Cariprazine in Autism Spectrum Disorder and Intellectual Disability Disorder
Cohen LS*, Pella C
Developmental Disability Center, Clinical Neuroscience Center- Mount Sinai West, New York
Submission:July 24, 2019; Published:October 14, 2019
*Corresponding author:Shahid H Sheikh, Senior Research Scientist, Seattle Institute of Nucleopathy, 126 Habitat Shadman II, Jail Road, LAHORE, Pakistan
How to cite this article:Cohen LS and Pella C. Cariprazine in Autism Spectrum Disorder and Intellectual Disability Disorder. Glob J Intellect Dev Disabil, 2019; 6(3): 555694. DOI: 10.19080/GJIDD.2019.06.555694
Abstract
This is the first clinical report of the use of Cariprazine, a second generation orally administered atypical antipsychotic compound, in patients with Autism Spectrum Disorder (ASD) and Intellectual Disability (ID). Risperidone and Aripiprazole have been studied in autistic patients and are FDA approved for the treatment of irritability associated with autism, but studies of newer agents are limited. We studied eight patients diagnosed with ASD and/or ID from our developmental disability clinic, some of whom have concomitant severe behavioral issues characterized by aggression, impulsivity, and self-injurious behavior. One case was co-morbid with cerebral palsy and three cases were co-morbid with seizure disorder or epilepsy. Cases were retrospectively chart ¬reviewed for Clinical Global Impression Severity Scale (CGI-S) before initiating Cariprazine and Clinical Global Impression Improvement Scale (CGI-I) and Clinical Global Impression Efficacy Scale (CGI-E) after initiating Cariprazine. Overall, 87.5% of patients treated with Cariprazine showed improvement in clinical functioning. Cariprazine may function as an alternative compound for improvement in impulsivity, aggression, and self-injurious behavior in the treatment of patients diagnosed with ASD or ID who have failed currently approved compounds. This retrospective chart review indicates the potential need for more controlled studies to evaluate Cariprazine in these diagnostic groups.
Keywords: Autism spectrum disorder; Intellectual disability; Cariprazine
Introduction
Autism spectrum disorder (ASD) includes a group of developmental disorders that can result in social, behavioral, and communication problems. The Diagnostic and Statistical Manual of Mental Disorders V (DSM-V) defines ASD as: Persistent deficits in social communication and social interaction across multiple contexts, as manifested by the following, currently or by history: Deficits in social-emotional reciprocity, ranging, for example, from abnormal social approach and failure of normal back-and-forth conversation; to reduced sharing of interests, emotions, or affect; to failure to initiate or respond to social interactions.
Deficits in nonverbal communicative behaviors used for social interaction, ranging, for example, from poorly integrated verbal and nonverbal communication; to abnormalities in eye contact and body language or deficits in understanding and use of gestures; to a total lack of facial expressions and nonverbal communication. Deficits in developing, maintaining, and understanding relationships, ranging, for example, from difficulties adjusting behavior to suit various social contexts; to difficulties in sharing imaginative play or in making friends; to absence of interest in peers [1].
The overall prevalence of ASD is estimated to be 1 of 68 children. The average ratio of ASD in males compared to femalesis 4.5:1. In ASD, common problems that arise are aggression, self-injury, and irritability [2]. These maladaptive behaviors can lead to patients with ASD to be placed in restrictive home or school settings, increase risk of victimization, and place severe burden on caretakers [3].
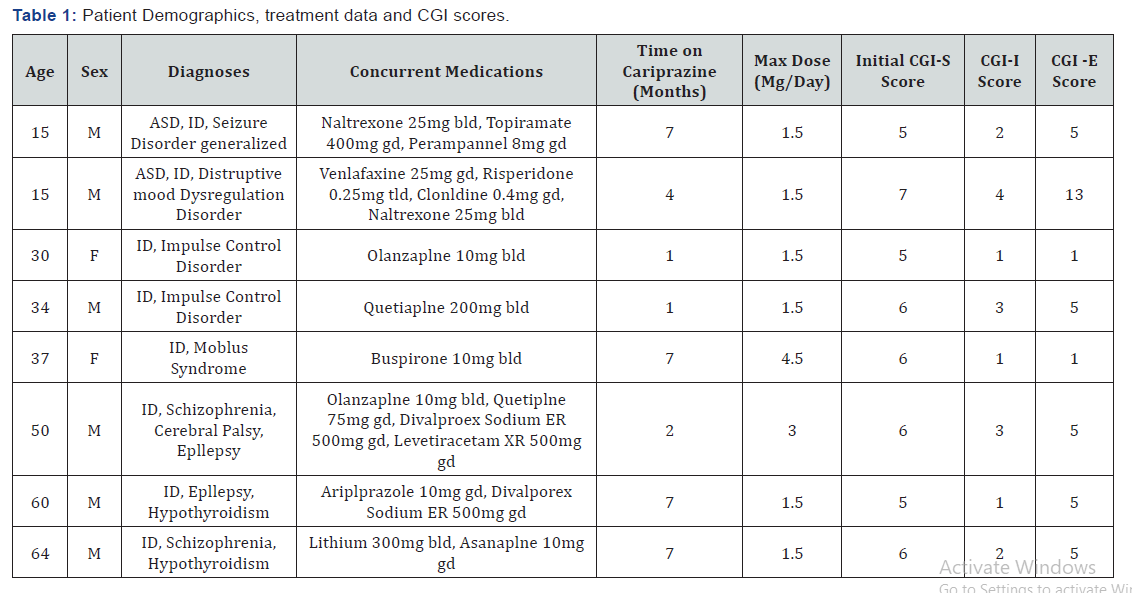
Intellectual disability (ID) is defined as deficits in intellectual functioning and adaptive behavior before the age of 18 in the DSM-V and by the American Association on Intellectual and Developmental Disabilities (AAIDD). Intellectual functioning or intelligence refers to mental tasks such as learning or reasoning, which can be assessed with an Intelligence Quotient (IQ) score that is usually 2 standard deviations below the mean in ID. Adaptive behavior refers to conceptual, social, and practical skills used in multiple settings, such as home or social situations. ID is more common in males than females and is seen more frequently in individuals of lower socioeconomic status (SES) than mid to high SES [4,5]. Aggressive behavior is cited as a common challenge in the individuals diagnosed with ID and behavioral disturbances are often treated with antipsychotics. However, there is no official treatment for aggression in patients diagnosed with ID and previous studies have shown that Risperidone and Haloperidol were not effective compared to placebo [6] (Table 1).
Atypical antipsychotics act via blockade of post-synaptic dopamine and serotonin receptors, or some may work by acting as a partial agonist. They are preferred over typical antipsychotics due their side effect profile and have been found useful in the treatment of severe maladaptive behavior in children with ASD. Currently, Risperidone and Aripiprazole, two atypical antipsychotics, have been approved for the treatment of tantrums, aggression, or self-injurious behavior in children with ASD [3, 7-9]. There is also preliminary evidence for the potential use of Simvastatin as an adjunct to Risperidone in the treatment of irritability in ASD patients [10]. Despite the limited data on atypical antipsychotics use in ASD and pediatric patients, there have been developments in the field. Lurasidone, an atypical antipsychotic, has been shown to cause clinical improvement in adolescents diagnosed with schizophrenia, leading to its approval for treatment in this population. Furthermore, Asenapine has also been approved to treat childhood bipolar I disorder. Asenapine is another atypical antipsychotic that has shown clinical effectiveness based on the Young-Mania Rating Scale (YMRS) [11-14].
Oxytocin and arginine vasopressin have become attractive potential therapeutic targets for ASD research because they are key regulators of social behavior [15]. Recent studies show evidence of the use of oxytocin metabolites, such as OT (4-9), to improve sociability in ASD patients. This discovery shows the possibility of a new drug discovery pathway for ASD [16]. Another approach for improving socialization and communication includes the use of vasopressin V1a receptor antagonists, such as Balovaptan, which demonstrated improvement in Vineland-II socialization and communication scores in a phase two clinical trial [17].Supplementation of Vitamin D and omega-3-fatty acids is correlated with improving the irritability symptoms of children with ASD. Vitamin D has also been shown to improve hyperactivity symptoms in children [18,19]. Insufficient Vitamin D is common in autistic children and may also be associated as a risk factor for developing ASD [20]. Additionally, Vitamin B supplementation is associated with decreasing behavioral problems in ASD patients, although it is unclear as to exactly how it is effective. Vitamin B6 is critical in the synthesis of several neurotransmitters; including serotonin, dopamine, and others; as a result, it is surmised that Vitamin B supplementation can treat impaired neurotransmitter systems [21].
There is still further research to be conducted on the use of pharmacotherapy in patients with ASD and ID. Cariprazine has not been studied. This retrospective study aims to analyze the efficacy of the second-generation atypical antipsychotic drug Cariprazine for the treatment of aggressive behavior in those with autism spectrum disorder and intellectual disability.
Cariprazine
Cariprazine is an atypical antipsychotic, FDA approved for the treatment of schizophrenia and acute treatment of manic or mixed episodes associated with bipolar I disorder. The precise mechanism of Cariprazine is unknown in schizophrenia and bipolar disorder. It is thought to be effective due to its partial agonist activity at the dopamine D2 receptor and serotonin 5HT1A receptor and antagonism at the serotonin 5HT2A receptor. Additionally, Cariprazine has high affinity for Dopamine D3 receptors acting as a partial agonist at D3 receptors. Cariprazine is primarily metabolized by CYP3A4 into Desmethyl Cariprazine (DCAR) and DCAR is then further metabolized by CYP3A4 into Didesmethyl Cariprazine (DDCAR). DCAR and DDCAR both display similar pharmacological potency as Cariprazine. The half-life of Cariprazine ranges from 2-4 days and its two active metabolites, DCAR and DDCAR, have half-lives ranging from 1-3 weeks, making it the atypical antipsychotic with the longest halflife. The peak plasma concentration of Cariprazine occurs in 3-6 hours. Cariprazine and DCAR reach steady state in 1-2 weeks, whereas DDCAR reaches steady state in 4-8 weeks.
Commonly observed adverse reactions (incidence ≥ 5% and at least twice that for placebo) were: Schizophrenia: extrapyramidal symptoms and akathisia Bipolar Mania: extrapyramidal symptoms, akathisia, dyspepsia, vomiting, somnolence and restlessness.
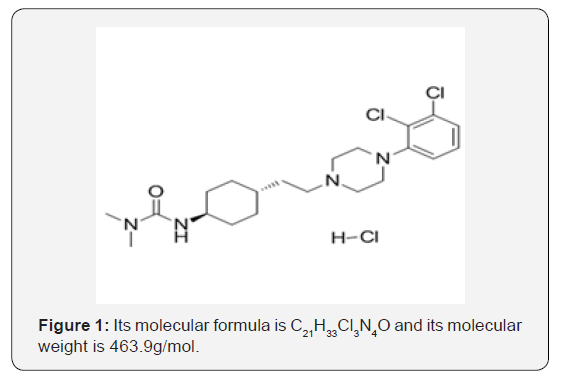
The chemical name of Cariprazine is trans-N-{4-[2-[4- (2,3-dichlorophenyl) piperazine-1-yl] ethyl] cyclohexyl}-N’, N’-dimethylurea hydrochloride. Its molecular formula is C21H33Cl3N4O and its molecular weight is 463.9 g/mol [22-24] (Figure 1).
Material and Methods
We reviewed the charts of over 120 patients at the Developmental Disabilities Center of Mount Sinai West Hospital and identified eight patients receiving the target drug (Cariprazine). We then retrospectively reviewed each patient’s chart to assign a CGI-S score to each patient before each was prescribed Cariprazine and again at the time of investigation to determine a CGI-I and CGI-E score. Co-morbid conditions and concurrent medications during treatment with Cariprazine were noted.
Clinical Global Impression (CGI) scales are used as measures of symptom severity alongside treatment response as judged by a trained clinician. The severity scale (CGI-S) is a seven-point scale used to assign symptom severity at time of assessment, usually prior to initiating a treatment modality. The values are defined as 1, normal or not at all ill; 2, borderline mentally ill; 3, mildly ill; 4, moderately ill; 5, markedly ill; 6, severely ill; or 7, among the most extremely ill.
The improvement scale (CGI-I) is a seven-point scale used to assess a patient’s improvement, if any, from the baseline CGI-S due to drug treatment. Its values are defined as 1, very much improved; 2, much improved; 3, minimally improved; 4, no improvement; 5, minimally worse; 6, much worse; or 7, very much worse.
The efficacy index (CGI-E) consists of a 4-point x 4-point rating scale and assesses the therapeutic effect and side effect of the treatment. The therapeutic effect can be scored as marked (vast improvement), moderate (partial remission), minimal (slight improvement), or unchanged or worse. The side effect can be scored as none, no significant interference with functioning, significant interfering with functioning, or side effect outweighs therapeutic effect. CGI-E score can range from 1-16. A score of 1 indicates vast improvement with no side effects and a score of 16 signifies unchanged or worsened functioning with side effects outweighing therapeutic effect [25].
Results and Discussion
The sample included 2 female and 6 male cases. Mean patient age of the sample was 38 years old (range 15 to 64 years old). Mean length of time on Cariprazine was 4.5 months (range 1 to 7 months). Mean titrated total daily dose was 1.88mg (range 1.5 to 3.0mg) (Figure 2). Cases were retrospectively chart reviewed for Clinical Global Impression Severity Scale (CGI-S) before initiating Cariprazine and Clinical Global Impression Improvement Scale (CGI-I) and Clinical Global Impression Efficacy Scale (CGI-E) after initiating Cariprazine. The mean CGI-S of the sample was 5.75 (range 5 to 7), which correlates with marked to severe illness, and mean CGI-I of the sample was 2.13 (range 1 to 4), which correlates with minimal to much improvement. Three patients were very much improved (1), two patients were much improved (2), two patients showed minimal improvement (3), and one showed no change (4) after clinical review by a board-certified psychiatrist. A similar pattern arose using the CGI-E. Mean CGI-E of the sample was 5.0 (range 1 to 13), which correlates with moderate therapeutic efficacy. One patient showed no efficacy of the drug (13), five patients showed moderate efficacy of the drug (5), and two patients showed marked efficacy (1).
The resulting scores based on the CGI metrics provides primitive evidence about the efficacy of Cariprazine on treating ASD and ID. Considering that the mean of the patients was ranked in the category of severe illness, we are given a good baseline to compare from. The CGI-I and CGI-E scales showed how most of the patients’ disease state did improve after treatment but at varying degrees. Although one patient did not improve after treatment, that patients’ symptoms did not worsen either and there were no adverse events reported. The various responses to the treatment hint at the possibility of some intrinsic factors that may be leading to different degrees of efficacy; a potential focal point for future studies. Additionally, the ages within the test group had a large range so treatment results on children and adults should be separated in future studies.

Conclusion
87.5% of patients treated with Cariprazine in this study demonstrated minimal to very much improvement as defined by a decrease in aggression, impulsivity, and/or self-injurious behavior using the CGI-I parameter. No patients being treated with Cariprazine exhibited worsening of clinical symptoms. There were no serious adverse drug reactions reported while patients were undergoing treatment with Cariprazine.
Cariprazine was combined with other atypical antipsychotics in six out of eight cases without evidence of cross-reactivity. Cariprazine was combined with anti-epileptic drugs in three patients without evidence of cross-reactivity. These findings suggest that Cariprazine, a second-generation atypical antipsychotic, may be effective in reducing symptoms of aggression, impulsivity, and self-injurious behavior in patients diagnosed with ASD and ID when other interventions have been tried unsuccessfully. Further investigations with Cariprazine in this patient population are warranted.
References
- (2019) Diagnostic Criteria | Autism Spectrum Disorder (ASD) | NCBDDD | CDC. (n.d.).
- Autism Spectrum Disorder (2017) National Health. USA.
- Fitzpatrick SE, Srivorakiat L, Wink LK, Pedapati EV, Erickson CA (2016) Aggression in autism spectrum disorder: presentation and treatment options. Neuropsychiatric Disease and Treatment 12: 1525-1538.
- American Association on Intellectual and Developmental Disabilities (AAIDD) (2017) Definition of Intellectual Disability.
- Boat TF, Wu JT (2015) Clinical Characteristics of Intellectual Disabilities. Mental Disorders and Disabilities Among Low-Income Children. Washington (DC): National Academies Press, USA.
- Tyrer P (2012) Risperidone, haloperidol, and placebo in the treatment of aggressive challenging behaviour in patients with intellectual disability: a randomised controlled trial. Lancet 371(9606): 57-63.
- Cohen S (1998) Risperidone for Aggression and Self-Injurious Behavior in Adults with Mental Retardation. Journal of Autism and Development Disorders 28(3): 229-33.
- McCracken J, McGough J, Shah B, Cronin P, Hong D, et al. (2002) Risperidone in Children with Autism and Serious Behavioral Problems. New England Journal of Medicine 347: 314-21.
- Owen R, Sikich L, Marcus RN, Corey-Lisle P, Manos G, et al. (2009) Aripiprazole in the Treatment of Irritability in Children and Adolescents with Autistic Disorder. Pediatrics 124(6): 1533-1540.
- Moazen Zadeh E, Shirzad F, Karkhaneh-Yousefi MA, Khezri R, Mohammadi MR, et al. (2018) Simvastatin as an Adjunctive Therapy to Risperidone in Treatment of Autism: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Journal of Child & Adolescent Psychopharmacology 28(1): 82-89.
- Findling RL, Landbloom RL, Mackle M, Wu X, Adami LS, et al. (2016) Long-term Safety of Asenapine in Pediatric Patients Diagnosed with Bipolar I Disorder: A 50-Week Open-Label, Flexible-Dose Trial. Paediatric Drugs 18(5): 367-378.
- Goldman Robert, Loebel Antony, Cucchiaro Josephine, Deng Ling, Findling Robert L (2017) Efficacy and Safety of Lurasidone in Adolescents with Schizophrenia: A 6-Week, Randomized Placebo-Controlled Study. Journal of Child and Adolescent Psychopharmacology 27(6): 516-525.
- Loebel A, Brams M, Goldman RS, Silva R, Hernandez D, et al. (2016) Lurasidone for the Treatment of Irritability Associated with Autistic Disorder. Journal of Autism and Developmental Disorders 46(4):1153-1163.
- Millard PH, McLaren JL, Coffey DBJ (2014) Lurasidone Treatment in a Child with Autism Spectrum Disorder with Irritability and Aggression. Journal of Child and Adolescent Psychopharmacology 24(6): 354-356.
- Lukas M, Neumann ID (2013) Oxytocin and vasopressin in rodent behaviors related to social dysfunctions in autism spectrum disorders. Behavioural Brain Research 251: 85-94.
- Moy SS, Nikolova VD, Riddick NV, Pedersen CA, Teng BL, et al. (2019) Prosocial effects of an oxytocin metabolite, but not synthetic oxytocin receptor agonists, in a mouse model of autism. Neuropharmacology 144: 301-311.
- Bolognani F, Rubido MD, Squassante L, Wandel C, Derks M, et al. (2019) A phase 2 clinical trial of a vasopressin V1a receptor antagonist shows improved adaptive behaviors in men with autism spectrum disorder. Science Translational Medicine 11(491).
- Mazahery H, Conlon CA, Beck KL, Mugridge O, Kruger MC, et al. (2019) A randomised controlled trial of vitamin D and omega-3 long chain polyunsaturated fatty acids in the treatment of irritability and hyperactivity among children with autism spectrum disorder. Journal of Steroid Biochemistry & Molecular Biology 187: 9-16.
- Saad K, Abdel Rahman AA, Elserogy YM, Al Atram AA, El Houfey AA, et al. (2018) Randomized controlled trial of vitamin D supplementation in children with autism spectrum disorder. Journal of Child Psychology & Psychiatry 59(1): 20-29.
- Jia F, Shan L, Wang B, Li H, Miao C, et al. (2018) Bench to bedside review: Possible role of vitamin D in autism spectrum disorder. Psychiatry Research 260: 360-365.
- Sato K (2018) Why is vitamin B6 effective in alleviating the symptoms of autism? Medical Hypotheses 115: 103-106.
- Earley W, Durgam S, Lu K, Laszlovszky I, Debelle M, et al. (2017) Safety and Tolerability of Cariprazine in Patients with Acute Exacerbation of Schizophrenia. International Clinical Psychopharmacology 32(6): 319-328.
- Stahl S (2016) Mechanism of action of cariprazine. CNS Spectrums 21(2): 123-127.
- Werner FM, Covenas R (2015) New developments in the management of schizophrenia and bipolar disorder: potential use of cariprazine. Therapeutics and Clinical Risk Management 11: 1657-1661.
- Guy W (2000) Clinical Global Impressions (CGI) Scale. Modified from: Rush J et al. Psychiatric Measures, APA, Washington DC.






























