Aerobic and Resistant Physical Exercise on Chronic Kidney Disease (CKD) Patients: are there any Benefits?
Pereira Pinto W*, Morales A, Motta Elias R, Tinucci T and Aparecida Dalboni M
Deparment of Medicine, Universidade Nove de Julho/ Uninove, Brazil
Submission:October 26, 2022;Published:November 10, 2022
*Corresponding author:Walter Pereira Pinto, Department of medicine, Universidade Nove de Julho/Uninove, Vergueiro street 235 Liberdade, Brazil
How to cite this article:Pereira Pinto W, Morales A, Motta Elias R, Tinucci T, Aparecida Dalboni M. Aerobic and Resistant Physical Exercise on Chronic Kidney Disease (CKD) Patients: are there any Benefits?. JOJ Urology & Nephrology, 2022; 7(5): 555721. DOI: 10.19080/JOJUN.2022.07.555721
Abstract
Chronic kidney disease (CKD) has become an epidemic condition, with increasing incidence worldwide, generating high mortality rates and high health care costs. Several studies have demonstrated that inflammation is common in patients with CKD and it is associated with a high mortality rate in this population. Regular exercise has been described as a therapeutic strategy for reducing the risk of chronic and metabolic diseases, in part because of exercise training seems to exert anti-inflammatory effects. Therefore, the aim of the present review was to analyze the effects of aerobic and resistance exercise on inflammatory biomarkers in patients with CKD. CKD results in serum accumulation of toxic substances, the uremic toxins. These activate immune and inflammatory response in experimental models and in patients with CKD. Several studies involving non-pharmacological interventions have focused on CKD patients, aiming at reducing the inflammatory state, improving quality of life and extending the survival of these patients. It has been found that using treadmill for walking in patients undergoing pre-dialysis treatment was effective in reducing CRP and IL-6, showing that this type of aerobic exercise may also promote an anti-inflammatory effect. In contrast, some studies have not been able to show any beneficial effect on inflammation; e.g., a study using an intradialytic cycling protocol for HD patients 3x / week for 6 months found no decrease in IL-6, CRP and TNF-α. There are a number studies reporting favorable effects of exercise training on the inflammatory response triggered by hypertension, diabetes, obesity, dyslipidemia and cardiovascular events in the general population; however, this review showed that the role of the anti-inflammatory effect of aerobic and resistance exercise remains controversial in CKD patients, particularly in those undergoing dialysis. Conversely, data from the literature found that high intensity aerobic exercise training for a sustained period of time seems to have anti-inflammatory effects when compared to resistance exercise, in particular when PCR is used as a marker of inflammation.
Keywords: Kidney Disease; Inflammation; Exercise Effect
Introduction
Chronic kidney disease (CKD) is now a leading cause of mortality and morbidity around the world with increasing worldwide incidence and high health care costs [1]. It is estimated that the prevalence of chronic kidney disease (CKD) affects between 11% and 13%. % of population [2], affecting around 750 million people worldwide [3]. Besides, it is estimated that five million are currently on dialysis (dialysis) [2] and the increased incidence and prevalence of CKD will have a 33% increase in the impact on health costs by 2020 [4]. CKD is defined by the presence of renal injury or glomerular filtration rate (GFR <60 ml/min per 1.73 m2) for ≥3 months, regardless of the underlying disease, and is classified into five stages, according to GFR [5]. CKD is a progressive condition, and it is characterized by a decrease in GFR <15 ml/min per 1.73m2 (uremia) due to the accumulation of toxic substances (uremic toxins) that should be excreted by the kidney.
GFR <15 ml / min per 1.73 m2 is incompatible with survival [6], requiring dialysis treatment or kidney transplantation [7]. The major risk factors involved in the decline of renal function include hypertension, diabetes, obesity and primary renal disorders [8]. These diseases share the pathophysiological mechanism of inflammation. Several studies have reported that CKD patients have higher concentrations of oxidative stress markers, C-reactive protein and inflammatory cytokines when compared to healthy individuals [9,10]. Moreover, inflammation is associated with high cardiovascular mortality rates [11-13] and muscle catabolism, due to protein restriction [14] and malnutrition [15], which also contribute to skeletal muscle mass loss, decreased muscle performance and worse quality of life in patients with CKD [16]. Uremia “per se” and inflammation appear to be associated with protein degradation and consequently marked catabolic state in this population [17]. Thus, patients with CKD often complain of weakness, fractures, functional disability [18], which are associated with the lack of regular physical activity [19]. Both low level of physical activity [18] and loss of physical function capacity [20] are independently associated with adverse outcomes that impair the quality of life of these individuals [20]. In recent years, several studies have proposed that exercise training in the general population reduces the risk of chronic and metabolic diseases, and has anti-inflammatory effects [21,22]. However, there are few studies addressing the effect of aerobic exercise and exercise resistance to inflammation modulation in CKD patients on dialysis and pre-dialysis [23].
Materials and Methods
Searches were performed in five databases; PubMed, ProQuest, Bireme, Scielo and ScienceDirect. Relevant publications from the last ten years (2009 to 2019) were selected. The keywords selected were: “Kidney disease and inflammation”, “physical exercise in kidney disease”, “physical exercise in inflammation”, “effects of physical exercise on chronic kidney disease”, “anti-inflammatory effects of physical exercise on kidney disease” and “anti-inflammatory effects of exercise”.
Hypertension
The progression of renal failure is associated with risk factors such as hypertension, diabetes and obesity [8,24]. Hypertension is characterized by increased blood pressure (BP) and the overall prevalence of this condition affects around 40% of the adult population worldwide [25]. Hypertension is the/a major risk factor associated with CKD, and as such strongly contributes to cardiovascular diseases (CDVs) and mortality [26,27]. In hypertension, renal function self-regulation is impaired, resulting in glomerular hypertension [28]. Elevated intraglomerular pressure signals local inflammatory responses with the production of proinflammatory cytokines and oxygen radicals, inducing glomerular injury and progressive loss of renal function [29]. The loss of renal function and the progression of CKD-related hypertension induce structural changes in the kidney, vascular sclerosis, glomerular hypertrophy, tubular injury and fibrosis, leading to kidney failure (Figure 1).
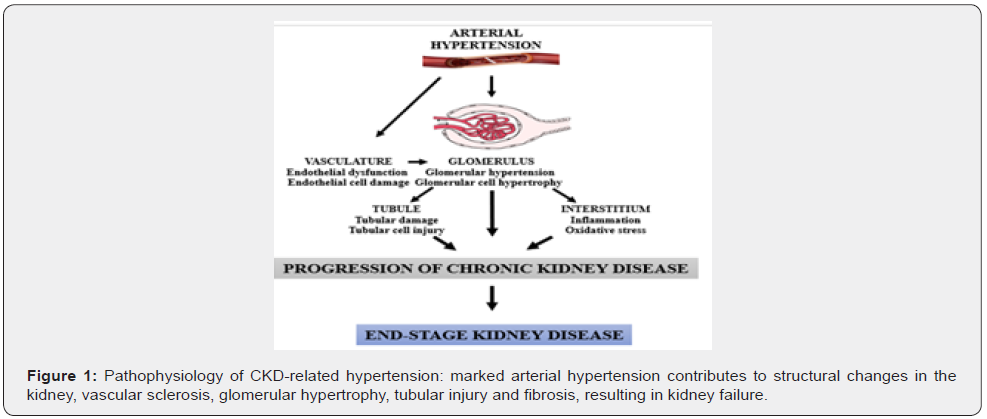
Diabetes
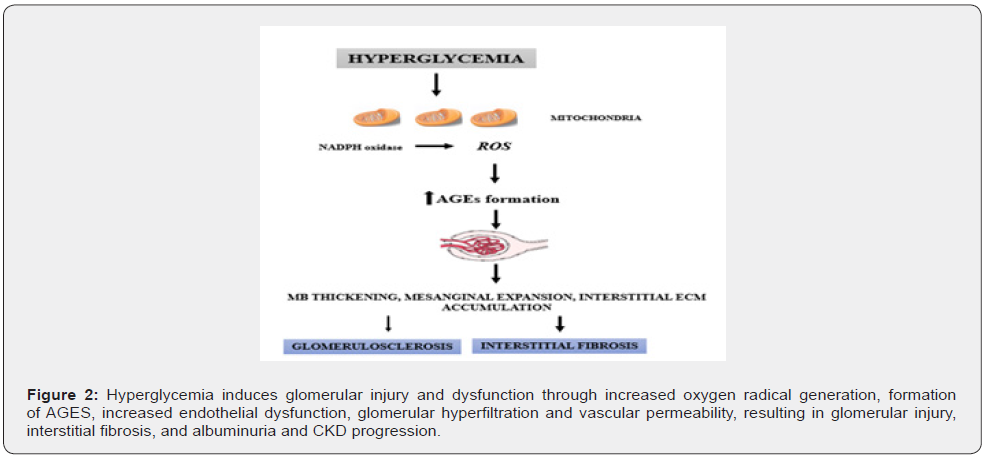
The number of individuals with diabetes is estimated to reach 642 million by 2040 [30]. Diabetes is also associated with an increased incidence of chronic kidney disease (CKD) [31,32]. Studies have found that hyperglycemia induces glomerular injury and dysfunction through increased oxygen radical generation and formation of AGES.
These mechanisms contribute to endothelial dysfunction, glomerular hyperfiltration and increased vascular permeability, resulting in glomerular injury and consequent albuminuria [33] and CKD progression [34,35]. (Figure 2).
Obesity
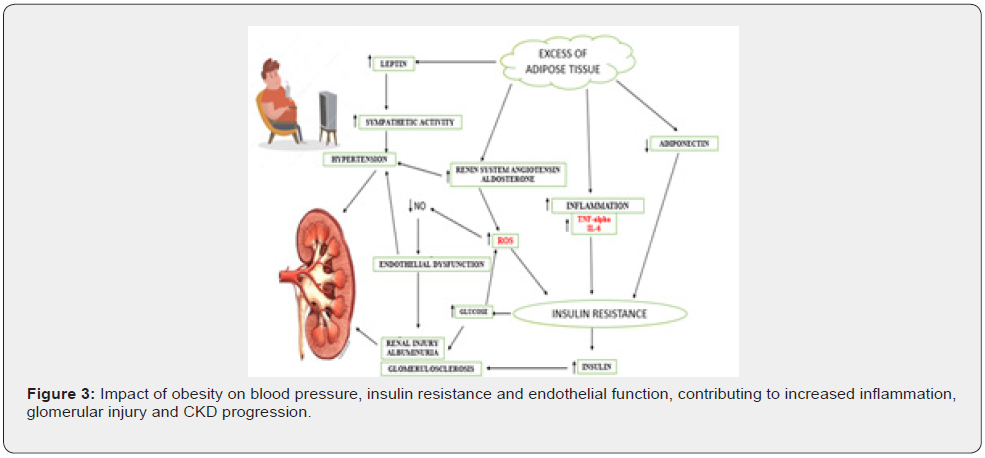
Obesity has also been found to be a risk factor for CKD. Adipose tissue consists of adipocytes, which in excess are capable of producing TNF-α, IL-6, oxygen radicals (ROS) having autocrine and paracrine action. These inflammatory mediators increase the activity of the renin angiotensin aldosterone system (RAAS) contributing to hypertension and promote insulin resistance, which results in hyperglycemia. In addition, obesity is associated with low levels of adiponectin, a hormone with a protective effect on the regulation of inflammatory response, including in endothelial cell. Thus, the adverse effects of obesity on blood pressure, insulin resistance, inflammation, dyslipidemia and systemic hemodynamic changes contribute to glomerular lesions and may eventually lead to CKD [36-39] (Figure 3).
Inflammation and CKD
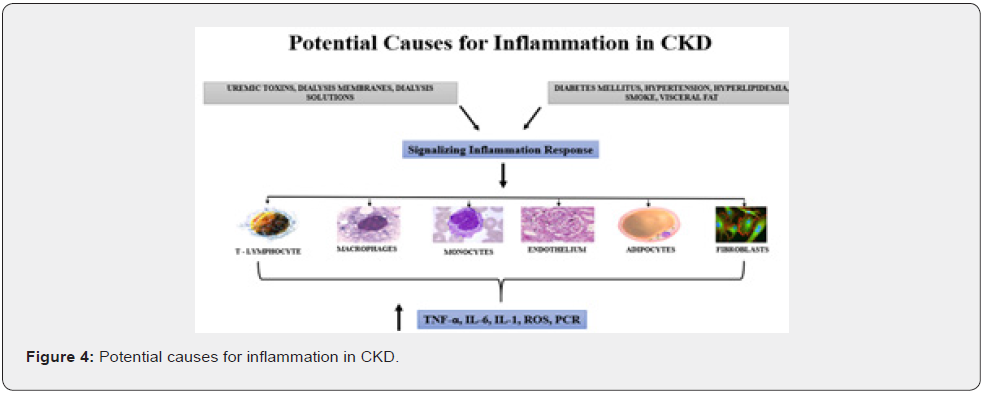
Chronic Kidney disease (CKD) results in serum accumulation of toxic substances, uremic toxins [40]. These toxins in turn activate immune and inflammatory response in experimental models and in patients with CKD [41-45], Dialytic procedures are the treatment necessary to maintain CKD patients alive. However, despite the technological advances in the dialysis procedures, peritoneal dialysis and hemodialysis are not able to remove all toxins, particularly those too large to be filtered and/or protein bounded 46 [46]. Therefore, patients on dialysis still have serum circulating uremic toxins. In addition, some studies have demonstrated a direct relationship between uremic toxins and inflammation, and impact on cardiovascular disease (CVD), the main cause of mortality of CKD patients [47, 48]. Uremic toxins , such as indoxyl sulfate and p-cresyl, which originate from the bowel metabolism, have been found to cause immune activation with production of proinflammatory cytokines, including TNF-α, IL-6 and MCP-1 [49, 50] mainly by monocytes [51,52]. Although these proinflammatory cytokines enhance host defense, excessive production leads to unresolved inflammation [53]. The causes of inflammation in CKD patients are multifactorial (Figure 4) and are associated with presence of uremic toxins, acidosis, dialytic procedures “per se”, bioincompatibility of the membrane, quality of water used in hemodialysis, besides traditional risk factors as hypertension, diabetes, obesity, smoking and dyslipidemia [54] (Figure 4).
Loss of Skeletal Muscle Mass in CKD
Loss of muscle mass in patients with CKD is common and is associated with low calorie intake, protein restriction, metabolic acidosis, hyperkalemia and uremia control) [55-57] and protein energy loss (PEW) [58]. These changes are common and increase the risk of mortality in this population, especially in the later stages of CKD with glomerular filtration rate (GFR) <60 ml / min / 1.73 and in dialysis patients [59-61]. PEW in CKD patients is a result of uremia [59], inadequate nutrition and inflammation, which contribute to a marked catabolic state [62], excessive energy loss and decreased muscle mass [63]. In addition, changes in insulin and/or insulin grow factor-1 (IGF-1) signaling are also risk factors for muscle loss [64-66] and mortality in this population [67]. In summary, CKD patients are more susceptible to skeletal muscle loss, with a consequent increased risk for mortality [67].
Physical Exercise and Inflammation in the Drc-Free Population
Cross-sectional and population cohort studies have consistently shown an inverse association between systemic inflammation and physical exercise; the lowest the concentration of inflammatory biomarkers is observed in individuals who exercise frequently [68,69]. Studies have demonstrated that both aerobic and acute exercise may play a proinflammatory role due to increased oxidative stress [70]. Immediately after exercise, the increased inflammatory response due to muscle stimulation stimulates repair and remodeling [71]. Thus, after adaptation, these responses may have a beneficial effect, resulting in reduced inflammatory response [70,72]. The repair mechanism of muscle tissue damage in response to exercise is divided into degeneration, regeneration and tissue remodeling in response to exercise [73]. After acute exercise and / or trauma, muscle fiber destruction, inflammation, injury and necrosis occur. Macrophages are then recruited into the lesion area and produce MCP-1 (Monocyte Chemotactic Protein-1) and IL-10 [74,75] to promote muscle tissue regeneration [76-78]. Besides macrophage recruitment , muscle regeneration also occurs in response to damage to myofibers and activation of myogenic cells which, after proliferation, differentiation and fusion, stimulate the formation of new fibers and the reconstitution of the injured tissue [79]. Therefore, physical exercise seems to play anti-inflammatory role, stimulating the increase of myocin production from muscle contraction, with a subsequent increase in plasma level of anti-inflammatory cytokines (IL-10 and IL-1ra) [80-83]. In addition, regular exercise can also reduce visceral fat and increase adiponectin release, thus promoting an antiinflammatory environment. In a study involving pre hypertensive young individuals, Beck et al. [84] have observed that endothelial function was improved after aerobic exercise. In another study, Beck et al. [85] have demonstrated that aerobic exercise increases the bioavailability of NO and prostaglandins, in addition to reducing circulating levels of endothelin 1 (ET-1; vasoconstrictor). Several studies have also reported the beneficial effects of aerobic exercise on decreasing CRP and IL-6 [86-89] and increasing of IL- 10 [90].
Effect of Physical Exercise On CKD
Several studies have demonstrated the positive impact of non-pharmacological interventions on the inflammatory state, quality of life and survival of CKD patients. Protocol have used both acute and resistance aerobic exercise or a combination of the two. In fact, Liao et al [91] have found that cycling (exercise on an exercise bike) during intradialytic periods for 3 months, 3x week reduced CRP and IL-6 levels. Moreover, in the same study patients who underwent cycling training presented increased endothelial progenitor cells and improved bone mineral density. In a study using treadmill for walking in patients undergoing pre-dialysis treatment, Zang et al [92] have demonstrated that exercise was effective in reducing CRP and IL-6, showing that this type of aerobic exercise may also have an anti-inflammatory effect.
However, some studies have not been able to show any beneficial effect on inflammation. Dungey et al. [93] have also evaluated the impact of an intradialytic cycling program on HD patients 3x week for 6 months and found no decrease in IL-6, CRP and TNF-α. Similarly, Wilund et al. [94], after 4 months of intradialytic cycling for 45 minutes 3x week, did not observe any effect on CRP and IL-6 decrease. Leehey et al. [95], in a randomized pilot study, have not observed any significant effects of treadmill for walking 3x week for 24 weeks on decreased CRP levels in CKD patients in pre-dialysis stages. There are few studies evaluating the acute effect of exercise in patients with CKD. Peres et al. [96] have observed increased IL-10 expression after 20 minutes of an acute intradialytic cycling session. In contrast, Dungey et al. [97] did not observe any significant impact of an acute 30 minute intradialytic cycling session on IL-6 and TNF-α. Moraes et al. [98] have reported a reduction in serum CRP levels after performing exercises with elastic (Theraband) on the lower limbs during the first 2 h of HD, 3x / week, for 6 months. In a study conducted by Barcellos et al. [99], CKD patients in pre-dialytic stages performed resistanceassociated aerobic training sessions for 60 min / 3x week for 16 weeks, and CRP was not reduced when compared to the control group. Likewise, Esgalhado et al (100), have found no changes in CRP levels after 30 min of acute session in a 3-set protocol with 10 repetitions of lower-limb resistance exercise.
Conversely, different results were observed in a study conducted by Afshar et al. [101], who compared the effects of cycling for 10 to 30 minutes vs. resistance exercise through the use of knee-extension ankle weights, hip abduction, and flexions in the patients hemodialysis for 8 weeks (3x wk). which CRP levels decreased in both groups (P = 0.005 and P = 0.036), but the reduction was greater for aerobic exercise training. Similarly, Moraes et al. [98] have found that a 6-month resistance exercise program reduced CRP levels, but had no impact on both IL-6 and TNF-α. However, Kopple et al. [102], did not observe changes in CRP, TNF-α and IL-6 concentrations in HD patients undergoing training for 20 - 40 min / 3x / 20 without / 20 weeks of aerobic, resistance and aerobic training + resistance. Similarly, other studies using aerobic + resistance exercise had no effect on decreasing inflammation biomarkers in HD patients [103-105]. Watson et al. [106] evaluated the effects of resistance exercise with 3 sets of 10 to 12 leg extension repetitions, 3x 8 week[s] in CKD patients undergoing pre-dialytic treatment and found no decrease in IL-6, MCP-1, and TNF-α or IL-15 expression.
Conclusion
There are several studies reporting the favorable effects of exercise counteracting the inflammatory response triggered by hypertension, diabetes, obesity, dyslipidemia and cardiovascular events in the general population. However, the present review in patients with CKD showed that the anti-inflammatory properties of aerobic and resistance exercise remains controversial in this population, particularly in dialysis patients. However, the practice of aerobic exercise with greater intensity and duration which does seem to have an anti-inflammatory effects when compared to resistance exercise, especially when PCR is used as a marker of inflammation. Further research is needed to fully elucidate the impact of exercise on the complex state of immune dysfunction of CKD patients.
References
- Pereira ERS, Pereira AdC, Andrade GBd, Naghettini AV, Pinto FKMS, et al. (2016) Prevalência de doença renal crônica em adultos atendidos na Estratégia de Saúde da Famí J Bras Nefrol 38(1): 22-30.
- Hill NR, Fatoba ST, Oke JL, Hirst JA, O'Callaghan CA, et al. (2016) Global Prevalence of Chronic Kidney Disease - A Systematic Review and Meta-Analysis. PloS one 11(7): e0158765.
- Collaborators GBDDaH (2016) Global, regional, and national disability-adjusted life-years (DALYs) for 315 diseases and injuries and healthy life expectancy (HALE), 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet (London, England) 388(10053): 1603-1658.
- Tucker PS, Kingsley MI, Morton RH, Scanlan AT, Dalbo VJ (2014) The increasing financial impact of chronic kidney disease in australia. Int J Nephrol 2014: 120537.
- Levey AS, de Jong PE, Coresh J, Nahas MEl, Astor BC, et al. (2011) The definition, classification, and prognosis of chronic kidney disease: a KDIGO Controversies Conference report. Kidney International 80(1):17-28.
- Pereira RMdP, Batista MA, Meira AdS, Oliveira MPd, Kusumota L (2017) Quality of life of elderly people with chronic kidney disease in conservative treatment. Revista Brasileira de Enfermagem 70: 851-859.
- Hoefield RA, Kalra PA, Baker P, Lane B, New JP, et al. (2010) Factors associated with kidney disease progression and mortality in a referred CKD population. Am J Kidney Dis. 56(6): 1072-1081.
- Gansevoort RT, Correa-Rotter R, Hemmelgarn BR, Jafar TH, Heerspink HJ, et al. (2013) chronic kidney disease and cardiovascular risk: epidemiology, mechanisms, and prevention. Lancet 382(9889): 339-352.
- Dounousi E, Papavasiliou E, Makedou A, Ioannou K, Katopodis KP, et al. (2006) Oxidative stress is progressively enhanced with advancing stages of CKD. Am J Kidney Dis 48(5): 752-760.
- Cachofeiro V, Goicochea M, de Vinuesa SG, Oubina P, Lahera V, et al. (2008) Oxidative stress and inflammation, a link between chronic kidney disease and cardiovascular disease. Kidney Int Suppl(111): 4-9.
- Sangeetha Lakshmi B, Harini Devi N, Suchitra MM, Srinivasa Rao PVLN, Siva Kumar V (2018) Changes in the inflammatory and oxidative stress markers during a single hemodialysis session in patients with chronic kidney disease. Renal failure 40(1): 534-540.
- Suliman M, Stenvinkel P, Qureshi AR, Kalantar-Zadeh K, Bárány P, et al. (2007) The reverse epidemiology of plasma total homocysteine as a mortality risk factor is related to the impact of wasting and inflammation. Nephrol Dial Transplant 22(1): 209-217.
- Sriram S, Subramanian S, Sathiakumar D, Venkatesh R, Salerno MS, et al. (2011) Modulation of reactive oxygen species in skeletal muscle by myostatin is mediated through NF-kappaB. Aging Cel 10(6): 931-948.
- Kalantar-Zadeh K, Fouque D (2017) Nutritional Management of Chronic Kidney Disease. N Engl J Med 377(18): 1765-1776.
- Garneata L, Stancu A, Dragomir D, Stefan G, Mircescu G (2016) Ketoanalogue-Supplemented Vegetarian Very Low-Protein Diet and CKD Progression. J Am Soc Nephrol 27(7): 2164-2176.
- Zhou Y, Hellberg M, Svensson P, Hoglund P, Clyne N (2018) Sarcopenia and relationships between muscle mass, measured glomerular filtration rate and physical function in patients with chronic kidney disease stages 3-5. Nephrol Dial Transplant 33(2): 342-348.
- Wang XH, Mitch WE (2014) Mechanisms of muscle wasting in chronic kidney disease. Nature reviews Nephrology 10(9): 504-516.
- Zelle DM, Klaassen G, van Adrichem E, Bakker SJ, Corpeleijn E (2017) Physical inactivity: a risk factor and target for intervention in renal care. Nat Rev Nephrol 13(3): 152-168.
- West SL, Ma C, Chaudhry M, Thomas SG, Lok CE (2017) The Association of Daily Activity Levels and Estimated Kidney Function in Men and Women with Predialysis Chronic Kidney Disease. Kidney Int Rep 2(5): 874-880.
- Roshanravan B, Robinson-Cohen C, Patel KV, Ayers E, Littman AJ, et al. (2013) Association between physical performance and all-cause mortality in CKD. J Am Soc Nephrol 24(5): 822-830.
- Batista Júnior ML, Lopes RD, Seelaender MCL (2009) Lopes AC Efeito anti-inflamatório do treinamento físico na insuficiência cardíaca: papel do TNF-± e da IL-10. Arquivos Brasileiros de Cardiologia 93: 692-700.
- Gleeson M, Bishop NC, Stensel DJ, Lindley MR, Mastana SS, ET AL. (2011) The anti-inflammatory effects of exercise: mechanisms and implications for the prevention and treatment of disease. Nat Rev Immunol 11(9): 607-615.
- Highton PJ, Neale J, Wilkinson TJ, Bishop NC, Smith AC (2017) Physical activity, immune function and inflammation in kidney patients (the PINK study): a feasibility trial protocol. BMJ Open 7(5): e014713.
- Jha V, Garcia-Garcia G, Iseki K, Li Z, Naicker S, et al. (2013) chronic kidney disease: global dimension and perspectives. Lancet 382(9888): 260-272.
- Worldwide trends in blood pressure from 1975 to 2015: a pooled analysis of 1479 population-based measurement studies with 19.1 million participants. Lancet 389(10064): 37-55.
- Hayashi M, Uchida S, Kawamura T, Kuwahara M, Nangaku M, et al. (2015) Prospective randomized study of the tolerability and efficacy of combination therapy for hypertensive chronic kidney disease: results of the PROTECT-CKD study. Clin Exp Nephrol 19(5): 925-932.
- Fukuma S, Shimizu S, Niihata K, Sada KE, Yanagita M, et al. (2017) Development of quality indicators for care of chronic kidney disease in the primary care setting using electronic health data: a RAND-modified Delphi method. Clin Exp Nephrol 21(2):247-256.
- Mori T, Polichnowski A, Glocka P, Kaldunski M, Ohsaki Y, et al. (2008) High Perfusion Pressure Accelerates Renal Injury in Salt-Sensitive Hypertension. Journal of the American Society of Nephrology 19(8): 1472-1482.
- Ruggenenti P, Cravedi P, Remuzzi G (2012) Mechanisms and Treatment of CKD. J Am Soc Nephrol 23(12): 1917-1928.
- Guariguata L, Whiting DR, Hambleton I, Beagley J, Linnenkamp U, et al. (2014) Global estimates of diabetes prevalence for 2013 and projections for 2035. Diabetes Res Clin Pract 103(2): 137-149.
- Thomas MC, Cooper ME, Zimmet P (2016) Changing epidemiology of type 2 diabetes mellitus and associated chronic kidney disease. Nat Rev Nephrol 12(2): 73-81.
- Collins AJ, Foley RN, Herzog C, Chavers BM, Gilbertson D, et al. (2010) Excerpts from the US Renal Data System 2009 Annual Data Report. Am J Kidney Dis: the official journal of the National Kidney Foundation 55(1 Suppl 1): S1-A7.
- De Cosmo S, Menzaghi C, Prudente S, Trischitta V (2013) Role of insulin resistance in kidney dysfunction: insights into the mechanism and epidemiological evidence. Nephrol Dial Transplant 28(1): 29-36.
- Donate-Correa J, Martin-Nunez E, Muros-de-Fuentes M, Mora-Fernandez C, Navarro-Gonzalez JF (2015) Inflammatory cytokines in diabetic nephropathy. J Diabetes Res 2015: 948417.
- Wada J, Makino H (2013) Inflammation and the pathogenesis of diabetic nephropathy. Clin Sci (Lond) 124(3): 139-152.
- Whaley-Connell A, Sowers JR (2017) Obesity and kidney disease: from population to basic science and the search for new therapeutic targets. Kidney Int 92(2): 313-323.
- Herrington WG, Smith M, Bankhead C, Matsushita K, Stevens S, et al. (2017) Body-mass index and risk of advanced chronic kidney disease: Prospective analyses from a primary care cohort of 1.4 million adults in England. PLoS One 12(3): e0173515.
- Garofalo C, Borrelli S, Minutolo R, Chiodini P, De Nicola L, et al. (2017) A systematic review and meta-analysis suggests obesity predicts onset of chronic kidney disease in the general population. Kidney Int 91(5): 1224-1235.
- Kovesdy CP, Furth SL, Zoccali C, World Kidney Day Steering Committee (2017) Obesity and kidney disease: hidden consequences of the epidemic. J Nephrol 30(1): 1-10.
- Vanholder RC, Glorieux GL (2003) An overview of uremic toxicity. Hemodialysis international International Symposium on Home Hemodialysis 7(2): 156-161.
- Borges NA, Barros AF, Nakao LS, Dolenga CJ, Fouque D, et al. (2016) Protein-Bound Uremic Toxins from Gut Microbiota and Inflammatory Markers in Chronic Kidney Disease. J Ren Nutr 26(6): 396-400.
- Ito S, Yoshida M (2014) Protein-bound uremic toxins: new culprits of cardiovascular events in chronic kidney disease patients. Toxins (Basel) 6(2): 665-678.
- Moradi H, Sica DA, Kalantar-Zadeh K (2013) Cardiovascular burden associated with uremic toxins in patients with chronic kidney disease. Am J Nephrol 38(2):136-148.
- Rossi M, Campbell KL, Johnson DW, Stanton T, Vesey DA, et al. (2014) Protein-bound uremic toxins, inflammation and oxidative stress: a cross-sectional study in stage 3-4 chronic kidney disease. Arch Med Res 45(4): 309-317.
- Kaminski TW, Pawlak K, Karbowska M, Mysliwiec M, Pawlak D (2017) Indoxyl sulfate - the uremic toxin linking hemostatic system disturbances with the prevalence of cardiovascular disease in patients with chronic kidney disease. BMC nephrology 18(1): 35.
- Claro LM, Moreno-Amaral AN, Gadotti AC, Dolenga CJ, Nakao LS, et al. (2018) The Impact of Uremic Toxicity Induced Inflammatory Response on the Cardiovascular Burden in Chronic Kidney Disease. Toxins 10(10): 384.
- Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY (2004) Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. The New England journal of medicine 351(13): 1296-1305.
- Lekawanvijit S, Krum H (2014) Cardiorenal syndrome: acute kidney injury secondary to cardiovascular disease and role of protein-bound uraemic toxins. The Journal of physiology 592(18): 3969-3983.
- Kracht M, Saklatvala J (2002) Transcriptional and post-transcriptional control of gene expression in inflammation. Cytokine 20(3): 91-106.
- Bhavsar P, Hew M, Khorasani N, Torrego A, Barnes PJ, et al. (2008) Relative corticosteroid insensitivity of alveolar macrophages in severe asthma compared with non-severe asthma. Thorax 63(9): 784-790.
- Kim HY, Yoo TH, Hwang Y, Lee GH, Kim B, et al. (2017) Indoxyl sulfate (IS)-mediated immune dysfunction provokes endothelial damage in patients with end-stage renal disease (ESRD). Sci Rep 7(1): 3057.
- Trojanowicz B, Ulrich C, Seibert E, Fiedler R, Girndt M (2014) Uremic conditions drive human monocytes to pro-atherogenic differentiation via an angiotensin-dependent mechanism. PloS one 9(7): e102137.
- Parrillo JE (1993) Pathogenetic mechanisms of septic shock. N Engl J Med 328(20): 1471-1477.
- Dai L, Golembiewska E, Lindholm B, Stenvinkel P (2017) End-Stage Renal Disease, Inflammation and Cardiovascular Outcomes. Contrib Nephrol 191: 32-43.
- Locatelli F, Del Vecchio L (2014) Protein restriction: a revisited old strategy with new opportunities? Nephrol Dial Transplant 29(9): 1624-1627.
- Fouque D, Mitch WE (2015) Low-protein diets in chronic kidney disease: are we finally reaching a consensus? Nephrol Dial Transplant 30(1): 6-8.
- Andrassy KM (2013) Comments on ‘KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease’. Kidney Int 84(3): 622-623.
- Obi Y, Qader H, Kovesdy CP, Kalantar-Zadeh K (2015) Latest consensus and update on protein-energy wasting in chronic kidney disease. Curr Opin Clin Nutr Metab Care 18(3): 254-262.
- Foley RN, Wang C, Ishani A, Collins AJ, Murray AM (2007) Kidney function and sarcopenia in the United States general population: NHANES III. Am J Nephrol 27(3): 279-286.
- Carrero JJ, Stenvinkel P, Cuppari L, Ikizler TA, Kalantar-Zadeh K, et al. (2013) Etiology of the protein-energy wasting syndrome in chronic kidney disease: a consensus statement from the International Society of Renal Nutrition and Metabolism (ISRNM). J Ren Nutr 23(2): 77-90.
- Gonzalez-Parra E, Gracia-Iguacel C, Egido J, Ortiz A (2012) Phosphorus and nutrition in chronic kidney disease. Int J Nephrol 2012: 597605.
- Fahal IH (2014) Uraemic sarcopenia: aetiology and implications. Nephrol Dial Transplant 29(9): 1655-1665.
- Streese L, Deiseroth A, Schafer J, Schmidt-Trucksass A, Hanssen H, et al. (2018) Exercise, Arterial Crosstalk-Modulation, and Inflammation in an Aging Population: The ExAMIN AGE Study. Front Physiol 9: 116.
- Peng H, Cao J, Yu R, Danesh F, Wang Y, et al. (2016) CKD Stimulates Muscle Protein Loss Via Rho-associated Protein Kinase 1 Activation. J Am Soc Nephrol 27(2): 509-519.
- Shin J, Tajrishi MM, Ogura Y, Kumar A (2013) Wasting mechanisms in muscular dystrophy. Int J Biochem Cell Biol 45(10): 2266-2279.
- Zha Y, Qian Q (2017) Protein Nutrition and Malnutrition in CKD and ESRD. Nutrients 9(3): 208.
- Gracia-Iguacel C, Gonzalez-Parra E, Perez-Gomez MV, Mahillo I, Egido J, et al. (2013) Prevalence of protein-energy wasting syndrome and its association with mortality in haemodialysis patients in a centre in Spain. Nefrologia 33(4): 495-505.
- Dungey M, Hull KL, Smith AC, Burton JO, Bishop NC (2013) Inflammatory Factors and Exercise in Chronic Kidney Disease. International Journal of Endocrinology 2013: 569831.
- Carney EF (2014) chronic kidney disease. Walking reduces inflammation in predialysis CKD. Nat Rev Nephrol 10(6): 300.
- Ribeiro-Samora G, Rabelo LA, Ferreira AC, M Favero M, Guedes GS (2017) Inflammation and oxidative stress in heart failure: effects of exercise intensity and duration Braz J Med Biol Res 50(9): e6393.
- Silva FOCd, Macedo DV (2011) Exercicio físico, processo inflamatorio e adaptacao: uma visão geral. Rev. bras. cineantropom. desempenho hum 13: 320-328.
- Falone S, Mirabilio A, Pennelli A, Cacchio M, Di Baldassarre A, et al. (2010) Differential impact of acute bout of exercise on redox- and oxidative damage-related profiles between untrained subjects and amateur runners. Physiol Res 59(6): 953-961.
- Smith LL (2004) Tissue trauma: the underlying cause of overtraining syndrome? J Strength Cond Res 18(1): 185-193.
- Zwetsloot KA, John CS, Lawrence MM, Battista RA, Shanely RA (2014) High-intensity interval training induces a modest systemic inflammatory response in active, young men. J Inflamm Res 7: 9-17.
- Hovanloo F, Arefirad T, Ahmadizad S (2013) Effects of sprint interval and continuous endurance training on serum levels of inflammatory biomarkers. J Diabetes Metab Disord 12(1): 22.
- Gleeson M (2007) Immune function in sport and exercise. J Appl Physiol (1985) 103(2): 693-639.
- Tidball JG (2005) Inflammatory processes in muscle injury and repair. Am J Physiol Regul Integr Comp Physiol 288(2): R345-353.
- Morgan JE, Partridge TA (2003) Muscle satellite cells. The International Journal of Biochemistry & Cell Biology 35(8): 1151-1156.
- Charge SB, Rudnicki MA (2004) Cellular and molecular regulation of muscle regeneration. Physiol Rev 84(1): 209-238.
- Pedersen BK (2011) Exercise-induced myokines and their role in chronic diseases. Brain Behav Immun 25(5): 811-816.
- Pedersen BK, Febbraio MA (2008) Muscle as an endocrine organ: focus on muscle-derived interleukin-6. Physiol Rev 88(4): 1379-1406.
- Petersen AM, Pedersen BK (2005) The anti-inflammatory effect of exercise. J Appl Physiol (1985) 98(4): 1154-1162.
- Steensberg A, Fischer CP, Keller C, Moller K, Pedersen BK (2003) IL-6 enhances plasma IL-1ra, IL-10, and cortisol in humans. Am J Physiol Endocrinol Metab 285(2): 433-437.
- Beck DT, Martin JS, Casey DP, Braith RW (2014) Exercise training improves endothelial function in resistance arteries of young prehypertensives. J Hum Hypertens 28(5): 303-309.
- Beck DT, Casey DP, Martin JS, Emerson BD, Braith RW, et al. (2013) Exercise training improves endothelial function in young prehypertensives. Experimental biology and medicine (Maywood, NJ) 238(4): 433-441.
- Geffken DF, Cushman M, Burke GL, Polak JF, Sakkinen PA, et al. (2001) Association between physical activity and markers of inflammation in a healthy elderly population. Am J Epidemiol 153(3): 242-250.
- Kwasniewska M, Kostka T, Jegier A, Dziankowska-Zaborszczyk E, Leszczynska J, et al. (2016) Regular physical activity and cardiovascular biomarkers in prevention of atherosclerosis in men: a 25-year prospective cohort study. BMC Cardiovasc Disord 16: 65.
- Bergstrom G, Behre CJ, Schmidt C (2012) Moderate intensities of leisure-time physical activity are associated with lower levels of high-sensitivity C-reactive protein in healthy middle-aged men. Angiology 63(6): 412-415.
- Reuben DB, Judd-Hamilton L, Harris TB, Seeman TE (2003) The associations between physical activity and inflammatory markers in high-functioning older persons: MacArthur Studies of Successful Aging. J Am Geriatr Soc 51(8): 1125-1130.
- Jankord R, Jemiolo B (2004) Influence of physical activity on serum IL-6 and IL-10 levels in healthy older men. Med Sci Sports Exerc 36(6): 960-964.
- Liao MT, Liu WC, Lin FH, Huang CF, Chen SY, et al. (2016) Intradialytic aerobic cycling exercise alleviates inflammation and improves endothelial progenitor cell count and bone density in hemodialysis patients. Medicine (Baltimore) 95(27): e4134.
- Zang L, Wang SQ, Mao N, Fu XJ, Lin XJ, et al. Effect of Different Walking Number on Inflammation and Nutrition in Patients with Chronic Kidney Disease]. Sichuan Da Xue Xue Bao Yi Xue Ban 50(2): 252-255.
- Dungey M, Young HML, Churchward DR, Burton JO, Smith AC (2017) Regular exercise during haemodialysis promotes an anti-inflammatory leucocyte profile. Clin Kidney 10(6): 813-821.
- Wilund KR, Tomayko EJ, Wu PT, Ryong Chung H, Vallurupalli S, et al. (2010) Intradialytic exercise training reduces oxidative stress and epicardial fat: a pilot study. Nephrol Dial Transplant 25(8): 2695-2701.
- Leehey DJ, Moinuddin I, Bast JP, Qureshi S, Jelinek CS, et al. (2009) Aerobic exercise in obese diabetic patients with chronic kidney disease: a randomized and controlled pilot study. Cardiovascular Diabetology 8: 62.
- Peres A, Perotto DL, Dorneles GP, Fuhro MI, Monteiro MB (2015) Effects of intradialytic exercise on systemic cytokine in patients with chronic kidney disease. Ren Fail 37(9): 1430-1434.
- Dungey M, Bishop NC, Young HM, Burton JO, Smith AC (2015) The Impact of Exercising During Haemodialysis on Blood Pressure, Markers of Cardiac Injury and Systemic Inflammation--Preliminary Results of a Pilot Study. Kidney Blood Press Res 40(6): 593-604.
- Moraes C, Marinho SM, da Nobrega AC, de Oliveira Bessa B, Jacobson LV, et al. (2014) Resistance exercise: a strategy to attenuate inflammation and protein-energy wasting in hemodialysis patients? Int Urol Nephrol 46(8):1655-1662.
- Barcellos FC, Del Vecchio FB, Reges A, Mielke G, Santos IS, et al. (2018) Exercise in patients with hypertension and chronic kidney disease: a randomized controlled trial. J Hum Hypertens 32(6): 397-407.
- Esgalhado M, Stockler-Pinto MB, de Franca Cardozo LF, Costa C, Barboza JE, et al. (2015) Effect of acute intradialytic strength physical exercise on oxidative stress and inflammatory responses in hemodialysis patients. Kidney Res Clin Pract 34(1): 35-40.
- Afshar R, Shegarfy L, Shavandi N, Sanavi S (2010) Effects of aerobic exercise and resistance training on lipid profiles and inflammation status in patients on maintenance hemodialysis. Indian J Nephrol 20(4): 185-189.
- Kopple JD, Wang H, Casaburi R, Fournier M, Lewis MI, et al. (2017) Exercise in maintenance hemodialysis patients induces transcriptional changes in genes favoring anabolic muscle. J Am Soc Nephrol 18(11): 2975-2986.
- Oliveros RM, Avendano M, Bunout D, Hirsch S, De La Maza MP, Pedreros C, et al. (2011) [A pilot study on physical training of patients in hemodialysis]. Rev Med Chil 139(8): 1046-1053.
- Molsted S, Eiken P, Andersen JL, Eidemak I, Harrison AP (2014) Interleukin-6 and vitamin D status during high-intensity resistance training in patients with chronic kidney disease. BioMed research international 2014: 176190.
- Cheema BS, Abas H, Smith BC, O'Sullivan AJ, Chan M, et al. (2011) Effect of resistance training during hemodialysis on circulating cytokines: a randomized controlled trial. Eur J Appl Physiol 111(7): 1437-1445.
- Watson EL, Viana JL, Wimbury D, Martin N, Greening NJ, et al. (2017) The Effect of Resistance Exercise on Inflammatory and Myogenic Markers in Patients with Chronic Kidney Disease. Frontiers in Physiology 8: 541.






























