD patients. Of the patients 56.3% (n= 71) were males. The median age of the patients was 58 years (Range= 30-81 years). According to ABPM results, 44.4% (n= 56) of the patients were in the non-dipping group, 34.9% (n= 44) were in the dipping group, and 20.6% (n= 26) were in the reverse-dipping group. Table 1 shows the comparison of the characteristic features of the patients in terms of ABPM groups.
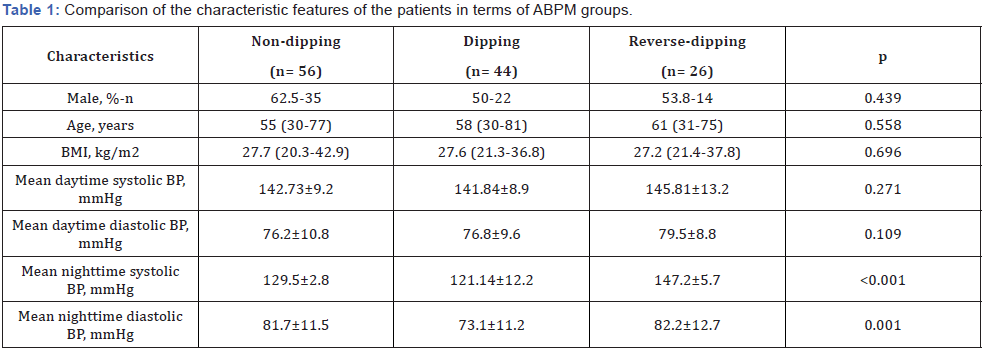
There was a statistically significant difference between groups in terms of fasting plasma glucose (FPG), HbA1c, and proteinuria (Figure 1-3).
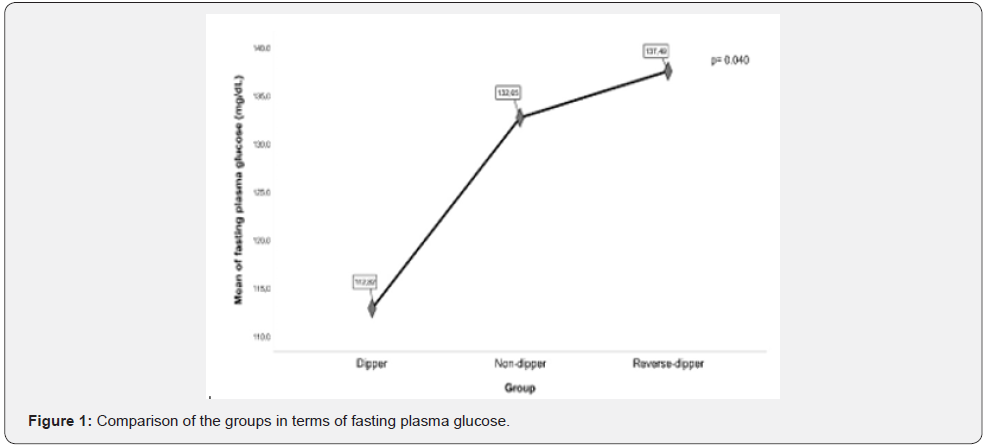
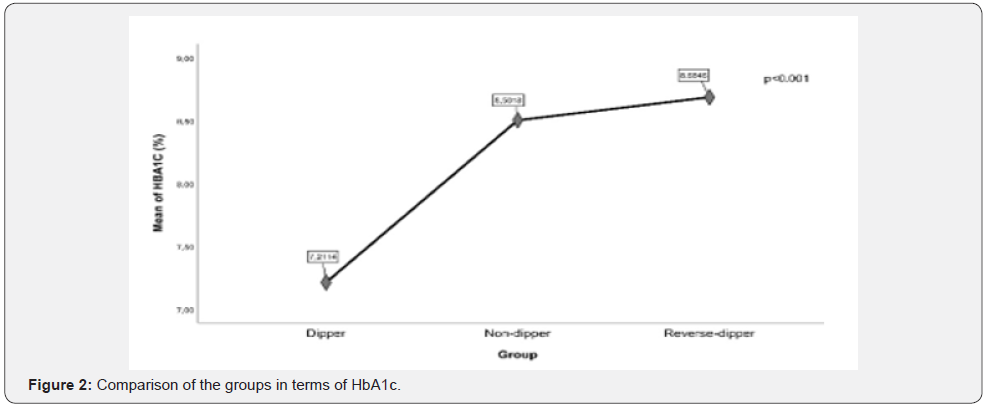
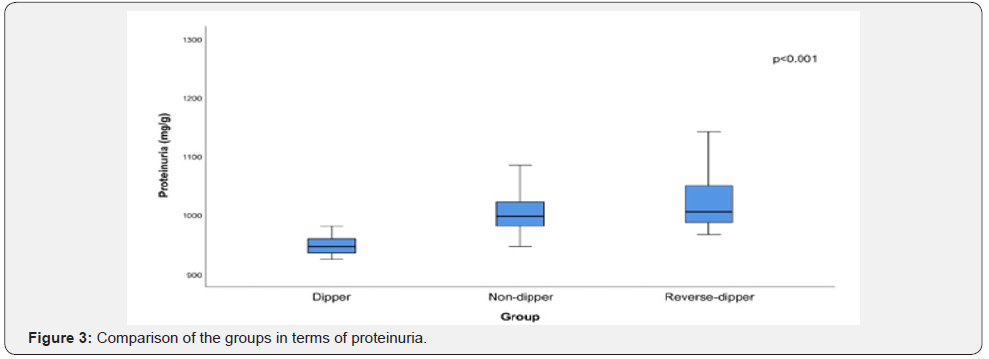
In the post-hoc analysis, it was determined that the difference between the groups was due to the fact that FPG, HbA1c, and proteinuria were lower in the dipping group than in the other two groups. The nondipping group and the reverse-dipping group were similar to each other in terms of FPG, HbA1c, and proteinuria (Table 2).
Discussion
The present study showed that patients with DKD and dipping blood pressure status have better glycemic control and less proteinuria than patients with DKD and non-dipping or reverse-dipping statuses. In a recent study, the non-dipping pattern was found to be a risk factor for kidney disease and mortality in patients with type 1 diabetes mellitus [11]. The situation is similar in patients with type 2 diabetes, and cardiovascular mortality and renal involvement are increased in patients without a dipping pattern [12-14]. It is assumed that the decrease in vascular tone and nocturnal dipping occurs as a result of the decrease in sympathetic discharge and the prominence of parasympathetic activity during sleep [15]. Li et al. [16] found that non-dipping status is common in DKD and it is correlated with 24-h protein excretion. 24-hour urine collection is not practical and is not a standard method for outpatients. In our study, albumin excretion in spot urine was found to be higher in patients without a dipping pattern than in patients with a dipping pattern. According to the findings of our study, it can be investigated whether there is a non-dipping pattern or a reverse dipping pattern in DKD patients with higher proteinuria.
While insulin, one of the endogenous hormones, takes the lead in glycemic regulation, almost all other hormones have an effect on raising blood glucose. High levels of stress hormones such as catecholamines and cortisol are among the factors that prevent dipping status in non-dipper and reverse-dipper patients [16,17]. The negative effects of these hormones on glycemic regulation may be a factor that complicates glycemic regulation in patients with impaired dipping patterns. Studies have shown that non-dipping status and impaired glycemic regulation are commonly accompanied in diabetic patients, and cardiovascular outcomes are poor in these patients [18-19]. This study also showed that non-dipping and reverse dipping patterns are accompanied by poor glycemic regulation and higher proteinuria in patients with type 2 diabetes mellitus.
While insulin, one of the endogenous hormones, takes the lead in glycemic regulation, almost all other hormones have an effect on raising blood glucose. High levels of stress hormones such as catecholamines and cortisol are among the factors that prevent dipping status in non-dipper and reverse-dipper patients [16,17]. The negative effects of these hormones on glycemic regulation may be a factor that complicates glycemic regulation in patients with impaired dipping patterns. Studies have shown that non-dipping status and impaired glycemic regulation are commonly accompanied in diabetic patients, and cardiovascular outcomes are poor in these patients [18-19]. This study also showed that non-dipping and reverse dipping patterns are accompanied by poor glycemic regulation and higher proteinuria in patients with type 2 diabetes mellitus.
The limitations of our study are that it was conducted with a limited number of patients for a common disease such as diabetes, that it was single-centered and retrospective, and that other diabetic complications such as neuropathy were not examined. Our study reaffirms the importance of 24-h ABPM measurement in DKD. Although the duration of action of antihypertensive drugs can be prolonged up to 24 hours, glycemic regulation and proteinuria can be kept under control more effectively with an antihypertensive treatment that can mimic the normal circadian rhythm.
Conclusion
We found that non-dipping and reverse-dipping statuses are accompanied by poor glycemic regulation and higher proteinuria. We think that there are several shared pathophysiological processes causing this accompaniment. Effective control of these shared pathophysiological processes that lead to this condition can improve both conditions.
Author Contributions
Tunca O, Kazan S, and Dizen Kazan E contributed to the design, implementation of the research, and to writing of the manuscript. Statistical analysis was done by Kazan S.
References
- Ghaderian SB, Hayati F, Shayanpour S, Beladi Mousavi SS (2015) Diabetes and end-stage renal disease; a review article on new concepts. J Renal Inj Prev 4(2): 28-33.
- American Diabetes Association Professional Practice Committee; Draznin B, Aroda VR, Bakris G, Benson G, Brown FM, Freeman R, Green J, Huang E, Isaacs D, Kahan S, Leon J, Lyons SK, Peters AL, Prahalad P, Reusch JEB, Young-Hyman D. 11. Chronic Kidney Disease and Risk Management: Standards of Medical Care in Diabetes-2022. Diabetes Care 2022; 45(Suppl 1): S175-S184.
- Kramer H (2005) Screening for kidney disease in adults with diabetes and prediabetes. Curr Opin Nephrol Hypertens 14(3): 249-252.
- Baskar V, Kamalakannan D, Holland MR, Singh BM (2006) Does ethnic origin have an independent impact on hypertension and diabetic complications? Diabetes Obes Metab 8(2): 214-219.
- Centers for Disease Control and Prevention. 2007 National diabetes fact sheet.
- Bachir Cherif A, Bennouar S, Bouamra A, Taleb A, Hamida F, et al. (2018)Prevalence of diabetes and dyslipidemia in hypertensive patients in the area of Blida (Algeria). Ann Cardiol Angeiol (Paris) 67(3): 198-203.
- Harjutsalo V, Groop PH (2014) Epidemiology and risk factors for diabetic kidney disease. Adv Chronic Kidney Dis 21(3): 260-266.
- Wang Z, do Carmo JM, Aberdein N, Zhou X, Williams JM, et al. (2017) Synergistic Interaction of Hypertension and Diabetes in Promoting Kidney Injury and the Role of Endoplasmic Reticulum Stress. Hypertension 69(5): 879-891.
- Patel A; ADVANCE Collaborative Group; MacMahon S, Chalmers J, Neal B, et al. (2007) Effects of a fixed combination of perindopril and indapamide on macrovascular and microvascular outcomes in patients with type 2 diabetes mellitus (the ADVANCE trial): a randomized controlled trial. Lancet 370(9590): 829-840.
- Nandhini H (2022) Abnormal Dipping Pattern of Blood Pressure in Diabetics-A Study. J Assoc Physicians India 70(4): 11-12.
- Hjortkjær HO, Persson F, Theilade S, Winther SA, Tofte N, et al. (2022) on-dipping and higher nocturnal blood pressure are associated with risk of mortality and development of kidney disease in type 1 diabetes. J Diabetes Complications 36(9): 108270.
- Sturrock ND, George E, Pound N, Stevenson J, Peck GM, et al. (2000) Non-dipping circadian blood pressure and renal impairment are associated with increased mortality in diabetes mellitus. Diabet Med 17(5): 360-364.
- Chiriacò M, Pateras K, Virdis A, Charakida M, Kyriakopoulou D, et al. (2019) Association between blood pressure variability, cardiovascular disease and mortality in type 2 diabetes: A systematic review and meta-analysis. Diabetes Obes Metab 21(12): 2587-2598.
- Ayala DE, Moyá A, Crespo JJ, Castiñeira C, Domínguez-Sardiña M, et al. (2013) Hygia Project Investigators. Circadian pattern of ambulatory blood pressure in hypertensive patients with and without type 2 diabetes. Chronobiol Int 30(1-2a): 99-115.
- Birkenhäger AM, van den Meiracker AH (2007) Causes and consequences of a non-dipping blood pressure profile. Neth J Med 65(4): 127-131.
- Lapiński M, Lewandowski J, Januszewicz A, Kuch-Wocial A, Symonides B, et al. (1993) Hormonal profile of dipper and non-dipper patients with essential hypertension. J Hypertens Suppl 11(5): S294-S295.
- Därr R, Bursztyn M, Pamporaki C, Peitzsch M, Siegert G, et al. (2016) Dipping in Ambulatory Blood Pressure Monitoring Correlates With Overnight Urinary Excretion of Catecholamines and Sodium. J Clin Hypertens (Greenwich) 18(9): 921-926.
- Sowers JR, Epstein M, Frohlich ED (2001) Diabetes, hypertension, and cardiovascular disease: an update. Hypertension 37(4): 1053-1059.
- Kimura G, Dohi Y, Fukuda M (2010) Salt sensitivity and circadian rhythm of blood pressure: the keys to connect CKD with cardiovascular events. Hypertens Res 33(6): 515-520.






























