Acute Hematocrit Change as an Indicator of Intracranial Hemorrhage on Neonatal Head Ultrasound
Matthew O Thompson1*, Atalie C Thompson2, Nathan Hull3, Joseph Davis4 and Gary Schooler5
1Department of Radiology, Atrium Wake Forest Baptist Health, USA
2Department of Ophthalmology, Atrium Wake Forest Baptist Health, USA
3Department of Radiology, Mayo Clinic, USA
4Department of Radiology, Riley Children’s Hospital, USA
5Department of Radiology, Children’s Medical Center Dallas, USA
Submission: September 24, 2022; Published: October 26, 2022
*Corresponding author: Matthew Olen Thompson, Wake Forest Baptist Hospital, Medical Center Drive, Winston-Salem, NC 27157, USA
How to cite this article: Matthew O T, Atalie C T, Nathan H, Joseph D, Gary S. K, Dimitris A, Marion F, Joao L. Acute Hematocrit Change as an Indicator of Intracranial Hemorrhage on Neonatal Head Ultrasound. Glob J Intellect Dev Disabil. 2022; 10(5): 555797. DOI:10.19080/GJIDD.2022.10.555797
Abstract
Purpose: To determine whether an indication related to a decrease in hematocrit is predictive of an intracranial hemorrhage (ICH) on neonatal head ultrasound (HUS).
Methods: This was a single center retrospective study of 215 neonatal head ultrasounds (HUS). The association between an ICH on HUS and indication related to hematocrit, a change in hematocrit in the 48 hours prior to HUS, gestational age, number of days since birth at time of HUS, designation of prematurity, or abnormal neurologic exam were examined.
Results: Thirty-seven (37/215, 17.2%) neonates had an ICH on their HUS. There was no significant association between studies that were ordered with an indication related to hematocrit (or similar indication) and the likelihood of having an ICH on HUS (OR 0.92, p=0.82). Lower gestational age, prematurity, and lower birthweight were associated with an ICH on HUS (p<0.05), but only gestational age remained statistically significant in a multivariable model (p<0.005).
Conclusion: Neither a decrease in hematocrit in the preceding 48 hours, nor an indication related to a decrease in hematocrit, was predictive of an ICH on HUS. Rather, prematurity, lower birth weight, early gestational age, and younger age were significantly associated with the presence of an ICH on neonatal HUS.
Keywords: Hemorrhage; Neonate; Ultrasound; Hematocrit; Prematurity
Introduction
Intracranial hemorrhage (ICH) is a devastating cause of neonatal morbidity and mortality that can result in substantial adverse outcomes during an important window for neurodevelopment. Prior investigations have shown that ICH is typically associated with prematurity (defined as a neonate born prior to 37 weeks), low birth weight, and anticoagulation [1,2]. Subdural and intraparenchymal hemorrhages are more commonly found in term infants, primarily related to birth trauma, while the most common type of intracranial hemorrhage in premature infants are germinal matrix hemorrhages (GMH) [3-5]. GMHs occur in preterm infants due to the thin-walled vessels found at the germinal matrix which are more susceptible to hemorrhage in the setting of hemodynamic changes [6-10]. The clinical data has shown that the majority of GMHs are identified during the first 4 days of life, with 40% detected within the first 5 hours of birth [2, 11-12]. GMHs are graded based on their severity as described in a landmark paper in 1978 (see Figure 1) [13]. Although most grade 1 and 2 GMHs are managed with observation, higher grade hemorrhages are associated with substantial neurodevelopmental sequelae, significant disability, and poor survival rates [14,15].
Head ultrasound (HUS) is the most common first line technique for imaging neonates suspected of having an ICH [16]. HUS affords real-time assessment of the brain using the fontanelles as a sonographic window. However, it is not always clear which neonates warrant an evaluation with HUS for ICH. We have observed that HUS are often ordered to look for an ICH when a change in hematocrit or hemoglobin has occurred on routine labs. But to our knowledge, no study to date has evaluated whether HUS ordered for indications related solely to a decrease in hematocrit are more likely to reveal an ICH than those ordered for alternative indications. The purpose of this study was to determine whether a decrease in hematocrit or a similar HUS indication (e.g., decrease in hematocrit or hemoglobin, bleeding, anemia, etc.) was predictive of an ICH on neonatal HUS.
Materials and Methods
This was a retrospective study of neonates that underwent HUS from 2009-2018. This study was conducted in accordance with the Declaration of Helsinki with approval by our Institutional Review Board. A waiver of informed consent was granted due to the retrospective nature of this work.
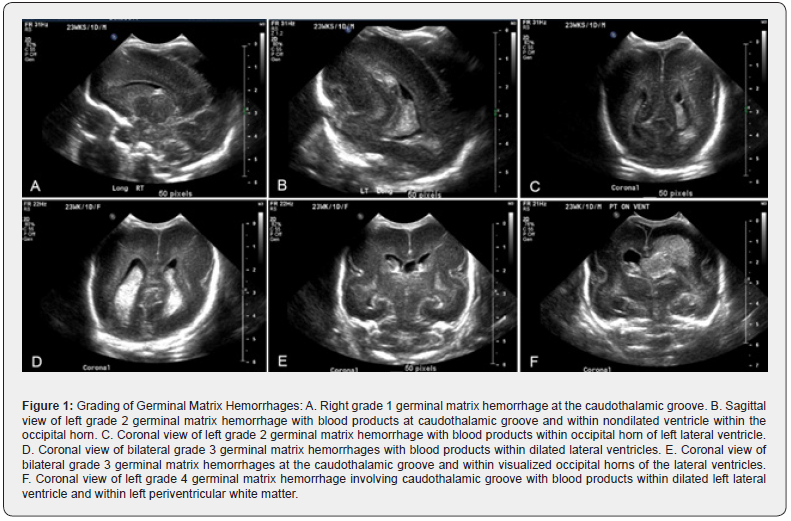
The PACS (Picture Archive and Communication System) at our institution (Centricity, General Electric) was searched using two search filters: head ultrasound, and the year (2009-2018). The indications for the head ultrasound examinations (HUS) were consecutively reviewed for any terms that were related to a decrease in hematocrit (or similar indication), including the following: “low”, “decreased”, “drop in”, “fall in”, or “dropping” hematocrit or hemoglobin, “hemoglobin drop”, “hematocrit drop”, “HH drop”, “bleeding”, or “anemia.” This resulted in approximately 116 patients with an indication for HUS that was related to a decrease in hematocrit or hemoglobin. A random sample of 101 patients from the same time period with indications unrelated to hematocrit, hemoglobin, bleeding, or anemia were selected for inclusion as a comparison group. The images of all HUS were initially reviewed, and a radiology report was generated by a board-certified radiologist to determine whether or not an ICH had occurred. The reports and images were then later reviewed by another board-certified radiologist for agreement of imaging findings. If there was concurrence of HUS findings, the examination was included in the study; if there was disagreement on HUS findings, the examination was excluded from the study. Only two HUS examinations were excluded from this study (2/217 = 0.9%). Those patients with an ICH on HUS (37/215 =17.2%) were then further classified into one of four groups based on known GMH grading (see Figure 1) [13].
The electronic medical records of the 215 patients included in this study were then reviewed and the following clinical data were collected: the change in hematocrit in the 48 hours prior to and at the time of HUS, whether or not there was a decrease in the hematocrit lab value in the 48 hours prior to HUS, the gestational age (GA) in weeks, whether the patient was classified as premature (< 37 weeks) or not (≥37 weeks), the birth weight in kilograms, the number of days since birth, and whether the result of a neurologic examination was abnormal or normal when it was performed. Premature neonates were further subclassified as term (>37 weeks), moderately preterm (32-37 weeks), very preterm (28-32 weeks), and extremely preterm (<28 weeks).
Statistical Analysis
The association between the presence or absence of an ICH on the HUS and each of the clinical and demographic independent variables was then analyzed using separate univariable logistic regression models to estimate the odds ratios (OR) and 95% confidence intervals. A multivariable model was constructed to evaluate the association between the likelihood of an ICH on HUS and an indication related to a decrease in hematocrit (or similar indication) as well as any clinical or demographic factors that had been statistically significant in univariable analysis.
Separate linear regression models were used to assess whether the magnitude of the change in hematocrit was associated with the category of prematurity (i.e., moderately or very or extremely preterm vs. term) or the grade of ICH on HUS (i.e., Grade 4 or 3 or 2 vs. 1). Similarly, a logistic regression model was constructed to assess whether the presence or absence of a measured drop in the hematocrit lab value was associated with the category of prematurity. A p-value of <0.05 was considered statistically significant. All statistical analyses were completed in Stata (version 16.0, College Station, TX).
Results
A total of 215 patients were included in our study, 112 male (52%) and 103 (48%) female. A total of 37/215 (17.2%) of patients had an ICH on HUS. The mean gestational age (GA) at birth was 32.6±5.4 weeks with a median of 34 weeks (Interquartile range 27, 37). The mean age at the time of HUS was 21.3±41.6 days with the median at 7 days (Interquartile range 2, 18). A majority of patients included in the study were premature, or < 37 weeks old at the time of birth) (145/192, 75.5%), and the average birthweight was 2.05±1.39 kg with a median of 1.9 kg (Interquartile range 0.9, 2.9) (Table 1).

There were 114/215 (53%) patients with an indication related to a decrease in hematocrit (or similar indication) and 101 patients with indication unrelated to hematocrit drop (Figure 2). Of those with a decrease in hematocrit, 19/114 (17%) had an intracranial hemorrhage and 95/114 (83%) did not. Most of the patients with an ICH were premature (32/36, 89%), and most patients with an ICH were ≤ 7 days old at the time of HUS (28/37, 75.7%).
Table 2 shows the association of each of the clinical or demographic factors with the presence or absence of an ICH on HUS. Studies that were ordered with an indication related to a decrease in hematocrit (or similar indication) were no more likely to have an ICH on HUS than those ordered for an alternative indication (OR 0.92, p=0.82). Also, there was no significant association between the likelihood of having an ICH on HUS and a decrease in the measured hematocrit lab value (OR 0.74, p=0.44), or the magnitude of the change in the hematocrit lab value (OR 1.0, p=0.90) during the 48 hours preceding HUS. The actual hematocrit lab value at the time of HUS also did not significantly differ between those with an ICH and without an ICH, suggesting no significant association between anemia and ICH (OR 0.97, p=0.14).
We also evaluated whether an abnormal neurologic examination was associated with an ICH on HUS and found that 12 patients had missing data, 25 patients were sedated at the time of HUS, 22 had abnormal neurologic exams, and 156 had normal neurologic exams. Thus, 6/37 (16.2%) of patients with ICH had an abnormal neurologic examination, and 16/178 (9.0%) of patients without an ICH had an abnormal neurologic examination. The odds of having an abnormal neurologic examination was higher in the group with an ICH compared to those without an ICH (OR 1.71, p=0.30), but the association was not statistically significant. Reasons for an abnormal neurologic examination included “hypotonia,” “decreased tone and flexation,” “hyperactive reflexes,” “decreased tone and reflexes,” “minimal response to stimulation,” “no spontaneous movement,” “no withdrawal to painful stimuli,” and “facial droop.”
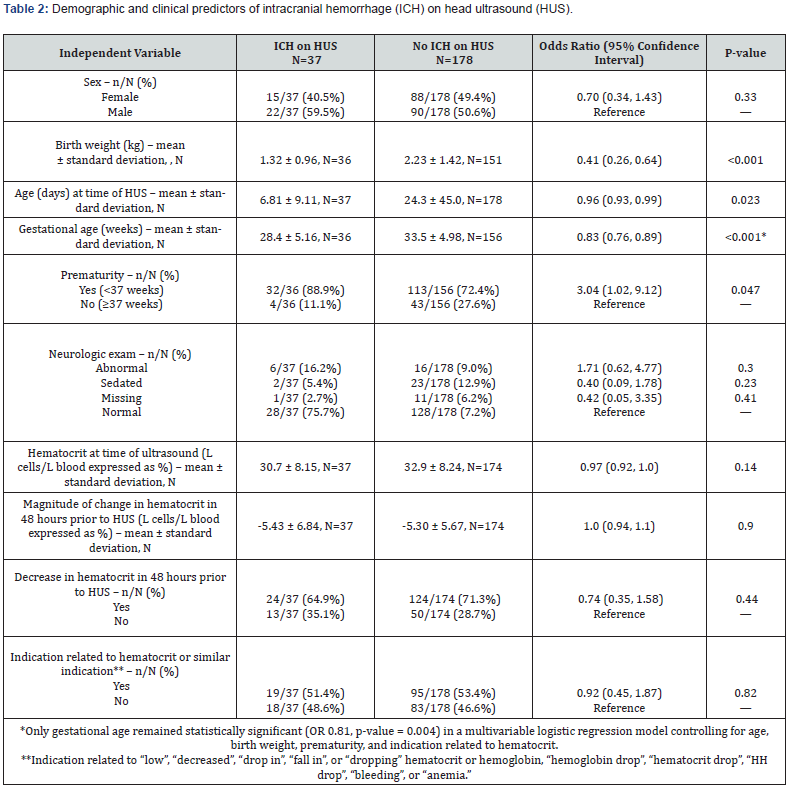
In univariable logistic regression analyses, prematurity (OR 3.04, p=0.04), lower gestational age in weeks (OR 0.83, p<0.001), younger age in days at the time of HUS (OR 0.96, p=0.02), and lower birth weight (OR 0.41, p<0.001) were each significantly associated with an ICH on HUS. However, only gestational age remained significantly predictive of ICH (p<0.005) in a multivariable logistic regression model that included indication related to hematocrit (or similar indication), and each of the factors that had been statistically significant (p<0.05) in univariable analysis (i.e., prematurity, younger age, and lower birth weight).
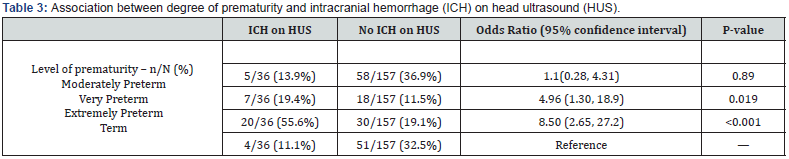
We next examined whether the level of prematurity was associated with a greater likelihood of having an ICH on HUS (Table 3). Patients that were very preterm (OR 4.96, p=0.02) or extremely preterm (OR 8.50, p<0.001) were at a significantly greater risk than those that were term. Moderately preterm infants did not have an increased risk of ICH (OR 1.1, p=0.89). However, there was no association between the level of prematurity and a measured drop in the hematocrit lab value (all p>0.60), nor the magnitude of that change in hematocrit in the 48 hours prior to HUS (all p>0.70).
Finally, we evaluated whether the grade of hemorrhage on HUS was related to a drop in hematocrit (all p>0.20) or the magnitude of the change in hematocrit during the 48 hours preceding the HUS (all p>0.40), and there was no significant association or trend to suggest any relationship between the change in the hematocrit lab value and the grade of ICH.
Discussion
Our study demonstrates that neither a decrease in the hematocrit lab value during the 48 hours prior to HUS nor an indication for HUS related to a decrease in hematocrit (or similar indication) is predictive of having an ICH on HUS. There was also no significant association between the change in hematocrit and the grade of ICH on HUS. Rather, younger age at the time of ultrasound, lower gestational age at birth, and prematurity were all associated with a greater likelihood of finding an ICH on HUS, regardless of whether the patient also had a decrease of hematocrit. Of these factors, lower gestational age was the strongest predictor of an ICH and was the only variable that remained significantly associated with ICH in a multivariable model. Infants with a higher degree of prematurity, such as very preterm or extremely preterm, were also at a substantially increased risk of having an ICH, but there was no relationship between the degree of prematurity and the change in hematocrit in the 48 hours prior to HUS. Thus, a decline in hematocrit alone on laboratory testing is unlikely to be explained by an ICH on HUS and may not be a sufficient indication for ordering this test. However, higher degrees of prematurity or lower gestational age are strong risk factors for an ICH and warrant thorough evaluation with HUS.
Head ultrasound is an inexpensive adjunct to MRI because of its wide availability, lack of side effects, and wealth of anatomical and functional information [17,18]. The threshold to screen neonates for abnormalities with HUS have been historically low since HUS is relatively inexpensive compared to brain MRI. In fact, in 2002 the American Academy of Neurology (AAN) and Child Neurology Society (CNS) recommended routine screening with HUS in all preterm neonates <30 weeks, and at 36- and 40-weeks gestational age, as well as routine Noncontrast CT in encephalopathic term infants with a history of birth trauma, low hematocrit, or coagulopathy [19]. However, this recommendation was retired in 2018 because the AAN and CNS did not update or reaffirm their recommendations. Thus, it may be time to reexamine whether such low thresholds for HUS examinations should be routinely recommended in most neonates [20].
Moreover, with rising healthcare costs, there is increasing concern regarding over-utilization of imaging studies in medicine. Since a hospital’s resources do not simply encompass the costs of imaging tests, but also the time and energy spent by experienced sonographers, support staff, and radiologists to acquire and interpret these tests, it is important that clinicians have clear guidelines regarding the appropriate indications for ordering radiologic tests for their patients. The results of our study suggest that ordering a HUS solely because the hematocrit has decreased or because there is a concern for new anemia may not be a costeffective use of hospital resources. We found that head ultrasounds ordered for an indication related to a decrease in hematocrit (or a similar indication) were no more likely to have an ICH on HUS than those ordered for other indications. Moreover, neonates that demonstrated a decrease in hematocrit in the 48 hours preceding the HUS were no more likely to have an ICH, nor were they at greater risk of having a higher-grade ICH, compared to those with a stable hematocrit.
On the other hand, our study confirmed several well-known risk factors for neonatal ICH, including lower gestational age, especially prematurity, younger age at the time of HUS, and low birthweight [21-23]. We found that gestational age was the most significant predictor of ICH, independent of the other factors in a multivariable model. There was a notable dose response with higher levels of prematurity, such as very and extremely preterm infants, showing increasing odds of having an ICH compared to those who were only moderately preterm or term. This result confirms the findings of recent studies investigating the incidence of GMH in premature neonates [2,3,6]. One such study evaluated a total of 101 neonates and found that those born at <30 weeks gestational age had a 47% chance of ICH, and those born weighing <1000 grams at birth had a 54% chance of ICH [24]. Even though we also found that neonates with lower gestational age (especially higher levels of prematurity) were more likely to have an ICH, we did not find that a change in hematocrit was associated with either the level of prematurity or the likelihood of an ICH. Thus, change in hematocrit neither confounds nor modifies the relationship between prematurity and ICH on HUS.
Our study also suggests that an abnormal neurologic examination may be associated with an increased likelihood of ICH, but the association was not very strong and did not reach statistical significance. Dubowitz et al. have previously shown that multiple neurologic signs can correlate with ICH [25]. Our study did not evaluate whether the many different types of neurologic examination findings correlate with the results of HUS because the neurologic examination findings were not consistently documented in the medical record, and a substantial proportion of patients were either sedated or on a ventilator at the time of HUS and thus lacked documentation of specific neurologic signs. Furthermore, since our sample size was small and our primary question was whether an indication related to hematocrit alone was associated with an ICH on HUS, our study was not powered to adequately study the predictive power of abnormal neurologic examinations for ICH.
Some organizations, such as the Choosing Wisely initiative and American College of Physicians, have attempted to identify imaging tests and procedures that are overused by medical professionals [20]. Moreover, the American College of Radiology (ACR) has developed appropriateness criteria for the use of several imaging examinations in an attempt to limit and properly direct the use of imaging resources [26]. Given the findings in our study, we suggest that a decline in hematocrit, hemoglobin or other laboratory indication of new anemia is unlikely to be predictive of an ICH and is not a sufficient indication for ordering a HUS in the absence of other well-known risk factors as described in this and several other studies [22,24].
This study has several limitations. The types and quality of the data available for review in the electronic medical record were limited by the retrospective nature of this study. Data regarding anticoagulation at the time of HUS was not consistently available and thus was not evaluated in this study. Although abnormal neurologic examination was associated with an ICH on HUS, missing data may have limited our power to detect a statistically significant p-value. Also, since we could not know the neurologic examination of patients that were sedated at the time of HUS, our analysis of this variable was limited. It can be difficult to compare the results of neurologic examinations among patients with ICH and those without ICH given the great variability in expertise and experience amongst neonatal care providers, and the level of expertise could not be ascertained from the medical record. Finally, though the included cases represent nearly a decade’s worth of data, the relatively small sample size of patients with an intracranial hemorrhage within a single institution are limiting factors. A larger sample of patients with ICH on HUS collected across multiple medical centers would allow greater generalizability as well as additional subgroup analyses.
Conclusion
In conclusion, this study demonstrates that a decrease in hematocrit (or similar indication) is not predictive of an ICH and should not be the sole indication for ordering a HUS to rule out ICH. When additional factors are present in the neonate, such as low gestational age, prematurity, lower birth weight, younger age at the time of HUS, or abnormal neurologic examination, then a HUS is more likely to detect an ICH. Infants who are very or extremely preterm appear to be at a substantially greater risk for disability resulting from intracranial hemorrhage than those who are moderately preterm or term, and thus a HUS should always be considered in these patients whether or not there is a clinically reported drop in hematocrit.
References
- Fleischmann AR, Clark SL, Oinuma M (2010) Rethinking the definition of “term pregnancy. Obstetrics and Gynecology 116(1): 136-139.
- Paneth N, Martin JP, Gardiner J (1993) Incidence and Timing of Germinal Matrix/Intraventricular Hemorrhage in Low-Birth Weight Infants. American Journal of Epidemiology 137(11): 1167-1176.
- Brouwer AJ, Floris Groenendaal, Corine Koopman, Rutger-Jan A Nievelstein, Sen K Han, et al. (2010) Intracranial hemorrhage in full-term newborns: a hospital-based cohort study. Neuroradiology 52(6): 567-576.
- Fumagalli M, Laura Bassi, Ida Sirgiovanni, Fabio Mosca, Andrea Sannia, et al. (2015) From germinal matrix to cerebellar haemorrhage. J Matern Fetal Neonatal Med 1: 2280-2285.
- Kriegstein AR, Noctor SC (2004) Patterns of neuronal migration in the embryonic cortex. Trends in Neurosciences 27(7): 392-399.
- Kadri H (2006) The incidence, timing, and predisposing factors of germinal matrix and intraventricular hemorrhage (GMH/IVH) in preterm neonates. Childs Nerv Syst 22(9): 1086-90.
- Alvarez MD, M Villamil, G Reyes (1994) Predictive factors in the genesis of intraventricular hemorrhage in premature infants. Puerto Rico Health Sciences Journal 13(4): 251-254.
- Talbert David (2016) Cerebral venous malformation as a cause of neonatal intra-ventricular hemorrhage and unexplained infant subdural hemorrhage. Anatomy and Physiology 6(2): 1-5.
- Praveen B (2014) Pathogenesis and prevention of intraventricular hemorrhage. Clinics in Perinatology 41(3): 47-67.
- Langley EA, Stephanie M Blake, Kristi L Coe (2022) Recent review of germinal matrix hemorrhage-intraventricular hemorrhage in preterm infants. Neonatal Netw 41(2): 100-106.
- Volpe JJ (2008) Intracranial hemorrhage: germinal matrix-intraventricular hemorrhage of the premature infant, in Neurology of the Newborn, (5th edn), Philadelphia, PA: Saunders-Elsevier, pp. 517-588.
- Barkovich, James, Raybaud, Charles. Pediatric Neuroimaging. Wolters Kluwer, 2018.
- Papile LA, J Burstein, R Burstein, H Koffler (1978) Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birth weights less than 1500gm. The Journal of Pediatrics 92(4): 529-534.
- Raets MMA, Dudink J, Govaert P (2015) Neonatal disorders of germinal matrix. The Journal of Maternal-Fetal and Neonatal Medicine 28(sup1): 2286-2290.
- Zhang (2018) Predictive value of cranial ultrasound for neurodevelopmental outcomes of very preterm infants with brain injury. Chin Med J 131(8): 920-926.
- Mack LA, K Wright, JH Hirsch, EC Alvord, RD Guthrie, et al. (1981) Intracranial hemorrhage in premature infants: accuracy in sonographic evaluation. American Journal of Roentgenology137: 245-250.
- Salas J, Tekes A, Hwang M, Northington FJ, Huisman TAGM (2018) Head ultrasound in neonatal hypoxic-ischemic injury and its mimickers for clinicians: a review of the patterns of injury and the evolution of findings over time. Neonatology 114(3): 185-197.
- Parodi A, Govaert P, Horsch S, Bravo MC, Ramenghi LA, et al. (2020) Cranial ultrasound findings in preterm germinal matrix haemorrhage, sequelae and outcome. Pediatr Res 87 (suppl 1): 13-24.
- Ment LR, HS Bada, P Barnes, PE Grant, D Hirtz, et al. (2002) Practice Parameter: Neuroimaging of the neonate: Report of the quality standards subcommittee of the American Academy of Neurology and the practice committee of the child neurology society. Neurology 58(12): 1726-1738.
- Rao V, Levin DC (2012) The overuse of diagnostic imaging and the choosing wisely initiative. Ann Intern Med 157(8):574-576.
- Haim B (2009) Intracranial hemorrhage in the preterm infant. Understanding it, and preventing it. Clinics in Perinatology 36(4): 737-762.
- Lu H, Wang Q, Lu J, Zhang Q, Kumar P (2016) Risk factors for intraventricular hemorrhage in preterm infants born at 34 weeks of gestation or less following preterm premature rupture of membranes. Journal of Stroke and Cerebrovascular Diseases 25(4): 807-812.
- Blencowe H, Cousens S, Chou D (2013) Born too soon: the global epidemiology of 15 million preterm births. Reprod Health 10(Suppl 1):S2, Nov 2013.
- Mancini MC, Barbosa NE, Banwart D (1999) Intraventricular hemorrhage in very low birth weight infants: associated risk factors and outcome in the neonatal period. Rev Hosp Clin Fac Med Sao Paulo 54: 151-154.
- Dubowitz, M I Levene, A Morante, P Palmer, V Dubowitz (1981) Neurologic signs in neonatal intraventricular hemorrhage: A correlation with real-time ultrasound. The Journal of Pediatrics 99(1): 127-133.
- Hendee WR, Becker GJ, Borgstede JP, Bosma J, Casarella WJ, et al. (2010) Addressing overutilization in medical imaging. Radiology 257(1): 240-245.






























