Abstract
Background: The global prevalence of diabetes mellitus and metabolic syndrome has reached epidemic proportions, driven by insulin resistance and chronic hyperglycemia. The skin, as the body’s largest border organ, often serves as an early, visible barometer of these internal metabolic
derangements.
Objective: This review highlights dermatological manifestations that mechanistically reflect underlying metabolic dysfunction and discusses
their significance in clinical screening and early intervention.
Discussion: Metabolic dysfunction impacts cutaneous biology through hyperinsulinemia, advanced glycation end products (AGEs), microvascular
dysfunction, and dyslipidemia. Proliferative markers such as acanthosis nigricans and acrochordons (skin tags) directly reflect compensatory
hyperinsulinemia. Microvascular damage and chronic glycemic burden manifest as necrobiosis lipoidica, diabetic dermopathy, and foot ulcers,
the latter representing the culmination of metabolic injury. Notably, eruptive xanthomas signal severe hypertriglyceridemia and an imminent
risk of acute pancreatitis. Recognition of these markers provides a critical window for diagnosis, as skin changes often precede overt laboratory
abnormalities.
Conclusion: Dermatologists and primary care clinicians play a pivotal role in the “Skin-to-System” diagnostic approach. Utilizing a structured
clinical algorithm and targeted laboratory workup-including HbA1c, fasting glucose, and lipid panels-enables early identification of at-risk
individuals. Interdisciplinary collaboration is essential to transition from cutaneous recognition to systemic management, ultimately reducing
the burden of cardiovascular and microvascular complications.
Keywords: Insulin Resistance; Metabolic Syndrome; Cutaneous Manifestations; Diabetes mellitus; Hyperinsulinemia
Abbreviations: HOMA-IR: Homeostatic Model Assessment for Insulin Resistance; IGF-1: Insulin-like growth factor-1; IR: Insulin receptor; IGF- 1R: Insulin-like growth factor-1 receptor; PI3K: Phosphoinositide 3-kinase; Akt: Protein kinase B; MAPK: Mitogen-activated protein kinase; AGEs: Advanced glycation end products; RAGE: Receptor for advanced glycation end products; NF-κB: Nuclear factor kappa-light-chain-enhancer of activated B cells; AN: Acanthosis Nigricans; ADA: American Diabetes Association; NL: Necrobiosis Lipoidica; HbA1c: Hemoglobin A1c (Glycated hemoglobin); BMI: Body Mass Index; OGTT: Oral glucose tolerance test; SHBG: Sex hormone-binding globulin
Introduction
Diabetes mellitus and metabolic syndrome have emerged as defining public health challenges of the twenty-first century. As of 2024, 589 million adults aged 20-79 years are living with diabetes- approximately 1 in every 9 adults worldwide- a figure projected to climb to 853 million by 2050 [1]. The economic toll is equally staggering: diabetes was responsible for 3.4 million deaths in 2024 alone and caused at least one trillion US dollars in health expenditure, representing a 338% increase over the past seventeen years [1]. The epidemic extends well beyond diabetes itself. Between 2000 and 2023, the global prevalence of metabolic syndrome rose from 14.7% to 31.0% among women and from 9.0% to 25.7% among men, translating to an estimated 1.54 billion affected adults in 2023 [2]. These trends reflect a broader metabolic crisis driven by urbanization, sedentary lifestyles, and caloric excess that no healthcare system can afford to ignore.
At the mechanistic core of this epidemic lies insulin resistancea state in which peripheral tissues fail to respond appropriately to insulin signaling, forcing compensatory hyperinsulinemia and ultimately precipitating overt hyperglycemia. Insulin resistance is a well-recognized pathophysiologic factor in the development of type 2 diabetes, characteristically appearing years before its clinical diagnosis, and is present in 70–80% of patients with obesity and virtually every patient with type 2 diabetes [3]. Despite its central importance, diagnosing insulin resistance in everyday clinical practice remains challenging: the euglycemic hyper insulinemic clamp, the gold standard measurement, is invasive, expensive, and logistically impractical, while derived indices such as HOMA-IR carry well-documented limitations in precision and reproducibility.
It is here that the skin assumes particular clinical significance. The skin is the major border organ of the human body and offers a window to internal processes, with changes at its surface frequently serving as markers of underlying systemic disease [4]. In the context of metabolic dysfunction, this translating role is remarkably consistent: between thirty and seventy percent of patients with diabetes mellitus, both type 1 and type 2, will present with a cutaneous complication at some point during their lifetime, and such skin changes can offer insight into glycemic control while sometimes constituting the first sign of metabolic derangement in undiagnosed patients [5]. Critically, skin manifestations of insulin resistance-including acanthosis nigricans, acrochordons, androgenetic alopecia, acne, and hirsutism-offer a reliable, straightforward, and real-time method to detect insulin resistance that requires no laboratory equipment and is immediately accessible during any physical examination [3].
Early recognition of these cutaneous signals carries direct clinical consequences. Prompt recognition of skin lesions is essential, as it enables early diagnostic testing and timely treatment, minimizing the long-term complications of diabetes mellitus [6]. Yet this opportunity remains underutilized: cutaneous manifestations of diabetes often serve as indicators of insulin resistance that vary with disease progression and highlight systemic involvement, but current challenges in their identification- including under detection in darker-skinned populations-mean that metabolic derangement frequently goes unrecognized at a stage when intervention could alter the disease course [7].
Numerous reviews have catalogued the broad spectrum of skin findings in diabetes, ranging from therapy-related reactions to infectious complications and neuropathic ulcers. The present review takes a more focused approach. We aim to highlight specifically those dermatological manifestations that directly and mechanistically reflect underlying metabolic derangementprincipally insulin resistance, hyperinsulinemia, dyslipidemia, and chronic hyperglycemia- and to discuss their recognition and clinical significance in everyday practice. By illuminating these cutaneous clues, we hope to equip clinicians across specialties to use the skin as it deserves to be used: as a readily accessible, realtime barometer of their patients’ metabolic health.
Core Pathophysiologic Mechanisms Linking Metabolic Dysfunction and the Skin
Metabolic dysfunction exerts multisystem effects that directly impact cutaneous biology through endocrine, inflammatory, oxidative, and microvascular pathways. Emerging evidence positions the skin not only as a passive target but also as an active participant in systemic insulin resistance and cardiometabolic disease [8,9]. The following sections summarize the principal biological mechanisms linking metabolic abnormalities to structural and functional alterations in the skin.
Hyperinsulinemia and IGF-1 Signaling
Insulin resistance induces compensatory hyperinsulinemia, leading to enhanced activation of insulin receptors (IR) and insulin-like growth factor-1 receptors (IGF-1R) expressed on keratinocytes and dermal fibroblasts [8,10]. Sustained stimulation of downstream PI3K/Akt and MAPK signaling cascades promotes cellular proliferation while inhibiting apoptosis within the epidermis [10].
This persistent mitogenic signaling contributes to epidermal hyperplasia, altered differentiation, and increased dermal extracellular matrix production. Clinical and translational studies demonstrate that hyperinsulinemia correlates with proliferative dermatologic conditions such as acanthosis nigricans and psoriasis, where insulin-mediated growth signaling augments keratinocyte turnover and fibroblast activity [8,9]. Collectively, these findings support a mechanistic model in which chronic insulin excess remodels cutaneous architecture and promotes hyperproliferative skin phenotypes.
Chronic Hyperglycemia and Advanced Glycation End Products (AGEs)
Chronic hyperglycemia promotes nonenzymatic glycation of long-lived structural proteins, including collagen and elastin, resulting in the formation of advanced glycation end products (AGEs) [11,12]. Glycation induces abnormal collagen cross-linking, decreasing solubility and disrupting dermal biomechanical properties [12]. These alterations lead to increased dermal stiffness, reduced elasticity, and impaired tensile strength. Beyond structural effects, AGEs bind to the receptor for advanced glycation end products (RAGE) on keratinocytes, fibroblasts, and endothelial cells, activating NF-κB–mediated inflammatory pathways [13]. This sustained inflammatory signaling, combined with extracellular matrix stiffening, contributes to delayed wound healing and increased susceptibility to ulceration in patients with diabetes and metabolic syndrome [11,13].
Microvascular Dysfunction
Metabolic dysfunction is strongly associated with endothelial injury mediated by oxidative stress, chronic lowgrade inflammation, and reduced nitric oxide bioavailability [14]. Microvascular dysfunction has been documented even in early insulin-resistant and prediabetic states [15]. In cutaneous tissue, endothelial injury promotes thickening of the capillary basement membrane, a hallmark of diabetic microangiopathy [6] Basement membrane thickening and capillary rarefaction impair oxygen and nutrient diffusion, thereby reducing tissue perfusion [15,16]. These alterations predispose patients to ischemic skin changes, delayed wound repair, and ulcer formation. Importantly, cutaneous microvascular dysfunction parallels microangiopathy observed in cardiac and renal tissues, underscoring a shared systemic pathophysiologic substrate [14].
Dyslipidemia and Lipid Deposition
Severe hypertriglyceridemia and dyslipidemia contribute to abnormal lipid handling within dermal macrophages. Elevated circulating triglyceride-rich lipoproteins enhance macrophage lipid uptake and foam cell formation within the dermis [17,18]. Clinically, this process manifests as eruptive or tuberous xanthomas, histologically characterized by aggregates of lipid-laden macrophages [18]. Systemic lipid abnormalities are also associated with cutaneous inflammatory activation and extracellular matrix remodeling, indicating that dyslipidemia exerts both structural and immunologic effects on the skin [19]. Thus, lipid metabolism disorders represent a direct mechanistic bridge between systemic metabolic dysfunction and dermatologic manifestations.
Dermatologic Manifestations Directly Reflecting Metabolic Dysfunction
Acanthosis Nigricans: The Prototype of Insulin Resistance
Acanthosis Nigricans (AN) presents as symmetric, velvety, hyperpigmented plaques most commonly located in intertriginous areas such as the posterior neck, axillae, groin, and inframammary folds. The affected skin appears thickened and papillomatous, often described by patients as having a “dirty” appearance that does not improve with washing [20]. Although frequently perceived as a cosmetic concern, AN should be approached clinically as a warning sign. In many patients, particularly adolescents and young adults, these cutaneous findings represent one of the earliest manifestations of insulin resistance [21]. The underlying mechanism is driven by hyperinsulinemia. Elevated circulating insulin levels stimulate insulin-like growth factor-1 (IGF-1) receptors on keratinocytes and dermal fibroblasts, promoting epidermal proliferation and dermal thickening [22]. In this context, the skin reflects chronic metabolic signaling before overt hyperglycemia may be detected on laboratory testing.
A strong association has been demonstrated between AN and obesity, elevated fasting insulin levels, metabolic syndrome, and Type 2 diabetes mellitus [23]. Its presence in both adults and adolescents correlates with impaired glucose metabolism and increased cardiometabolic risk. According to the American Diabetes Association (ADA), metabolic screening is recommended in overweight or obese individuals who also exhibit clinical signs of insulin resistance, including AN [24]. In practice, evaluation should include fasting plasma glucose, hemoglobin A1C, and a lipid panel, along with assessment of other cardiovascular risk factors. Recognizing AN during physical examination, therefore, provides an opportunity for early identification and intervention.
Acrochordons (Skin Tags)
While AN is a more conspicuous marker, other subtle dermatologic findings can carry similar metabolic implications. Acrochordons, commonly referred to as skin tags, are benign, pedunculated lesions composed of a fibrovascular core. Increasing evidence supports their association with insulin resistance and impaired carbohydrate metabolism [25,26]. Patients with multiple skin tags have been shown to exhibit higher fasting insulin levels and elevated markers of insulin resistance when compared with controls [27]. These lesions are most frequently located in areas of friction, including the neck, axillae, groin, eyelids, and inframammary folds.
The pathophysiologic overlap with AN is notable. Hyperinsulinemia appears to stimulate IGF-1 receptors, promoting fibroblast and epidermal proliferation [26]. This shared mechanism supports the concept that acrochordons may represent a milder phenotypic expression of insulin-driven proliferative signaling. Although often dismissed as incidental findings, the presence of multiple acrochordons, particularly in patients with central obesity, should prompt consideration of metabolic evaluation. In this setting, they may function as early clinical markers of insulin resistance and provide another opportunity for preventive intervention [25].
Necrobiosis Lipoidica
In contrast to the proliferative lesions associated with hyperinsulinemia, necrobiosis lipoidica (NL) represents a less common but clinically significant dermatologic manifestation of diabetes. NL is characterized by well-demarcated, yellowbrown atrophic plaques, most frequently located on the pretibial surfaces. Lesions typically evolve from erythematous papules into plaques with central thinning, telangiectasias, and a waxy appearance [28]. NL is strongly associated with diabetes mellitus, more commonly type 1 than type 2, although it may occur in either population [29]. In some cases, these lesions precede the formal diagnosis of diabetes and serve as a visible indicator of underlying metabolic dysfunction.
The pathogenesis is believed to involve diabetic microangiopathy, immune-mediated vascular injury, collagen degeneration, and granulomatous inflammation [30]. Chronic hyperglycemia contributes to endothelial dysfunction and impaired tissue perfusion, leading to the characteristic dermal changes observed clinically. Prompt recognition of NL is essential due to the risk of ulceration, which may result in secondary infection and delayed wound healing [28]. For this reason, identification of NL should prompt careful assessment of glycemic control and evaluation for additional microvascular complications.
Diabetic Dermopathy
Diabetic dermopathy represents the most common cutaneous finding in diabetes mellitus, occurring in approximately 40% of patients with diabetes and manifesting as atrophic, hyperpigmented macules characteristically located on the pretibial surfaces-hence the colloquial term “shin spots.” These lesions appear as well-circumscribed, round to oval, light brown patches that develop asymmetrically on the anterior lower legs, though they may occasionally appear on the forearms, thighs, or lateral malleoli [31-33].
The pathophysiology of diabetic dermopathy reflects underlying microvascular injury, with histologic examination revealing thickening of capillary walls, endothelial proliferation, and extravasation of red blood cells [31]. The presence of diabetic dermopathy correlates significantly with other microvascular complications of diabetes, including retinopathy, nephropathy, and neuropathy [32,34,35]. In a prospective study of 173 patients with diabetes, the frequency of diabetic dermopathy increased proportionally with the number of microvascular complications present in each patient, suggesting that common pathophysiologic mechanisms-likely related to chronic hyperglycemia and microvascular damage-contribute to both dermopathy and internal complications [32].
Diabetic dermopathy serves as a clinical marker of chronic glycemic burden and disease duration. Patients with dermopathy demonstrate significantly longer diabetes duration compared to those without these lesions, and the condition is more prevalent in individuals aged 50 years and older [32]. The association extends beyond microvascular disease; studies have also demonstrated a relationship between diabetic dermopathy and coronary artery disease, suggesting that these seemingly benign skin lesions may indicate more extensive vascular pathology [31]. Recognition of diabetic dermopathy should prompt comprehensive evaluation for associated systemic complications and intensification of glycemic control strategies.
Eruptive Xanthomas: Marker of Severe Dyslipidemia
Eruptive xanthomas represent a dermatologic emergency, signaling severe hypertriglyceridemia that places patients at imminent risk for life-threatening acute pancreatitis [36,37]. These lesions manifest as sudden-onset yellow papules with characteristic erythematous halos, typically measuring 1-4 mm in diameter and appearing in crops on the extensor surfaces of the extremities, buttocks, and back [37,38]. The lesions may be accompanied by pruritus or tenderness and develop when triglyceride levels exceed 1000-2000 mg/dL, though they can occur at lower levels in susceptible individuals [36,39].
The pathophysiology involves leakage of triglyceride-rich lipoproteins through capillaries into the dermis, where they are phagocytosed by macrophages, creating foam cells that accumulate as visible xanthomatous deposits [38]. Eruptive xanthomas are frequently associated with the chylomicronemia syndrome, which encompasses not only the cutaneous manifestations but also abdominal pain, lipemia retinalis, hepatosplenomegaly, and most critically, the risk of acute pancreatitis [36,39,40]. The risk of pancreatitis increases markedly when triglyceride levels exceed 2000 mg/dL, though it can occur at levels above 1000 mg/dL [36].
Eruptive xanthomas may be the first visible sign of lifethreatening hypertriglyceridemia, particularly in patients with uncontrolled diabetes mellitus, as insulin deficiency impairs lipoprotein lipase activity and triglyceride clearance [36,38,41]. In the context of diabetes, eruptive xanthomas often present alongside diabetic ketoacidosis, representing a metabolic crisis requiring urgent intervention [38,41]. Recognition of eruptive xanthomas mandates immediate metabolic evaluation, including fasting lipid panel, comprehensive metabolic panel, hemoglobin A1c, and assessment for secondary causes of hypertriglyceridemia (the “Four D’s”: Diet/Lifestyle, Drugs/Medications, and Diseases/ Disorders of metabolism) [37]. Treatment requires aggressive triglyceride reduction through severe dietary fat restriction (typically <20 g/day), insulin therapy in diabetic patients, and pharmacologic intervention with fibrates, omega-3 fatty acids, or other lipid-lowering agents to prevent pancreatitis [36-41].
Diabetic Foot Ulcers as the Endpoint of Metabolic Injury
Diabetic foot ulcers represent the culmination of multiple intersecting pathophysiologic processes, reflecting long-standing metabolic dysregulation and its devastating consequences on peripheral nerves, blood vessels, and tissue healing capacity [42,43]. These ulcers develop in approximately 19-34% of patients with diabetes during their lifetime and account for the majority of non-traumatic lower extremity amputations in developed countries [42].
The pathogenesis of diabetic foot ulcers involves a complex interplay between hyperglycemia, neuropathy, and microangiopathy, each contributing distinct but synergistic mechanisms of tissue injury. Diabetic neuropathy-affecting sensory, motor, and autonomic nerve fibers-serves as the primary predisposing factor in the majority of diabetic foot ulcers [42- 45]. Sensory neuropathy causes loss of protective sensation, rendering patients unable to perceive repetitive trauma, excessive pressure, or thermal injury that would normally trigger protective responses [42-44]. Motor neuropathy leads to intrinsic foot muscle atrophy, resulting in biomechanical abnormalities, foot deformities (including claw toes and prominent metatarsal heads), and altered gait patterns that create abnormal pressure points [42,45]. Autonomic neuropathy impairs sweating and causes viscoelastic changes in the skin, leading to dry, fissured skin that is more susceptible to breakdown [42,44].
Peripheral artery disease and microvascular dysfunction compound the neuropathic injury by reducing tissue perfusion and oxygen delivery, critically impairing the wound healing response [43,45,46]. Chronic hyperglycemia drives microvascular injury through multiple pathways, including advanced glycation end-product formation, oxidative stress, endothelial dysfunction, and impaired neurovascular coupling [46-57]. These processes reduce capillary density, impair vasodilation, and limit the angiogenic response necessary for wound repair [43,46]. Macrovascular disease, frequently involving below-knee arteries in diabetes and often complicated by medial artery calcification (Mönckeberg’s sclerosis), further reduces blood flow to the distal extremities [45].
Chronic inflammation and impaired wound healing mechanisms characterize the diabetic foot ulcer environment, reflecting systemic metabolic derangement [43,46]. Hyperglycemia impairs multiple aspects of the healing cascade, including neutrophil and macrophage function, fibroblast proliferation and migration, collagen synthesis, and epithelialization [43,44]. Diabetic wounds demonstrate dysregulated inflammatory responses with prolonged pro-inflammatory cytokine expression, excessive matrix metalloproteinase activity, and impaired growth factor signaling [43]. The combination of defective white cell function, reduced tissue perfusion, and loss of protective sensation creates an environment highly susceptible to infection, which further perpetuates the non-healing state and increases amputation risk [44,45].
Diabetic foot ulcers thus represent a visible manifestation of long-standing metabolic dysregulation, integrating the cumulative effects of chronic hyperglycemia on multiple organ systems [42-47]. The presence of foot ulceration correlates with diabetes duration, glycemic control (as measured by hemoglobin A1c), and the presence of other microvascular complications, including retinopathy and nephropathy [47]. Prevention requires comprehensive risk factor management, including intensive glycemic control, regular foot examination, appropriate footwear, patient education, and early intervention for pre-ulcerative lesions such as calluses [42,45]. Once ulceration occurs, successful management demands a multidisciplinary approach addressing metabolic optimization, infection control, pressure off-loading, vascular assessment with revascularization when indicated, and advanced wound care strategies.
Clinical Screening Implications
Dermatologic manifestations of metabolic dysfunction frequently represent the first visible clinical features of diabetes mellitus and insulin resistance, often appearing before formal diagnosis and providing a critical window for early intervention [48]. Recognition of these cutaneous markers by dermatologists and other clinicians offers an opportunity to identify at-risk individuals, initiate timely metabolic screening, and prevent the development of serious systemic complications, including cardiovascular disease, nephropathy, and neuropathy [48-49].
Early Detection Through Cutaneous Markers
Acanthosis nigricans serves as one of the most important dermatologic indicators warranting immediate metabolic evaluation, particularly in overweight or obese individuals [50-52]. Current guidelines from the American Diabetes Association and the American Association of Clinical Endocrinology specifically identify acanthosis nigricans as a physical sign associated with insulin resistance that should prompt screening for type 2 diabetes, even in asymptomatic individuals [51-53]. In pediatric populations, the presence of acanthosis nigricans in children with BMI at or above the 85th percentile is considered a risk factor that triggers diabetes screening recommendations starting at age 10 years or at the onset of puberty [50].
Multiple skin tags (acrochordons), particularly when numerous and located in intertriginous areas, should similarly prompt consideration of metabolic screening, as these lesions demonstrate strong associations with insulin resistance, impaired glucose tolerance, and dyslipidemia [49]. The presence of skin tags in conjunction with other cutaneous markers such as acanthosis nigricans substantially increases the likelihood of underlying metabolic dysfunction and warrants comprehensive evaluation [49].
Necrobiosis lipoidica, given its strong association with diabetes mellitus (occurring in 0.3-1.6% of diabetic patients), should trigger glycemic assessment even in patients without known diabetes, as approximately 15% of cases precede the diagnosis of diabetes [42,52,54]. The appearance of this distinctive dermatosis-particularly the characteristic yellow-brown plaques with atrophic centers and telangiectasias on the pretibial surfacesnecessitates evaluation of glycemic status including hemoglobin A1c and fasting glucose [54].
Eruptive xanthomas represent a dermatologic emergency requiring urgent metabolic evaluation, as they signal severe hypertriglyceridemia (typically >1000-2000 mg/dL) and imminent risk for acute pancreatitis [54,55]. The sudden appearance of yellow papules with erythematous halos on extensor surfaces mandates immediate laboratory assessment including fasting lipid panel, comprehensive metabolic panel, and hemoglobin A1c, particularly in patients with diabetes or suspected metabolic dysfunction [55].
Diabetic dermopathy, while highly prevalent in established diabetes, serves as a clinical marker of chronic glycemic burden and microvascular complications [56]. When identified, it should prompt a comprehensive assessment for other microvascular complications, including retinopathy, nephropathy, and neuropathy, as well as evaluation of long-term glycemic control [56].
Interdisciplinary Referral and Collaboration
Dermatologists function as key figures in the early detection of metabolic syndrome and diabetes through recognition of cutaneous manifestations [49]. The identification of dermatologic markers of metabolic dysfunction should trigger a systematic approach to interdisciplinary care involving primary care physicians, endocrinologists, and other specialists as appropriate [49,57]. For patients with psoriasis and features of metabolic syndrome, guidelines from the American Academy of Dermatology and National Psoriasis Foundation specifically recommend that dermatologists inform patients of the association and refer them to appropriate healthcare providers for treatment of metabolic components [57].
The Endocrine Society emphasizes that physicians across specialties can screen for metabolic risk factors during routine clinical visits through history and physical examination [44]. Dermatologists encountering cutaneous markers of insulin resistance or metabolic dysfunction should either initiate metabolic screening themselves or facilitate prompt referral to primary care or endocrinology for comprehensive evaluation [48,49]. This collaborative approach ensures that patients benefit from both dermatologic treatment and systemic metabolic management.
Prevention of Systemic Complications
Early identification and treatment of metabolic dysfunction based on dermatologic findings can delay or prevent the onset of type 2 diabetes and reduce cardiovascular risk [40,48]. Individuals with prediabetes identified through screening prompted by cutaneous markers benefit from interventions including lifestyle modification, dietary changes, and pharmacologic therapy when indicated [40]. The Endocrine Society recommends screening for all five components of metabolic risk (elevated blood pressure, increased waist circumference, elevated fasting triglycerides, low HDL cholesterol, and elevated glycemia) in individuals aged 40-75 years, with repeat screening every 3 years in those with at least one risk factor [40].
Recognition of cutaneous markers enables intervention before the development of irreversible complications including diabetic nephropathy, retinopathy, neuropathy, and cardiovascular disease [48]. For patients with severe hypertriglyceridemia manifesting as eruptive xanthomas, urgent triglyceride reduction through dietary fat restriction, insulin therapy, and lipid-lowering medications prevents life-threatening acute pancreatitis [44,45]. Similarly, identification of acanthosis nigricans in adolescents facilitates early lifestyle interventions that may prevent or delay progression to type 2 diabetes [50].

The American Diabetes Association recommends that screening be repeated at 3-year intervals in individuals with normal initial results, with more frequent screening in those with prediabetes or multiple risk factors [36,41]. Patients identified through dermatologic screening should receive comprehensive metabolic evaluation and ongoing monitoring to optimize longterm outcomes and prevent complications [40,51]. To facilitate this systematic approach, a practical clinical algorithm for diagnosing metabolic dysfunction via cutaneous markers is presented in Figure 1.
Additional Considerations for Metabolic Screening
In patients with clinical suspicion of severe insulin resistance (acanthosis nigricans, abnormal adipose distribution, family history of type 2 diabetes, severe fatty liver or dyslipidemia), comprehensive biochemical workup should include paired fasting insulin and glucose, HbA1c, fasting lipid profile, liver function tests, sex hormone-binding globulin (SHBG), liver ultrasound, and consideration of serum leptin and adiponectin levels [44].
For pediatric patients, screening for type 2 diabetes should be considered in youth with overweight (≥85th percentile) or obesity (≥95th percentile) who have acanthosis nigricans or other signs of insulin resistance, with testing initiated at age 10 years or at onset of puberty [50,52]. In these patients, oral glucose tolerance testing may be more sensitive than HbA1c alone for detecting early type 2 diabetes or prediabetes [50].
Patients with eruptive xanthomas require immediate evaluation and should be assessed for secondary causes of hypertriglyceridemia, including uncontrolled diabetes, hypothyroidism, nephrotic syndrome, alcohol use, and medications that may elevate triglycerides [55,56]. The urgency of this evaluation cannot be overstated, as triglyceride levels exceeding 2000 mg/dL carry substantial risk for acute pancreatitis [55]. A summary of the suggested laboratory workup for each of these dermatologic findings is provided in (Table 1) [58-61].
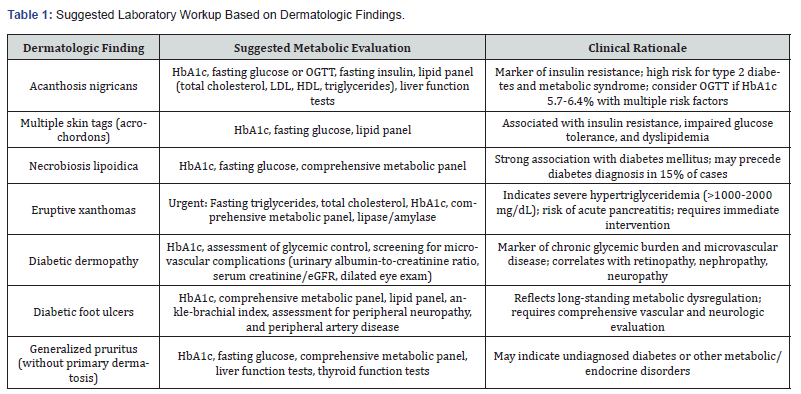
Abbreviations: ABI: Ankle-Brachial Index; eGFR: Estimated Glomerular Filtration Rate; HbA1c: Glycated Hemoglobin; HDL: High-Density Lipoprotein; LDL: Low-Density Lipoprotein; OGTT: Oral Glucose Tolerance Test. Adapted from references [51-60].
Conclusion
The skin is not merely a passive target of metabolic disease but a highly accessible, real-time indicator of a patient’s internal metabolic status. Manifestations such as acanthosis nigricans, diabetic dermopathy, and eruptive xanthomas provide distinct “cutaneous clues” that reflect specific pathophysiologic processes-namely, insulin resistance, microvascular injury, and severe dyslipidemia. As the global metabolic crisis intensifies, the ability to recognize these markers becomes a vital clinical skill. By integrating dermatological physical findings into a proactive screening framework, clinicians can identify metabolic dysfunction years before irreversible complications arise. Transitioning from a dermatologic diagnosis to a multidisciplinary care modellinking dermatology, primary care, and endocrinology-is the most effective strategy to alter the disease course of diabetes and metabolic syndrome.
References
- International Diabetes Federation (2024) IDF Diabetes Atlas. 11th ed International Diabetes Federation.
- Despres M (2025) Worldwide trends in metabolic syndrome from 2000 to 2023: a systematic review and modelling analysis. Nat Commun 16(1): 67268.
- Rodríguez CM (2017) Skin manifestations of insulin resistance: from a biochemical stance to a clinical diagnosis and management. Dermatol Ther (Heidelb) 7(1): 37-51.
- Wollina U (2025) Skin manifestations of diabetes mellitus- what is new? Front Med (Lausanne) 12: 1640144.
- Napolitano M (2025) Cutaneous manifestations of diabetes mellitus: a narrative review. JAAD Int 18: 11908747.
- Vâță D (2023) Cutaneous manifestations associated with diabetes mellitus- a retrospective study. Diseases 11(3): 106.
- Napolitano M (2025) Dermatological manifestations of diabetes mellitus and its complications. Dermatopathology 6(3): 18.
- Napolitano M, Megna M, Monfrecola G (2015) Insulin resistance and skin diseases. Scientific World Journal 2015: 479354.
- Gisondi P, Bellinato F, Targher G, Girolomoni G (2022) Psoriasis and metabolic syndrome: mechanisms and clinical implications. J Clin Med 11(9): 2434.
- Vahlquist Å, Törmä H, Carlsson T (2023) Role of insulin and IGF-1 in epidermal proliferation and differentiation. Cells 12(4): 623.
- Chen C (2022) Advanced glycation end products in the skin: mechanisms and pathological effects. Biomedicines 10(5): 1062.
- Gkogkolou P, Böhm M (2012) Advanced glycation end products: key players in skin aging? Dermato-endocrinal 4(3): 259-270.
- Yamagishi S, Matsui T (2022) Role of AGE-RAGE signaling in inflammatory and vascular complications of diabetes. Int J Mol Sci 23(12): 6741.
- Beckman JA, Creager MA (2022) Vascular complications of diabetes: mechanisms and clinical implications. Circ Res 130(8): 1216-1235.
- Sørensen BM, Houben AJHM, Berendschot TTJM (2023) Microvascular dysfunction in insulin resistance and type 2 diabetes. Diabetologia 66(1): 1-13.
- Veves A, Akbari CM, Primavera J (2022) Diabetic microangiopathy in the skin: pathophysiology and clinical relevance. J Diabetes Complications 36(4): 108145.
- Berglund L, Brunzell JD, Goldberg AC (2022) Hypertriglyceridemia: pathophysiology and clinical implications. Endocr Rev 43(4): 679-720.
- Zuber J, Lemaire R, Bruckert E (2023) Xanthomas and lipid metabolism disorders: clinical and molecular insights. Front Cardiovasc Med 10: 1158702.
- Toth PP, Banach M (2022) Lipid disorders and cutaneous manifestations. J Clin Lipidol 16(6): 799-812.
- Schwartz RA (1994) Acanthosis nigricans. J Am Acad Dermatol 31(1): 1-19.
- Sinha S, Schwartz RA (2007) Juvenile acanthosis nigricans. J Am Acad Dermatol 57(3): 502-508.
- Phiske MM (2014) An approach to acanthosis nigricans. Indian Dermatol Online J 5(3): 239-249.
- Kong AS, Williams RL, Smith M (2007) Acanthosis nigricans and diabetes risk factors. J Pediatr 150(6): 652-657.
- American Diabetes Association Professional Practice Committee (2024) Standards of care in diabetes-2024. Diabetes Care 47(suppl 1): S1-S350.
- Rasi A, Soltani AR, Shahbazi N, Aryanejad F (2007) Skin tag as a cutaneous marker for impaired carbohydrate metabolism. Acta Diabetol 44(4): 222-225.
- Tamega AA, Aranha AM, Guiotoku MM, Miot LDB, Miot HA, et al. (2010) Association between skin tags and insulin resistance. An Bras Dermatol 85(1): 25-31.
- Sudhakar RGS, Pai GS, Bhat M (2013) Skin tags as markers of insulin resistance. J Clin Diagn Res 7(12): 2858-2861.
- Lowitt MH, Dover JS (1991) Necrobiosis lipoidica. J Am Acad Dermatol 25(5): 735-748.
- Müller SA, Winkelmann RK (1966) Necrobiosis lipoidica diabeticorum: a clinical and pathological investigation of 171 cases. Arch Dermatol 93(3): 272-281.
- Cohen O, Yaniv R, Karasik A, Trau H (1998) Microangiopathy in necrobiosis lipoidica. Diabetes Care 21(3): 419-420.
- Morgan AJ, Schwartz RA (2008) Diabetic dermopathy: a subtle sign with grave implications. J Am Acad Dermatol 58(3): 447-451.
- Shemer A, Bergman R, Linn S, Kantor Y, Friedman BR, et al. (1998) Diabetic dermopathy and internal complications in diabetes mellitus. Int J Dermatol 37(2): 113-115.
- Lima AL, Illing T, Schliemann S, Elsner P (2017) Cutaneous manifestations of diabetes mellitus: a review. Am J Clin Dermatol 18(4): 541-553.
- Marinescu M, Botnariu GE, Vâță D (2025) Diabetic dermopathies as predictive markers of cardiovascular and renal complications: a narrative review. J Clin Med 14(21): 7719.
- Demirseren DD, Emre S, Akoglu G (2014) Relationship between skin diseases and extracutaneous complications of diabetes mellitus: clinical analysis of 750 patients. Am J Clin Dermatol 15(1): 65-70.
- Berglund L, Brunzell JD, Goldberg AC (2012) Evaluation and treatment of hypertriglyceridemia: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 97(9): 2969-2989.
- Marogi EP, Ohiomoba RO, Stone NJ (2022) Eruptive xanthomas: importance of recognition to reduce delay of effective triglyceride reduction. Am J Med 135(6): e163-e164.
- Kashif M, Kumar H, Khaja M (2016) An unusual presentation of eruptive xanthoma: a case report and literature review. Medicine (Baltimore) 95(6): e2630.
- Leaf DA (2008) Chylomicronemia and the chylomicronemia syndrome: a practical approach to management. Am J Med 120(1): 10-12.
- Saadatagah S, Larouche M, Naderian M (2025) Recognition and management of persistent chylomicronemia: a joint expert clinical consensus by the National Lipid Association and the American Society for Preventive Cardiology. J Clin Lipidol 19(1): 12-34.
- Stark M, Stuart J (2018) Eruptive xanthoma in the setting of hypertriglyceridemia and pancreatitis. Am J Emerg Med 36(8): 1526.e5-1526.e6.
- Armstrong DG, Tan TW, Boulton AJM, Bus SA (2023) Diabetic foot ulcers: a review. JAMA 330(1): 62-75.
- Aditya C, Bukke SPN, Anitha K (2025) A comprehensive review on diabetic foot ulcer addressing vascular insufficiency, impaired immune response, and delayed wound healing mechanisms. Front Pharmacol 16: 1421033.
- Singer AJ, Tassiopoulos A, Kirsner RS (2017) Evaluation and management of lower-extremity ulcers. N Engl J Med 377(16): 1559-1567.
- Jeffcoate W, Boyko EJ, Game F (2024) Causes, prevention, and management of diabetes-related foot ulcers. Lancet Diabetes Endocrinol 12(7): 472-482.
- Gallagher KA, Mills JL, Armstrong DG (2024) Current status and principles for the treatment and prevention of diabetic foot ulcers in the cardiovascular patient population: a scientific statement from the American Heart Association. Circulation 149(4): e232-e253.
- Das SR, Bonaca MP, Creager MA (2025) Management of peripheral artery disease in adults with diabetes: 2025 ACC scientific statement: a report of the American College of Cardiology. J Am Coll Cardiol 85(4): 442-468.
- Karadag AS, Ozlu E, Lavery MJ (2018) Cutaneous manifestations of diabetes mellitus and the metabolic syndrome. Clin Dermatol 36(1): 85-93.
- Uzuncakmak TK, Akdeniz N, Karadag AS (2017) Cutaneous manifestations of obesity and the metabolic syndrome. Clin Dermatol 36(1): 81-88.
- Hannon TS, Arslanian SA (2023) Obesity in adolescents. N Engl J Med 389(3): 251-261.
- Blonde L, Umpierrez GE, Reddy SS (2022) American Association of Clinical Endocrinology clinical practice guideline: developing a diabetes mellitus comprehensive care plan-2022 update. Endocr Pract 28(10): 923-1049.
- American Diabetes Association Professional Practice Committee (2025) 2. Diagnosis and classification of diabetes: standards of care in diabetes-2026. Diabetes Care 49(suppl 1): S27-S49.
- Department of Veterans Affairs, Department of Defense (2023) VA/DoD Clinical Practice Guideline for the Management of Type 2 Diabetes Mellitus. Department of Veterans Affairs.
- Elmets CA, Leonardi CL, Davis DMR (2019) Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with awareness and attention to comorbidities. J Am Acad Dermatol 80(4): 1073-1113.
- Rosenzweig JL, Bakris GL, Berglund LF (2019) Primary prevention of ASCVD and T2DM in patients at metabolic risk: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 104(9): 3939-3985.
- American Diabetes Association Professional Practice Committee (2025) 4. Comprehensive medical evaluation and assessment of comorbidities: standards of care in diabetes-2026. Diabetes Care 49(suppl 1): S52-S78.
- Elhassan YS, Hawley JM, Cussen L (2025) Society for Endocrinology clinical practice guideline for the evaluation of androgen excess in women. Clin Endocrinol (Oxf) 103(4): 540-566.
- Zhang H, Zhou XD, Shapiro MD (2024) Global burden of metabolic diseases, 1990-2021. Metabolism 160: 155999.
- Vlahovic TC (2025) Skin manifestations of diabetes mellitus. In: Feingold KR, Anawalt B, Blackman MR, et al, eds. Endotext MDText.com, Inc.
- Quartier J (2026) Insulin resistance, metabolic syndrome, and inflammatory skin disease. J Clin Med 15(1): 330.
- Sacks DB, Arnold M, Bakris GL (2023) Guidelines and recommendations for laboratory analysis in the diagnosis and management of diabetes mellitus. Diabetes Care 46(10): e151-e199.






























