Mixed Epithelial and Stromal Tumor of the Kidney Case Report
Ktari kamel*1, Hamdouni Wadiaa1, Moez Hamdani2, Saidi Radhia1, Zakhama Abdelfateh2 and Saad Hamadi1
1 Department of Urology, Hospital Fattouma Bourguiba, Monastir, Tunisia
2 Department of Pathology, Hospital Fattouma Bourguiba, Monastir, Tunisia
Submission: June 08, 2018; Published:July 19, 2018
*Corresponding author: Ktari Kamel, epartment of Urology, Hospital Fattouma Bourguiba, Tunisia, Email: ktari-kamel@hotmail.fr
How to cite this article:Ktari k, Hamdouni W, Moez H, Saidi R, Zakhama A, Saad H. Mixed Epithelial and Stromal Tumor of the Kidney Case report. 002 JOJ uro & nephron. 2018; 5(5): 555675. DOI: 10.19080/JOJUN.2018.05.555675
Abstract
In recent years, a rare distinctive kidney tumor composed of a mixture of stroma and epithelium with solid and cystic architecture has been recognized, which has to be distinguished from other renal neoplasms. The term mixed epithelial and stromal tumor was first introduced by Michal and Syrucek in 1998. The vast majority of cases show a benign course without tumor recurrence. Here, we present a case of this entity, found incidentally.
Keywords: Kidney; Benign; Mixed epithelial and stromal tumor
Abbreviations: MESTK: Mixed Epithelial and Stromal Tumor of Kidney; KUB: Kidney Ureter Bladder; RCC: Renal Cell Carcinoma
Introduction
Mixed epithelial and stromal tumor of kidney (MESTK) is a rare complex renal neoplasm, composed of stromal and epithelial elements, and a mixture of cystic and solid areas [1,2]. It was originally described in 1973 by Block et al. as a congenital mesoblastic nephroma.
The name mixed epithelial and stromal tumor of the kidney “MESTK” was first introduced by Michal and Syrucek in 1998 [1]. Cystic hamartoma of the renal pelvis, leiomyomatous renal hamartoma, adult mesoblastic nephroma, multilocular renal cyst with Mullerian-like stroma, and solid and cystic biphasic tumor were designations for this condition in the past [1,3]. Recent advances in molecular-level studies of the pathogenesis have resulted in an increased awareness of this type of renal tumor. In this article, we present a case of this entity, found accidentally and a review of the literature.
Case Report
A 48-year-old woman, she is menopaused, has ten children and has a history of oral contraceptive use during five years. She was followed at our service since 2000 for management of recurrent bilateral nephritic lithiasis. She had presented to our clinic with left intermittent flank pain. Physical examination was unremarkable, except mild left flank pain. Her routine blood investigations were normal, routine urine analysis revealed no hematuria.
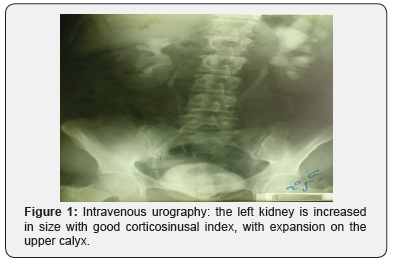
Kidney-ureter-bladder (KUB) radiograph had showed calcification in the left kidney. Intravenous urography showed bilateral and symmetrical secretion and renal excretion, with delayed opacification of the renal pelvis and the ureter to the left side, up to 10 minutes, the left kidney is increased in size with good corticosinusal index, with expansion on the upper calyx (Figure 1).
The patient had a dorsal lumbotomy.!
On surgical exploration there was incidentally a whitish solid nodule of 2 cm in diameter, round shaped, and well circumscribed with a thin fibrous capsule. There were no signs of infiltrative
growth into the surrounding renal parenchyma. The tumor was situated in the renal parenchyma and compressed the adjacent calyceal system. A lumpectomy was performed. Grossly the tumor had a solid appearance without cyst formation, white color, soft to elastic consistency without hemorrhage and necrosis.
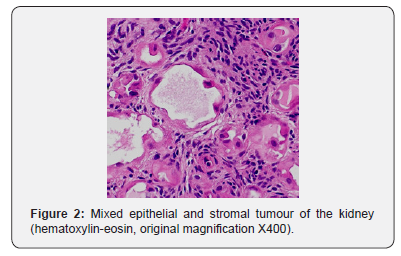
Histologically (Figure 2), the tumor was biphasic comprising epithelial and stromal elements. It includes microcysts and tubules, and ovarian-like stroma. The epithelial component shows flattened cuboidal or hobnail cells, their cytoplasm ranges from clear to pale eosinophilic. The architecture of the microcysts varied from simple microcysts with abundant stroma between them, to densely packed clusters of microcysts to complex branching channels. The stroma consists of variably cellular proliferation of spindle cells with plump nuclei and abundant cytoplasm. Areas of myxoid stroma and smooth muscle cells also identified at places. No dysplasia increased mitotic activity, or tumor necrosis as signs for malignant transformation were observed. The immunohistochemical investigations was not performed. The final histopathological diagnosis was MESTK (mixed epithelial and stromal tumor of the kidney).
The postoperative course was uneventful.
The patient has received regular follow-up at our out-patient department. No evidence of tumor recurrence has been found after 10 years.
Discussion
MESTK is a distinct category of renal neoplasm. Patients’ ages range from 19 to 84 years (mean, 46 years), with a female/male incidence ratio of 10-6:1. It often affects women with prolonged estrogen exposure and rarely occurs in male patients who have undergone androgen deprivation therapy. Most patients typically present with flank pain, a palpable abdominal or flank mass, hematuria, or symptoms related to genitourinary infections, although up to 25% of tumors are identified incidentally [1,2,4,5]. MESTK is typically a centrally located, tan to yellow, well-circumscribed, solid cystic lesion, with 1.7 to 20cm being the largest. In this case, owing to loss of the perirenal fascia tension, a CT scan showed that the surgical specimen was larger than the size of the mass. Microscopically, the MESTK contained an admixture of stroma and epithelial lined large cysts, microcysts, and tubules. In general, ER, PR, CD10, Vimentin, Desmin, SMA, and inhibin expression was seen in the stromal component. HMB-45, S-100, CD99, CD117, and CD34 stains were negative. The epithelial components were positive for CK7, PCK, cytokeratin AE1/3, Ulex europaeus, and calretinin [1,2,5,6].
MESTKs have certain features in imaging and are more common in Bosniak III/IV. Most MESTKs can be described as well circumscribed in the renal pelvis or protruding from the cortex, and show multiseptate cystic renal masses with thick septa and solid components. The emphasis here is on enhancing the image of the solid components with the delayed enhancement during the nephrographic phase in the contrast-enhanced CT images. The different extent of the delayed enhancement may depend on the spindle cell components of these masses, which exhibit variable patterns and cellularity ranging from paucicellular fibrosis to densely cellular areas [7,8].
However, the pathogenesis of MESTK is still not clear. There are several theories to explain the possible histogenesis mechanism. One hypothesis is the presence of multidifferentiation potential fetal primitive mesenchyme or metanephric blastema in the kidney, which may be an epithelialestromal interaction given its status of hormonal hyperstimulation, which may then cause the proliferation to form a mass [3,9]. Another hypothesis is the abnormal migration of ovarian stromal cells during embryogenesis. Because of the close proximity of mesonephric ducts and Mullerian ducts during the 4th week of development, ovarian stromal cells could become incorporated in the ureteric bud and metanephric mesoderm. These cells may become activated in the steroid hormonal stimulation, secrete paracrine factors, and induce proliferation of the
adjacent epithelium [3]. Above all, although the histogenesis of MESTK is unknown, in view of the frequent expression of ER and PR in the stromal component and the existence of ovariantype stroma, both theories suggest that the steroid hormones might play a role in the evolution of these tumors.
Tickoo et al. [10] believed that ER/PR-positive stromal proliferation can be present in some nonneoplastic kidneys and the part of neoplastic kidneys away from the tumor, such as ureterepelvic junction stenosis, benign multilocular cyst, benign ureteric stricture, acquired cystic disease-associated renal cell carcinoma (RCC), urothelial carcinoma ureter, urothelial carcinoma bladder, chromophobe RCC, clear cell RCC, except for MEST and cystic nephroma. However, they observed that the diseases mentioned above had a common findingda generalized or segmental hydronephrosis, or tumor compression-related focal obstruction. Based on this finding, they reached a conclusion that renal obstructive changes may represent a metaplastic change in the renal interstitial cells surrounding these obstructed epithelial structures. Moreover, the ER/PR stoma seen in association with renal tumors may also be a secondary (metaplastic) response to their cystic epithelial components and not necessarily a neoplastic constituent. Similarly, using electron microscopy, Picken and Fresco [11] found that the tubules in MESTK are entrapped rather than neoplastic. To summarize, we postulate that the histogenesis of MEST results not only from the stimulation of the hormones, which cause the proliferation to form a mass, but also that the cysts’ compression-related obstructive changes may likewise play an important role in its development. Through a synergistic action, the MESTK may grow larger and lead to further clinical symptoms. We argue that this requires further study.
Tumor spillage or positive surgical margins are presumably risk factors for local recurrence [12]. Hence, complete surgical resection is the preferred treatment for MESTK. According to the patient’s condition, partial or complete nephrectomy was chosen. And when a partial nephrectomy was performed to exclude a positive margin, an intraoperative frozen section was helpful [2]. Because the tumor was rare and the preoperative diagnosis was difficult, we could not rule out malignancy. At this point, the treatment of choice was nephrectomy and the complete removal of the tumor. For the diagnosis, preoperative biopsy may cause the cyst to rupture and increase the risk of recurrence. Therefore, preoperative biopsy should be conservative. In terms of clinical behavior, rare aggressive behavior has been reported [13]; however, most cases of MESTK exhibit a benign histologic picture and clinical course, and both neoplasms are benign and surgical excision results in restoration [14]. If diagnosed preoperatively, for the patient who has a high risk for an operation, follow-up without any operation might be an alternative.
Conclusion
MESTK is relatively rare, usually presents in perimenopausal women as a partially cystic mass, and its growth may be influenced by hormones. Any cystic tumor presenting in perimenopausal women should undergo the same series of tumor surveys as for malignant tumors. A mixed epithelial and stromal tumor of the kidney should be considered one of the possibilities. Therefore, pathogenesis and preoperative diagnosis should be further explored.
http://sci-hub.io/
References
- Montironi R, Mazzucchelli R, Lopez-Beltran A, Martignoni G, Cheng L, et al. (2008) Cystic nephroma and mixed epithelial and stromal tumour of the kidney: opposite ends of the spectrum of the same entity? Eur Urol 54(6): 1237-1246.
- Mohanty SK, Parwani AV (2009) Mixed epithelial and stromal tumors of the kidney: an overview. Arch Pathol Lab Med 133(9): 1483-1486.
- Turbiner J, Amin MB, Humphrey PA, Srigley JR, De Leval L, et al. (2007) Cystic nephroma and mixed epithelial and stromal tumor of kidney: a detailed clinicopathologic analysis of 34 cases and proposal for renal epithelial and stromal tumor (REST) as a unifying term. Am J Surg Pathol 31(4): 489-500.
- Jevremovic D, Lager DJ, Lewin M (2006) Cystic nephroma (multilocular cyst) and mixed epithelial and stromal tumor of the kidney: a spectrum of the same entity? Ann Diagn Pathol 10(2): 77-82.
- Portier BP, Hansel DE, Zhou M (2009) Mixed epithelial and stromal tumor of the kidney. J Urol 181: 1879-1880.
- Antic T, Perry KT, Harrison K, Zaytsev P, Pins M, et al. (2006) Mixed epithelial and stromal tumor of the kidney and cystic nephroma share overlapping features: reappraisal of 15 lesions. Arch Pathol Lab Med 130(1): 80-85.
- Lang N, Li J, Liu JY, Zeng XZ, Yang Y (2010) Mixed epithelial and stromal tumor of the kidney: an analysis of multidetector computed tomography manifestations and clinicopathologic findings. J Comput Assist Tomogr 34(2): 177-181.
- Sahni VA, Mortele KJ, Glickman J, Silverman SG (2010) Mixed epithelial and stromal tumour of the kidney: imaging features. BJU Int 105(7): 932-939.
- Buritica C, Serrano M, Zuluaga A, Arrabal M, Regauer S, et al. (2007) Mixed epithelial and stromal tumour of the kidney with luteinised ovarian stroma. J Clin Pathol 60(1): 98-100.
- Tickoo SK, Gopalan A, Tu JJ, Harik LR, Al-Ahmadie HA, et al. (2008) Estrogen and progesterone-receptor-positive stroma as a nontumorous proliferation in kidneys: a possible metaplastic response to obstruction. Mod Pathol 21(1): 60-65.
- Picken MM, Fresco R (2005) Mixed epithelial and stromal tumor of the kidney: preliminary immunohistochemical and electron microscopic studies of the epithelial component. Ultrastruct Pathol 29: 283-286.
- Yap YS, Coleman M, Olver I (2004) Aggressive mixed epithelialestromal tumour of the kidney treated with chemotherapy and radiotherapy. Lancet Oncol 5: 747-749.
- Jung SJ, Shen SS, Tran T, Jun SY, Truong L, et al. (2008) Mixed epithelial and stromal tumor of kidney with malignant transformation: report of two cases and review of literature. Hum Pathol 39(3): 463-468.
- Michal M, Syrucek M (1998) Benign mixed epithelial and stromal tumor of the kidney. Pathol Res Pract 194: 445-448.






























