Pyro- Versus Hydrometallurgy or Dry Versus Wet Methods
Fathi Habashi*
Department of Mining, Metallurgical, and Materials Engineering, Laval University, Canada
Submission: December 19, 2017; Published: January 08, 2018
*Corresponding author: Fathi Habashi, Department of Mining, Metallurgical, and Materials Engineering, Laval University, Quebec City, Canada, Email: Fathi.Habashi@arul.ulaval.ca
How to cite this article: Fathi H. Pyro- Versus Hydrometallurgy or Dry Versus Wet Methods. JOJ Material Sci. 2017; 3(4): 555618. DOI: 10.19080/JOJMS.2018.03.555618
Abstract
Processes using hydrometallurgical methods have replaced many using pyrometallurgical techniques in the alumina, zinc, copper, and nickel industries. In the chemical industry earlier, the Solvay wet process of 1865 replaced the Leblanc dry process of 1782 for producing sodium carbonate.
Keywords: Sodium carbonate; Elemental sulphur; Matte formation; Pressure leaching; Precipitation
Introduction
Pyrometallurgy was most successful when high grade massive ores were treated in a blast furnace, because such a furnace has maximum heat economy being itself a heat exchanger. Dust problems were also minimum because the ore was in the form of large lumps. With the exhaustion of such raw material, metallurgists turned their attention towards the treatment of low-grade ores. This necessitated extensive grinding and flotation, which resulted in finely divided concentrates as raw material which could not be charged to a blast furnace. Hence the birth of the fossil-fired horizontal reverberatory furnace for melting sulfide concentrates. This was a turning point for the worst with respect to pollution of the environment, high energy consumption, and excessive dust formation.
Treatment of Sulfide Ores
Pyrometallurgical treatment of sulfide ore is plagued by the problem of sulfur dioxide generation. If it is in high enough concentration, it must be used for making acid and a nearby market for this acid must be found. If the sulfur dioxide concentration is too low for making acid the gas is simply emitted to the atmosphere. On the other hand, sulfides can be treated by hydrometallurgical methods without generating sulphur dioxide thus eliminating the need to manufacture sulfuric acid. The sulfur can be recovered in the elemental form which can be easily stock-piled, or transported at low cost [1].
Material Handling
In pyrometallurgical processes, the metallurgist is forced to transfer molten slags and matte from one furnace to the other in large, heavy, refractory-lined ladles. Besides the inconvenience and the cost of handling these materials, there is also the inevitable gas emission from them because they are usually saturated with sulfur dioxide and during transfer they cool down a little, resulting in decreased gas solubility and emission, hence the inconvenient working condition. In hydrometallurgical plants, solutions and slurries are transferred by pipelines without any problem.
Energy
Because ofthe high temperature involved in pyrometallurgical processes, usually around 1500°C, the reaction rates are high but much fuel is needed. To make a process economical, heat recovery systems are essential. Heat can be readily recovered from hot gases, but rarely from molten material like slag or metal. Thus, a great deal of energy is lost. Further, the equipment needed for heat economy is bulky and expensive. In hydrometallurgical processes, on the other hand, less fuel is needed because of the low temperature involved (usually below 100 °C) and heat recovery is usually no problem [2].
Dust
Combustion of fossil fuels in a reverberatory furnace results in the formation of a large volume of gases that carry over large amounts of dust. This must be recovered to abate pollution and because the dust itself is also a valuable material. The technology of dust recovery is well established but the equipment is bulky and expensive. In hydrometallurgical processes this is no problem because wet material is usually handled.
Treatment of Low-Grade Ores
Treatment of low-grade ores by pyrometallurgy is unsuitable because of the large amount of energy required to melt the gangue minerals. On the other hand it is especially suitable to use hydrometallurgy since a selective leaching agent that can solubilize the valuable minerals, and not the gangue, is usually available [3].
Economics
The economics of a pyrometallurgical process is usually suitable for large scale operations and this requires a large capital investment. On the other hand, hydrometallurgical processes are suitable for small scale operations and low capital investment. The hydrometallurgical units can be increased in number when the need arises without any economic disadvantage [4].
Residues
Many residues of pyrometallurgical processes are coarse and harmless. For example, slags which are a silicate phase can be stored in piles exposed to air and rain without the danger of dissolution and contaminating the streams. They are just unacceptable from the aesthetic point of view. On the other hand most residues of hydrometallurgical processes are finely divided solids. If they are dry, they create dust problems when the wind blows and when wet they will gradually release metal ions in solution which will contaminate the environment. Hence well prepared storage sites must be created.
Sodium Carbonate
Leblanc versus Solvay process
Leblanc Process: In 1782, Nicholas Leblanc (1742-1806) made sodium carbonate by reacting sodium chloride with H2SO4, reducing the resultant sodium sulfate by carbon in a furnace to form sodium sulfide, which was then reacted with CaCO3 in another furnace to form Na2CO3 and CaS. Sodium carbonate is then leached away from the product and evaporated (Figure 1):
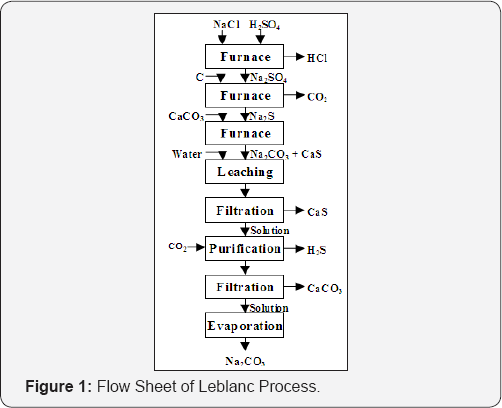
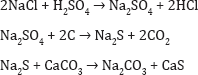
Any calcium sulfide dissolved with the sodium carbonate is converted to CaCO3 by blowing CO2 in the solution. This will also removes H2S that may form which must be collected and converted to elemental sulfur:
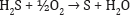
The process marks the beginning of the chemical industry and dominated the world until it was superseded by the Solvay process about a century later.
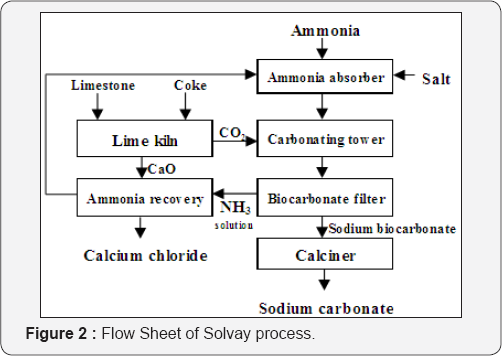
Solvay process: Ernest Solvay (1838-1922) built the first plant for the production of sodium carbonate by the wet method in 1865. The process proved to be more economical than the Leblanc Process, and by the end of the century it had displaced it completely. In this process ammonia is absorbed in a concentrated brine solution in an absorption tower. Carbon dioxide is then absorbed in the ammoniated brine in another tower whereby sodium bicarbonate precipitates:
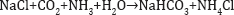
The carbon dioxide required for reaction is produced by calcination of the limestone. The sodium bicarbonate precipitate is then converted to the final product, sodium carbonate by calcination at 160-230oC:
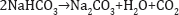
The ammonium chloride solution is then reacted with the quicklime to recover ammonia for recycle:
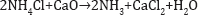
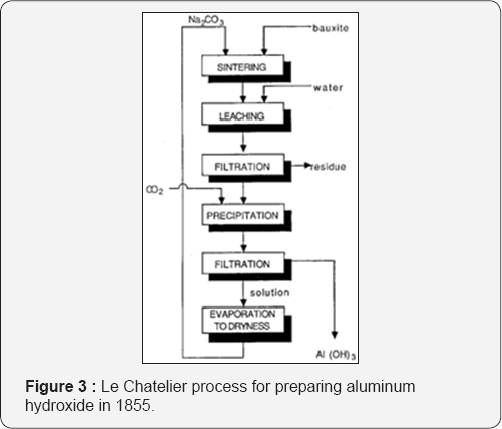
An only small amount of additional ammonia is used to make up for losses. A flow sheet of the process is shown in (Figure 2). Louis Le Chatelier (1815-1873) in France, father of Henri Le Chatelier at the School of Mines in Paris, produced alumina for the new aluminum industry by sintering bauxite with sodium carbonate at high temperature to make sodium aluminates and leaching the product with water. After filtering off the insoluble residue the solution was then carbonated with CO2 to precipitate aluminium hydroxide which was then calcined to alumina (Figure 3). The process was used by Saint Claire Deville (1818-1881).
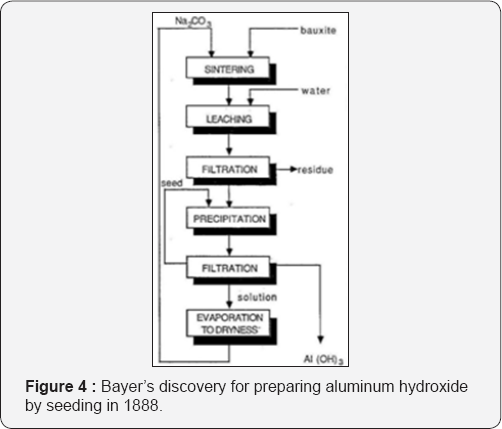
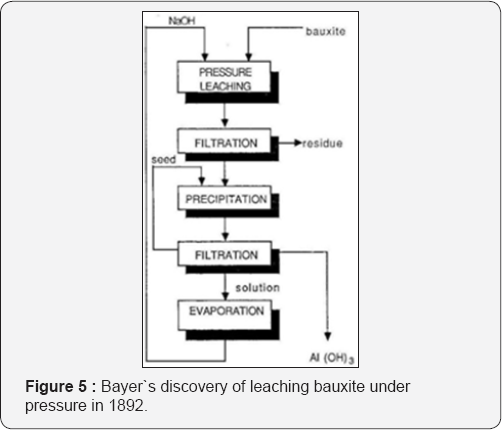
Karl Josef Bayer (1847-1904) in Russia produced of pure aluminum hydroxide in 1888 by precipitation from sodium aluminate solution by a seed of a freshly precipitated aluminum hydroxide when agitated vigorously in the cold solution (Figure 4). The product was pure and can be easily filtered and washed. Four years later in 1892 he made his second discovery that alumina contained in bauxite could be dissolved selectively by heating with a solution of sodium hydroxide under pressure in an autoclave to form sodium aluminate solution. He found also that the alkaline mother liquor obtained after the precipitation of aluminium hydroxide could be used (Figure 5). Bayer introduced pressure reactors as well as precipitation tanks for seeding.
Zinc
Retort versus Pressure Leaching.
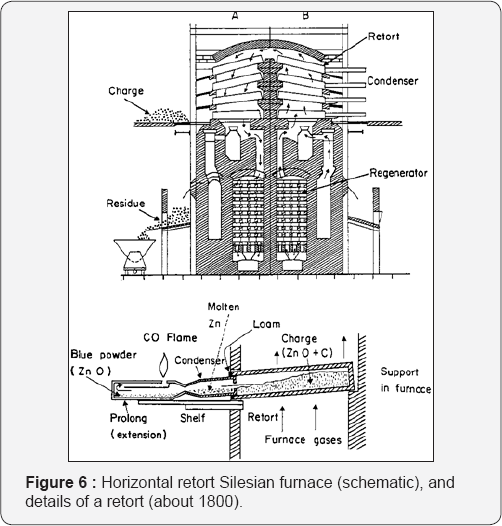

Retort Proces: The first important zinc smelting works in Upper Silesia in 1798 built by Johann Ruberg (1751-1807) used the horizontal retort process (Figure 6). Zinc sulfide, the main zinc ore, was roasted previously to zinc oxide which was then mixed with carbon and reduced in the retorts to metallic zinc. Any carbon monoxide produced during the reduction was burnt outside the retorts as a flame (Figure 7). The retorts were fixed horizontally into the furnace allowing them to be charged and discharged without cooling. The condensers were luted onto the open ends and this also simplified the task of metal recovery. By placing the retorts in large banks, fuel efficiency was greatly increased. A large plant was built in 1810 in Belgium which few years later became the largest zinc-producing country in the world. This was the predecessor of the Societe de la Vieille Montagne. The reactions involved:
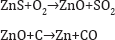
Zinc obtained by this process is refined by distillation in vacuum. It was shut down when a hydrometallurgical was later adopted based on roasting-leaching-electro winning process which in 1980's became a pressure leaching process.

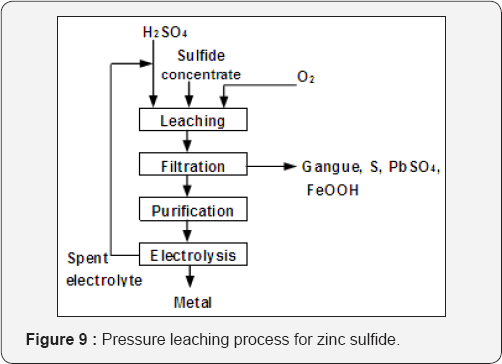
Pressure Leaching Process: The aqueous oxidation of zinc sulfide concentrates introduced in the 1980s solved numerous problems associated with the roasting-leaching-electro winning process (Figure 8). The zinc industry became independent of the necessity to fabricate and market H2SO4 (Figure 9). The reactions involved:
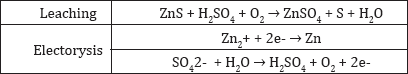
Copper
Matt formation versus Pressure leaching
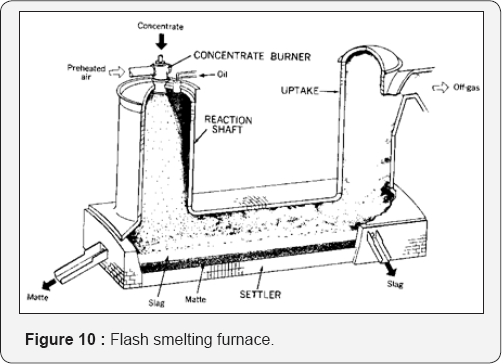
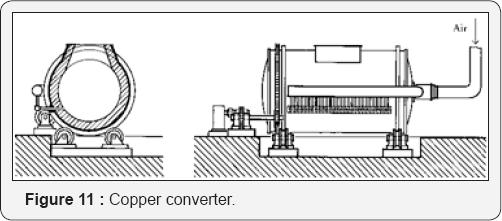
Matte Formation: Copper sulfide concentrate is melted in a flash smelting furnace (Figure 10) to produce matte. This must be treated further in a converter (Figure 11) to remove the iron then to produce blister copper for refining. The process produces much dust, SO2, and heat recovery systems must be installed. The slag must be treated in an electric furnace to recover as much as possible of the suspended matte. It involves also extensive ladles to transfer the molten material from one reactor to the other Pressure hydrometallurgy offers a better solution.
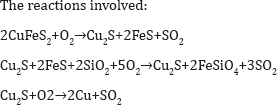
Pressure Leaching
A process for pressure leaching of chalcopyrite concentrates has been installed in Arizona. It is similar to the pressure leaching of zinc sulfide shown above.
Nickel
Reduction versus Precipitation
Reduction of Oxide: There are many methods for treating nickel sulfide concentrates. One of them is the oxidation to oxide then reduction of the oxide by carbon to metal which is then cast into anodes for electrolytic refining [1-4].
Precipitation from Solution: Nickel sulfide concentrate is leached in ammonia and oxygen in an autoclave then the pure nickel is precipitated from solution by hydrogen under pressure:
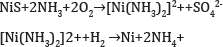
The solution contains ammonium sulfate which is marketed as a fertilizer. Canadian coins were produced by this method (Figure 12).

References
- Métallurgie Extractive Québec, Québec City, Canada.
- F Habashi (2002) Textbook of Pyrometallurgy, Metallurgie Extractive Québec, Québec City, Canada.
- F Habashi, (2003) Metals from Ores. An Introduction to Extractive Metallurgy, Métallurgie Extractive Québec, Québec City, Canada.
- F Habashi (2006) Readings in Historical Metallurgy. Selected Topics for Metallurgists, Métallurgie Extractive Québec, Québec City, Canada 2.






























