- Mini Review
- Abstract
- Introduction
- Computation and Methods for Calculating ATP in Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency for Converting Light Energy into Electrochemical Energy
- Efficiency for Converting Light Energy into ATP
- Comparing Power and Efficiency of Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency of Converting Chemical to Electrical Energy
- Functional and Structural Differences of Bacteriorhodopsin and Bacterial Reaction Center
- Conclusion
- References
Photochemistry Governing Bacteriorhodopsin and Bacterial Reaction Center
Bernadine G Ang* and Friedrich Schotte
University of the Philippines Diliman, Philippines
Submission: February 24, 2018; Published: May 11, 2018
*Corresponding author: Bernadine Ruiza G Ang MS Candidate, University of the Philippines Diliman, Manila, Philippines; Tel: +63-2-981-8500; Email: angbernadine@hotmail.com
How to cite this article: Bernadine G Ang, Friedrich Schotte. Photochemistry Governing Bacteriorhodopsin and Bacterial Reaction Center. JOJ scin. 2018; 1(2): 555559. DOI: 10.19080/JOJS.2018.01.555559
- Mini Review
- Abstract
- Introduction
- Computation and Methods for Calculating ATP in Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency for Converting Light Energy into Electrochemical Energy
- Efficiency for Converting Light Energy into ATP
- Comparing Power and Efficiency of Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency of Converting Chemical to Electrical Energy
- Functional and Structural Differences of Bacteriorhodopsin and Bacterial Reaction Center
- Conclusion
- References
Abstract
Bacteriorhodopsin (bR) is a light driven proton pump that converts sunlight to chemical energy. BR is an integral membrane structured protein found in the purple membrane of Halobacterium halobium. It is composed of 248 amino acids and a chromophore in the middle which captures light. Electricity can be generated through the process of light-chemical conversion, when photons are absorbed by thechromophore, the photo cycle begins. Bacterial Reaction Center (bRC) is a light driven electron transfer reaction that converts solar energy to chemical energy. bRC are integral membrane structured proteins found in the purple membrane of Rhodobacter sphaeroides. It is composed of 3 major co-factors such as bacteriochlorophylls, bacteriopheophytin and quinone. Its primary mechanism is to execute photo synthesis. In this interaction, electron transfer occurs through light ejection of electron that passes through the membrane. Conversion of sunlight to chemical energy simultaneously precipitates. This review aims to compare the function and structure of Bacteriorhodopsin and bacterial reaction centers, underscoring the energy generated in both membranes. Through calculating the ATP, protons and photons that cross the membrane, exact value of energy emission in the order of electron volts present the energy generated. Advantages and mechanisms of photoreactions including bioelectronic, bio energy production in bacteriorhodopsin and bacterial reaction centers will be exemplified. Ultimately, energy efficiency of bacteriorhodopsin and bacterial reaction center will be determined.
Keywords: Bacteriorhodopsin; Bacterial reaction center; Photochemistry; Electrochemical efficiency
- Mini Review
- Abstract
- Introduction
- Computation and Methods for Calculating ATP in Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency for Converting Light Energy into Electrochemical Energy
- Efficiency for Converting Light Energy into ATP
- Comparing Power and Efficiency of Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency of Converting Chemical to Electrical Energy
- Functional and Structural Differences of Bacteriorhodopsin and Bacterial Reaction Center
- Conclusion
- References
Introduction
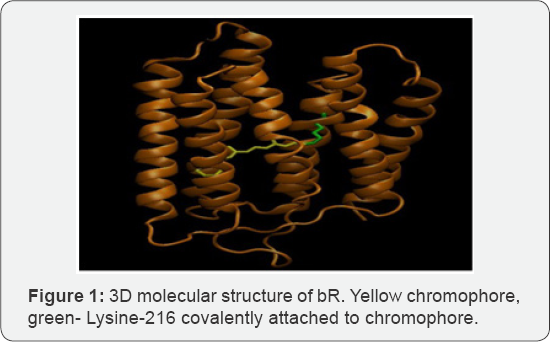
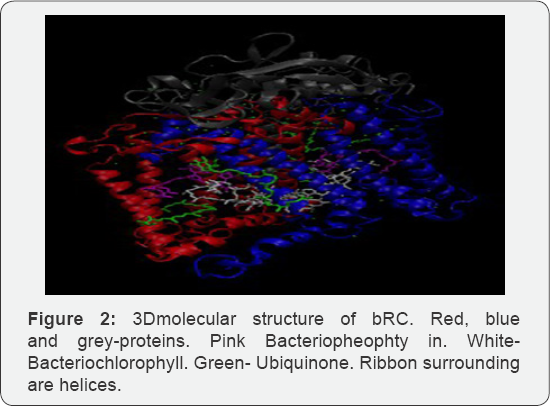
Bacteriorhodopsin (bR) is an integral membrane protein found in the purple membrane of the Halobacterium halobium. As a light driven proton pump, bRs capture photons in the order of 500nm-650nm. Existing research found that 300mv of electricity generated per purple membrane [1]. Conversely, bacterial reaction center (bRC) is found in Rhodobacter sphaeroides. It generates 800mv of electricity in total, considering the transfer of 4 protons in the membrane. Multiple interventions to denature and modulate the structure of bR should be considered, including pH, temperature, detergent and wavelength of laser light. Bacteriorhodopsin has been known to function between0C to 45C at a pH of 1-11. It tolerates temperature over 80C in water and up to 140C in dry [2]. Conventionally, laser is utilized to precipitate the photoisomerization of bR, consequently shifting all trans-13 to structure. The chromophore which is covalently attached to Lys216 through a Schiff base is modulated through this method. Bacterial Reaction Center (bRC) is a light driven electron transfer reaction that converts solar energy to chemical energy. bRC are integral membrane structured proteins found in the purple membrane of Rhodobacter sphaeroides. It is composed of 3 protein subunits--L, M and H. It has 3 major co-factors composed of 4bacterio-chlorophylls, 2 bacteriopheophytins and 2 quinones [3]. In this interaction, electron transfer occurs through light ejection of electron that passes through them embrane. Conversion of sunlight to chemical energy simultaneously precipitates. Both bR and bRC from Halobacterium halobium and Rhodobacter sphaeroides respectively generate a considerable worth of electricity that can be used in the industry today. Although bRC emits power in a rather limited merit, future research would determine its potential for catalyzing electrically reliant applications such as optics, instrumentation and therapeutic values. bRC can be utilized for solar energy and drive photovoltaic cells. Its usage is ubiquitous at this point and has generated multitude of photovoltaic and solar energy driven applications. Further research will enhance the efficacy of bRC generated solar cells and related interventions.
- Mini Review
- Abstract
- Introduction
- Computation and Methods for Calculating ATP in Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency for Converting Light Energy into Electrochemical Energy
- Efficiency for Converting Light Energy into ATP
- Comparing Power and Efficiency of Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency of Converting Chemical to Electrical Energy
- Functional and Structural Differences of Bacteriorhodopsin and Bacterial Reaction Center
- Conclusion
- References
Computation and Methods for Calculating ATP in Bacteriorhodopsin and Bacterial Reaction Center
Bioenergetic quantification through converting and comparing the proton, photon, and voltage in bR and bRC. Chromophore absorption of photon varies between bR and bRC. Permeability of membrane may affect absorption and transport of protons. Hence, data for photo cycle bR efficiency is 60% reliable, while bRC efficiency is 100%.
- Mini Review
- Abstract
- Introduction
- Computation and Methods for Calculating ATP in Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency for Converting Light Energy into Electrochemical Energy
- Efficiency for Converting Light Energy into ATP
- Comparing Power and Efficiency of Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency of Converting Chemical to Electrical Energy
- Functional and Structural Differences of Bacteriorhodopsin and Bacterial Reaction Center
- Conclusion
- References
Efficiency for Converting Light Energy into Electrochemical Energy
Electrochemical energy produce electric energy from chemical energy.bR emits 1 proton/photon, cross-membrane voltage 300mV: 300meV/photon [4] while bRC generates 4 protons/photon equal to 200mV multiplied by 4 resulting to 800meV/photon [5].
- Mini Review
- Abstract
- Introduction
- Computation and Methods for Calculating ATP in Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency for Converting Light Energy into Electrochemical Energy
- Efficiency for Converting Light Energy into ATP
- Comparing Power and Efficiency of Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency of Converting Chemical to Electrical Energy
- Functional and Structural Differences of Bacteriorhodopsin and Bacterial Reaction Center
- Conclusion
- References
Efficiency for Converting Light Energy into ATP
BR generates 22 photons per ATP while bRC generates 1 photon per ATP. Therefore, bR constitutes 22 photons multiplied by 300meV equals to 6600meV which is equivalent to 6.6V [6]. BRC generates 1 photon multiplied by 800meV is equal to 800meV equivalent to .8V [5].
- Mini Review
- Abstract
- Introduction
- Computation and Methods for Calculating ATP in Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency for Converting Light Energy into Electrochemical Energy
- Efficiency for Converting Light Energy into ATP
- Comparing Power and Efficiency of Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency of Converting Chemical to Electrical Energy
- Functional and Structural Differences of Bacteriorhodopsin and Bacterial Reaction Center
- Conclusion
- References
Comparing Power and Efficiency of Bacteriorhodopsin and Bacterial Reaction Center
bR and bRC are both directly correlated with power and efficiency. The higher the unit of power (i.e. milli volts), the more efficient it is in driving industrial applications. Typical instrumentations driven by bR and bRC include bioreactors for hydrogen production, molecular detection and biosensing to name a few [2]. Calculating the power and efficiency of bR and bRC involve the formulas below Figure 1.
P=iV
i= current, V= voltage
Efficiency= Work Output/Work Input * 100% Efficiency= Energy Output/ Energy Input *100 %( Thermodynamic Efficiency) [7].
W=Fd
To get electrochemical efficiency, photo cycle efficiency multiplied by cross membrane voltage gradient times number of protons per photo cycle divided by photon energy. For chemical efficiency, photo cycle efficiency is multiplied by energy per ATP which is always 300meV, divided by number of photons per ATP multiplied by number of protons per photon divided by photon energy. Hence, bR electrochemical efficiency is 7 % (.6*300meV* 1 proton/2.5eV) while bRC electrochemical efficiency is 1 6% (.200meV*1*1*1proton/2.5eV). Chemical efficiency of bR is .54% (.300meV/ 22 * 2.5eV) while bRC chemical efficiency is 3% (.300meV/ (2.5 eV* 4 protons) [5]. Photon energy (2.5eV) was obtained by using the formula E= hc/ λ, where h is the Planck's constant (6.63x10-34), c is the speed of light (3.0x108m/s) and wavelength in the order of 500nm, when green light is emitted, blue and red light are absorbed Figure 2.
- Mini Review
- Abstract
- Introduction
- Computation and Methods for Calculating ATP in Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency for Converting Light Energy into Electrochemical Energy
- Efficiency for Converting Light Energy into ATP
- Comparing Power and Efficiency of Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency of Converting Chemical to Electrical Energy
- Functional and Structural Differences of Bacteriorhodopsin and Bacterial Reaction Center
- Conclusion
- References
Efficiency of Converting Chemical to Electrical Energy
Kim YC et al. [8] this equation derived from Hummer et al. entails the mechanism of converting chemical to electrical energy: γ= (1+η)Vm/ΔGp Chemical Energy symbolizes Δ Gp, which is equal to (.5eV) (mimic Cco) ((1 + 1).150mv/.5)-100%= 40% while electrical energy symbolizes (1+η )Vm. Vm is them embrane potential, η is pumping efficiency; equivalent to the equation J pump/Jel. And bR constant value is 300meV while bRC constant value is 80meV. BR efficiency of converting chemical to electrical energy is ((1+1).300meV/6.6V)=9% while bRC efficiency of converting chemical to electrical energy is (1+1).200meV/.8V)= 50%. (bR: 100%) (bRC:) [9,10].
- Mini Review
- Abstract
- Introduction
- Computation and Methods for Calculating ATP in Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency for Converting Light Energy into Electrochemical Energy
- Efficiency for Converting Light Energy into ATP
- Comparing Power and Efficiency of Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency of Converting Chemical to Electrical Energy
- Functional and Structural Differences of Bacteriorhodopsin and Bacterial Reaction Center
- Conclusion
- References
Functional and Structural Differences of Bacteriorhodopsin and Bacterial Reaction Center
BR and bRC are both proton pumps that utilize solar energy and convert it to chemical energy Table 1. Although the function of both proteins are the same, they have distinct structural features and consequently have differing energy emissions.BR has a simpler structure than bRC. It is composed of 1740 atoms, while bRC contains 7155 atoms. BR has a chromophore in the middle while bRC contains Bph, Bchl and Q chromophores. BR is composed of seven transmembrane helices; while bRC compose five helices in the L and M subunits and one in the H subunit [3] (Solution structure of the loops of bacteriorhodopsin closely resembles the crystal structure). Both proteins, bR and bRC, come from purple membranes of Halobacterium halobium and Rhodobacter sphaeroides, respectively [11-20].

- Mini Review
- Abstract
- Introduction
- Computation and Methods for Calculating ATP in Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency for Converting Light Energy into Electrochemical Energy
- Efficiency for Converting Light Energy into ATP
- Comparing Power and Efficiency of Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency of Converting Chemical to Electrical Energy
- Functional and Structural Differences of Bacteriorhodopsin and Bacterial Reaction Center
- Conclusion
- References
Conclusion
bRC is more efficient in converting solar energy to chemical energy and solar energy to ATP than bR, while bR generates more electricity, considering the photons emitted by each. BR generates 22 photons, multiplied by 300meV equaling to 6600meV equivalent to 6.6V, while bRC generates 1 photon, multiplied by 800meV equaling to 800meV equivalents to .8V. With 6.6 voltages, lanterns, flashlights and automobiles can be powered.
- Mini Review
- Abstract
- Introduction
- Computation and Methods for Calculating ATP in Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency for Converting Light Energy into Electrochemical Energy
- Efficiency for Converting Light Energy into ATP
- Comparing Power and Efficiency of Bacteriorhodopsin and Bacterial Reaction Center
- Efficiency of Converting Chemical to Electrical Energy
- Functional and Structural Differences of Bacteriorhodopsin and Bacterial Reaction Center
- Conclusion
- References
References
- Lórenz-Fonfría VA, Heberle J (2014) Proton Transfer and Protein Conformation Dynamics in Photosensitive Proteins by Time-resolved Step-scan Fourier-transform Infrared Spectroscopy. J Vis Exp 88: eS1622.
- Seema Trivedi, Om Prakash Choudhary, Jitendra Gharu (2011) Different Proposed Applications of Bacteriorhodopsin. Recent Patents on DNA & Gene Sequences 5(1): 35-40.
- Allen JP, Feher G, Yeates TO, Komiya H, Rees DC (1987)Structure of the Reaction Center from Rhodobacter sphaeroides R-26: The Cofactors. Proc Natl Acad Sci USA 84(16): 5730-5734.
- Heberle (2000) Proton Transfer Reactions Across Bacteriorhodopsin and Along the Membrane. Biochim Biophys Acta 1458(1): 135-147.
- Geyer T, Helms V (2006) Reconstruction of a Kinetic Model of the Chromatophore Vesicles from Rhodobacter sphaeroides. Biophys J 91(3): 927-937.
- 6. Hartmann R, Oesterhelt D (1977) Bacteriorhodopsin mediated photophosphorylation in Halobacterium halobium. Eur J Biochem 77(2): 325-335.
- Juretiç D, Zupanovic P (2003) Photosynthetic Models with Maximum Entropy Production in Irreversible Charge Transfer Steps. Comput Biol Chem 27(6): 541-553.
- Kim YC, Hummer G (2012) Proton-pumping Mechanism of Cytochrome c Oxidase: A Kinetic Master-Equation Approach. Biochim Biophys Acta 1817(4): 526-536.
- Kim YC, Wikström M, Hummer G (2007) Kinetic Models of Redoxcoupled Proton Pumping. Proc Natl Acad Sci USA 104(7): 2169-2174.
- Kim YC, Wikström M, Hummer G (2009) Kinetic gating of proton pump in cytochrome c oxidase. Proc Natl Acad Sci USA 33: 13707-13712.
- Michael L Cartron, John D Olsen, Melih Sener, Philip J Jackson, Amanda A Brindley, et al. (2014) Integration of Energy and Electron Transfer Processes in the Photosynthetic Membrane of Rhodobacter sphaeroides. Biochim Biophys Acta 1837(10): 1769-1780
- Gerencsér L, Maróti P (2006) Uncoupling of Electron and Proton Transfers in the Photo cycle of Bacterial Reaction Centers under High Light Intensity. Biochemistry 45(17): 5650-5662.
- Tiede (1996) Time-Resolved Electrochromism Associated with the Formation of Quinone Anions in the Rhodobacter sphaeroides R26 Reaction Center. Biochemistry 35(33): 1076375.
- Crofts AR, Meinhardt SW, Jones KR, Snozzi M (1983) The Role of the Quinone Poolin the Cyclic Electron Transfer Chain of Rhodopseudomonas sphaeroides. Biochim Biophys Acta 723(2): 202218.
- Murchie (2011) Manipulation of Photoprotectionto Improve Plant Photosynthesis. Plant Physiol 155(1): 86-92.
- Goss R, Lepetit B (2015) Biodiversity of NPQ. J Plant Physiol 172: 1332.
- Rhinow D, Imhof M, Chizhik I, Baumann RP, Hampp N (2012) Structural Changes in bR Caused by Two Photon Induced Photo bleaching. J Phys Chem B 116(25): 7455-7462.
- Vos MH, Lambry JC, Robles SJ, Youvan DC, Breton J, et al. (1992) Fem to second Spectral Evolutionof the Excited State of Bacterial Reaction Centers at 10K. Proc Natl Acad Sci USA 89(2): 613-617.
- Berger Gérard, Tiede , David M, Breton Jacques (1984) HPLC Purification of a Biologically Active Membrane Protein: The Reaction Center from Photosynthetic Bacteria Biochemical and Biophysical Research Communications12(1) : 47-54.
- Katragadda M, Alderfer JL, Yeagle PL (2000) Solution structure of the loops of bacteriorhodopsin closely resembles the crystal structure 1466: 1-6.






























