Clinical Efficacy of SelectMDx for the Detection of Clinically Significant Prostate Cancer
Perez-Lanzac A*
Department of Urology, Hospital Ruber International, Madrid, Spain
Submission:January 18, 2022;Published:January 26, 2022
*Corresponding author:Alberto Perez-Lanzac, Servicio de Urologia, Hospital Ruber International, Spain
How to cite this article:Perez-Lanzac A. Clinical Efficacy of SelectMDx for the Detection of Clinically Significant Prostate Cancer. JOJ Urology & Nephrology, 2022; 7(4): 555718. DOI: 10.19080/JOJUN.2022.07.555718
Abstract
Prostate Cancer (PCa) is the second most frequent cancer diagnosed in men. Elevated serum PSA levels (>4.0 ng/mL) detection is commonly used as a threshold for recommending the tissue biopsy. However, PSA serum levels are not adequate to correctly localize and stage PCa, and to determine its volume and aggressiveness. Therefore, there is a need for healthcare specialists to base the therapeutic decisions on new biomarkers and advanced imaging findings. SelectMDx is a novel non-invasive urine-based assay that measures the expression of two mRNA cancer-related biomarkers (HOXC6 and DLX1) combined with traditional clinical risk factors. Validation studies evidenced the higher precision of SelectMDx urine test to predict PCa. Combining the assay with multi-parametric magnetic resonance imaging (mpMRI) can improve PCa diagnosis and therefore the treatment. Compared to traditional diagnosis strategy, the use of the SelectMDx test in men with elevated PSA levels may lead to a reduction in overdiagnosis and overtreatment. SelectMDx has to be tested in larger and heterogeneous populations in diverse clinical settings to further assess its clinical validity and utility.
Keywords: Prostate; Cancer; Biomarker; Urine; SelectMDx
Introduction
Prostate Cancer (PCa) is the second most common and fifth most aggressive cancer diagnosed among men worldwide. According GLOBOCAN data, in 2018, PCa represented the 7.1% of all cancer diagnoses, and 13.5% of all male cancer diagnoses (after lung cancer). Mortality rates in PCa have declined in many countries, partly related to earlier diagnosis and the improved management of the disease [1]. Survival rates have also increased in the last years. The 5-year survival rate for men diagnosed is approximately 98% in US and 76-88% in Europe [2]. However, PCa still accounted for 3.8% of all cancer-related deaths (6.7% in men) in 2018 [1]. PCa is especially incident in countries with a high development index. The highest incidence rates were observed in Australia/New Zealand population (86.4/100,000), whereas South Central populations have the lowest rates (5.0/100,000) [1-3]. Moreover, mutations in BRCA1 and BRCA2 genes increase the PCa risk by 2.64-fold in men. PCa shows clinical/pathological and progression variability and at early stages may be asymptomatic [2]. The disease is supposed to initiate with a pre-malignant lesion characterized by the presence of abnormalities in the prostate epithelial cells in the glands or ducts.
Localized PCa is the transition phase between benign epithelium and invasive cancer and represents an early stage. The malignancy is confined to the prostate and is classified as indolent or aggressive according to clinical/pathological features [2]. Metastatic PCa occurs when it spreads to bones, lymph nodes and bladder. To this state, cancer may respond to androgen deprivation therapy, but it can also progress to a castration resistant PCa [4]. PCa is normally diagnosed by Transrectal Ultrasound (TRUS)-guided biopsy, after detecting abnormal serum Prostate-Specific Antigen (PSA) levels, and/or Digital Rectal Examination (DRE) [5]. Blood level of PSA is often elevated in men with PCa, although there are other non-cancerous conditions that can cause serum PSA increase [6-8]. Even though, PSA serum value >4.0 ng/mL is commonly used by healthcare specialists as a threshold for recommending the tissue biopsy [7]. Since 1990s, PSA has been long employed as a PCa biomarker [9,10]. Despite data supporting the usefulness of PSA for early detection of the disease, its used has become controversial. This biomarker has a Positive Predictive Value (PPV) of 25-40% to detect PCa. Eventually, 65-70% of the patients presenting increased PSA level (4.0-10.0 ng/ml) obtain a negative result in biopsy[7]. Therefore, PSA has a limited specificity for detecting a clinically significant PCa (csPCa; grade group [GG] ≥2) leading to overdiagnosis and overtreatment in patients with indolent disease [9,10].
Moreover, 15% of men diagnosed with PCa have PSA levels <4.0 ng/ml [7]. New PCa biomarkers have emerged to cope with PSA limitations. An ideal PCa biomarker would provide diagnostic and prognostic information to guide in treatment decisions. The biomarker should have high specificity and sensitivity, with a PPV and Negative Predictive Value (NPV) for its outcome, and should differentiate a csPCa (GG ≥2) from indolent disease, inexpensive, and easily accessible. Additionally, a biomarker should be easily measured, preferably non-invasive [11,12]. Over the last years, several PCa tests have been commercialized, including urinebased (ExoDx Prostate IntelliScore, EPI, SelectMDx, prostate cancer antigen 3, PCA3, Mi‑Prostate Score, MiPS) blood-based (Four Kallikrein Score, Prostate Health Index), and tissue-based ones (Oncotype DX, Prolaris, ConfirmMDx, Decipher).
Evidence on SelectMDx
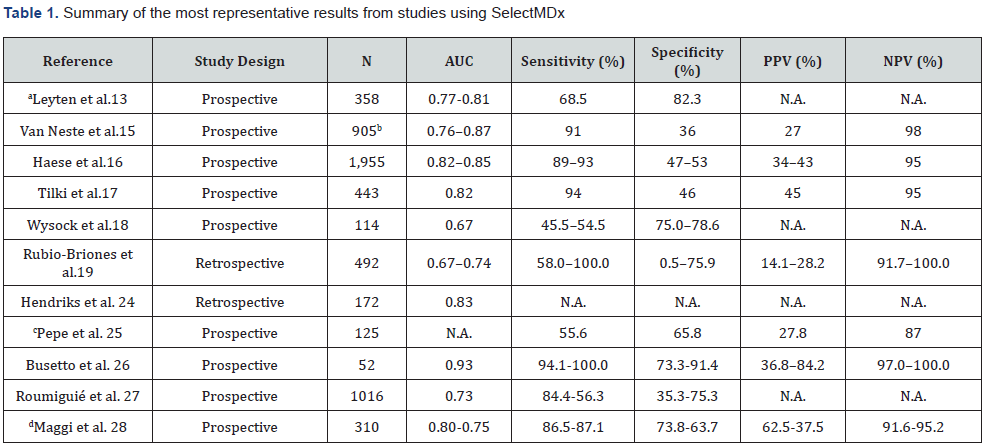
AUC (Area Under The Curve); PPV (Positive Predictive Value); NPV (Negative Predictive Value); N.A (Not Available)
aIncluding the analysis of TDRD1 mRNA levels;
bTotal patients (training and validation cohorts);
cUse of SelectMDx in individuals in active surveillance (off-label);
dObtained values in predicting PCa (first) and csPCa
The SelectMDx urine-based assay combines molecular test with clinical factors of age, PSA, prostate volume, and DRE findings to estimate the likelihood of PCa and GG ≥2 on biopsy [9]. SelectMDx molecular test detects HOXC6 (homeobox C6) and DLX1 (distal-less homeobox 1) mRNA levels in urine sample obtained after DRE [12]. The most representative results from SelectMDx validation studies are summarized in Table 1. In 2015, Leyten et al. identified three genes in urinary sediment for the early diagnosis of PCa: HOXC6, DLX1 and TDRD1 (Tudor Domain- Containing Protein 1) [13]. Authors suggested that this novel three-gene panel had higher precision to predict high-grade PCa cancer [13] (Gleason Score [GS] ≥7 which is equivalent to GG ≥2 [14]).
The Area Under The Curve (AUC) was 0.77 (95% Confidence Interval [CI]: 0.71–0.83), better than urinary test PCA3 (AUC: 0.68; 95% CI: 0.62–0.75) or serum PSA (AUC: 0.72; 95% CI: 0.65–0.78). Combining the three-gene panel with PSA, the predictive accuracy improved (AUC: 0.81; 95% CI: 0.75–0.86) [13]. The precision of the new model was maintained in groups with different PSA levels, noting predictive accuracy is barely affected by PSA [13]. In 2016, Van Neste et al. developed a multimodal model, incorporating mRNA of HOXC6 and DLX1 and traditional risk factors, to identify patients with high-grade PCa (GS7 ≥7) on prostate biopsy [15]. HOXC6 and DLX1 mRNA levels were measured in urine samples collected after DRE and prior to prostate biopsy. The multimodal risk score was developed on a cohort of 519 patients and then, clinically validated in an independent cohort (n=386) [15].
In the first cohort (n=519) the exploratory analysis of RNA levels (HOXC4, HOXC6, TDRD1, DLX1, KLK3, and PCA3 gene) was performed. The combination of HOXC6 and DLX1 mRNA levels displayed the highest AUC (0.76; 95% CI: 0.71–0.81) for detecting high-grade PCa [15]. The combination was validated in the 386-patients cohort (AUC: 0.73; 95% CI: 0.67–0.78; p = 0.4 difference of AUCs). The combination model reached an overall AUC of 0.90 (95% CI: 0.85–0.95) in the validation cohort (AUC=0.86 in the training cohort) [15]. The mRNA signature, PSA density, and previous cancer-negative prostate biopsies were the most-significant components. By contrast, PSA, age, and family history had nonsignificant contributions to the model. The model which included DRE as an additional risk factor obtained an AUC of 0.86 (95% CI: 0.80–0.92; AUC 0.90 in the training cohort). Both models were validated, with no significant changes in AUC in the validation cohort [15].
According to these published data, SelectMDx could reduce the number of unnecessary prostate biopsies and potential overtreatment [7]. Haese et al. in a recent multicenter and multinational study (The Netherlands, France, and Germany), SelectMDx was evaluated in biopsy-naïve patients divided into two groups (a training cohort and a validation cohort) [16]. The SelectMDx model consisted of urinary HOXC6 and DLX1 mRNA in addition to age, PSAD, and DRE result (without incorporating history of prior biopsy) [16]. Considering patients with PSA level <10 ng/ml in the validating cohort (n=715), the AUC was 0.82 (95% CI: 0.79–0.86), with 89% sensitivity, 53% specificity, and 95% NPV. In the complete validation cohort (n=916) which included all PSA levels reached similar results, in terms of AUC (0.85), sensitivity (93%), specificity (47%), and NPV (95%; 95% CI: 0.83–0.88) [16]. Notably, when not considered prostate volume (i.e., PSA density was replaced with PSA), SelectMDx show decreased sensitivity (87%), specificity (38%), and NPV (92%). The inclusion of prostate volume in the SelectMDx model has clinical implications, particularly in biopsy-naïve men, given that measurement of prostate volume requires ultrasound or Magnetic Resonance Imaging (MRI) [16]. Tilki et al. evaluated SelectMDx clinical performance in a German cohort undergoing initial prostate biopsy [17].
For the detection of PCa GG ≥2, AUC of SelectMDx was 0.82 (95% CI: 0.78-0.86). Sensitivity, specificity, PPV, and NPV were 94% (95% CI: 89–98%), 46% (95% CI: 40–52%), 45% (95% CI: 42–47%), and 95% (95% CI: 90–97%), respectively. A total of 34% of biopsies would have been avoided. Therefore, results validated the clinical significance of SelectMDx [17]. Wysock et al. compared the blood-based test, 4Kscore, with SelectMDx in newly-diagnosed patients (referred with elevated PSA) [18]. Although limited by a sample size of 50 men who underwent prostate biopsy, AUC for SelectMDx was 0.67 (95% CI: 0.52–0.83) for detecting a PCa GG≥2. Authors also reported a discordance between the two biomarkers in guiding decision to biopsy, suggesting the need for studies to optimize its clinical application [18]. In a recent study, Rubio- Briones et al. performed an external validation of the urine-based SelectMDx test [19]. They analyzed 492 archived samples from patients who underwent a prostate biopsy in an opportunistic screening scenario, with PSA 3-10 ng/mL and/or suspicious DRE and without previous multi‐parametric MRI (mpMRI). SelectMDx test yield an AUC for PCa GG ≥2 of 0.75 (95% CI: 0.690‐0.807 [19]). This AUC was lower compared with Van Neste et al. [15,19].
Authors explained that the discrepancy in the results were based on differences between the two cohorts. They conclude that SelectMDx test could be a potential and useful tool, avoiding around 37% of biopsies in men with PSA between 3 to 10 ng/mL and/or suspicious DRE. In addition, those cases could be selected for a mpMRI (before the first biopsy) to optimize its use [19]. Quintana et al. performed a real-world evidence study analyzing the usefulness and applicability of the SelectMDx marker in a clinical practice settings [20] . The probability of PCa or csPCa after biopsy was 88% and 63%, respectively.
Selectmdx and Multiparametric MRI
The emergence of new biomarkers and the advances in MRI and functional imaging have improved diagnostic protocols [21]. One of the most notable imaging advances has been the mpMRI. This imaging technique has demonstrated its value as a PCa diagnostic tool, as well as an instrument for target biopsy [21, 22]. Indeed, the European Association of Urology guidelines recommend performing mpMRI before prostate biopsy in biopsynaïve patients when PCa is suspected [23]. Combining mpMRI with urine-based and blood-based biomarkers could improve the csPCa detection by avoiding unnecessary biopsies. Hendriks et al. evaluated this association in 172 patients from the study of Van Neste et al. [15] who had undergone this imaging technique [24]. The AUC of SelectMDx with mpMRI was 0.83 (95% CI: 0.77– 0.89), in comparison with 0.66 (95% CI: 0.58–0.74) of PSA and 0.65 (95% CI: 0.57–0.74) with PCA3 [24]. Pepe et al. evaluated” the diagnostic precision of SelectMDx for detecting csPCa in 125 patients in active surveillance [25]. Post-DRE urine was obtained from 45 patients, and 20% of them were classified as csPCa (GG ≥2). Sensitivity results were 55.6% with SelectMDx vs. 66.6% with mpMRI, 65.8% vs. 87.7% of specificity, 27.8% vs. 54.5% of PPV, 87% vs. 92.3% of NPV, and 70.3% vs. 84.9% of diagnostic accuracy [25].
The study carried out by Busetto et al. evaluated the diagnostic precision of SelectMDx and its association with mpMRI for predicting PCa and csPCa [26]. The study included 52 men scheduled for initial prostate biopsy, based on elevated total PSA level (> 3 ng/ml) or abnormal DRE. All subjects underwent mpMRI, SelectMDx, and PSA determination. Prostate imagingreporting and data system score (PI-RADS) was used for grading lesion (1 to 5). Urine samples were collected after standardized DRE [26]. SelectMDx showed the same sensitivity (94.1%) but higher specificity (91.4%) than mpMRI (80.0%) in predicting PCa at biopsy. The combination of SelectMDx and mpMRI showed higher specificity (91.4%) compared to the combination with PSA (85.7%). SelectMDx showed higher specificity (73.3%) than PSA (13.3%) or mpMRI (64.4%) in csPCa prediction [26].
The association of SelectMDx with mpMRI had higher specificity (75.6%) than PSA plus mpMRI (68.9%). Decision Curve Analyses (DCA) were performed to evaluate clinical benefit of both tests (alone or together). DCA for SelectMDx alone revealed the highest net benefit for the detection of PCa. In this study, the combination of mpMRI and PSA density was revealed as the best strategy for the detection of csPCa only slightly superior to SelectMDx and mpMRI combination [26]. Roumiguié et al. evaluated” SelectMDx in a cohort of upfront mpMRI and imageguided biopsy patients [27]. The study included 1,016 patients and reported a similar accuracy between tools (AUC: 0.67, for both). In their analysis combining PI-RADS score with either SelectMDx or the sole clinical variables resulted in a higher performance compared to either test separately (AUC: 0.73 and 0.80, respectively) [27]. In a recent study, suggested that SelectMDx could be a reliable predictor after an initial negative mpMRI [28]. The prospective study included 310 patients to evaluate the diagnostic accuracy of SelectMDx and its association to mpMRI for predicting PCa in biopsies.
All patients underwent SelectMDx and mpMRI before performing prostate biopsy [28]. The AUC with SelectMDx for PCa was 0.80 (95% CI: 0.76–0.85) and 0.75 (95% CI: 0.70–0.81) for csPCa. The AUC when using together mpMRI and SelectMDx was 0.72 (95% CI: 0.67–0.77) to detect PCa, and 0.73 (95% CI: 0.67–0.80) for csPCa [28]. The best combination for the detection of PCa and csPCa was SelectMDx plus mpMRI if compared to the association of mpMRI with other diagnostic tools (i.e., PSA) [28]. Authors proposed the best clinical strategy, consisting of getting a mpMRI first and, if positive (considered as PI-RADS score 3–5), perform a biopsy. If negative, using SelectMDx and doing a biopsy only if positive. By using this strategy, it would be possible to avoid 40.0% of biopsies, and detect 98.1% of PCa (96.8% of csPCa) [28].
Selectmdx for Active Surveillance
Overtreatment is a consequence of the PCa overdetection, particularly problematic in individuals with low risk of aggressive or lethal disease. In these cases, patients might be exposed to the treatment’s adverse effects, with minimal benefits in terms of cancer-specific survival. Active Surveillance (AS) is focused on the prevention of overtreatment by selecting low-risk PCa patients [30]. Thus, the challenge is to identify patients with csPCa in an early stage and follow-up those with a lower risk to avoid unnecessary treatments. Clinical guidelines recommend the AS of PCa patients with <10 ng/ml PSA serum levels and GS ≤6. The AS programs also recommend serial PSA blood tests (about every 6 months), DRE (at least once per year) and prostate biopsies and imaging tests (every 1 to 3 years) to identify indications of tumor progression [30, 31].
In a recent study, Fiorella et al. evaluated the predictive value for pathological progression of PCA3 and SelectMDx, in an individual and combined way, in a standard AS program [32]. Authors correlated PCA3 and SelectMDx scores, as well as clinical and pathological variables with pathological progressionfree survival (PPFS). SelectMDx showed statistically significant differences related to PPFS (HR 1.04) with an AUC of 0.71 at 5 years. This study concluded that in lower risk PCa, a SelectMDx value >5 predicted 5 years of PPFS with a moderate discrimination ability superior to PCA3 “test. Pepe et al. determined the diagnostic accuracy of the SelectMDx test in the diagnosis of csPCa in men enrolled in the AS program [25].
Authors suggested that transperineal saturation biopsy combined with MRI/TRUS fusion biopsy significantly overcome the diagnostic accuracy of SelectMDx (70.3%) in the diagnosis of csPCa. Thus, they stated that SelectMDx may be useful for assisting in the re-evaluation of men in AS to select those at risk in need of early repeat prostate biopsy. Zhao et al. also indicated that novel noninvasive urine tests are promising alternatives to monitor patients with clinically insignificant cancer [33]. Those tests should be considered to identify and preclude AS patients with indolent cancer and to personalize monitoring strategies for AS patients.
Strengths and Limitations of Selectmdx
One of the main challenges associated with SelectMDx in clinical practice is to select the adequate patients for biopsy and, among diagnosed, differentiate tumors with an indolent course from those with an unfavorable prognosis. This will help to take the best therapeutic decision for each case, avoiding unnecessary interventions. Several blood-based and urine-based biomarkers have been designed to help in this decision [34]. SelectMDx is a fast, simple, and reliable test to dismiss PCa, with high sensitivity for the detection of high-grade PCa, and can be used to select those patients who should undergo mpMRI and/or prostate biopsy [13,15,16,24]. PCA3 assay is another urine-based biomarker, that measures PCA3 mRNA levels in urine after DRE, and extrapolates a PCA3 score [29,30]. PCA3 test is more reliable than PSA and it is independent of prostate volume. However, predictive tests that include clinical variables, such as SelectMDx, are considered more reliable than those which do not so, like PCA3 [30].
SelectMDx was found to significantly improve the detection of csPCa with an AUC of 0.77 compared to PSA (AUC: 0.72) and PCA3 (AUC: 0.68) [13]. Moreover, PCA3 assay may be useful for monitoring patients with low-grade PCa, but the predictive value for aggressive disease has been contradictory [31,34]. In a recent study, the impact of PCA3 and SelectMDx, individually or combined, in predicting pathological progression was evaluated [35]. The study revealed that SelectMDx has a discriminatory ability superior to PCA3. SelectMDx predicted five years of pathological progression free survival with a moderate discrimination ability outperforming PCA3 [34]. Dijkstra et al. evaluated the costeffectiveness of SelectMDx to identify patients for TRUS-guided biopsy and compared with PSA to select TRUS-guided biopsy [36]. Authors demonstrated cost-effectiveness of SelectMDx as a second step diagnostic decision tool. Compared to traditional PSA strategy, 41% of biopsies could be prevented [36].
Authors concluded that the use of the SelectMDx test in men with PSA levels >3 ng/mL may lead to a reduction in total costs per patient and in overdiagnosis and overtreatment, as well as a gain in Quality-Adjusted Life Years (QALYs) [36]. Govers et al. evaluated the cost-effectiveness of SelectMDx for PCa in four European countries (France, Germany, Italy, and Spain), and concluded that using SelectMDx could gained QALYs while saving healthcare costs in the initial diagnosis of PCa [37].
Most of the published studies have described the development and initial clinical validation. Nevertheless, few of them have evaluated the diagnostic performance characteristics of SelectMDx, and compared with mpMRI, PCA3, or other biomarkers results. Despite the positive results obtained for SelectMDx, the overall evidence is limited, especially for determining its real clinical validity and utility [13,15-20,24-28,36] . In addition, methodological limitations of studies (such as the retrospective design, multiple studies evaluating the same cohort, failure to report confidence intervals, and small sample sizes) also restrict further and stronger conclusions. Furthermore, most of the studies have been conducted in European studies, thus results cannot be easily extrapolated worldwide.
Conclusion
SelectMDx is a novel and reliable PCa biomarker which improves PCa detection and reduces unnecessary biopsies and overdiagnoses. However, SelectMDx has to be tested in larger and heterogeneous populations in diverse clinical settings to further assess its clinical validity and utility.
Acknowledgement
Authors also express gratitude to Meisys (Madrid, Spain) for writing assistance.
Conflict of Interest
No conflict of interst
References
- Bray (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians 68 (6): 394–424.
- Rawla P (2019) Epidemiology of Prostate Cancer. World J Oncol 10(2): 63-89.
- Barsouk A (2020) Epidemiology, Staging and Management of Prostate Cancer. Med Sci (Basel) 8(3):28.
- Arora K, Barbieri CE (2018) Molecular Subtypes of Prostate Cancer. Curr Oncol Rep 20(8):58.
- Shore N (2014) Management of early-stage prostate cancer. Am J Manag Care 20 (12 Suppl):S260-2
- Thompson IM, Pauler DK, Goodman PJ (2004) Prevalence of prostate cancer among men with a prostate-specific antigen level < or =4.0 ng per milliliter. New England Journal of Medicine 350(22):2239–2246.
- Hendriks RJ, van Oort IM, Schalken JA (2017) Blood-based and urinary prostate cancer biomarkers: a review and comparison of novel biomarkers for detection and treatment decisions. Prostate Cancer Prostatic Dis 20(1):12-19.
- Schatten H (2018) Brief Overview of Prostate Cancer Statistics, Grading, Diagnosis and Treatment Strategies. Adv Exp Med Biol 1095:1-14.
- Eyrich NW, Morgan TM, Tosoian JJ (2021) Biomarkers for detection of clinically significant prostate cancer: contemporary clinical data and future directions. Transl Androl Urol 10(7):3091-3103.
- Alford AV, Brito JM, Yadav KK, Yadav SS, Tewari AK et al.(2017) The Use of Biomarkers in Prostate Cancer Screening and Treatment. Rev Urol 19(4):221-234.
- Prensner JR, Rubin MA, Wei JT, Chinnaiyan AM (2012) Beyond PSA: the next generation of prostate cancer biom Sci Transl Med 4(127):127rv3.
- Narayan VM (2020) A critical appraisal of biomarkers in prostate cancer. World Journal of Urology 38(3): 547–554.
- Leyten GH, Hessels D, Smit FP, Jannink SA, de Jong H et al. (2015) Identification of a Candidate Gene Panel for the Early Diagnosis of Prostate Clin Cancer Res 21(13):3061-3070.
- Sehn JK (2018) Prostate Cancer Pathology: Recent Updates and Controversies. Mo Med 115(2):151-155.
- Van Neste L, Hendriks RJ, Dijkstra S, Trooskens G, Cornel EB et al. (2016) Detection of High-grade Prostate Cancer Using a Urinary Molecular Biomarker-Based Risk Score. Eur Urol 70(5):740-748.
- Haese A, Trooskens G, Steyaert S, Hessels D, Brawer M et al. (2019) Multicenter Optimization and Validation of a 2-Gene mRNA Urine Test for Detection of Clinically Significant Prostate Cancer before Initial Prostate Biopsy. J Urol 202(2):256-263.
- Tilki, D (2019 )Validation of a two-gene mRNA urine test for detection of high-grade prostate cancer in German men. Journal of Clinical Oncology 37(7).
- Wysock, J.S, Becher E, Persily J, Loeb S, Lepor H (2020) Concordance and performance of 4Kscore and SelectMDx for informing decision to perform prostate biopsy and detection of prostate cancer. Urology141:119–124.
- Rubio-Briones J (2020) Validation of a 2-gene mRNA urine test for the detection of ≥GG2 prostate cancer in an opportunistic screening population. The Prostate 80(6):500–507.
- Quintana LM, Fernández Pascual E, Linares Espinos E, Martinez-Ballesteros C, Martin-Vivas C et al. (2020) Initial experience with SelectMDx® in the diagnosis of prostate cancer in a real-world evidence clinical practice setting. Actas Urol Esp (Engl Ed) 44(6):400-407.
- Litwin MS, Tan HJ (2017) The Diagnosis and Treatment of Prostate Cancer: A Review. JAMA 317(24):2532-2542.
- Panebianco V, Barchetti F, Sciarra A, Ciardi A, Indino EL, et al. (2015) Multiparametric magnetic resonance imaging vs. standard care in men being evaluated for prostate cancer: a randomized study. Urol Oncol 33(1):17.e1-17.e7.
- Mottet N, van den Bergh R.C.N, Briers E, Van den Broeck T, Cumberbatch M.G, et al. (2020) EAU-EANM-ESTRO-ESUR-SIOG Guidelines on Prostate Cancer—2020 Update. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur. Urol 79(2):243–262.
- Hendriks R.J. (2017) A urinary biomarker-based risk score correlates with multiparametric MRI for prostate cancer detection. The Prostate 77(14):1401–1407.
- Pepe P, Dibenedett G, Pepe L, Pennisi, M. (2020) Multiparametric MRI versus SelectMDx accuracy in the diagnosis of clinically significant PCa in men enrolled in active surveillance. In Vivo 34(1):393–396.
- Busetto GM, Del Giudice F, Maggi M, De Marco F, Porreca A et al. (2020) Prospective assessment of two-gene urinary test with multiparametric magnetic resonance imaging of the prostate for men undergoing primary prostate biopsy. World J Urol 39(6):1869-1877.
- Roumiguié M, Ploussard G, Nogueira L, Bruguière E, Meyrignac O et al. (2020) Independent Evaluation of the Respective Predictive Values for High-Grade Prostate Cancer of Clinical Information and RNA Biomarkers after Upfront MRI and Image-Guided Biopsies. Cancers (Basel) 12(2):285.
- Maggi M, Del Giudice F, Falagario UG, Cocci A, Russo GI et al. (2021) SelectMDx and Multiparametric Magnetic Resonance Imaging of the Prostate for Men Undergoing Primary Prostate Biopsy: A Prospective Assessment in a Multi-Institutional Study. Cancers (Basel) 13(9):2047.
- Carneiro A, Priante Kayano P, Gomes Barbosa ÁR, Langer Wroclawski M, Ko Chen C et al. (2018) Are localized prostate cancer biomarkers useful in the clinical practice?. Tumour Biol 40(9):1010428318799255.
- Bruinsma SM, Bangma CH, Carroll PR, Leapman MS, Rannikko A et al. (2016). Active surveillance for prostate cancer: a narrative review of clinical guidelines. Nat Rev Urol 13(3):151-67.
- American Cancer Society [Internet]. Observation or Active Surveillance for Prostate Cancer [Consulted on 13/12/2021].
- Fiorella D, Marenco JL, Mascarós JM, Borque-Fernando Á, Esteban LM, et al. (2021) Role of PCA3 and SelectMDx in the optimization of active surveillance in prostate cancer. Actas Urol Esp (Engl Ed) 45(6):439-446. English,
- Zhao F, Vesprini D, Liu RSC, Olkhov-Mitsel E, Klotz LH, et al. (2019) Combining urinary DNA methylation and cell-free microRNA biomarkers for improved monitoring of prostate cancer patients on active surveillance. Urol Oncol 37(5):297.e9-297.e17.
- Leslie SW, Soon-Sutton TL, Sajjad H, Siref LE (2021) Prostate Cancer. 2021 Sep 17. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; PMID: 29261872.
- Kohaar I, Petrovics G, Srivastava S. (2019) A Rich Array of Prostate Cancer Molecular Biomarkers: Opportunities and Challenges. Int J Mol Sci 20(8):1813.
- Dijkstra S, Govers TM, Hendriks RJ, Schalken JA, Van Criekinge et al. (2017). Cost-effectiveness of a new urinary biomarker-based risk score compared to standard of care in prostate cancer diagnostics - a decision analytical model. BJU Int 120(5):659-665.
- Govers TM, Hessels D, Vlaeminck-Guillem V, Schmitz-Dräger BJ, Stief CG, et al. (2019) Cost-effectiveness of SelectMDx for prostate cancer in four European countries: a comparative modeling study. Prostate Cancer Prostatic Dis 22(1):101-109.
- EviCore Healthcare [Internet]. SelectMDx Lab Management Guidelines [Consulted on 05/11/2021].






























