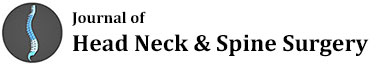Biopsy-Proven Brain Metastases from Prostate Adenocarcinoma on 68Ga PSMA PET/CT: Case Series and Review of the Literature
Charlotte Yin1*, Biyi Chen2, Yael Barnett3, Bao Ho1,3, Cecelia Gzell4 and Louise Emmett1
1 Department of Nuclear Medicine and Theranostics, St Vincent’s Hospital, Australia
2Faculty of Medicine, Sydney University, Australia
3 Department of Medical Imaging, St Vincent’s Hospital, Australia
4 Genesis Cancer Care, St Vincent’s Clinic, Australia
Submission: August 13, 2018; Published: September 19, 2018
*Corresponding author: Charlotte Yin, Department of Nuclear Medicine and Theranostics, St Vincent’s Hospital Sydney, 390 Victoria St, Darlinghurst, NSW 2010, Australia.
How to cite this article: Charlotte Y, Biyi C, Yael B, Bao H, Cecelia G, Louise E. Biopsy-Proven Brain Metastases from Prostate Adenocarcinoma on 68Ga PSMA PET/CT: Case Series and Review of the Literature.J Head Neck Spine Surg. 2018; 3(4): 555616. DOI: 10.19080/JHNSS.2018.03.555616
Abstract
Prostate cancer brain metastases are extremely rare and typically occur at a late stage in the course of the disease with poor prognosis. However, the incidence is rising as novel antiandrogens and radionuclide therapy prolong survival and change the natural course of the disease. Surveillance imaging of the brain is not the current standard of care. We present two cases of patients who had brain metastases from prostate adenocarcinoma initially detected on prostate specific membrane antigen (PSMA) positron emission tomography/computed tomography (PET/CT) and also provide a review of the epidemiology, pathogenesis, imaging features, pathogenesis and current treatment modalities of prostate cancer brain metastases. Our patient with multiple brain metastases is still alive three and a half years post initial diagnosis after being successfully treated with surgery, androgen deprivation therapy and radiosurgery. This is the longest survival time of any patient with multiple brain metastases and systemic disease. We postulate that overall survival will increase with earlier detection and treatment of brain metastases from a prostate cancer primary and that scanning vertex to mid-thigh should be standard practice with PSMA PET imaging.
Keywords: Brain metastasis; Prostate cancer; Neuroradiology; Ga68PSMA PET/CT
Abbrevations: PCa: Prostate Cancer; Ga: Gallium; PSMA: Prostate Specific Membrane Antigen; PET/CT: Positron Emission Tomography/Computed Tomography; MRI: Magnetic Resonance Imaging; SRS: Stereotactic Radiosurgery; Gy: Gray; PSA: Prostate Specific Antigen; PAP: Prostatic Acid Phosphatase; ADT: Androgen Deprivation Therapy
Case 1
A neurologically intact 66-year-old man presented for a Ga68 prostate specific membrane antigen (PSMA) positron emission tomography (PET) scan due to a rising prostate specific antigen (PSA) level of 7.4ng/ml thirteen years after a successful radical prostatectomy for a Gleason 7 prostate cancer (PCa).
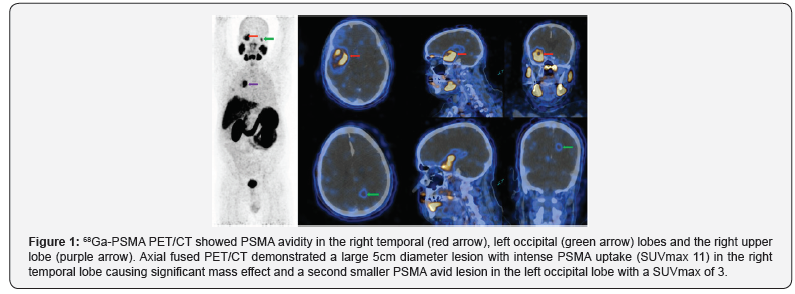
PSMA demonstrated an unusual distribution of disease with intensely PSMA avid foci in an enlarged mediastinal lymph node, within small lung nodules and in two PSMA avid brain foci within the right temporal and left occipital lobes. Brain MRI revealed a large 6.5cm complex enhancing lesion in the right temporal lobe compressing the temporal horn and the body of the right lateral ventricle. The cystic component of the mass showed no restricted diffusion. A second 1.1cm lesion was noted in the left occipital lobe superior to the ventricular trigone with peripherally restricted diffusion and increased diffusibility centrally (Figure 1).
The patient underwent a right temporal craniotomy and excision with gross total resection achieved (Figure 2). Histology confirmed metastatic prostatic adenocarcinoma with positive immunohistochemistry for PAP and PSA. The patient received adjuvant stereotactic radiosurgery (SRS) to the right temporal lobe cavity, left parietal lobe mass and hormonal manipulation with androgen deprivation therapy (ADT).
Nine months postoperatively, the patient remained clinically well with no neurological symptomology and undetectable PSA with ongoing ADT. MRI brain confirmed no recurrence in the right temporal lobe cavity but an increase in the size of the left occipital lobe lesion. PSMA PET/CT demonstrated mild persistent increased PSMA avidity in a solitary focus in the left occipital lobe. Complete metabolic response at all other metastatic sites with no new disease found. The patient was treated with further SRS (14Gy in a single fraction) to the region of PSMA avidity in the left occipital lobe. Three and a half years post operatively, he remains well with good disease control.
Case 2
A 71-year-old man with metastatic castration resistant prostate cancer previously treated with external beam radiotherapy, ADT, radium 223 therapy and docetaxel presented with a three-week history of increasing confusion and ataxia.
MRI brain revealed a well circumscribed 2.4 x 2.0cm cystic, enhancing mass in the left para-median thalamus and midbrain compressing the cerebral aqueduct and five other lesions throughout the bilateral cerebellar hemispheres. The thalamic cyst was biopsied and aspirated, but his symptoms persisted. Histologic examination confirmed metastatic prostatic adenocarcinoma. He was treated with whole-brain radiotherapy but passed away two months following radiation treatment.
Introduction
Intraparenchymal metastases from a prostate adenocarcinoma primary is rare and only occur in an estimated 0.6-1.9% of patients [1-3]. However, the incidence is increasing as novel antiandrogens and radionuclide therapy prolong survival and change the natural course of the disease.
On the 1st of May 2018, a comprehensive literature search examining peer-reviewed, English language articles from 1982 to 2018 was performed on multiple databases, yielding 1286 articles. These articles were reviewed and selected for studies that met the following inclusion criteria;
a) Patients with intraparenchymal metastases from prostate adenocarcinoma proven either on antemortem resection or stereotactic biopsy and
b) Metastases confirmed on CT or MR brain imaging.
Additional relevant studies were also searched manually in the reference lists of identified studies and by using the “related articles” tool in PubMed. Patients with dural or skull-based tumours extending into the brain were excluded from the review. We found 1286 articles and considered 29 articles eligible. A total of 47 men with brain metastases from prostate adenocarcinoma origin confirmed on radiological imaging and antemortem biopsy was thus identified in the literature (including our 2 cases reported here).
Discussion
Epidemiology
For patients with brain parenchymal metastases from prostate adenocarcinoma of any subtype, the mean at diagnosis of brain metastasis was 66.3 years (range, 48-88 years) and the median age was 66 years.
35 of the 47 men had known prostate adenocarcinoma at the time of their cerebral metastasis diagnosis whilst for 12 men this was the initial presentation of their primary prostate tumour. The mean age at diagnosis of brain metastasis for men with known prostate cancer (PCa) was 65.5 years (range, 48- 88 years). In patients with cerebral metastases as the initial presentation of their primary tumour, the mean age of brain metastasis diagnosis was 66.4 years (range, 56-75 years).
Hatzoglou et al. [4] study of 7 patients with biopsy proven brain metastasis from prostate adenocarcinoma was not included as we were unable to differentiate the patients’ mean age at diagnosis of brain metastasis from other patients in their cohort who had biopsy from other sites of distant disease confirming metastatic PCa.
Timing and symptoms
Patients with metastatic disease to the brain developed disease an average of 4.4 years (range, 3 days to 13 years) after the initial diagnosis of their PCa. New therapies increasing overall survival time gives the tumour enough time to develop brain metastases, that is usually a late event of the disease [5,6].
Neurological manifestations on presentation varied according to the anatomical site with most metastases situated in arterial border zones and the junction between the cortex and subcortical white matter. The majority of patients (95.7%) presented with overlapping neurological signs and symptoms. 22 patients presented with headache, the most common presenting complaint at diagnosis followed by motor weakness (n=16), ataxia (n=13), confusion (n=10), seizures (n=8), speech impairment (n=7) and visual field disturbances(n=4). In the two neurologically asymptomatic patients, the brain metastases were diagnosed on surveillance imaging after a rise in PSA.
In 11 patients, the brain was the sole site of distant metastasis [7-16]. In patients with metastatic disease, the most common extra prostatic sites were bone (61.7%), lungs (26.4%), liver (16.1%) and lymph nodes (13%).
Lesion characteristics
A total of 73 metastases was seen in the 47 patients. 39 patients (83%) had a solitary brain metastasis whereas the other 8 patients (17%) had multiple metastases. Four patients (8.5%) had two metastases and 4 patients (8.5%) had 6 or more brain metastases.
The frontal lobe (n=15) and the cerebellum (n=15) were the most prevalent sites of metastases followed by the parietal (n=14), temporal (n=12) and occipital (n=6) lobes. One patient had a solitary metastasis in the temporo-parietal lobe and one case involved the parieto-occipital lobe. Five patients had 6 metastases in the brainstem. Two cases involved the cavernous sinus and one patient (our case) had a metastasis in the thalamus. 49 patients (67.1%) had parenchymal metastases located in the cerebral hemispheres which corresponds to what is known about the distribution of parenchymal metastases from other primary cancers.
15 patients (20.5%) with brain metastases from PCa origin were found in the cerebellum and only 8.2% of metastases were found in the brainstem. This distribution of intracranial metastases is similar to metastases from breast, lung and melanoma where approximately 15% of metastases are found in the cerebellum. Brainstem are uncommon sites especially for solitary lesions and account for <1% of all brain metastases. Interestingly 3 patients (4.1%) in our review of all the literature to date had solitary metastases in the brainstem [8-9,17].
31 patients (68.9%) had one or more metastatic brain lesions in the supratentorial compartment [7,9-13,16,18-25], 11 patients (24.4%) in the infratentorial compartment [8- 10,13-14,17,23,26] and 3 patients (6.7%) in both compartments [27-29]. Two patients with lesions in the cavernous sinus were excluded.
Intraparenchymal cerebral metastases from prostate adenocarcinoma are rare and multiple metastases without systemic disease is exceedingly uncommon. In the largest case series to date of 16280 patients with brain metastases from prostate cancer by Tremont-Lukats et al. [1] only 103 patients (0.6%) had parenchymal brain metastases. In most cases the metastases were singular (86%) and supratentorial (76%). Only 3 patients (2.9%) of the cohort had both infratentorial and supratentorial metastases.
A more recent case series by Hatzoglou et al. [4] found 10 patients (47.6%) of their cohort had both supratentorial and infratentorial metastases. This is significantly higher than other case series to date and is likely due to MRI images from Hatzoglou’s case series being reviewed by a neuroradiologist versus some cases in our review being limited to only single slice images of patients’ CT or MRI which limits our ability to ascertain the distribution of lesions outside of our two cases.
Gross pathology
Parenchymal metastases are generally round, discrete lesions. The metastases in our review had variable peritumoural oedema, necrosis and mass. Non-uniformity in the spatial distribution of the parenchymal metastases suggests that vulnerability to metastases differ according to its anatomical location. Our review found parenchymal metastases from PCa had a predilection for the frontal lobe and cerebellum (n=15, 20.5%) which is consistent with other large case series which found 17-25% of metastases from PCa were found in the cerebellum [1,4]. Unlike melanoma, renal cell carcinoma and choriocarcinoma which are particularly prone to developing intratumoural haemorrhages, brain metastases from PCa was not found to have intratumoural haemorrhage on imaging or histopathology. All of the patients included in our review had biopsy confirmed adenocarcinoma with positive immunohistochemical staining for PSA and PAP.
Imaging features
The cerebral metastases had highly variable imaging appearance and was difficult to differentiate from metastases originating from other primary tumour sites. Eight patients in our review only had CT imaging of their cerebral metastases [7,9,18-21,26]. On non-contrast CT imaging, most metastases were isodense to slightly hypodense relative to grey matter. The majority of parenchymal metastases enhanced strongly following contrast administration.
On axial T1-weighted MRI, most of the parenchymal lesions were mildly hypointense. On post gadolinium imaging, nearly all of the non-haemorrhagic metastases showed enhancement with patterns of disease varying from solid uniform enhancement to nodular or ring like lesions. FLAIR imaging also demonstrated significant variability in lesion cellularity, presence of haemorrhage and amount of peritumoural oedema. On diffusion weighted imaging (DWI), well differentiated adenocarcinoma metastases generally showed no diffusion restriction compared to highly cellular metastases which demonstrated diffusion restriction.
Cystic intraparenchymal metastases from PCa are rare with only eight cases documented in literature to date, including our two patients [11,19,22-23,27,30]. Five patients had solitary cystic lesions and three patients had multiple cystic lesions. Intralesional restricted diffusion was present in both our patients.
Unlike true intraparenchymal cerebral metastases of prostate adenocarcinoma origin, prostate cancer is the second most common primary after breast cancer to metastasize to the dura [5] and poses a radiological diagnostic challenge especially when it presents as a solitary lesion which can be mistaken for a meningioma as up to 44% of prostate metastases have a dural tail [6,31,32]. Distinguishing between the two lesions is important due to the poor prognosis of intracranial metastatic PCa and the potential for conservative management versus active treatment if a radiological diagnosis is made. The incidence of brain metastases in the most recent series of 16,280 prostate cancer patients is reported to be around 0.63% [1] which is less than the 1-2.4% incidence reported in autoptic series [3,33-34] with the incidence of brain metastases being detected in the pre-MRI era the same as in the post MRI era [1]. We postulate that advances in MR imaging such as triple dose gadolinium and or 3.0Tesla (T) MRIs have led to earlier detection of metastases in PCa patients which allow earlier treatment and thus decrease the potential for further extraprostatic spread.
Ga68 PSMA PET/CT imaging appears to have superseded F18 FDG PET/CT, CT and MR imaging not only in the staging of PCa but also in the detection of PSMA-avid disease and is increasingly being used for restaging recurrent PCa. Our case demonstrates the importance of scanning from vertex to mid-thigh as albeit rare, PCa can metastasize to the brain and earlier detection and treatment correlates directly with improved survival time and quality of life.
Treatment
All of the patients included in our review underwent either surgical resection (n=37) or stereotactic biopsy (n=9) of their intracranial lesion which confirmed their diagnosis on histopathology. 31 patients (66%) underwent whole-brain irradiation; 22 patients had adjuvant whole-brain radiotherapy post-surgical resection of their lesion and 9 patients had whole-brain radiotherapy as their primary treatment. Three patients had stereotactic radiosurgery (SRS) to their lesion; 1 in conjunction with whole-brain radiotherapy post-surgical resection and 2 patients had SRS post-surgical resection of the main symptomatic lesion. 6 patients had trimodality treatment with surgical resection, whole-brain radiotherapy and ADT [10,13,18-19,24-26]. One patient had surgical resection of the dominant metastasis, SRS to another metastasis and also received concurrent androgen deprivation therapy (our case).
Surgical resection with adjuvant whole-brain radiotherapy has been the Gold standard for treating solitary metastasis in the brain. This combined strategy has been evaluated in randomised controlled trials to significantly prolong survival, alleviate neurological symptoms and reduce the risk of recurrence when compared with surgical resection or whole-brain radiotherapy alone [35].
ADT have also been found to bring both symptomatic and radiological improvement [36-37] leading not only to an increase in the overall survival time but also an improved quality of life and is thus used in both the curative and palliative settings in patients with prostate adenocarcinoma.
Prognosis
For the 41 patients who had documented survival times from the initial detection of their brain metastases, the mean survival time was 13.7 months (range, 3 days to 7 years). The patient who had the longest survival time of 7 years in our series had a solitary metastasis in the cerebellum that was resected en bloc and also underwent a bilateral orchiectomy. Previous case series have reported a median duration from diagnosis to death of between 3.5 to 31 months [1,38-39]. Patients without systemic disease were less likely to have brain lesions [4]. Patients with brain metastasis as the sole site of extra-prostatic disease had a mean overall survival time of 24.6 months (range, 1month to 7 years) compared to 13.4 months (range, 3 days to 5 years) in patients with systemic disease.
We postulate that advances in imaging such as Ga68 PSMA PET/CT, triple dose gadolinium and 3.0 Tesla (T) MRIs have led to earlier detection of metastases in PCa patients which allow the patient to be treated earlier thus decreasing the potential for further extraprostatic spread and the increased incidence of patients presenting with brain metastases as the sole site of disease from their primary PCa.
Solitary brain metastasis has better prognosis than patients with multiple brain metastases. The overall survival time for patients with a solitary brain metastasis was 14.3 compared to 7.2 months (range, 3 days to 29 months) for patients with multiple metastases. Our patient had the longest survival time for a patient with multiple brain metastases of three years likely due to low volume disease in the brain post-surgical resection and exceptional response to ADT at the extracranial metastatic sites.
The mean overall survival time for patients who had surgical resection was 20.3months (range, 3 weeks to 7 years). The mean overall survival time for patients who had stereotactic biopsies was considerably shorter at 6.25 months (range, 1month to 2years). This latter cohort of patients either had disease in the brainstem that was unresectable [8,17,21] or had multiple cerebral metastases [13,16,28].
Prior to the introduction of docetaxel in 2002, the incidence of prostatic brain metastasis from 1994 to 2002 was 0.8%. In the post-docetaxel era (2002-2011), this incidence had increased to 2.8%. This represents a 239% increase in the frequency of brain metastases from PCa between the two observation periods [40]. As the appearance of parenchymal metastases usually occurs in the late phase of the disease process it appears that the increase in frequency may actually reflect a gain in overall survival. In general patients are living longer with brain metastases in the context of PCa due to advances in imaging ability, systemic treatment and increased surveillance.
Pathogenesis
PCa rarely metastasizes to the brain with the incidence in large case series ranging from 0.63 to 1.1% [2,41] which suggests that the brain parenchyma is resistant to the establishment of metastatic foci by prostate carcinoma cells.
Currently the pathogenesis of cerebral metastases from PCa is unknown however in summary there are two main mechanisms postulated;
a) Single step spread via Batson’s paravertebral venous plexus draining the prostate. Low pressure in the large venous plexus allowing Valsalva maneuver to generate enough pressure to reverse blood flow from the IVC to the venous plexus, avoiding the lung and reaching the CNS. This mechanism however does not explain the absence of vertebral metastases and
b) Multi step haematogenous spread where secondary seeding of tumour cells to the brain occur from a primary metastatic focus involving the lungs or bones with brain metastasis usually a late event in the course of prostate cancer.
Haematogenous metastases have a special predilection for arterial border zones and the junction between the cortex and subcortical white matter. PSA is a sensitive indicator of the presence of disease however serum levels of PSA did not correlate to the development of brain metastases in our cases which is consistent with what is found in other case series [11,22].
Conclusion
Intraparenchymal spread of prostate cancer should be considered in men over the age of 60 years as a treatable cause of gradual neurological deterioration especially if a cranial malignancy or hyperostosis is found. The incidence of intraparenchymal brain metastasis is only expected to increase due to the longer life expectancy of patients with prostate adenocarcinoma with novel therapies. Patients undergoing Ga68-PSMA PET/CT for staging of PCa or when there is a PSA rise should be scanned from vertex to mid-thigh as albeit rare, prostate cancer brain metastases is a not to be missed differential in this particular group of patients.
Consent
The patients provided informed consent to the publication of their data, de-identified PET and MRI scans. No ethics approval through an institutional committee on human research was required.
References
- Tremont-Lukats IW, Bobustuc G, Lagos GK, Lolas K, Kyritsis AP, et al. (2003) Brain metastasis from prostate carcinoma: The M. D. Anderson Cancer Center experience. Cancer 98: 363-368.
- McCutcheon IE, Eng DY, Logothetis CJ (1999) Brain metastasis from prostate carcinoma: antemortem recognition and outcome after treatment. Cancer 86(11): 2301-2311.
- Catane R, Kaufman J, West C, Merrin C, Tsukada Y, et al. (1976) Brain metastasis from prostatic carcinoma. Cancer 38(6): 2583-2587.
- Hatzoglou V, Patel GV, Morris MJ, Curtis K, Zhang Z, et al. (2014) Brain metastases from prostate cancer: an 11-year analysis in the MRI era with emphasis on imaging characteristics, incidence, and prognosis. J Neuroimaging 24(2): 161-166.
- Nayak L, Abrey LE, Iwamoto FM (2009) Intracranial dural metastases. Cancer 115(9): 1947-1953.
- Lath CO, Khanna PC, Gadewar S, Patkar DP (2005) Intracranial metastasis from prostatic adenocarcinoma simulating a meningioma. Australas Radiol 49(6): 497-500.
- Kasabian NG, Previte SR, Kaloustian HD, Ganem EJ (1992) Adenocarcinoma of the prostate presenting initially as an intracerebral tumor. Cancer 70(8): 2149-2151
- Gupta A, Baidas S, Cumberlin RK (1994) Brain stem metastasis as the only site of spread in prostate carcinoma. A case report. Cancer 74(9): 2516-2519.
- Sutton MA, Watkins HL, Green LK, Kadmon D (1996) Intracranial metastases as the first manifestation of prostate cancer. Urology 48(5): 789-793.
- Salvati M, Frati A, Russo N, Brogna C, Piccirilli M, et al. (2005) Brain metastasis from prostate cancer. Report of 13 cases and critical analysis of the literature. J Exp Clin Cancer Res 24(2): 203-207.
- Behrens B, Husain MM, Schmidley JW (2001) Cystic solitary intracerebral metastasis from prostate adenocarcinoma. Neuroradiology 43: 162-164.
- Sweets T, Bracken RB, Geisler EJ, Warnick R (2009) Intracranial treatment for solitary prostatic adenocarcinoma brain metastasis is curative. Urology 73(3): 681e7-681e9.
- Gzell CE, Kench JG, Stockler MR, Hruby G (2013) Biopsy-proven brain metastases from prostate cancer: a series of four cases with review of the literature. Int Urol Nephrol 45(3): 735-742.
- Craig J, Woulfe J, Sinclair J, Malone S (2015) Isolated brain metastases as first site of recurrence in prostate cancer: case report and review of the literature. Curr Oncol 22(6): e493-e497.
- Mithal P, Gong Y, Sirkis H, Aronowitz (2016) A brain lesion as the sole metastasis of prostate cancer. Journal of Clinical Urology 9(5): 348-350.
- Chang DS, Hwang SL, Howng SL, Chai CY (1998) Prostatic carcinoma with brain metastasis presenting as a tumor hemorrhage. Kaohsiung J Med Sci 14: 247-250.
- Salvati M, Cervoni L (1997) Carcinoma of the prostate: brain stem metastasis as the only site of spread. Tumori 83(4): 776-778.
- Baumann MA, Holoye PY, Choi H (1984) Adenocarcinoma of prostate presenting as brain metastasis. Cancer 54(8): 1723-1725.
- Bland LI, Welch WC, Okawara SH (1992) Large cystic intraparenchymal brain metastasis from prostate cancer. Neuroradiology 34(1): 70-72.
- Kunkler RB, Cooksey G, Millac P (1993) Carcinoma of the prostate presenting with a cerebral metastasis. Br J Urol 71(1): 103-104.
- Zhang X, Tsukuda F, Yamamoto N, Takenaka (1997) Brain metastasis from prostate cancer: a case report. Int J Urol 4: 519-521.
- Fervenza FC, Wolanskyj AP, Eklund HE, Richardson RL (2000) Brain metastasis: an unusual complication from prostatic adenocarcinoma. Mayo Clin Proc 75(1): 79-82.
- Tsai V, Kim S, Clatterbuck RE, Ewend MG, Olivi A (2001) Cystic prostate metastases to the brain parenchyma: report of two cases and review of the literature. J Neurooncol 51(2): 167-173.
- Castro Gomez JE, Anido Herranz U, Carballo Castro A, Gómez Caamaóo A, León Mateos LA et al. (2009) Brain metastases from prostate adenocarcinoma. Clin Transl Oncol 11(1): 63-64.
- Grenader T, Shavit L, Lossos A, Pizov G, Wygoda M (2007) Brain metastases: a rare initial presentation of prostate cancer. Int Urol Nephrol 39(2): 537-539.
- Lynes WL, Bostwick DG, Freiha FS, Stamey TA (1986) Parenchymal brain metastases from adenocarcinoma of prostate. Urology 28(4): 280-287.
- Mackay DD, Diamond EL, Klein JP (2012) Widespread intracerebral metastases from prostate adenocarcinoma. Pract Neurol 12(4): 257- 258.
- Taddei G, Marzi S, Coletti G, De Paulis D, Ricci A, et al. (2012) Brain Metastasis From Prostate Adenocarcinoma: Case Report and Review of Literature. World J Oncol 3(2): 83-86.
- Lam A, Gan PY (2017) Metastatic Prostate Adenocarcinoma to the Brain: Case Reports and Literature Review. J Neurol Surg Rep 78(1): e62-e65.
- Chitale S, Webb R, MacIver D, Curtis K, Zhang Z, et al. (2008) Multifocal symptomatic intracerebral metastases as the first manifestation of prostatic carcinoma: a report and literature review. Int Urol Nephrol 40(3): 675-678.
- Tagle P, Villanueva P, Torrealba G, Huete I (2002) Intracranial metastasis or meningioma? An uncommon clinical diagnostic dilemma. Surgical neurology 58(3-4): 241-245.
- Mitchell RA, Dimou J, Tsui A, Kavar B (2011) Metastatic prostate adenocarcinoma invading an atypical meningioma. Journal of clinical neuroscience : official journal of the Neurosurgical Society of Australasia 18(12): 1723-1725.
- Demierre B, Berney J (1988) Intracranial metastasis of prostatic adenocarcinoma. Journal of neurosurgery 69: 644.
- Chung TS, Thannikkary C (1986) Carcinoma of the prostate with brain metastasis. J Surg Oncol 33(2): 103-105.
- Kocher M, Soffietti R, Abacioglu U, Villà S, Fauchon F, et al. (2011) Adjuvant whole-brain radiotherapy versus observation after radiosurgery or surgical resection of one to three cerebral metastases: results of the EORTC 22952-26001 study. J Clin Oncol 29(2): 134-141.
- Ameur A, Touiti D, el Mostarchid B, el Alami M, Jira H, et al. (2001) Brain metastasis of prostatic cancer: regression under hormonal treatment. Prog Urol 11(6): 1298-1301.
- Kohri K, Yamate T, Tsujihashi H, Nakao Y, Katayama Y, et al. (1991) Effect of endocrine therapy on a brain metastatic lesion of prostatic carcinoma. Urol Int 47(2): 100-102.
- Kim SH, Chao ST, Toms SA, Vogelbaum MA, Barnett GH, et al. (2008) Stereotactic radiosurgical treatment of parenchymal brain metastases from prostate adenocarcinoma. Surgical neurology 69(6): 641-646.
- McDermott RS, Anderson PR, Greenberg RE, Milestone BN, Hudes GR (2004) Cranial nerve deficits in patients with metastatic prostate carcinoma: clinical features and treatment outcomes. Cancer 101(7): 1639-1643.
- Caffo O, Veccia A, Fellin G, Mussari S, Russo L, et al. (2013) Frequency of brain metastases from prostate cancer: an 18-year single-institution experience. J Neurooncol 111(2): 163-167.
- Saitoh H, Hida M, Shimbo T, Nakamura K, Yamagata J, et al. (1984) Metastatic patterns of prostatic cancer. Correlation between sites and number of organs involved. Cancer 54(12): 3078-3084.