Pesticide Residues in Fruits and Vegetables from Farmers’ Markets
Beatrice N Dingha* and Louis EN Jackai
Department of Natural Resources and Environmental Design, USA
Submission: November 29, 2018; Published: December 20, 2018
*Corresponding author: Beatrice N Dingha, Department of Natural Resources and Environmental Design, USA.
How to cite this article: Beatrice N D, Louis E J. Pesticide Residues in Fruits and Vegetables from Farmers’ Markets. Int J Environ Sci Nat Res. 2018; 16(1): 555927. DOI: 10.19080/IJESNR.2018.16.555927.
Abstract
Pesticides are important in crop production worldwide. Many commonly grown fruits and vegetables require pesticide protection. The consequences of using pesticides for food production can be far-reaching, and farm produce may contain pesticide residues that pose serious health and environmental risks. In this paper, we review the Electrical Penetration Graph (EPG) and explore the potential for its use in food safety. Several pesticide residues were found on produce obtained from farmers’ markets. The results indicate that the EPG could be a useful tool for detecting pesticide residues. In conclusion, we emphasize the need for case-driven Integrated Pest Management (IPM) approaches that produce safe food and high yield.
Keywords: Food safety; Electrical penetration graph (EPG); Insecticide; Pesticide Residue; Integrated pest management (IPM)
Introduction
Food safety concerns continue to be important to agriculture in many parts of the world. Many farmers use chemical pesticides to control insect pests, weeds and diseases to obtain high yields, quality and blemish free appearance to meet standards imposed by consumers. Residues from pesticides may still be present on produce and may have long-term adverse effects on human health [1]. It is therefore not surprising that over 90 percent of consumers are concerned about pesticide and fertilizer residues in food and food products [2,3]. In the USA, farmers’ markets represent an attractive outlet where consumers purchase fresh produce directly from the producer. The number of farmers’ market has grown significantly, with a 79% increase from 1994 to 2002 [4]. A major reason for this trend is the fact that consumers believe produce from farmers’ markets are fresher, pesticide-free and of higher quality [5,6] and there is little data available to support all of these claims. Pesticide detection methods are generally expensive, time consuming, and not suitable for field measurements. Simpler, yet innovative devices that are easy to use and capable of providing reliable information are needed.
The EPG was originally developed by McLean and Kinsey [7-9] with a number of recent enhancements [10,11] used to analyze, quantify, and compare feeding behaviors of hemipteran insects. It works when a test insect is attached to the monitor through a gold wire glued to its dorsum with conductive silver paint. Low-voltage current is introduced into the test plant via an electrode to the soil or plant’s tissues. When the insect’s stylets penetrate the electrified plant, the circuit is closed. Changes in voltage across the stylets are amplified and recorded as a time-varying waveform [12,13] and can be correlated with many types of feeding and non-feeding behaviors. The EPG has been used to investigate host plant resistance [14,15] transmission of plant viruses [16,17] action of insecticides [18-24] etc. In the area of food safety, EPG technology can be used to study the response of insect pests on fresh produce with the goal of determining the least amount of pesticide residue that will elicit feeding without killing the test insect. Waveforms obtained will be compared with those obtained with untreated produce and thus develop standard waveforms that would serve as the litmus test for detecting pesticide residue on produce.
In one study [25] the southern green stink bug Nezara viridula L was exposed to fresh fruits and vegetables from farmers’ markets or roadside stands situated along the I-85 and I-40 transect at the following locations: Auburn, Tuskegee and Montgomery (AL), Gaffney (SC) and Greensboro (NC), and pesticide residue of these produce analyzed. A dose-response curve for the neonicotinoid Imidacloprid (Provado®, Bayer Crop Science, NC) was obtained by exposing stink bugs to a range of doses (0.08-0.75mg/ml). The feeding behavior of adult stink bugs on cowpea pods treated with 0.08 and 0.75mg/ml Imidacloprid and untreated pods (control) was evaluated using the EPG.
Discussion
From the dose response curve, the LD50 for Imidacloprid at 24, 48 and 72h was 0.1195mg/ml, 0.0580mg/ml and 0.0168mg/ml respectively. Mortality increased with increasing dose to 100% after 72h in all doses tested. From the bioassay experiment no mortality was observed on tomato, squash, green beans and okra from all locations after 24 and 48h. After 24h, mortality for Auburn cucumbers was (11%); Montgomery and Auburn peaches (22%); and 33% for Tuskegee peaches. After 48h, mortality on peaches from Tuskegee was (67%); Auburn and Montgomery (56%); Gaffney (22%); Greensboro (11%) and 33% for apples from Greensboro. Chlorpyrifos, Thiamethoxam, Endosulfan, Bifenthrin and Esfenvalerate residues were found on produce; however, peaches had Chlorpyrifos residue (0.074ppm) above the EPA tolerance level (0.05ppm). EPG waveform patterns indicated that, when at rest, (i.e. when the insect was not moving), a straight line was recorded at the base. Stink bugs did not feed immediately when placed on treated cowpea pods; they crawled and pulled the gold wire and made repeated attempts at penetrating pod wall with their stylet for a few minutes (Figure 1a). After stylet penetrated the pod wall, a stable voltage output with little variation in amplitude was observed (salivation). After this, either the stylet was withdrawn, or variable spiky waveform patterns (Figures 1b-1d) which varied over time were formed. The stink bug fed on untreated pea and exhibited feeding waveforms (Figures 1b-1d). However, when transferred to pod treated with 0.08mg/ml and 0.77mg/ ml feeding waveform patterns were disrupted and insect became paralyzed. EPGs with feeding disruption and distorted patterns have been reported by other workers [18,20-24,26].
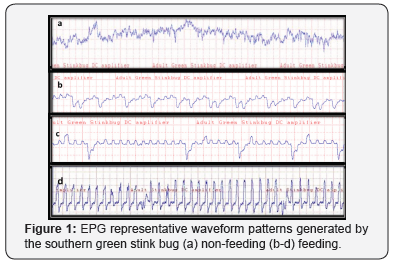
Conclusion
These results indicate that fruits and vegetables sold in farmers’ markets used in our study contained insecticide residues at levels that would kill stink bugs. These levels cannot be considered safe for human consumption. EPG can thus be used as a reliable tool to detect presence of pesticide residue levels that are dangerous to humans in a fairly short amount of time. Further studies using minimal doses that would allow feeding but not paralyze or kill the insect are needed.
References
- Bolognesi C, Morasso G (2000) Genotoxicity of pesticides: potential risk for consumers. Food Sci Technol 11: 18-27.
- Collins JK, Cuperus GW, Cartwright B, Stark J, Ebro LL (1992) Consumer attitudes on pesticide treatment histories of fresh produce. J Sustain Agric 3(1): 81-98.
- Anderson MD, Hollingsworth CS, van Zee V, Coli WM, Rhodes M, et al. (1996) Consumer response to integrated pest management and certification. Agric Ecosyst Environ 60(2-3): 97-106.
- (2002) Agricultural Marketing Service. AMS Farmers’ Market Facts.
- Andreatta S, Wickliffe W (2002) Managing Farmer and Consumer Expectations: A Study of a North Carolina Farmers’ Market. Hum Organ 61(2): 167-176.
- Wolf MM, Spittler A, Ahern J (2005) A Profile of Farmers’ Market Consumers and the Perceived Advantages of Produce Sold at Farmers’ Markets. J Food Distri Res 36(1): 192-201.
- McLean DL, Kinsey MG (1964) A technique for electronically recording aphid feeding and salivation. Nature 202: 1358-1359.
- Mc Lean DL, Kinsey MG (1967) Probing behavior of the pea aphid, Acyrthosiphon pisum. I. Definitive correlation of electronically recorded waveforms with aphid probing activities. Annals Entomol Soc Am 60(2): 400-405
- Mc Lean DL Weigt WA (1968) An electronic measuring system to record aphid salivation and ingestion. Ann Entomol Soc Am 61: 180-185.
- Tjallingii WF (1978) Electronic recording of penetration behaviour by aphids. Entomol Exp Appl 24: 521-530.
- Backus EA, Bennett WH (2009) The AC-DC Correlation Monitor: New EPG design with flexible input resistors to detect both R and emf components for any piercing-sucking hemipteran. J Insect Physiol 55(10): 869-884.
- Tjallingii WF, Hogen Esch T (1993) Fine structure of aphid stylet routes in plant tissues in correlation with EPG signals. Physiol Entomol 18: 317-328.
- Backus EA, Lin PA, Chang CJ, Shih HT (2016) Electropenetrography: A new diagnostic technology for study of feeding behavior of piercing- sucking insects. J Taiwan Agric Res 65(3): 219-237.
- Lightle DM, Dossett M, Backus EA, Lee JC (2012) Location of the mechanism of resistance to Amphorophora agathonica (Hemiptera: Aphididae) in red raspberry. J Econ Entomol 105(4): 1465-1470.
- Garzo E, Moreno A, Hernando S, Mariño V, Torne M, et al. (2016) Electrical penetration graph technique as a tool to monitor the early stages of aphid resistance to insecticides. Pest Manag Sci 72: 707-718.
- Fereres A, Moreno A (2009) Behavioural aspects influencing plant virus transmission by homopteran insects. Virus Res 141(2): 158-168.
- Moreno A, Tjallingii WF, Fernandez-Mata G, Fereres A (2012) Differences in the mechanism of inoculation between a semi-persistent and a non-persistent aphid-transmitted plant virus. J gen Virol 93: 662- 667.
- Harrewijn P, Kayser H, Pymetrozine (1997) Pymetrozine, a fast-acting and selective inhibitor of aphid feeding. In situ studies with electronic monitoring of feeding behaviour. Pest Sci 49: 130-140.
- Costa RR, Moraes JC DaCosta RR (2011) Feeding behaviour of the green- bug Schizaphis graminum on wheat plants treated with imidacloprid and/or silicon. J Appl Entom 135: 115-120.
- He Y, Chen L, Chen J, Zhang J, Chen L et al. (2011) Electrical penetration graph evidence that pymetrozine toxicity to the rice brown planthopper is by inhibition of phloem feeding. Pest Manag Sci 67(4): 483-491.
- Butler CD, Walker GP, Trumble JT (2012) Feeding disruption of potato psyllid, Bactericera cockerelli, by imidacloprid as measured by electrical penetration graphs. Entomol Exp Applic 142: 247-257.
- Miao J, Du ZB, Wu YQ, Gong ZJ, Jiang YL, et al. (2013) Sub-lethal effects of four neonicotinoid seed treatments on the demography and feeding behaviour of the wheat aphid Sitobion avenae. Pest Manag Sci 70(1): 55-59.
- Jacobson AL, Kennedy GG (2013) Electrical penetration graph studies to investigate the effects of cyantraniliprole on feeding behaviour of Myzus persicae (Hemiptera: Aphididae) on Capsicum annuum. Pest Manag Sci 70(5): 836-840.
- Civolani S, Cassanelli S, Chicca M, Rison JL, Bassi A, et al. (2014) An EPG study of the probing behaviour of adult Bemisia tabaci biotype Q (Hemiptera: Aleyrodidae) following exposure to cyantraniliprole. J Econ Entomol 107(3): 910-919.
- Dingha BN, Jackai LEN, Shirley VB, Sintim OH (2011) Vegetables and Fruits from Farmers’ Markets Are Not Free of Pesticide Residues. Association of Research Directors (ARD) 16th Biennial Research Symposium. April 9-13, 2011, Georgia.
- Kang MA, Seo MJ, Hwang IC, Jang C, Park HJ, et al. (2012) Insecticidal activity and feeding behavior of the green peach aphid, Myzus persicae, after treatment with nano types of pyrifluquinazon. J Asia Pac Entomol 15(4): 533-541.






























