Robotic Cystectomy with Intracorporeal Urinary Diversion is Associated with Decreased Narcotic Consumption
Charles M Ellis1, Emily Roebuck1, Jiaxian He2, Megan Mcdonald2, Hamza Beano1, William Worrilow1, Alexander Sinks1, Peter E Clark1, Kris E Gaston1, James T Kearns1 and Stephen B Riggs1*
1Department of Urology, Levine Cancer Institute/Atrium Health, Charlotte, USA
2Department of Cancer Biostatistics, Levine Cancer Institute/Atrium Health, Charlotte, USA
Submission:January 30, 2023;Published:February 14, 2023
*Corresponding author:Stephen B Riggs, Department of Urology, Levine Cancer Institute/Atrium Health, Charlotte, USA
How to cite this article:Charles M Ellis, Emily Roebuck, Jiaxian He, Megan Mcdonald, Hamza Beano, William Worrilow, Alexander Sinks, Peter E Clark, Kris E Gaston, James T Kearns and Stephen B Riggs. Robotic Cystectomy with Intracorporeal Urinary Diversion is Associated with Decreased Narcotic Consumption. JOJ Urology & Nephrology, 2023; 7(5): 555725. DOI: 10.19080/JOJUN.2023.07.555725
Abstract
Introduction: Improvements in robotic technology and surgeon experience have made intracorporeal ileal conduit urinary diversion (ICUD) following robot-assisted radical cystectomy (RARC) a reality. Thus, it is essential to understand the benefits and drawbacks to this technically challenging approach. This study investigates surgical and pain-related perioperative outcomes in patients who underwent ICUD compared to extracorporeal urinary diversion (ECUD) at a tertiary care center.
Methods: We reviewed a prospectively maintained database of 198 patients who underwent robot-assisted radical cystectomy between January 2015 and October 2020. Surgical, functional, and pain-related perioperative outcomes were compared between 177 patients who received either ICUD or ECUD.
Results and Conclusion: Among 177 patients, 107 (60.4%) patients underwent ECUD and 70 (39.6%) underwent ICUD. Patients who received ICUD had significantly lower narcotic consumption (P=.031). There were no significant differences across cohorts in operative time (P=.10), estimated blood loss (P=.958), blood transfusion rate (P=.267), overall survival (P=.485), LOS (P=.652), or 30-day readmissions (P=.080). Our study suggests that RARC with intracorporeal ileal conduit reduces post-operative narcotic use as compared to extracorporeal diversion. Further validation across multiple institutions will be required to elucidate the full advantage and/or disadvantage to this approach.
Keywords: Radical cystectomy; Intracorporeal urinary diversion; Robot-assisted; Perioperative outcomes; Narcotics
Introduction
Radical cystectomy with extended pelvic lymphadenectomy is the standard of care treatment for muscle-invasive and high-grade non-muscle-invasive bladder cancer at high risk for progression [1]. With advancing technology, robot-assisted radical cystectomy (RARC) has emerged as an increasingly utilized approach, offering the surgical and postoperative advantages of minimally invasive surgery while maintaining comparable oncologic efficacy to the open surgery [2-5]. In the context of RARC, extracorporeal urinary diversion (ECUD) has been the mainstay method for urinary diversion as a completely intracorporeal procedure involves a steeper learning curve [6]. Nonetheless, improvements in robotic technology and surgeon experience have led to exploration of an intracorporeal urinary diversion (ICUD), offering potential benefits as an entirely minimally invasive approach relative to ECUD [7,8]. Previous studies have compared outcomes in robotic ICUD versus ECUD, though they have primarily focused on acute perioperative measures. Available literature has suggested both benefits and drawbacks to ICUD including shorter operative time and less blood loss, yet higher complications rates; however, results have been inconsistent [9-13]. There remains a paucity of literature evaluating pain-related measures such as pain scores and narcotic use, which are significant potential advantages to completely minimally invasive surgery and, given the current opioid crisis, beg to be explored.
In recent years, there has been a concerted effort among surgeons to combat the opioid epidemic [14] Oxycodone and hydrocodone represent over 80% of opioid prescriptions written after surgery and are also among the most common culprits in opioid-related overdoses which have nearly tripled in frequency since 1999 [15,16]. Studies have shown that even opioid-naïve patients are at risk for persistent postoperative opioid use [17,18]. In response to the current crisis, surgeons across fields have adopted various tools and techniques to minimize narcotic consumption, combatting the opioid epidemic within their own realm of influence [19-21]. As ICUD is less invasive than ECUD, we hypothesized that ICUD in the setting of a dedicated Enhanced Recovery After Surgery (ERAS) protocol driving post-operative pain management would reduce postoperative pain and narcotic consumption, offering urologists another valuable tool to combat this epidemic and contribute to improvement on a broader scale.
Understanding the challenges and advantages of performing a robot assisted ICUD will inform whether its learning curve is worthwhile. Given the lack of literature around pain and narcotic use comparing ICUD versus ECUD, we sought to assess perioperative outcomes in ICUD and ECUD, comparing surgical, functional, and pain-related measures.
Materials and Methods
Patients
We retrospectively analyzed our prospectively maintained, Institutional Review Board approved, database of patients undergoing RARC between January 2017 and October 2020. The data were obtained from our automated perspective data collection that is supplemented with manual data entry and housed in Redcap. We included all adult patients (age > 18 years) who underwent RARC with ileal conduit diversion. We excluded patients who underwent cystectomy for benign disease and patients who underwent concurrent nephrectomy.
Technique
Surgery was performed by two experienced, fellowship-trained urologic oncology surgeons (SR and KG) who currently diverge in their approach to ileal conduit urinary diversion (ICUD vs ECUD). Our surgical technique for RARC utilizes 6 ports (4 robotic and 2 assistant) and is performed in a similar fashion between surgeons. The suprapubic ports measured 15 mm in length. The cephalad extent of the lymph node dissection is the aortic bifurcation. ICUD and ECUD use a running technique for the ureteroenteric anastomosis. Following the removal of the bladder and lymph nodes for RARC ECUD patients, the robot was undocked, and the specimen was retrieved through extension of the camera port via a 5–7 cm peri or sub-umbilical midline incision. Urinary diversion with an ileal conduit was then performed extracorporeally. The ureteroenteric anastomosis was performed with 5-0 dissolvable monofilament suture in a combined interrupted and running fashion. In the case of ICUD, all procedures, including isolation of the ileal segment and uretero-enteric anastomosis, were performed intracorporeally. The specimen for intracorporeal diversion is removed through extension of the suprapubic port that is used for the stapled bowel anastomosis in male patients and through the vagina in female patients.
In April 2018, we instituted intraoperative transversus abdominis plane (TAP) blocks using liposomal bupivacaine, 0.25% bupivacaine and normal saline. TAP blocks were performed by each surgeon using the same technique. Recognizing TAP block as a potential covariate, we adjusted for this difference in our analyses as described below. Additionally, since January 1st 2017, all patients undergoing cystectomy at our center were enrolled in an ERAS program, which standardizes many facets of patient care, including analgesic administration from preoperative preparation through to post-operative follow up after discharge. The postoperative analgesic regimen consists of scheduled acetaminophen with oxycodone and hydromorphone administered on an as-needed basis. Due to the protocolized nature of the ERAS program, the provision of as needed narcotics in the postoperative period is largely nurse-directed, with little to no influence from individual surgeons.
Statistical Analysis
Patient baseline characteristics including demographics, ASA score, Charlson Comorbidity Index (CCI), body mass index (BMI), tumor clinical stage, history of pelvic radiation, and neoadjuvant chemotherapy were compared between ICUD and ECUD cohorts via descriptive statistics. Corresponding p-values were determined using Fisher’s exact test for categorical variables, and two-sample t-tests or non-parametric Mann-Whitney U tests were used for continuous variables. All patients were censored at time of death or last follow-up to calculate overall survival. The Kaplan-Meier method was utilized to estimate the probabilities of overall survival, and the differences between ICUD and ECUD were assessed via a log-rank test. Cox regression model was used to analyze risk factors for overall survival. Possible risk factors included surgery approach (ICUD vs ECUD), patient demographics, neoadjuvant chemo, pathologic stage, and TAP block. Postoperative complications included those occurring within 30 days of surgery. Univariate and multivariate logistic regression models were performed to evaluate 30-day readmission (adjusted for TAP block) and postoperative complications as a function of surgery approach (ICUD vs ECUD) and patient baseline characteristics. Other categorical outcomes such as blood transfusion, ureteral anastomotic stricture, and hernia were analyzed using the same method. A verbal numeric rating scale (NRS) 0-10, which is administered by nurses at regular intervals throughout a patient’s stay, was used to measure pain scores for each patient on postoperative day (POD) 1-3. Average pain score was summarized for each patient and a linear regression model was performed to analyze average pain score adjusted for TAP block. The same method was used to determine risk factors associated with continuous outcomes such as length of stay, operative time, EBL, and lymph nodes removed. Daily narcotic dosing was summed across dosing routes to calculate a morphine milligram equivalent dosing (MME). Total narcotic use was logtransformed (log[x+1]) to help reduce data variability and meet the normality model assumption. Risk factors associated with narcotic use were investigated using a linear regression model. All regression analyses were performed via a stepwise model selection procedure, where all covariates were first included in the univariate analysis, and covariates with P-value < 0.1 were entered in the multivariate model and only covariates with P-value < 0.05 were retained in the final model. All statistical tests were two sided, and a P-value < 0.05 was considered to indicate statistical significance.
Results
Analysis included 177 bladder cancer patients following application of inclusion/exclusion criteria. Of the 177 patients, 107 (60.4%) patients underwent ECUD and 70 (39.6%) underwent ICUD. Patient baseline characteristics were similar with regards to gender, age, race, body mass index (BMI), American Society of Anesthesiologists (ASA) score, history of pelvic radiation or neoadjuvant chemotherapy, pre-operative clinical stage and Charleston co-morbidity index (CCI) (Table 1, P>.05).
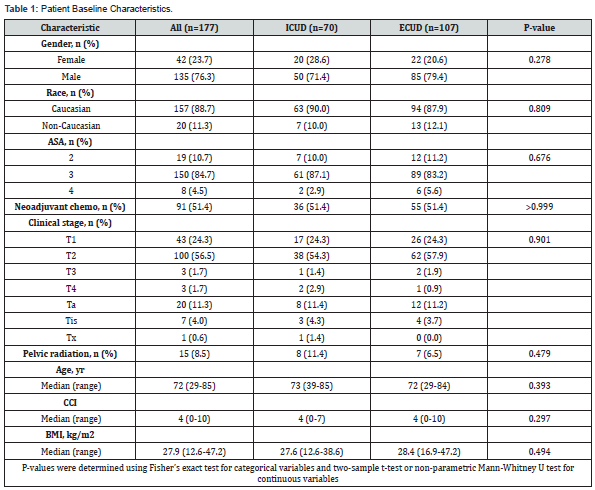
There were no differences between ICUD and ECUD in operative times (beta=-19.1, 95%CI [-41.96, 3.71], P=.10), estimated blood loss (beta=-2.23, 95%CI [-85.14, 80.69], P=.958), blood transfusion rate (OR=0.59, 95%CI [0.23, 1.5], P=.267), and lymph node yield (beta=1.95, 95%CI [-1.04, 4.9], P<.195) (Table 2). Additionally, 30-day readmissions (OR=0.55, 95%CI [0.29, 1.07] P=.080) and LOS (beta=-.33, 95%CI [-1.79, 1.12], P=.652) were not significantly different across ICUD and ECUD cohorts.
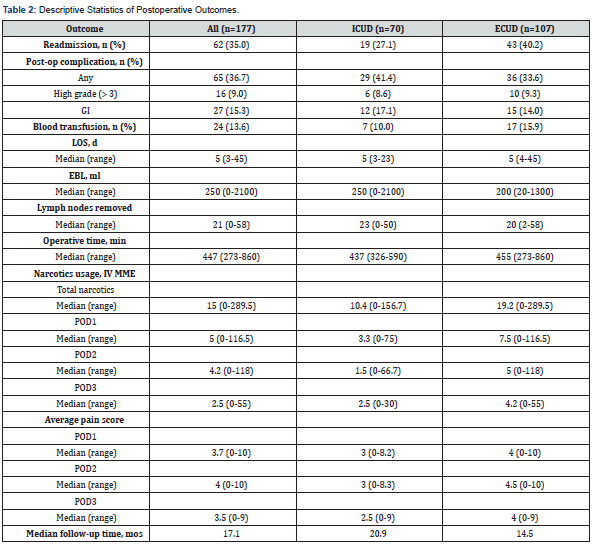
In the multivariate Cox model, no significant difference in OS was found between ICUD and ECUD cohorts (HR=0.52, 95%CI [0.24, 1.13], P=.485). Pathologic stage (stage 0 vs III-IV: HR=0.19, 95%CI [0.08, 0.47], P<.001, Stage I-II vs III-IV: HR=0.29, 95%CI [0.12, .7], P=.006), gender (HR=2.45, 95%CI [1.03, 11.53], P=.044), and pelvic radiation (HR=3.25, [1.3, 8.13], P=.012) were significantly associated with OS (Table 3).
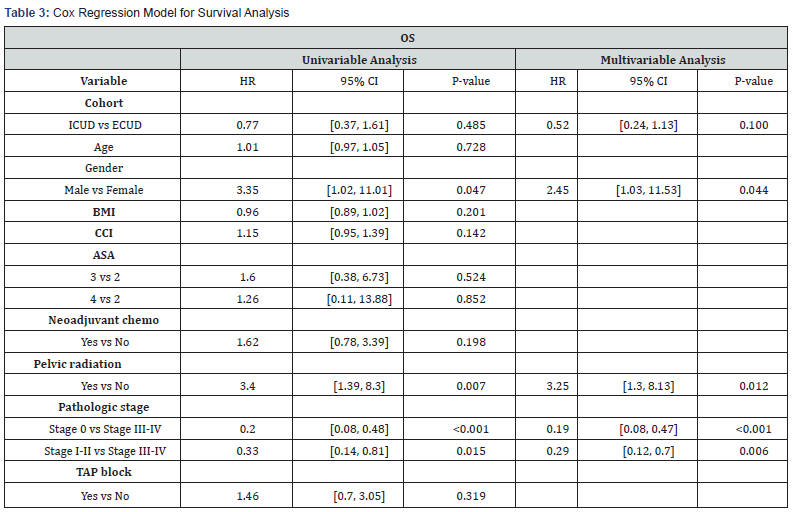
On log-transformed multivariate linear regression modeling of total narcotic use (POD1-3), patients who underwent ICUD had significantly lower total narcotic usage even after adjusting for TAP block (Table 4). Compared to ECUD, ICUD patients were estimated to consume 52.3% less opioids (beta=-0.74, 95%CI [-1.41, -0.07], P=.031). Pain scores were significantly lower among patients who received ICUD on POD2 (beta=-1.10, 95%CI [-1.95, -0.25], P=.012); however, pain scores were not significantly different across cohorts on POD1 (beta=-0.71, 95%CI [-1.56, 0.13], P=.098) or POD3 (beta=-0.39, 95%CI [-1.28, 0.5], P=.386).
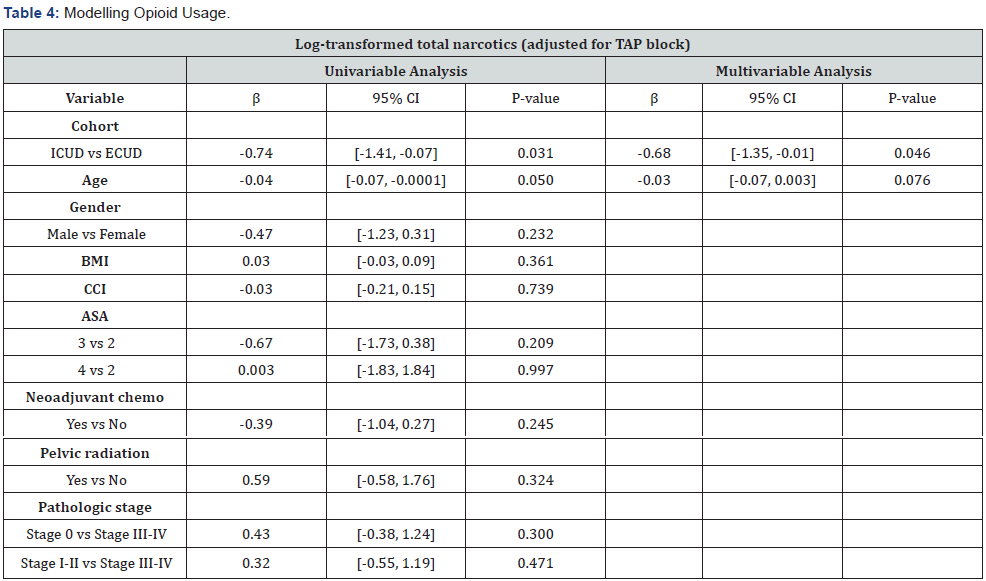
There was no difference between ICUD and ECUD with regards to rate of any postoperative complications at 30 days (OR=1.40, 95%CI [0.75, 2.6], P=.294). Furthermore, no significant difference was found between either technique with regards to high grade (OR=0.91, 95%CI [0.32, 2.63], P=.861) or GI complications (OR=1.27, 95%CI [0.56, 2.9], P=.573). However, BMI was significantly correlated with high grade complications (OR=1.11, 95%CI [1.01, 1.22], P=.039).
Discussion
In this study, we assessed perioperative and pain-related outcomes between ECUD and ICUD following RARC for bladder cancer. Our study is the first to our knowledge to explore narcotic consumption and pain scores as outcomes related to the use of ICUD. Our results demonstrate that despite similar pain scores between the cohorts, patients who underwent ICUD experienced a nearly 50% reduction in post-operative narcotic consumption compared to patients who underwent ECUD.
Though narcotic consumption has not yet been compared between ICUD and ECUD following RARC, previous studies have highlighted the advantage of minimally invasive surgeries in reducing postoperative narcotic usage [22-25]. Our data indicate no significant difference between ICUD and ECUD pain scores on POD1 and POD3. We postulate this could be due to the diminished effect of the TAP block by POD2 leading to the observed difference in pain scores on this day, followed by compensatory narcotic usage among ECUD patients resulting in a re-alignment of pain scores between the cohorts by POD3. Self-rating pain scales including NRS used at our institution are limited by subjectivity in measuring pain, as studies have shown factors such as mood can influence these pain scores [26-28]. Thus, it is possible that narcotic consumption is a more objective indicator of pain relative to the NRS. The observed differences in narcotic usage therefore may be attributable to the minimally invasive approach of ICUD. Additionally, it is important to note that all patients included in this study were enrolled in our ERAS program, of which a basic tenet is reducing narcotic consumption. This goal is in part achieved by standardizing a significant amount of post-operative care, with the effect that opioid administration is largely driven outside of individual surgeon influence.
Our data suggest that complication rates, operative times, 30-day readmission, LOS, and estimated blood loss were not significantly different across cohorts. Previous studies have provided conflicting results regarding these outcomes in patients undergoing ICUD versus ECUD [9-13]. A 2013 study found no difference in operative times and EBL between groups, while a more recent study demonstrated shorter operative time and less blood loss in ICUD compared to ECUD patients [9,10]. Nonetheless, our data are in concordance with the general finding that ICUD is at least non-inferior to ECUD in terms of operative time and EBL.
In our study, complication rates were not significantly different across cohorts, which we surmise could be due to the high level of experience among the surgeons. Supporting this possibility, one study demonstrated shorter operative time and reduced complications associated with increased experience, mentoring, and team-approaches to robotic surgery [29]. Similarly, another study found that while ECUD complication rates remained relatively stable, ICUD complications decreased over time, attributing the trend to increased surgeon experience with the technique [9].
Despite a steep learning curve, ICUD is becoming increasingly utilized given the possible benefits associated with a completely minimally invasive approach [9]. Our findings of reduced narcotic consumption and lower readmission rates further support investment in ICUD. Our study is limited by modest sample size and single institution data. Additionally, readmissions rates and complications were only assessed out to 30 days postoperatively, which excludes adverse events that might occur longer after surgery. Finally, selection bias may limit this study as receipt of ICUD versus ECUD was not randomized. While all ICUDs and ECUDs were performed by separate surgeons, overall operative characteristics and postoperative complications rates were similar. Additionally, as noted above, postoperative pain control is managed primarily by departmental ERAS nurses via a protocolized order set. Therefore, it is likely the results stem from procedural rather than surgeon differences.
Nonetheless, we demonstrate valuable advantages to the adoption of ICUD following RARC including decreased narcotic consumption and lower risk for readmission. Future studies are needed to further evaluate these perioperative outcomes and in addition to the effect of increased surgeon experience on perioperative outcomes.
Conclusion
Our study suggests that robotic cystectomy with intracorporeal ileal conduit reduces post-operative narcotic use as compared to extracorporeal diversion. Further validation across multiple institutions will be required to elucidate the full advantage and/ or disadvantage to this minimally invasive approach.
Acknowledgement
Hailey Holck and Fardous Ahmed and The Shelton Bladder Cancer Fund.
References
- Chang SS, Boorjian SA, Chou R, Clark PE, Daneshmand S, et al. (2016) Diagnosis and Treatment of Non-Muscle Invasive Bladder Cancer: AUA/SUO Guideline. J Urol 196: 1021-1029.
- Parekh DJ, Messer J, Fitzgerald J, Ercole B, Robert Svatek R, et al. (2013) Perioperative outcomes and oncologic efficacy from a pilot prospective randomized clinical trial of open versus robotic assisted radical cystectomy. J Urol 189: 474.
- Tan WS, Tan M-Y, Lamb BW, Sridhar A, Anna Mohammed A, et al. (2018) Intracorporeal robot-assisted radical cystectomy, together with an enhanced recovery programme, improves postoperative outcomes by aggregating marginal gains. BJU Int 121: 632-639.
- Parekh DJ, Reis IM, Castle EP, Gonzalgo ML, Woods ME, et al. (2018) Robot-assisted radical cystectomy versus open radical cystectomy in patients with bladder cancer (RAZOR): an open-label, randomised, phase 3, non-inferiority trial. Lancet 391: 2525.
- Nazzani S, Mazzone E, Preisser F, Bandini M, Tian Z, et al. (2018) Comparison of Perioperative Outcomes Between Open and Robotic Radical Cystectomy: A Population-Based Analysis. J Endourol 32: 701-709.
- Lobo N, Thurairaja R, Nair R, Dasgupta P, Shamim Khan M, et al. (2018) Robot-assisted radical cystectomy with intracorporeal urinary diversion – The new ‘gold standard’? Evidence from a systematic review. Arab J Urol 16: 307.
- Collins JW, Tyritzis S, Nyberg T, Schumacher M, Laurin O, et al. (2013) Robot-assisted radical cystectomy: description of an evolved approach to radical cystectomy. Eur Urol 64: 654-663.
- Murthy PB, Bryk DJ, Lee BH, Haber GP, et al. (2020) Robotic radical cystectomy with intracorporeal urinary diversion: beyond the initial experience. Transl Androl Urol 9: 942.
- Hussein AA, May PR, Jing Z, Ahmed YE, Wijburgt CJ, et al. (2018) Outcomes of Intracorporeal Urinary Diversion after Robot-Assisted Radical Cystectomy: Results from the International Robotic Cystectomy Consortium. J Urol 199: 1302.
- Ahmed K, Khan SA, Hayn MH, Agarwal PK, Badani KK, et al. (2014) Analysis of intracorporeal compared with extracorporeal urinary diversion after robot-assisted radical cystectomy: results from the International Robotic Cystectomy Consortium. Eur Urol 65: 340.
- Tan TW, Nair R, Saad S, Thurairaja R, Shamim Khan M et al. (2019) Safe transition from extracorporeal to intracorporeal urinary diversion following robot-assisted cystectomy: a recipe for reducing operative time, blood loss and complication rates. World J Urol 37: 367.
- Pyun JH, Kim HK, Cho S, Gu Kang S, Cheon J et al. (2016) Robot-Assisted Radical Cystectomy with Total Intracorporeal Urinary Diversion: Comparative Analysis with Extracorporeal Urinary Diversion. J Laparoendosc Adv Surg Tech A 26: 349-355.
- Lenfant L, Verhoest G, Campi R, Parra J, Vivien Graffeille et al. (2018) Perioperative outcomes and complications of intracorporeal vs extracorporeal urinary diversion after robot-assisted radical cystectomy for bladder cancer: a real-life, multi-institutional french study. World J Urol 36: 1711.
- Hah JM, Bateman BT, Ratliff J, Curtin C, Sun E, et al. (2017) Chronic Opioid Use After Surgery: Implications for Perioperative Management in the Face of the Opioid Epidemic. Anesth Analg 125: 1733-1740.
- Rudd RA, Aleshire N, Zibbell JE, Gladden RM et al. (2016) Increases in Drug and Opioid Overdose Deaths--United States, 2000-2014. MMWR Morb Mortal Wkly Rep 64: 1378-1382.
- Wunsch H, Wijeysundera DN, Passarella MA, Neuman MD et al: Opioids Prescribed After Low-Risk Surgical Procedures in the United States, 2004-2012. JAMA 2016; 315: 1654-1657.
- Calcaterra SL, Yamashita TE, Min S-J, Keniston A, Frank JW, et al. (2016) Opioid Prescribing at Hospital Discharge Contributes to Chronic Opioid Use. J Gen Intern Med 31: 478-485.
- Sun EC, Darnall BD, Baker LC, Mackey S, et al. (2016) Incidence of and Risk Factors for Chronic Opioid Use Among Opioid-Naive Patients in the Postoperative Period. JAMA Intern Med 176: 1286.
- Horattas SA, Bahr KJ, Reissig J, James, Horattas Mark C, et al. (2018) Achieving the Goal of Opioid-Free Surgery: Lessons Learned from More than One Hundred Narcotic-Free Enhanced Recovery After Surgery Colectomies. J Am Coll Surg 227: S58.
- Meyer LA, Lasala J, Iniesta MD, Alpa M Nick, Mark F Munsell, et al. (2018) Effect of an Enhanced Recovery After Surgery Program on Opioid Use and Patient-Reported Outcomes. Obstet Gynecol 132: 281-290.
- Matulewicz RS, Patel M, Jordan BJ, Morano J, Frainey B, et al. (2018) Transversus Abdominis Plane Blockade as Part of a Multimodal Postoperative Analgesia Plan in Patients Undergoing Radical Cystectomy. Bladder Cancer 4: 161-167.
- Vora D, Kinnard M, Falk D, Hoy M, Gupta S, et al. (2018) A comparison of narcotic usage and length of post-operative hospital stay in open versus minimally invasive lumbar interbody fusion with percutaneous pedicle screws. J Spine Surg 4: 516-521.
- Harel M, Herbst KW, Silvis R, Makari JH, Ferrer FA, et al. (2015) Objective pain assessment after ureteral reimplantation: Comparison of open versus robotic approach. J Pediatr Urol 11(2): 82.e1-8.
- Schlatter MG, Nguyen LV, Tecos M, Kalbfell EL, Gonzalez-Vega O, et al. (2019) Progressive reduction of hospital length of stay following minimally invasive repair of pectus excavatum: A retrospective comparison of three analgesia modalities, the role of addressing patient anxiety, and reframing patient expectations. J Pediatr Surg 54: 663.
- Goebel S, Steinert AF, Schillinger J, Eulert J, Broscheit J, et al. (2012) Reduced postoperative pain in total hip arthroplasty after minimal-invasive anterior approach. Int Orthop 36: 491.
- Tang NKY, Salkovskis PM, Hodges A, Wright KJ, Hanna M, et al. (2008) Effects of mood on pain responses and pain tolerance: an experimental study in chronic back pain patients. Pain 138: 392-401.
- Taenzer P, Melzack R, Jeans ME (1986) Influence of psychological factors on postoperative pain, mood and analgesic requirements. Pain 24: 331-342.
- Middendorp HV, Lumley MA, Jacobs JWG, J Bijlsma JW, Geenen R, et al. (2010) The effects of anger and sadness on clinical pain reports and experimentally-induced pain thresholds in women with and without fibromyalgia. Arthritis Care Res 62: 1370-1376.
- Collins JW, Sooriakumaran P, Wiklund NP (2014) Launching and evolving a robotic cystectomy service by developing your ‘FORTE.’ BJU Int 113: 520-522.






























