The Renal Effects of Neprilysin Inhibition in Heart Failure and Hypertension
Girish Singhania1*, Subhankar Samal2 and Namrata Singhania3
1Department of Hospital Medicine, CHI St Vincent Infirmary, USA
2 Department of Medicine, University of Florida, USA
3Mount Carmel East Hospital, Columbus, USA
Submission: August 02, 2019; Published: Septembre 23, 2019
*Corresponding author:Girish Singhania, Department of Hospital Medicine, CHI St Vincent Infirmary, Little Rock, USA
How to cite this article:Girish Singhania, Subhankar Samal, Namrata Singhania. The Renal Effects of Neprilysin Inhibition in Heart Failure and Hypertension. 2019; 7(1): 555701. DOI: 10.19080/JOJUN.2019.07.555701
Abstract
Cardiovascular disease is one of the major causes of mortality throughout the world. Renin Angiotensin blockers are well known for their beneficial effects in patients with heart failure. New emerging medications like neprilysin inhibitors have shown trend towards positive benefit in patients with heart failure. Data on the effect on renal function is still limited. Through this article, we are reviewing multiple randomized clinical trials involving Neprilysin and summarize the renal effects of the medicine.
Keywords: Neprilysin; heart failure; hypertension; creatinine
Abbreviations: NEPi: Neprilysin inhibitors; HF: Heart failure; ACE: Angiotensin converting enzyme; ARB: Angiotensin receptor blocker HFREF: Heart Failure with Reduced Ejection Fraction
Introduction
Neprilysin inhibitors (NEPi) represent an emerging therapeutic option for treatment of congestion in heart failure (HF) as well as hypertension (HTN). While these agents have shown promising results regarding HF events, lowering blood pressure, and mortality, their impact on the kidney remains largely unknown. The aim of this study is to evaluate the currently available evidence on the effect of NEPi use on kidney-related parameters in patients with HF or HTN.
Methods
A search of articles cited in PubMed database using key words “neprilysin or neutal endopeptidase”, “heart failure” and “hypertension” was performed. Animal studies were excluded. Only those studies containing kidney-related parameters such as serum creatinine or estimated glomerular filtration rate were selected. Relevant data including changes in renal function, blood pressure and mortality were extracted and compared.
Discussion
A total of 43,368 patients from 13 randomized controlled trials with data pertaining to NEPi use (4 studies used LCZ696, 3 used Omapatrilat, 2 used Sampatrilat, 1 used Daglutril, 1 used Sacubitril and 1 used GW660511X) in HF (5 studies) and HTN (8 studies) were included in this study (Table 1). 8 studies used ACE-I while 4 used ARBS as control. The mean age of the patients was 59.19 years with a mean baseline systolic blood pressure of 118 to 162 mm Hg. The follow up periods were between 7 days and 27 months. Concerning the reduction in BP, 11 studies reported better or similar BP lowering effect of NEPi compared to control drugs (ACE-I, ARBs or placebo).
Table 1: Review of randomized controlled trials showing renal effects of Neprilysin inhibitors.
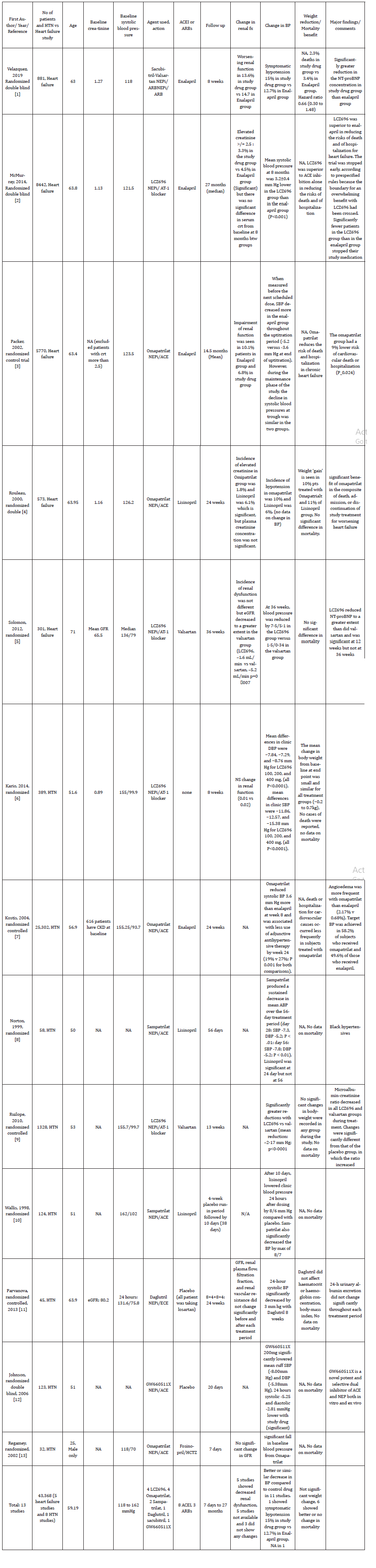
One study reported symptomatic hypotension in 15% in study drug (Sacubitril/Valsartan) group vs 12.7% in Enalapril group. BP data was unavailable in 1 study. Although the data regarding renal dysfunction was unavailable in 5 out of 13 studies, 4 reported less renal dysfunction with NEPi, 3 did not show any significant change compared to the control drug and 1 showed less frequent worsening renal function (13.6%) in study drug group (Sacubitril/Valsartan) vs Enalapril group (14.7%). 6 studies showed better or at least no change in mortality (unavailable in 7).
Conclusion
Current evidence suggests that the novel angiotensin receptor-NEPi can lower HF events and mortality in patients with HF while having a modest impact on lowering their blood pressure. Although it is mechanistically conceivable that these medications portend favorable effects on the kidneys, so far studies have not shown any evidence of improvement in kidney-related parameters in these patients. There is one more randomized controlled trial “EntrestoTM (LCZ696) In Advanced Heart Failure (LIFE Study - NCT02816736) which is currently enrolling patients with advanced HFrEF and will assess the safety, tolerability, and efficacy of ARNI. Hopefully we will have more data from this upcoming study.
References
- E J Velazquez, D A Morrow, A D DeVore, C I Duffy, A P Ambrosy, et al. (2019) Angiotensin-Neprilysin Inhibition in Acute Decompensated Heart Failure. N Engl J Med 380: 539–548.
- J J V Mcmurray, M Packer, A S Desai, J Gong, M P Lefkowitz, et al. (2014) Angiotensin–Neprilysin Inhibition versus Enalapril in Heart Failure. New J Med 11: 993–1004.
- M Packer, R M Califf, M A Konstam, H Krum, J J McMurray, et al. (2002) Comparison of omapatrilat and enalapril in patients with chronic heart failure: the Omapatrilat Versus Enalapril Randomized Trial of Utility in Reducing Events (OVERTURE). Circulation 106: 920–6.
- J L Rouleau, M A Pfeffer, D J Stewart, D Isaac, F Sestier, et al. (2000) Comparison of vasopeptidase inhibitor, omapatrilat, and lisinopril on exercise tolerance and morbidity in patients with heart failure: IMPRESS randomised trial. Lancet 356: 615–20.
- S D Solomon, M Zile, B Pieske, A Voors, A Shah, et al. (2012) The Angiotensin Receptor Neprilysin Inhibitor LCZ696 in Heart Failure with Preserved Ejection Fraction (PARAMOUNT). Lancet 380(9851): 1387-95.
- K Kario, N Sun, F T Chiang, O Supasyndh, S H Baek, et al. (2014) Efficacy and safety of LCZ696, a first-in-class angiotensin receptor neprilysin inhibitor, in Asian patients with hypertension: a randomized, double-blind, placebo-controlled study. Hypertens 63: 698–705.
- H R Black, D Henry, J B Kostis, E Levy, M Packer, et al. (2004) Omapatrilat and enalapril in patients with hypertension: The Omapatrilat Cardiovascular Treatment vs Enalapril (OCTAVE) trial, Am J Hypertens 17(2): 103-11.
- G R Norton, A J Woodiwiss, C Hartford, B Trifunovic, S Middlemost, et al. (1999) Sustained antihypertensive actions of a dual angiotensin-converting enzyme neutral endopeptidase inhibitor, sampatrilat, in black hypertensive subjects. Am J Hypertens 12: 563–71.
- L M Ruilope, A Dukat, M Bohm, Y Lacourciere, J Gong, et al. (2010) Blood-pressure reduction with LCZ696, a novel dual-acting inhibitor of the angiotensin II receptor and neprilysin: a randomised, double-blind, placebo-controlled, active comparator study. Lancet 375(9722): 1255-66.
- E J Wallis, L E Ramsay, J Hettiarachchi (1998) Combined inhibition of neutral endopeptidase and angiotensin-converting enzyme by sampatrilat in essential hypertension. Clin Pharmacol Ther 64: 439–49.
- Parvanova, I M van der Meer, I Iliev, A Perna, F Gaspari, et al. (2013) Effect on blood pressure of combined inhibition of endothelin-converting enzyme and neutral endopeptidase with daglutril in patients with type 2 diabetes who have albuminuria: a randomised, crossover, double-blind, placebo-controlled trial. Lancet Diabetes Endocrinol 1: 19–27.
- A G Johnson, G L Pearce, T M Danoff (2006) Original Article controlled, parallel-group study to assess the efficacy and safety of dual ACE / NEP inhibitor GW660511X in mild-to-moderate hypertensive patients 496–503.
- F Regamey, M Maillard, J Nussberger, H R Brunner, M Burnier (2002) Renal hemodynamic and natriuretic effects of concomitant Angiotensin-converting enzyme and neutral endopeptidase inhibition in men. Hypertens 40: 266–72.






























