Nephrotoxicity and Hepatotoxicity of Capparis Spinosahydro-Alcoholic Extract in Mice
Sahar Fanoudi1, Hasan Rakhshandeh1,2, Amir R Afshari1, Hamid Mollazadeh3 and Mohammad Taher Boroushaki1,2*
1Department of Pharmacology, School of Medicine, Mashhad University of Medical Sciences, Iran
2Pharmacological Research Center of Medicinal Plants, Mashhad University of Medical Sciences, Iran
3Department of Physiology and Pharmacology, School of Medicine, North Khorasan University of Medical Sciences, Iran
Submission: October 07, 2017; Published: November 29, 2017
*Corresponding author: Mohammad Taher Boroushaki, Department of Pharmacology, School of Medicine, Mashhad University of Medical Sciences, Mashhad, Iran, Tel: +98 513 8828566; Fax: +98 513 8828567; E-mail: BoroushakiMT@mums.ac.ir
How to cite this article: Sahar F, Hasan R, Amir R A, Hamid M, Mohammad T B. Nephrotoxicity and Hepatotoxicity of Capparis Spinosahydro-Alcoholic Extract in Mice. JOJ uro & nephron. 2017; 4(2): 555638. DOI: 10.19080/JOJUN.2017.04.555638
Abstract
Background: Caper (Capparis Spinosa) plant extracts have been related with different pharmacological activities including anti- histaminic, anti-oxidant, hypolipidemic, and anti-mutagenic. Nevertheless, its side-effects are not yet studied especially nephrotoxicity and hepatotoxicity. So, the point of this study was to examine the acute and sub-chronic toxicity of hydro-alcoholic extract of the Capparis spinosa on the liver, kidney, and serum enzymes.
Methods and Materials: In the experimental design, thirty-two male mice were randomly isolated into four groups of one control and three experimental. The doses of 200, 400 and 800 mg/kg of hydro-alcoholic extract of Capparis spinosa were administrated by oral gavages for 28 consecutive days in mice. Normal saline, 5ml/kg was given to the control group. Each group contained 8 male mice. at 14th day, for half of each group and at 28th day for the rest of them, serum samples were collected for liver function tests (ALT, AST) and renal function tests (BUN, Cr). The livers and kidneys were isolated for histopathological and biochemical studies.
Results: The results of this study have shown that Capparis spinosa can cause nephrotoxicity and hepatotoxicity especially during subchronic consumption, dose-dependently.
Conclusion: The extracts of Capparis spinosa must be used with caution especially in renal and liver pathologic conditions. However, the reasons and mechanisms of this toxicity need further investigation.
Keywords: Capparis spinosa; Malondialdehyde; Thiol; Nephrotoxicity; Hepatotoxicity
Abbreviations: UEC: University Ethics Committee; Na2EDTA: Ethylene Diamine Tetraacetic Acid Disodium; HCl: Hydrochloric Acid; TBA: 2-Thiobarbituric Acid; KCl: Potassium Chloride; TCA: Trichloroacetic Acid; DTNB: 2,20 -Dinitro-5,50 -Dithiodibenzoic Acid; MDA: Malondialdehyde; TBARS: Thiobarbituric Acid Reactive Substance
Introduction
Herbal medicines can be a good alternative to treat diseases because of their low costs, availability, and lack of undesirable side effects [1-5]. Capparis spinosa L. (Family: Capparidaceae) is a plant found in the areas of focal or west Asia and is generally developed especially in the Mediterranean basin and its fruit, roots and barks are utilized for medical purposes [6]. Chemical studies on Capparis spinosa have shown the presence of, alkaloids, indole, flavonoids, lipids, aliphatic glucosinolates and polyphenols [7]. It is additionally recognized as the herb-rich source of flavonoids such as rutin, kaempferol, quercetin and its derivatives [8]. These constituents display a significant role in the pharmacological activity of Capparis spinosa including: antioxidant [9], anti-inflammatory [10], anti-allergic, anti- histaminic [11], hypolipidemic [12], anti-mutagenic, antiproliferative [13], anti-microbial [14], anti-helminthic [15], hepato-protective [16] and anti-nociceptive [17] effects. It has been shown that alcoholic extract of this plant have high level of antioxidant [18].
In this decade, certain herbal medicines are being marketed without standard toxicological studies, although reports indicate the potential toxicity of herbal medicines [19]. Given that no comprehensive study of the toxicity of the plant has been done, the point of this research was to assess toxicity of the hydroalcoholic extract of Capparis spinosa that has traditionally used to treat numerous diseases in Iranian folk medicine.
Materials and Methods
Extract preparation
The hydro-alcoholic extract of the plant was prepared as revealed previously [20].
Animals
Thirty-two adult male mice (Animal House, Faculty of Medicine, Mashhad University of Medical Sciences, Mashhad, Iran) weighing 25-30g were used for all experiments. Animals were housed in pathogen-free facilities on a 12-hour light/ dark cycle, with ad libitum access to food and water. All animal procedures were affirmed by the University Ethics Committee (UEC) and were in compliance with National Laws and the National Institutes of Health guidelines for the utilization and care of laboratory animals.
Chemicals
Na2EDTA (ethylenediaminetetraacetic acid disodium salt), Trizma base [Tris (hydroxymethyl) aminomethane], n-butanol, TMP (tetra methoxy-propane), HCl (hydrochloric acid), TBA (2-thiobarbituric acid), KCl (potassium chloride), TCA (Trichloroacetic acid), phosphoric acid (1%), DTNB (2,20 -dinitro-5,50 -dithiodibenzoic acid), ether, and methanol were purchased from Merck (Darmstadt, Germany).
Experimental design
After acclimatization, animals were randomly divided into four groups (eight each). Group I (control) was treated with distilled water (5ml/kg). Groups 2, 3 and 4 were treated with 200, 400, and 800mg/kg [16] of hydro-alcoholic extract of Capparis spinosa by gavage for 28 days. On day 14, four animals in each group and on day 28 the rest of animals were killed, blood samples were collected by cardiac puncture, for measuring serum AST, ALT, Urea and Creatinine. The kidneys and livers were removed; the right kidney and a piece of each liver were fixed in 10% neutral buffered formalin for histological studies. The left kidneys and the rest of livers were homogenized in cold KCl solution (1.5%, pH=7) to give a 10% homogenate suspension to use for measuring malondialdehyde (MDA) and sulfhydryl (thiol) contents.
Biochemical methods
Urea and creatinine measurement: Serum level of Urea was determined calorimetrically using the urea kit (Man Lab Company, Tehran, Iran) and Auto-analyzer (Technicon RA-1000, London, England). Creatinine concentration was measured by the Jaffe's method [21].
Calculating MDA content: The lipid peroxidation levels of the liver and kidney tissues were measured by MDA, which is the final product of lipid peroxidation and reacts with TBA as a thiobarbituric acid reactive substance (TBARS) to produce a red-colored complex that has a peak absorbance at 532nm [22].
Briefly, 3mL phosphoric acid (1%) and 1mL TBA (0.6%) were added to 0.5mL of homogenate in a centrifuge tube and the mixture was heated for 45min in a boiling water bath. Subsequent to cooling, 4mL of n-butanol was added to the mixture, vortexed for 1min, and centrifuged at 20,000rpm for 20min. The organic layer was exchanged to a fresh tube and its absorbance was measured at 532nm. The MDA content was calculated from the following equation:
MDA (mmol/gr tissue) = absorbance/1.56 x105
Calculation of total thiol content: Total thiol content was measured utilizing DTNB as a reagent. This reagent reacts with thiol groups to produce a yellow-colored complex that shows a peak absorbance at 412nm. Precisely, 1ml Tris-EDTA buffer (pH=8.6) was added to 50|il kidney homogenate in 2ml cuvettes and absorbance was read at 412nm against the Tris-EDTA buffer alone (A1). At that point, 20μL DTNB reagent (10mM in methanol) was added to the mixture, and after 15min (put away in room temperature), the sample absorbance was read again (A2). The absorbance of DTNB reagent was additionally read as a blank (B). Total thiol concentration (mM) was calculated from the following equation [23]:
Thiol concentration (mM):(A2 - A1 - B) x (1.07/0.05) x13.6
Histological method
The livers and right kidneys were removed and fixed in 10% neutral buffered formalin, sectioned at 5μ and stained in Hematoxylin and Eosin (H&E) for histopathological studies.
Statistical Analysis
Data were expressed as mean ± SEM. Statistical examination was performed utilizing the Two-way analysis of variance (ANOVA) followed by the post hoc Bonferroni test for different correlations. The p-values less than 0.05 were thought to be measurably significant.
Results


Effect of Capparis spinosa on BUN and creatinine::A shown in Figure 1, there is no significant difference in serum creatinine level on day 14 but on day 28 the difference is significant, particularly in the high dose group (p<0.001). As shown in Figure 2, BUN levels showed significant increase only with 800mg/kg dose, on 14 and 28 days after treatment (p<0.05 and p<0.01, respectively).
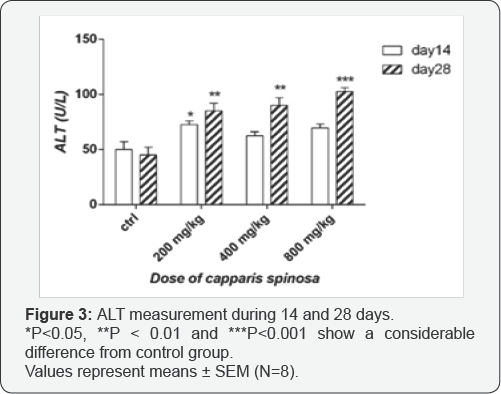
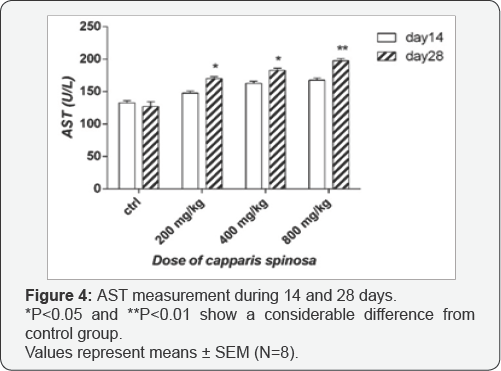
Effect of Capparis spinosa on ALT and AST: As shown in Figure 3, there are no significant difference in ALT levels after 14 days treatment with doses 400 and 800mg/kg of the extract compare to the control group, but with dose 200mg/kg this difference was significant (p<0.05). Data showed that there are significant difference in ALT levels after 28 days in different extract treated groups, particularly in the high dose treated one (p<0.01 for 200 and 400mg/kg and p<0.001 for 800mg/ kg) compare to control. As shown in Figure 4, there are no significant difference in AST levels after 14 day treatment, but after 28 days treatment there are significant difference in levels of AST especially with the high dose (p<0.01).
Oxidative stress measurement results

Effect of Capparis spinosa on renal MDA content: The results of measuring MDA indicate no significant increase after 14 and 28 days treatment with 200mg/kg. Whilst, by increasing the doses, particularly after 28 day treatment, a significant difference (p<0.001) was observed between treated groups compare to control (Figure 5).
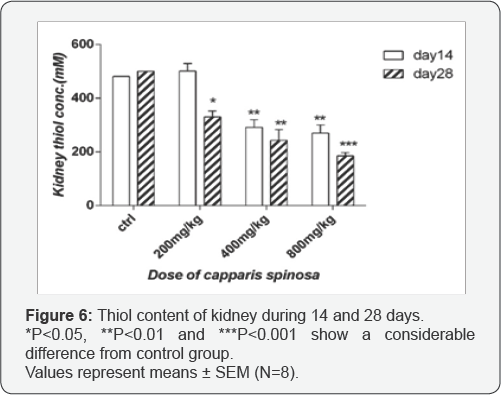
Effect of Capparis spinosa on renal thiol content: Data indicated that there is no significant decrease in thiol content the extract compare to the control group, but with dose 200mg/kg after 14 days treatment with 200mg/kg extract but after 28 days it was significant (p<0.05). Levels of renal thiol content were significantly decreased after 14 and 28 days treatment with 400 and 800mg/kg (p<0.01 and p<0.001) compare to control (Figure 6).
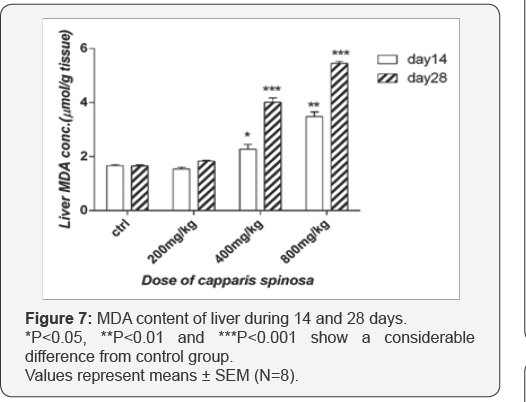
Effect of Capparis spinosa on liver MDA content: Data shown that, there is no significant difference in liver MDA content with 200mg/kg neither 14 nor 28 days after treatment. Whilst with increasing the doses to 400 and 800mg/kg, liver MDA contents are significantly elevated, especially after 28 days treatment (p<0.001) compare to the control group (Figure 7).
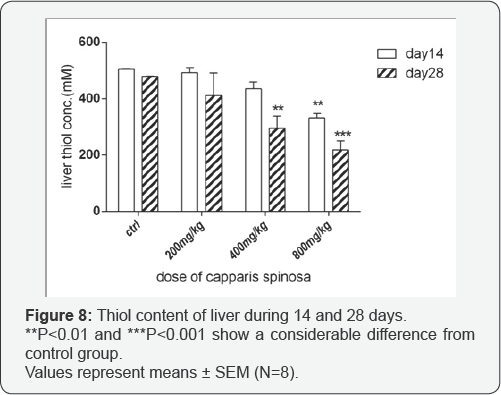
Effect of Capparis Spinosa on liver thiol content: Thiol measurement did not show any remarkable decrease with the doses 200 and 400mg/kg after 14 days, but within 28 days with increased dosage, especially at 800mg/kg, a significant reduction was observed (p<0.001) (Figure 8).
Histological results
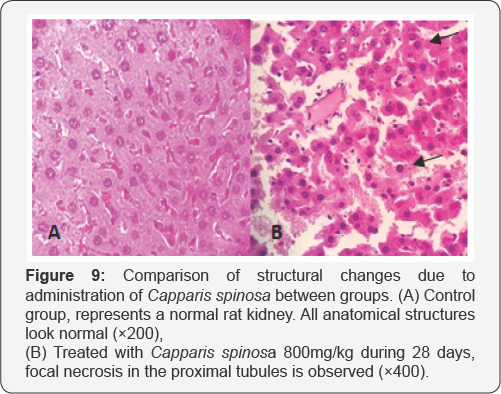
The kidney: Assessment of pathological observations in the kidney did not show any remarkable toxicity on the day 14. In addition, the doses of 200 and 400mg/kg did not show a significant change after 28 days administration. However, with 800mg/kg in some samples focal necrosis in the proximal tubules were observed (Figure 9).
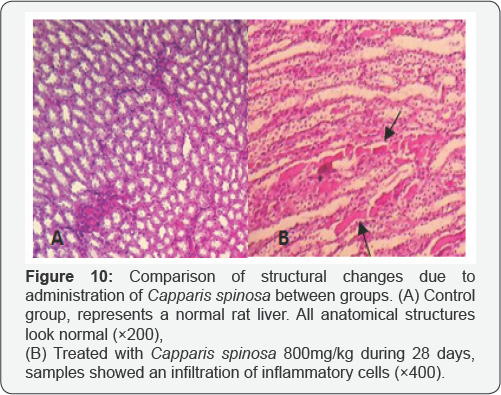
The liver: Similar the kidney, after 14 and 28 days treatment with 200 and 400 mg/kg extract, no damage in livers was observed. However, with 800mg/kg infiltration of inflammatory cells was observed (Figure 10).
Discussion
According to investigations conducted in this study, it is clear that the hydro-alcoholic extract of Capparis spinosa has some toxic effects on the kidney and liver. Regarding hepatotoxicity, liver enzyme measurements have shown that there was a significant difference only in ALT levels with dosages of 200, 400 and 800mg/kg after 14 days consumption. However, after 28 days consumption, especially with increased doses significant changes have been observed. Since the AST and particularly ALT, are indicators of liver damage [24-26] we can conclude that the used doses have a toxic effect on the liver parenchyma and hepatic cells. Also, increased levels of ALT could likely be due to toxicity of the extract on bile duct epithelium and cholestasis [27]. Levels of AST after 28 day treatment with all doses were increased, which is another reason for hepatotoxic effect of extract.
In the literature review, we did not find any similar studies that compared the findings of liver toxicity of hydro-alcoholic extract of Capparis spinosa. However, one study showed that the protective effects of Capparis spinosa was attributed to para-methoxy benzoic acid fractions extracted from methanolic extract [28]. In another study, the hepato-protective effect of Capparis spinosa against carbon tetrachloride-induced toxicity has been reported [16].
Regarding nephrotoxicity, serum creatinine and BUN data can be cited. The results showed that the extract with all doses did not significantly change in serum creatinine on day 14, compared to the control group. However, after 28 days the administered doses 200, 400, and 800mg/kg showed a significant increase in creatinine levels, especially with 800mg/kg.
In addition, the results showed that the levels of BUN increased only with the dose 800mg/kg, after 14 and 28 days treatment compared to the control group.
There are probably a series of materials in the extract that have BUN enhancer effects; hence, this study highlights the need for more studies in this field. Since the BUN and especially serum creatinine are the most common laboratory markers to identify renal function and glomerular filtration, it seems that the doses of 400and especially 800mg/kg exert toxic effects on glomerular function. However, increased levels of creatinine and BUN as an indirect marker of GFR do not necessarily indicate renal injury, but may be secondary indicators of dehydration, protein catabolism, and hypovolemia. When the effects of extract on kidney and liver are compared together, it showed that liver is likely more sensitive to toxic effect of extract than kidney.
MDA is a consistent metabolite of lipid peroxidation due to oxidative stress in cells. The evaluation of the MDA test in the kidney showed a significant increase with 800mg/kg after 14 and 28 days treatment, indicating lipid peroxidation. Likewise, thiol substance is an imperative piece of the basic protein and non-protein compounds and assumes an essential part in cellular processes for example, restoration of cellular pathways, enzyme activity, and mechanisms in cell detoxification [29,30]. In the present study, cellular thiol contents decreased when doses were increased. These results demonstrate expanded oxidative stress and diminished antioxidant enzyme activity, which are the main causes of renal toxicity. These biochemical parameters were related to the renal histological results. Additionally, the liver tests have shown a noteworthy increment in MDA and a significant decrease in cellular thiol content particularly after 14 and 28 days treatment, in a dose-dependent manner.
In pathological studies, we observed focal necrosis in proximal tubules and infiltration of inflammatory cells in kidney with high dose (800mg/kg) after 28 days treatment, which indicates the toxicity of Capparis spinosa.
Conclusion
The results of this study have shown that Capparis spinosa can cause nephrotoxicity and hepatotoxicity as dose-dependently. So, the extracts of Capparis spinosa must be use with caution especially in renal and liver pathologic conditions. However, the mechanisms of this toxicity need further investigations.
References
- Dirin MM, Mousavi S, Afshari AR, Tabrizian K, Ashrafi MH (2014) Potential drug-drug interactions in prescriptions dispensed in community and hospital pharmacies in East of Iran. J Res Pharm Pract 3(3): 104-107.
- Afshari AR, Sadeghnia HR, Mollazadeh H (2016) A review on potential mechanisms of Terminalia chebula in alzheimer's disease. Advances in Pharmacological Sciences 2016(2016).
- Boroushaki MT, Mollazadeh H, Afshari AR (2016) Pomegranate seed oil: a comprehensive review on its therapeutic effects. International Journal of Pharmaceutical Sciences and Research 7(2): 430.
- Sadeghnia HR, Jamshidi R, Afshari AR, Mollazadeh H, Forouzanfar F, et al. (2017) Terminalia chebula attenuates quinolinate-induced oxidative PC12 and OLN-93 cell death. Multiple Sclerosis and Related Disorders 14: 60-67.
- Shafiee-Nick R, Afshari AR, Mousavi SH, Rafighdoust A, Askari VR, et al. (2017) A comprehensive review on the potential therapeutic benefits of phosphodiesterase inhibitors on cardiovascular diseases. Biomedicine & Pharmacotherapy 94: 541-556.
- Goel A, Garg A, Kumar A (2016) Effect of Capparis spinosa Linn. extract on lipopolysaccharide-induced cognitive impairment in rats. Indian J Exp Biol 54(2): 126-132.
- Mansour RB, Jilani IB, Bouaziz M, Gargouri B, Elloumi N, et al. (2016) Phenolic contents and antioxidant activity of ethanolic extract of Capparis spinosa. Cytotechnology 68(1): 135-142.
- Sharaf M, El-Ansari, Saleh NA (1997) Flavonoids of four Cleome and three Capparis species. Biochemical Systematics and Ecology 25(2): 161-166.
- Bonina F, Puglia C, Ventura D, Aquino R, Tortora S, et al. (2002) In vitro antioxidant and in vivo photoprotective effects of a lyophilized extract of Capparis spinosa L. buds. J Cosmet Sci 53(6): 321-336.
- Zhou HF, Xie C, Jian R, Kang J, Li Y, Zhuang CL, et al. (2011) Biflavonoids from Caper (Capparis spinosa L.) fruits and their effects in inhibiting NF-kappa B activation. J Agric Food Chem 59(7): 3060-3065.
- Trombetta D, Occhiuto F, Perri D, Puglia C, Santagati NA, et al. (2005) Antiallergic and antihistaminic effect of two extracts of Capparis spinosa L. flowering buds. Phytotherapy Res 19(1): 29-33.
- Eddouks M, Lemhadri A, Michel JB (2005) Hypolipidemic activity of aqueous extract of Capparis spinosa L. in normal and diabetic rats. J Ethnopharmacol 98(3): 345-350.
- Lam SK, Ng TB (2009) A protein with antiproliferative, antifungal and HIV-1 reverse transcriptase inhibitory activities from caper (Capparis spinosa) seeds. Phytomedicine 16(5): 444-450.
- Boga C, Forlani L, Calienni R, Hindley T, Hochkoeppler A, et al. (2011) On the antibacterial activity of roots of Capparis spinosa L. Nat Prod Res 25(4): 417-421.
- Mali R, Mahajan S, Patil K (2005) Anthelmintic activity of root bark of Capparis spinosa. Indian J Nat Prod 21(4): 50-51.
- Aghel N, Rashidi I, Mombeini A (2010) Hepatoprotective activity of Capparis spinosa root bark against CCl4 induced hepatic damage in mice. Iranian journal of pharmaceutical research 6(4): 285-290.
- Arslan R, Bektas N (2010) Antinociceptive effect of methanol extract of Capparis ovata in mice. Pharm Biol 48(10): 1185-1190.
- Germano MP, De Pasquale R, D'Angelo V, Catania S, Silvari V, et al. (2002) Evaluation of extracts and isolated fraction from Capparis spinosa L. buds as an antioxidant source. J Agri Food Chem 50(5): 1168-1171.
- Stickel F, Shouval D (2015) Hepatotoxicity of herbal and dietary supplements: an update. Archives of toxicology 89(6): 851-865.
- Rahmani R, Mehdi M , Mehrnaz K, Fateme H, Rahim H, et al. (2013) Effect of Hydroalcoholic Extract of Capparis Spinosa Fruit on Blood Sugar and Lipid Profile of Diabetic and normal Rats. Zahedan J Res Med Sci 15(11): 34-38.
- Slot C (1965) Plasma creatinine determination a new and specific Jaffe reaction method. Scand J Clin Lab Invest 17(4): 381-387.
- Hosseinzadeh H, Sadeghnia HR, Ziaee T, Danaee A (2005) Protective effect of aqueous saffron extract (Crocus sativus L.) and crocin, its active constituent, on renal ischemia-reperfusion-induced oxidative damage in rats. J Pharm Pharm Sci 8(3): 387-93.
- Boroushaki MT, Asadpour E, Sadeghnia HR, Dolati K (2014) Effect of pomegranate seed oil against gentamicin-induced nephrotoxicity in rat. J Food Sci Technol 51(11): 3510-3514.
- Mollazadeh H, Taher BM, Soukhtanloo S, Amir RA, Mahdi MV, et al. (2017) Effects of pomegranate seed oil on oxidant/antioxidant balance in heart and kidney homogenates and mitochondria of diabetic rats and high glucose-treated H9c2 cell line. Avicenna J Phytomed 7(4): 317-333.
- Ozer J, Ratner M, Shaw M, Bailey W, Schomaker S (2008) The current state of serum biomarkers of hepatotoxicity. Toxicology 245(3): 194205.
- Hyder MA, Hasan M, Mohieldein AH (2013) Comparative levels of ALT, AST, ALP and GGT in liver associated diseases. European Journal of Experimental Biology 3(2): 280-284.
- Jüngst C, Berg T, Cheng J, Green RM, Jia J, et al. (2013) Intrahepatic cholestasis in common chronic liver diseases. Eur J Clin Invest 43(10): 1069-1083.
- Gadgoli C, Mishra S (1999) Antihepatotoxic activity of p-methoxy benzoic acid from Capparis spinosa. J Ethnopharma 66(2): 187-192.
- Chen Q, Jones TW, Brown PC, Stevens JL (1990) The mechanism of cysteine conjugate cytotoxicity in renal epithelial cells. Covalent binding leads to thiol depletion and lipid peroxidation. J Biol Chem 265(35): 21603-21611.
- Ziegler D (1985) Role of reversible oxidation-reduction of enzyme thiols-disulfidesin metabolic regulation. Ann Rev Biochem 54(1): 305329.






























