Carbon Dioxide Digital Subtraction Angiography (CO2 DSA) in Peripheral Intervention-The Malaysian Experience
Alan Koay Choon Chern1*, Johan Rizwal Ismail1,2, Kumara Gurupparan1 and Shaiful Azmi Yahaya1
1Peripheral and Vascular Unit, National Heart Institute (IJN), Malaysia
2Department of Cardiology, UiTM, Malaysia
Submission: November 15, 2017; Published: November 27, 2017
*Corresponding author: Alan Koay Choon Chern, Peripheral and Vascular Unit, National Heart Institute (IJN), Malaysia, Email: alankoay81@gmail.com
How to cite this article: Alan K C C, Johan R I, Kumara G, Shaiful A Y. Carbon Dioxide Digital Subtraction Angiography (CO2 DSA) in Peripheral Intervention-The Malaysian Experience. J Cardiol & Cardiovasc Ther 2017; 8(3): 555738. DOI: 10.19080/JOCCT.2017.08.555738
Introduction
Carbon dioxide (CO2) was first introduced as a non- vascular contrast agent in the 1920s when retroperitoneal pneumatography was used to visualise the kidneys and adrenal masses. While Paul Rosentein employed oxygen (O2) as a contrast agent, it was Humberto Horacio Carelli who propagated the use of CO2, which he perceived to be absorbed faster than O2, thus conferring a lower risk of air emboli [1]. Thirty to forty years on, the use of CO2 expanded into the realm of the cardiovascular system, firstly to evaluate for pericardial effusion then subsequently for veno cavography [2]. A decade later through a stroke of serendipity, Irvin Hawkins discovered that CO2 can also be safely applied in arteriography [3]. With the advent of digital subtraction angiography (DSA) and better delivery systems in the 1980s, the use of CO2 as a contrast agent continued to make headway in the field of peripheral intervention, particularly in patients with chronic kidney disease (CKD) and iodinated contrast allergy [4,5].
To the best of our knowledge, CO2 DSA has never been utilized in Malaysia until recently on 26 May 2017, when the Peripheral and Vascular Unit of the National Heart Institute (IJN) explored its use as an alternative contrast agent for diagnostic and therapeutic endovascular procedures. The traditional method of using iodinated (ionic or non-ionic) contrast medium (ICM) has its risk and limitation especially in the majority of diabetic patients with peripheral arterial disease who also have some degree of renal insufficiency. As such, the use of CO2 DSA has granted us more leeway when dealing with this group of patients.
Properties of Carbon Dioxide
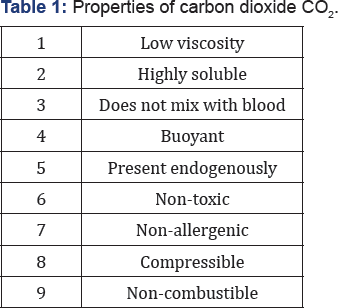
Suffice to say, a basic understanding of CO2 properties would enable the operator to utilize it with confidence and also caution (Table 1). Its low viscosity (1/400 of ICM) and high solubility (30 times O2), allows it to be rapidly dissolved within 30-60 seconds and removed from the lungs. Having mentioned that, CO2 does not mix with blood and together with its buoyancy, a blood-gas interface can be exploited to generate a negative image with DSA. It is thus important to note that because of its buoyancy, CO2 angiography can only safely be recommended for angiography below the diaphragm and preferably in a Trendelenburg position, to avert the risk of spinal, coronary and cerebral gas embolism [6]. Being endogenously present, there is no concern whatsoever with allergy or renal toxicity. Its compressibility and non-combustibility allow for optimal storage and delivery via dedicated systems. Such systems are designed to avoid excessive compression of CO2, which if otherwise delivered can result in pain due to an explosive gaseous expansion [7].
Concerns with Carbon Dioxide
Arguably, the most worrying among all concerns is gas embolism especially if used for arterial study above the diaphragm and should therefore be avoided. Otherwise, its rapid solubility should render it reasonably safe for studies below the diaphragm. Intuitively, some operators are wary of the use of CO2 particularly in patients with respiratory compromise such as chronic obstructive pulmonary disorder (COPD). Studies so far have not shown CO2 DSA to be detrimental in such patients, as the volume of CO2 used in imaging is relatively small compared to endogenous CO2 production. It is also recommended to allow a longer interval between injections (2 minutes), instead of the routine 30-60 seconds. Discomfort or pain is another concern that is related to gaseous expansion within the blood vessels, although the vast majority of patients only have minimal or no sensation at all. With the development of new delivery systems and stacking software, the volume of CO2 used can be reduced significantly without compromising image quality, making the procedure more tolerable to patients [8].
Another rare but real complication is the "vapour lock" phenomenon that can occur due to air trapping in nondependent vessels primarily during aortic intervention. Hawkins [3] reported only one incident of transient mesenteric ischemia resulting from this phenomenon, which eventually normalized after 3 days when endoscopy was repeated. Although mitigated by its high solubility, its risk increases when large volumes of CO2 are injected or when air contamination room occurs. Operators are often oblivious to radiation exposure and rightly need not be unduly worried if appropriate radiation protection is practised. Notwithstanding, it has to be emphasized that the acquisition of CO2 images requires double the frame rate that of ICM.
Delivery System

The CO2 delivery method has evolved significantly since its discovery as a contrast agent. At its inception, it was delivered via a hand-held syringe that was then substituted by a liquid contrast injector. These are susceptible to room air contamination and hence, the next phase of development saw the connection of the CO2 cylinder directly to the patient through a series of stopcocks. A non-pressurized reservoir (blood bag) was soon introduced to reduce the risk of overloading the vascular system with pressurized gas (Figure 1) [9,10]. In recent years, more sophisticated and compact dedicated delivery systems were developed, among which the user-friendly Angiodroid automated digital CO2 injector appears most outstanding-safe, reliable and less painful.

The Angiodroid CO2 injector was first launched in 2013 and two years later, its second version was introduced (Figure 2). It has successfully automated the process of CO2 delivery, making it independent from healthcare professionals and therefore, reduces potential for errors. Its main technical aspects comprise digital volume and pressure settings (touch screen console), fast automatic reload (20 seconds) for repeatable injection, and high accuracy of volume and pressure injection, dual microcontroller to ensure high performance and safety; and remote controller for injection.
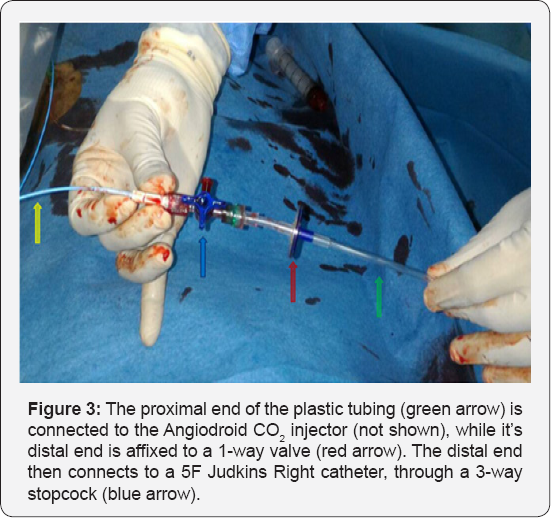
The system comes with disposable tubing affixed to a 1-way valve at its distal end to prevent room air contamination and 3-way stopcock, which allows for supplementary use of ICM at the operator’s discretion. The 3-way stopcock is then connected to the angiography catheter and the entire system is purged with 10cc of CO2 before it is ready for use (Figure 3). How much CO2 volume and pressure to be injected would depend on the patient's systolic blood pressure and body region of interest (provided in a chart). Most of the latest imaging hardware in the invasive laboratory is equipped with software that is compatible with the Angiodroid system, namely the CO2 protocol. Compared with ICM, the CO2 angiographic images are lower in spatial resolution [11]. Once acquired, the frames are integrated using stacking software to produce a final image with an improved resolution.
Case Illustration
A 56-year-old gentleman with a history of type 2 diabetes mellitus, hypertension, coronary artery disease (PCI in 2013), dyslipidemia and chronic smoking presented with a right foot non-healing ulcer. Physical examination revealed absent pulse at both popliteal arteries and below, with an arterial ulcer beneath the right big toe. X-ray did not show osteomyelitis at the affected area. Computed tomography angiography revealed bilateral superficial femoral arteries (SFA) occlusion and diffuse disease distally. Right lower limb percutaneous transluminal angioplasty (PTA) was offered to improve distal perfusion with the hope of expediting wound healing. Baseline renal function was normal with a creatinine level of 54μmol/L, white cell count was 13.6x103μL with predominantly neutrophils and elevated ESR/CRP.
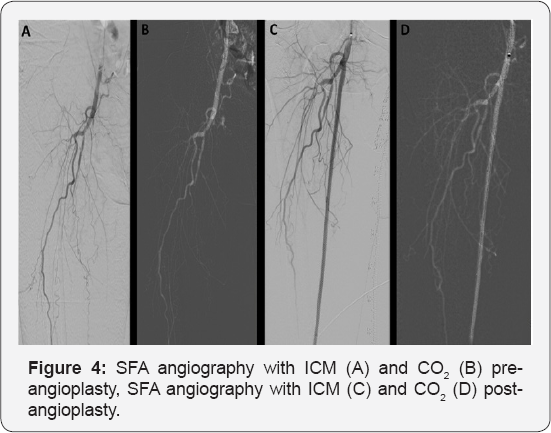
PTA was carried out with CO2 as the contrast agent using the Angiodroid CO2 injector. Left femoral artery was accessed with a 6F Flexor® Ansel guiding sheath (Cook Medical, Bloomington, IN) crossing over to the right femoral artery. Angiography showed occluded right SFA from the ostium. Intravenous fentanyl with a cumulative dose of 100mg was administered for pain relief. The lesion was successfully crossed and predilated with an Armada 5.5x200mm. It was then stented with Supera 5.0x200mm distally and overlapped with Absolute Pro 6.0x80mm proximally up to the SFA ostium. This was followed by post dilatation with Armada 5.0x80mm and 6.0x80mm. Final angiography showed improved flow to the distal vessels (Figure 4). The total ICM used was only 100cc while CO2 volume was 680cc with a total procedural time of 3.5 hours. Serial arterial blood gases (ABG) that were taken demonstrated the absence of CO2 retention. Pre-procedural ABG showed pH 7.48, p CO2 40.4, pO2 87.6, H CO3 29.6, BE 5.7. Post-procedural ABG showed pH 7.43, p CO2 40.4, pO2 89.8, HCO3 26.0, BE 1.6. Renal profile 24 hours after the procedure showed no evidence of contrast-induced nephropathy (CIN). Our experience also showed that smaller vessels (<10mm) had better correlation with ICM than CO2. Many experts opined that it is because CO2 dissolves too quickly and largely due to its low density rather than lack of perfusion (Figure 5) [12]. This case illustrated the feasibility and safety of CO2 in a successful PTA with the use of supplementary ICM.

Conclusion
CO2 DSA has great benefits in a variety of clinical scenarios, specifically CKD andiodinated contrast allergy, which are the bane of ICM. The unique properties of CO2 make it a safe alternative contrast agent in both diagnosis and intervention in this group of patients. Besides peripheral arterial disease, its use in the realm of cardiovascular system has now also expanded into endovascular abdominal aneurysmal repair, venous intervention, transarterial chemo-embolization, vena cava filter placement, renal artery ablation, detection of arteriovenous fistula and diagnosis of acute arterial hemorrhage (2.5 times more sensitive than ICM) [13]. The state-of-the-art Angiodroid technology has revolutionized CO2 DSA and our recent experience with it has witnessed a learning curve that was not at all steep.
References
- Buzzi AE, Gotta C (2014) Humberto Horacio Carelli: pionero de la radiologia argentina. Rev Argent Radiol 78(1): 49-59.
- Scatliff JH, Kummer AJ, Janzen AH (1959) The diagnosis of pericardial effusion with intracardiac carbon dioxide. Radiology 73(6): 871-883.
- Hawkins IF, Caridi JG (1998) Carbon dioxide (CO2) digital subtraction angiography: 26-year experience at the University of Florida. Eur Radiol 8(3): 391-402.
- Hawkins IF, Cho KJ, Caridi JG (2009) Carbon dioxide in angiography to reduce the risk of contrast-induced nephropathy. Radiol Clin 47(5): 813-825.
- Chao A, Major K, Kumar SR, Patel K, Trujillo I, et al. (2007) Carbon dioxide digital subtraction angiography-assisted endovascular aortic aneurysm repair in the azotemic patient. J Vasc Surg 45(3): 451-460.
- Cho KJ (2015) Carbon dioxide angiography: scientific principles and practice. Vasc Spec Int 31(3): 67-80.
- Caridi JG (2015) Vascular imaging with carbon dioxide: confidence in a safe, efficacious, user-friendly system. Semin Interv Radiol 32(4): 339342.
- Moos JM, Ham SW, Han SM, Lew WK, Hua HT, et al. (2011) Safety of carbon dioxide digital subtraction angiography. Arch Surg 146(12): 1428-1432.
- Cherian MP, Mehta P, Gupta P, Kalyanpur TM, Jayesh S, et al. (2009) Technical note: A simple and effective CO2 delivery system for angiography using a blood bag. Indian J Radiol Imaging 19(3): 203205.
- Cho KJ, Hawkins IF (2011) Discontinuation of the plastic bag delivery system for carbon dioxide angiography will increase radio contrast nephropathy and life-threatening complications. Am J Roentgenol 197(5): W940-W941.
- Bees NR, Beese RC, Belli AM, Buckenham TM (1999) Carbon dioxide angiography of the lower limbs: initial experience with an automated carbon dioxide injector. Clin Radiol 54(12): 833-888.
- Rolland Y, Duvauferrier R, Lucas A, Gourlay C, Morcet N, et al. (1998) Lower limb angiography: a prospective study comparing carbon dioxide with iodinated contrast material in 30 patients. Am J Roentgenol 171(2): 333-337.
- Hawkins IF, Caridi JG, LeVeen RF, Khoze SD, Mladinich CRJ (2000) Use of carbon dioxide for the detection of gastrointestinal bleeding. Tech Vasc Interv Radiol 3(3): 130-138.






























