Abstract
Insulin resistance (IR) is a complex metabolic disorder with profound implications for human health, extending beyond glucose regulation. This review provides a comprehensive examination of IR, encompassing its physiological mechanisms, associated hazards, and implications for conditions such as type 2 diabetes, cardiovascular disease (CVD), hypertension, polycystic ovary syndrome (PCOS), dyslipidemia, non-alcoholic fatty liver disease (NAFLD), and cancer risk. Emphasizing the urgent need for targeted interventions, including lifestyle modifications and pharmacological therapies, this review underscores the importance of understanding the intricate interplay between IR and systemic health outcomes
Keywords: Insulin Resistance; Metabolic Disorders; Cardiovascular Disease; Polycystic Ovary Syndrome (PCOS); Non-Alcoholic Fatty Liver Disease (NAFLD)
Abbrivation: IR: Insulin resistance; CVD: cardiovascular disease; PCOS: polycystic ovary syndrome; NAFLD: non-alcoholic fatty liver disease; ROS: Reactive oxygen species; FFA: free fatty acids; CETP: cholesteryl ester transfer protein; HTGL: hepatic triglyceride lipase; RAS: renin-angiotensin system; ASCVD: atherosclerotic cardiovascular disease; LDL: low-density lipoprotein; HDL: high-density lipoprotein; NGT: normal glucose-tolerant; ARIC: Atherosclerosis Risk in Communities
Introduction
Insulin resistance, which is a complicated metabolic disorder, has consequences that go far beyond just the change in blood sugar. But as complicated as it sounds, this condition links multiple physiological pathways, which makes a huge difference in our well-being. Although insulin resistance itself is the major cause, it refers to the decreased reaction of cells to the hormone insulin, which is one of the key players in glucose metabolism and regulation. This process results into the body’s inefficiency in utilizing the glucose for energy or storing it for later use, leading to a series of metabolic disruptions. Insulin resistance does not affect glucose dysregulation only; it is one of the factors in metabolic syndrome development, which is a set of interconnected conditions including central obesity, dyslipidemia, and hypertension. This condition brings with it the tendency of people to a higher predisposition to cardiovascular diseases including heart attacks and strokes as well as type 2 diabetes. In addition to this, insulin resistance has been considered as a major factor in the development of polycystic ovary syndrome (PCOS), which is a very common endocrine disorder found among premenopausal women. PCOS is a condition where hormones are out of balance, menstrual cycles are irregular or there is infertility, all of which are caused by the presence of insulin resistance. The teasing out of the intricate mechanisms of insulin resistance would be of paramount importance in the design of specific therapeutic interventions and preventive measures. Lifestyle changes, such include eating a balanced diet and regular physical activity, proved to be effective in the reduction of insulin resistance and its complications. Particularly, on-going research is focused on figuring out if insulin sensitizers and novel drug targets which aim at the mechanism of insulin resistance can be helpful in the management of various organ systems. Through gaining the insights into the complex intrinsic relationship between insulin resistance and the systemic ramifications that occur in metabolic, cardiovascular, and reproductive health, researchers and healthcare professionals become able to design personalized, individual, and holistic approaches to disease treatment and prevention.
Objective
The objective of this comprehensive review is to provide a thorough examination of insulin resistance, its associated hazards, and its implications for human health. By synthesizing existing research, this review aims to elucidate the multifaceted nature of insulin resistance, including its physiological mechanisms, risk factors, and downstream health consequences. Additionally, it seeks to explore potential strategies for prevention, management, and treatment of insulin resistance-related conditions.
Methods
Literature Search: Conduct a systematic search of electronic databases (such as PubMed, Google Scholar, and Scopus) using relevant keywords including “insulin resistance,” Type 2 diabetes, cardiovascular disease, Hypertension, PCOS, Dyslipidemia, NAFLD and increase risk of cancer.
Criteria: Include studies published in peer-reviewed journals that investigate insulin resistance in human subjects. Exclude studies that do not directly address the hazards or consequences of insulin resistance. Studies published in languages other than English will also be excluded unless an English translation is available. Summaries findings from selected studies to provide a comprehensive overview of the hazards associated with insulin resistance. Organise information according to themes such as Type 2 diabetes, cardiovascular disease, hypertension, PCOS, dyslipidemia, NAFLD and increased risk of cancer.
Results
Cardiovascular disease
Hyperglycemia and compensatory hyperinsulinemia are two aspects of the disease known as insulin resistance. It happens when the liver, adipose tissue, and skeletal muscles are among the target areas where insulin is unable to have the fullest impact. The development of cardio-metabolic diseases, such as obesity, dyslipidemia, low-grade inflammation, endothelial dysfunction, and hypertension, is caused by this modification of insulin signaling pathways. These conditions are risk factors for atherosclerosis and cardiovascular disease [1]. Thus, insulin resistance raises the risk of CVD through a variety of routes. We will discuss each route in more detail later, but for now, let’s concentrate on endothelial dysfunction and low-grade inflammation.
The role of insulin in inflammation and vasodilation: insulin resistance is linked to atherogenesis [2]. Insulin reduces inflammation in the body through its effects on macrophages and endothelial cells. Insulin promotes the production of endothelial nitric oxide synthase (eNOS) in endothelial cells. Nitric oxide (NO), which causes vasodilation, is released by eNOS. Endothelial cells’ intracellular nuclear factor-kappa-B (NF-kB) is suppressed by insulin. Reactive oxygen species (ROS) and O2 radical production are inhibited by insulin [3]. Mechanisms by which hyperglycemia induces endothelial damage due to ROS. Elevated glucose levels primarily cause vascular damage due to an imbalance in the endothelium’s generation of ROS and NO bioavailability. These are results of atherosclerosis of coronary vessels and May end with acute coronary syndrome due to rupturing of atherosclerosis of the coronary artery.
Diabetes mellitus
The development of T2D is linked to ROS generation, mitochondrial malfunction, ER stress, and changes in autophagy, all of which are essential for β-cell activity. In these cells, ROS and ROS-induced disruptions are mostly responsible for the start of diabetes. The primary role of β-cells is to respond to rises in blood glucose concentration by secreting insulin, which helps the body maintain proper glucose homeostasis. In short, glucose produces NADH upon entry, which is facilitated by GLUT2 (glucose transporter-2). This is the physiological mechanism of insulin release. The mitochondrial ETC metabolizes this NADH to produce ATP. In addition, the mitochondrial membrane potentialdependent Ca2+ uniporter can be activated by hyperpolarization of the inner membrane of the mitochondria, which will raise mitochondrial Ca2+ and promote TCA cycle activity. The exocytosis of insulin-containing granules is eventually caused by an increase in the ATP/ADP ratio, which also causes the plasma membrane to depolarize, voltage-dependent Ca2+ channels to open, and ATPsensitive K+ channels to close.
Due to these characteristics, β-cells are subjected to elevated glucose levels, which in turn exposes them to the harm caused by reactive oxygen species (ROS) resulting from hyperglycemia (Figure 1) [4-6].
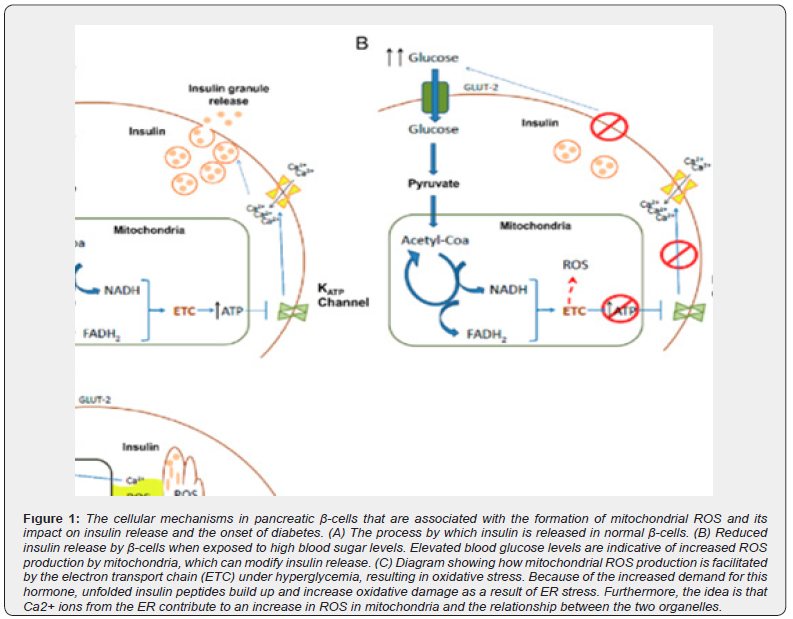
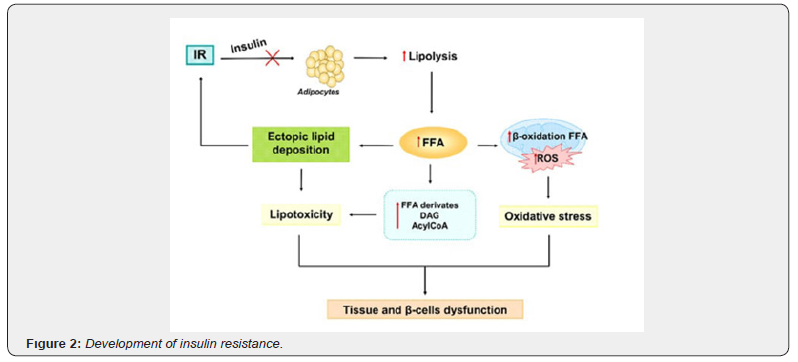
At the vascular level, inflammation plays an important role in microvascular complications and lipotoxicity is strongly associated with macrovascular complications [7]. Thus, it is evident that the pathophysiology of diabetes and insulin resistance includes fat as a risk factor. Furthermore, an additional result of inadequate insulin response is elevated blood levels of free fatty acids (FFA), primarily because insulin’s antilipolytic function on adipocytes is disrupted. In response, a significant amount of free fatty acid (FFA) is released into the bloodstream by fat cells. This leads to the onset of systemic lipotoxic consequences, such as ectopic lipid deposition and consequent disruption of insulin signaling. Thus, lipotoxicity has been identified as a major factor in the development of insulin resistance recently [8] (Figure 2).
Dyslipidemia
Insulin resistance affects all components of the lipid profile, including HDL, LDL, VLDL, TG and chylomicron, here we talk about all about separately.
a) TG and VLDL metabolism of IR has a significant impact on the metabolism of VLDL, including effects that raise the synthesis of TG in the liver. There is then varying evidence linking increased hepatic apo B-100 production to enhanced VLDL TG synthesis. Moreover, it has been suggested recently that HTGL activity is a key regulator of insulin clearance [9- 12].
b) Chylomicrons metabolism in the postprandial state, chylomicrons, which are produced and secreted by the colon, facilitate the transfer of TG obtained from meals to various organs. While apo B-100 is present in VLDL, the only apo B found in chylomicrons is apo B-48, which is a shortened version of the holoprotein. Chylomicrons are digested by LPL in the vasculature, releasing their fatty acids to nearby cells. Hydrolysis of chylomicron TGs is similarly influenced by the IR-related decrease in LPL activity. This is especially clear if all of the endothelium’s accessible LPL binding sites are saturated by high levels of hepatic VLDL [13-15].
c) HDL metabolism of Insulin has a significant role in HDL metabolism, and IR states are frequently associated with low HDL-C concentrations. Low HDL-C concentrations are hypothesized to be caused by IR through a number of different ways. First, IR is linked to an increased exchange of VLDL for cholesterol esters from HDL particles and TG from chylomicrons, which lowers HDL-C. This process is controlled by the cholesteryl ester transfer protein (CETP). Second, lower LPL activity leads to less TG being hydrolyzed from VLDL and chylomicrons, which may further restrict the amount of TG-rich lipoprotein-derived HDL particles. Third, Moreover,hepatic triglyceride lipase (HTGL) elevations are linked to IR and may cause HDL to be cleared more quickly and HDL-C levels to drop. Fourth, decreased apo A-I production and release from the liver and intestine may also be the cause of low HDL-C concentrations [16-19].
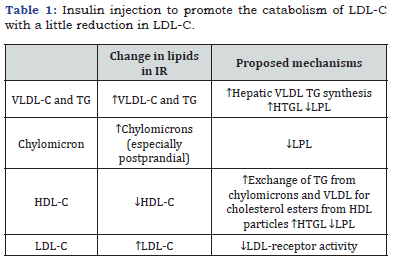
d) LDL metabolism of IR seems to have a less significant impact on LDL metabolism than it does on VLDL metabolism. Insulin injection has been shown to promote the catabolism of LDL-C with a little reduction in LDL-C. Insulin is known to upregulate LDL receptor function. (Table 1) provides a summary of these [20-23].
PCOS
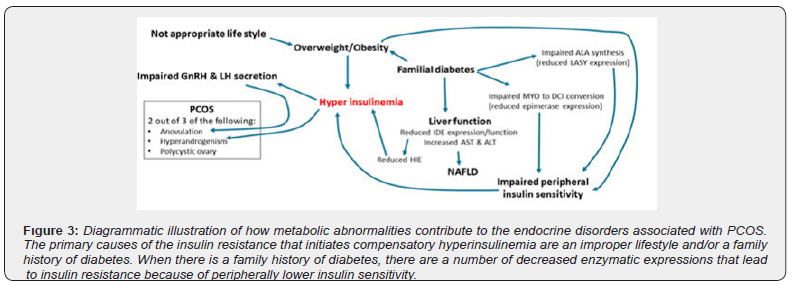
Polycystic ovarian syndrome (PCOS) is characterized by high androgen levels, irregular ovulation, and morphological abnormalities. PCOS affects 6-10% of women in their reproductive stages, with a potential twofold prevalence rate. Numerous pathophysiological variables contribute significantly to the anomalies observed in each patient with PCOS. PCOS causes a notable increase in androgen, which leads to significant suffering and issues with infertility. Insulin resistance and hyperinsulinemia are directly associated with androgen overexposure [24].
Insulin resistance in PCOS has a strong relation with Anovulation, reduced SHBG levels, increased androgens, especially free testosterone, increased truncal-abdominal fat mass, and high free fatty acid levels. It is still unclear what causes PCOS-related insulin resistance. Insulin resistance in women with PCOS may be caused by an increase in truncal-abdominal fat mass and consequent elevation in free fatty acid levels, according to one line of evidence. The abnormal body fat distribution appears to be caused by increased corticosteroid effects and a relative decrease in estrogen and progesterone [25]. This is summed up in (Figure 3) [26].
Hypertension
A number of metabolic disorders, including hyperglycemia and the production of excess free fatty acids, are brought on by insulin resistance. These disorders also affect the vascular wall by causing endothelial dysfunction, platelet hyperactivity, oxidative stress, and low-grade inflammation. When these processes are activated, vasoconstriction is further enhanced and thrombus formation is encouraged, which eventually leads to the development of atherosclerosis. Clinical research demonstrating the significance of platelet hyperactivity and endothelial dysfunction in the pathophysiology of atherosclerotic vascular problems, corroborate all of these findings [27]. Furthermore, Endothelial dysfunction and the emergence of hypertension have been linked to changes in both ROS generation and mitochondrial dynamics [28]. Endothelial dysfunction is independently linked to an elevated risk of cardiovascular disease and may have a role in the onset and progression of vascular inflammation, vascular remodeling, and atherosclerosis in hypertension [29]. Many complicated disorders have been related to mitochondrial dysfunction as its etiology, but many other factors have also been implicated in the pathophysiology of hypertension, such as vitamin D insufficiency and the activation of the renin-angiotensin system (RAS) [30].
Increase risk cancer
Insulin resistance is a global risk factor for the two most serious categories of human diseases: cancer and cardiovascular diseases. The five primary criteria for the insulin resistance syndrome include visceral obesity, low HDL cholesterol (dyslipidemia), excessive serum triglyceride levels, low blood sugar, and high blood pressure. When combined, these characteristics provide a multifactorial risk for cancer, as each is a risk factor in and of itself. Because of the increased beta cell secretory activity caused by insulin resistance in the liver, skeletal muscles, and adipose tissue, reactive hyperinsulinemia results. Insulin has diverse metabolic effects, and at the same time is a growth factor. It causes abnormal cell proliferation and increases the synthesis and mitogenic activity of other growth factors that resemble insulin. Hyperglycemia, which arises in the uncompensated phase of insulin resistance, is known to promote tumor genesis through many routes. The raised blood glucose level benefits the tumor cells’ enhanced DNA production. It initiates the production of free radicals, which destabilize DNA and the enzymes involved in the repair processes [31]. A plethora of research in recent years has demonstrated the synergistic association that exists between cancer, type 2 diabetes mellitus, and IR. While the actual reason or mechanisms for this relationship are yet unknown, it is widely known that hyperinsulinemia, a characteristic of insulin resistance, may contribute to carcinogenesis. However, IR is closely linked to systemic inflammation and dysfunctional visceral adiposity, two factors that promote the development of a pro-tumorigenic environment [32]. Coffee drinking and physical exercise are inversely linked to these malignancies, as these factors are known to lower the risk of type 2 diabetes. Insulin resistance, chronic hyperinsulinemia that results, and a rise in bioavailable insulin-like growth factor 1 (IGF1) are thought to be the mechanisms underlying these effects [33].
NAFLD
NASH (fat accumulation with hepatocellular damage and inflammation), liver cirrhosis, hepatocellular cancer, and simple hepatic steatosis are all examples of the wide spectrum of nonalcoholic fatty liver disease (NAFLD) [34]. Hepatic steatosis, a liver malfunction brought on by obesity, contributes to the development of NAFLD. Hyperinsulinemia and dyslipidemia are more severe in obese people with concurrent type 2 diabetes and NAFLD than in those without NAFLD [35]. Insulin resistance is caused by the accumulation of oxidized and excess fatty acids in peripheral organs such as the liver and adipose tissue. These fatty acids are produced via lipogenesis and fatty acid synthesis.[36] A modulator of systemic lipid accumulation is adipose tissue; it secretes cytokines such as Adiponectin. Additionally, adiponectin sustains hepatic insulin sensitivity and overall glucose homeostasis in the body.[37] In the development of non-alcoholic fatty liver disease (NAFLD) or type 2 diabetes, hypoadiponectinemia hinders fatty acid metabolism and encourages a chronic inflammatory state in the liver [38,39]. Therefore, keeping adiponectin levels stable may help patients with non-alcoholic fatty liver disease avoid fibrosis and inflammation. Adipose tissue grows as a result of malnutrition or inadequate exercise, and the hypertrophic adipocytes that result secrete TNF-α, IL-1 β, and IL-6. Through the stimulation of pro-inflammatory signaling and the blockage of insulin receptor signaling, these pro-inflammatory cytokines reduce the hepatic insulin sensitivity. As a result, fibrosis and steatosis of the liver develop [40].
Discussion
An inadequate physiologic response to insulin is known as insulin resistance. It is a defining trait of diabetes mellitus type 2. It is associated with several deleterious consequences such as cardiovascular diseases, hypertension and PCOS.
Cardiovascular disease
Patients with type 2 diabetes have a higher risk of cardiovascular disease due to a number of cardio-metabolic risk factors that are linked to insulin resistance. Insulin resistance has been found to be a robust predictor of atherosclerotic cardiovascular disease (ASCVD) in several studies [41-52]. recently summarized these findings in a meta-analysis. The Insulin Resistance Atherosclerosis Study was the first epidemiologic study to document the relationship between insulin resistance and CVD in a large multi-ethnic cohort [45], after correcting for confounding factors such as glucose tolerance, fasting insulin, low-density lipoprotein (LDL) and high-density lipoprotein (HDL) cholesterol, smoking, hypertension, and body mass index. Bressler et al. [43] used the euglycemic insulin clamp to conclusively demonstrate that normal glucose-tolerant (NGT) individuals with diffuse coronary artery disease were significantly more insulinresistant than NGT individuals with clean coronary arteries.
Similarly, throughout a follow-up period of 6.9 years in the Bothnia research [46], insulin resistance was an independent predictor of a higher risk of CVD in participants who were not diabetic. The Verona Diabetes research [47], the Bruneck research [48], the Malmö study [49], and the Atherosclerosis Risk in Communities (ARIC) study [51] have all reported similar findings. The strong correlation observed between ASCVD and insulin resistance can be explained by three mechanisms: (i) the fundamental molecular etiology of the resistance [43-62] (ii) the compensatory hyperinsulinemia resulting from the resistance [60,63-69]; and (iii) the correlation between the resistance and a group of cardiometabolic abnormalities, each of which is a separate risk factor for ASCVD [63-65,69-71] Although the use of drugs like statins, angiotensin-converting enzyme inhibitors, other antihypertensive drugs, and platelet inhibitory agents has decreased the incidence of atherosclerotic CV complications,there are still unknown CV risk factors in addition to the traditional risk factors that raise the risk of cardiovascular disease in patients receiving optimal treatment. The majority of the time, medical therapy targets one or more CV risk factors rather than the underlying pathophysiological defect-insulin resistance-that gives rise to the cardiometabolic abnormalities primarily.
This is demonstrated by a new Publication from the National Swedish Registry [72], which shows that between 1998 and 2014, the rate of CV death in people with T2DM decreased dramatically, although it remained noticeably higher and plateaued above NGT individuals. Conversely, Insulin resistance was found to be independently correlated with the coronary calcium score, a robust indicator of coronary artery disease, in a retrospective examination of 10,153 occupational individuals. This correlation was maintained even after controlling for other cardiovascular risk variables and previous cardiovascular disease [73]. Early identification of insulin resistance greatly reduces liver and cardiac damage.
Hypertension
The processes by which insulin resistance affects blood pressure are not entirely understood. High insulin secretion in response to insulin resistance has been shown to impact renal sodium excretion[74-76] the sympathetic nervous system [77], and the renin-angiotensin-aldosterone system.[78, 79] Insulin resistance has been linked to poor endothelial-dependent vasodilation, atherosclerosis, and inflammation.[80] Over time, the deleterious consequences of insulin resistance on the vasculature may lead to blood pressure increase, and as revealed in the current study, this appears to be independent of diabetes status. The link between insulin resistance and DBP in people with normal blood glucose levels shows that insulin resistance may alter DBP before hyperinsulinemia, which is linked with prediabetes. These findings may help explain why DBP is the biggest predictor of coronary heart disease in young and middle-aged adults. [81,82] Previous research indicates a potential relationship between BMI and IR in predicting hypertension. Lytsi et al. (n=1846) found that those with IR and normal BMI were not at a higher risk of hypertension. However, those with IR and overweight/obesity had a higher chance of developing hypertension [74]. A Greek study (n=141) found that women with IR have a 98% higher risk of hypertension. However, when the model was adjusted for fat, the association was no longer significant [75].
NAFLD, IR and liver disease association
Numerous studies have demonstrated the close relationship between metabolic disorders, including obesity, insulin resistance, type 2 diabetes mellitus, and hypertriglyceridemia, and non-alcoholic fatty liver disease (NAFLD). As a result, NAFLD is thought to be the liver’s manifestation of metabolic syndrome [83-85]. Because it permits the liver to store free fatty acids,insulin resistance plays a significant role in the pathophysiology of non-alcoholic fatty liver disease (NAFLD) [86-88]. The rise in insulin resistance in industrialized nations is one of the main causes of the increased prevalence of NAFLD [83]. According to reports, NAFLD was identified in over 75% of diabetic patients. [85] According to [89], it may contribute to hepatic fibrosis by either causing insulin to overstimulate connective tissue growth factor or causing liver stellate cells to be incubated with glucose. Research indicates that the persistent inflammation brought on by insulin resistance causes inflammatory markers to be elevated in individuals with non-alcoholic fatty liver disease [90, 91]. While some studies express ideas to the contrary, the majority of studies demonstrate that insulin resistance has a prognostic value for fibrosis [92-96] It has long been known that insulin resistance and obesity are related [97]. Furthermore, strong evidence points to the presence of metabolic dysfunction as an additional risk factor for the advancement of liver disease and extrahepatic clinical consequences [98-102].
Reproductive health and insulin resistance
“Excess insulin shifts the sex hormone balance to promote testosterone production over estrogen production,” is one of how insulin resistance relates to infertility. Furthermore, current research indicates that elevated average blood sugar levels may affect the quality of eggs. It can even prevent ovulation. [103] found that SHBG had a strong inverse correlation with IR in PCOS when compared to controls. This is in line with mechanistic research showing that insulin resistance (IR) and hyperinsulinemia [103] inhibit the formation of SHBG and that treatment with insulin sensitizers raises SHBG concentrations [104]. The research validates the use of SHBG in PCOS-affected women as a straightforward clinical indicator of IR. We further verify that, in comparison to controls, SHBG is not only lower but also less variable in PCOS [105] However, larger research is now required to determine precise SHBG cut-off values in order to predict IR. Moreover, decreased SHBG is linked, independently of obesity, to metabolic syndrome, diabetes, and unfavorable cardiovascular risk factors. Elevated “free testosterone” or the computed free androgen index, which incorporates SHBG in the calculation, is the most frequently seen androgenic aberration in PCOS [106] This presents a confounding factor, and since IR significantly affects SHBG, studies on the connections between androgens and IR have to be conducted apart from SHBG. Here, the studies that provided total testosterone levels were the main focus. This contrasts with the way we currently think about how androgens and IR interact [107] But, care must be taken when interpreting these results because the majority of the androgens were measured using radioimmunoassay (n = 10) and chemiluminescence immunoassays (n = 9). These methods are less sensitive than mass spectrometry (n = 2) in identifying androgen levels in women [108] Greater understanding of the interactions between hyperandrogenism and IR in PCOS may be attained as we move towards more precise techniques. [109-111]
Conclusion
In conclusion, insulin resistance (IR) is a multifaceted metabolic disorder with far-reaching implications for human health. This review has comprehensively examined the physiological mechanisms underlying IR and its association with various health hazards, including type 2 diabetes, cardiovascular disease, hypertension, polycystic ovary syndrome (PCOS), dyslipidemia, non-alcoholic fatty liver disease (NAFLD), and increased cancer risk. Through an exploration of the intricate interplay between IR and systemic health outcomes, it becomes evident that targeted interventions, such as lifestyle modifications and pharmacological therapies, are urgently needed. By understanding the complex relationship between IR and its associated health risks, researchers and healthcare professionals can develop personalized approaches to disease prevention and treatment, thereby improving the overall well-being of individuals affected by this condition.
References
- Kosmas CE, Bousvarou MD, Kostara CE, Papakonstantinou EJ, Salamou E, et al. (2023) Insulin resistance and cardiovascular disease. J Int Med Res 51(3):3000605231164548.
- Dandona P, Aljada A, Mohanty P, Ghanim H, Hamouda W, et al. (2001) Insulin inhibits intranuclear nuclear factor kappaB and stimulates IkappaB in mononuclear cells in obese subjects: evidence for an anti-inflammatory effect? J Clin Endocrinol Metab 86(7): 3257-3265.
- Vargas E, Joy NV, Carrillo SMA (2024) Biochemistry, Insulin Metabolic Effects. StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing.
- Lenzen S, Drinkgern J, Tiedge M (1996) Low antioxidant enzyme gene expression in pancreatic islets compared with various other mouse tissues. Free Radic Biol Med 20: 463-466.
- Tiedge M, Lortz S, Drinkgern J, Lenzen S (1997) Relation between antioxidant enzyme gene expression and antioxidative defense status of insulin-producing cells. Diabetes 46(11): 1733-1742.
- Robertson RP, Harmon J, Tran PO, Tanaka Y, Takahashi H (2003) Glucose toxicity in beta-cells: Type 2 diabetes, good radicals gone bad, and the glutathione connection. Diabetes 52: 581-587.
- Dinesh SA, Langenberg C, Rapsomaniki E, Denaxas S, Pujades RM, (2015) Type 2 diabetes and incidence of a wide range of cardiovascular diseases: A cohort study in 1.9 million people. Lancet 3(2): 105-113.
- Weinberg JM (2006) Lipotoxicity. Kidney Int 70(9): 1560-1566.
- Grundy SM, Mok HY, Zech L, Steinberg D, Berman M, et al. (1979) Transport of very low-density lipoprotein triglycerides in varying degrees of obesity and hypertriglyceridemia. J Clin Invest 63(6): 1274-1283.
- Kissebah AH, Alfarsi S, Adams PW (1981) Integrated regulation of very low-density lipoprotein triglyceride and apolipoprotein-B kinetics in man: normolipemic subjects, familial hypertriglyceridemia and familial combined hyperlipidemia. Metabolism 30(9): 856-868.
- Sparks JD, Sparks CE, Adeli K (2012) Selective hepatic insulin resistance, VLDL overproduction, and hypertriglyceridemia. Arterioscler Thromb Vasc Biol 32(9): 2104-2112.
- Baynes C, Henderson AD, Anyaoku V, Richmond W, Hughes CL, et al. (1991) The role of insulin insensitivity and hepatic lipase in the dyslipidaemia of type 2 diabetes. Diabet Med 8(6): 560-566.
- Haas ME, Attie AD, Biddinger SB (2013) The regulation of ApoB metabolism by insulin. Trends Endocrinol Metab 24(8): 391-397.
- Goldberg IJ, Eckel RH, Abumrad NA (2009) Regulation of fatty acid uptake into tissues: lipoprotein lipase- and CD36-mediated pathways. J Lipid Res 50 Suppl: S86-S90.
- Brown CM, Layman DK (1988) Lipoprotein lipase activity and chylomicron clearance in rats fed a high fat diet. J Nutr 118(11): 1294-1298.
- Garg A (1996) Insulin resistance in the pathogenesis of dyslipidemia. Diabetes Care 19(4): 387-389.
- Garg A, Haffner SM (1996) Insulin resistance and atherosclerosis. Diabetes Care 19(3): 274.
- de Vries R, Borggreve SE, Dullaart RP (2003) Role of lipases, lecithin: cholesterol acyltransferase and cholesteryl ester transfer protein in abnormal high density lipoprotein metabolism in insulin resistance and type 2 diabetes mellitus. Clin Lab 49(11-12): 601-613.
- Brinton EA, Eisenberg S, Breslow JL (1994) Human HDL cholesterol levels are determined by apoA-I fractional catabolic rate, which correlates inversely with estimates of HDL particle size. Effects of gender, hepatic and lipoprotein lipases, triglyceride and insulin levels, and body fat distribution. Arterioscler Thromb 14(5): 707-720.
- Wade DP, Knight BL, Soutar AK (1988) Hormonal regulation of low-density lipoprotein (LDL) receptor activity in human hepatoma Hep G2 cells. Insulin increases LDL receptor activity and diminishes its suppression by exogenous LDL. Eur J Biochem 174(1): 213-218.
- Verges B (2015) Pathophysiology of diabetic dyslipidaemia: where are we? Diabetologia 58(5): 886-899.
- Gerber PA, Thalhammer C, Schmied C, Spring S, Spinas GA, et al. (2013) Small, dense LDL particles predict changes in intima media thickness and insulin resistance in men with type 2 diabetes and prediabetes--a prospective cohort study. PLoS One 8(8): e72763.
- Bjornstad P, Eckel RH (2018) Pathogenesis of Lipid Disorders in Insulin Resistance: A Brief Review. Curr Diab Rep. 18(12): 127.
- Xu Y, Qiao J (2022) Association of Insulin Resistance and Elevated Androgen Levels with Polycystic Ovarian Syndrome (PCOS): A Review of Literature. J Healthc Eng 2022: 9240569.
- Holte J (1996) Disturbances in insulin secretion and sensitivity in women with the polycystic ovary syndrome. Baillieres Clin Endocrinol Metab 10(2): 221-247.
- Petrillo T, Semprini E, Tomatis V, Arnesano M, Ambrosetti F, et al. (2022) Putative Complementary Compounds to Counteract Insulin-Resistance in PCOS Patients. Biomedicines 10(8): 1924.
- Kaur R, Kaur M, Singh J (2018) Endothelial dysfunction and platelet hyperactivity in type 2 diabetes mellitus: molecular insights and therapeutic strategies. Cardiovasc Diabetol 17(1): 121.
- Lahera V, de Las HN, López FA, Manucha W, Ferder L, et al. (2017) Role of Mitochondrial Dysfunction in Hypertension and Obesity. Curr Hypertens Rep 19(2): 11.
- Gallo G, Volpe M, Savoia C (2022) Endothelial Dysfunction in Hypertension: Current Concepts and Clinical Implications. Front Med (Lausanne) 8: 798958.
- Manucha W, Ritchie B, Ferder L (2015) Hypertension and insulin resistance: implications of mitochondrial dysfunction. Curr Hypertens Rep 17(1): 504.
- Suba Z, Ujpál M (2006) Correlations of insulin resistance and neoplasms. Magy Onkol 50(2):127-135.
- Chiefari E, Mirabelli M, La Vignera S, Tanyolaç S, Foti DP, et al. (2021) Insulin Resistance and Cancer: In Search for a Causal Link. Int J Mol Sci 22(20): 11137.
- Tsugane S, Inoue M (2010) Insulin resistance and cancer: epidemiological evidence. Cancer Sci 101(5): 1073-1079.
- Brunt EM (2005) Nonalcoholic steatohepatitis: pathologic features and differential diagnosis. Semin Diagn Pathol 22(4): 330-338.
- Mendez SN, Arrese M, Zamora VD, Uribe M (2007) Current concepts in the pathogenesis of nonalcoholic fatty liver disease. Liver Int 27(4): 423-433.
- Lomonaco R, Bril F, Portillo SP, Ortiz LC, Orsak B, et al. (2016) Metabolic impact of nonalcoholic steatohepatitis in obese patients with type 2 diabetes. Diabetes Care 39(4): 632-638.
- Berg AH, Combs TP, Du X, Brownlee M, Scherer PE, et al. (2001) The adipocyte-secreted protein ACRP30 enhances hepatic insulin action. Nat. Med 7(8): 947-953.
- Bugianesi E, Pagotto U, Manini R, Vanni E, Gastaldelli A, et al. (2005) Plasma adiponectin in nonalcoholic fatty liver is related to hepatic insulin resistance and hepatic fat content, not to liver disease severity. J. Clin. Endocrinol. Metab 90(6): 3498-3504.
- Pagano C, Soardo G, Esposito W, Fallo F, Basan L, et al. (2005) Plasma adiponectin is decreased in nonalcoholic fatty liver disease. Eur. J. Endocrinol 152(1): 113-118.
- Hotamisligil GS (2006) Inflammation and metabolic disorders. Nature 444(7121): 860-867.
- Reaven GM, Knowles JW, Leonard D, Barlow CE, Willis BL, et al. (2017) Relationship between simple markers of insulin resistance and coronary artery calcification. J Clin Lipidol 11(4): 1007-1012.
- Fakhrzadeh H, Sharifi F, Alizadeh M, Arzaghi SM, Tajallizade KY, et al. (2016) Relationship between insulin resistance and subclinical atherosclerosis in individuals with and without type 2 diabetes mellitus. J Diabetes Metab Disord 15(1): 41.
- Bressler P, Bailey SR, Matsuda M, DeFronzo RA (1996) Insulin resistance and coronary artery disease. Diabetologia 39(11): 1345-1350.
- Haffner SM, Lehto S, Rönnemaa T, Pyörälä K, Laakso M, et al. (1998) Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction. N Engl J Med 339(4): 229-234.
- Howard G, O’Leary DH, Zaccaro D, Haffner S, Rewers M, et al. (1996) The Insulin Resistance Atherosclerosis Study (IRAS) Investigators. Insulin sensitivity and atherosclerosis. Circulation 93(10): 1809-1817.
- Isomaa B, Almgren P, Tuomi T, Forsén B, Lahti K, et al. (2001) Cardiovascular morbidity and mortality associated with the metabolic syndrome. Diabetes Care 24(4): 683-689.
- Bonora E, Formentini G, Calcaterra F, Lombardi S, Marini F, et al. (2002) HOMA-estimated insulin resistance is an independent predictor of cardiovascular disease in type 2 diabetic subjects: prospective data from the Verona Diabetes Complications Study. Diabetes Care 25(7): 1135-1141.
- Bonora E, Kiechl S, Willeit J, Oberhollenzer F, Egger G, et al. (2007) Insulin resistance as estimated by homeostasis model assessment predicts incident symptomatic cardiovascular disease in Caucasian subjects from the general population: the Bruneck study. Diabetes Care 30(2): 318-324.
- Hedblad B, Nilsson P, Janzon L, Berglund G (2000) Relation between insulin resistance and carotid intima-media thickness and stenosis in non-diabetic subjects. Results from a cross-sectional study in Malmö, Sweden. Diabet Med 17(4): 299-307.
- Golden SH, Folsom AR, Coresh J, Sharrett AR, Szklo M, et al. (2002) Risk factor groupings related to insulin resistance and their synergistic effects on subclinical atherosclerosis: the atherosclerosis risk in communities’ study. Diabetes 51(10):3069-3076.
- Hanley AJ, Williams K, Stern MP, Haffner SM (2002) Homeostasis model assessment of insulin resistance in relation to the incidence of cardiovascular disease: the San Antonio Heart Study. Diabetes Care 25(7): 1177-1184.
- Gast KB, Tjeerdema N, Stijnen T, Smit JW, Dekkers OM, et al. (2012) Insulin resistance and risk of incident cardiovascular events in adults without diabetes: meta-analysis. PLoS One 7(12): e52036.
- Petersen MC, Shulman GI (2018) Mechanisms of insulin action and insulin resistance. Physiol Rev 98(4): 2133-2223.
- Bajaj M, Defronzo RA (2003) Metabolic and molecular basis of insulin resistance. J Nucl Cardiol 10(3): 311-323.
- Rask MC, Li Q, Freund B, Feather D, Abramov R, et al. (2010) Loss of insulin signaling in vascular endothelial cells accelerates atherosclerosis in apolipoprotein E null mice. Cell Metab 11(5): 379-389.
- Jiang ZY, Lin YW, Clemont A, Feener EP, Hein KD, et al. (1999) Characterization of selective resistance to insulin signaling in the vasculature of obese Zucker (fa/fa) rats. J Clin Invest 104(4): 447-457.
- Cusi K, Maezono K, Osman A, Pendergrass M, Patti ME, et al. (2000) Insulin resistance differentially affects the PI 3-kinase- and MAP kinase-mediated signaling in human muscle. J Clin Invest 105(3): 311-320.
- Sasaoka T, Rose DW, Jhun BH, Saltiel AR, Draznin B, et al. (1994) Evidence for a functional role of Shc proteins in mitogenic signaling induced by insulin, insulin-like growth factor-1, and epidermal growth factor. J Biol Chem 269(18): 13689-13694.
- Hsueh WA, Law RE (1999) Insulin signaling in the arterial wall. Am J Cardiol 84(1): 21-24.
- Wang CC, Goalstone ML, Draznin B (2004) Molecular mechanisms of insulin resistance that impact cardiovascular biology. Diabetes 53(11): 2735-2740.
- Draznin B (2006) Molecular mechanisms of insulin resistance: serine phosphorylation of insulin receptor substrate-1 and increased expression of p85α: the two sides of a coin. Diabetes 55(8): 2392-2397.
- King GL, Park K, Li Q (2016) Selective insulin resistance and the development of cardiovascular diseases in diabetes: the 2015 Edwin Bierman Award Lecture. Diabetes 65(6): 1462-1471.
- DeFronzo RA (2009) From the triumvirate to the ominous octet: a new paradigm for the treatment of type 2 diabetes mellitus. Diabetes 58(4): 773-795.
- DeFronzo RA (2010) Insulin resistance, lipotoxicity, type 2 diabetes and atherosclerosis: the missing links. The Claude Bernard Lecture 2009. Diabetologia 53(7): 1270-1287.
- Reaven GM (1988) Banting lecture 1988. Role of insulin resistance in human disease. Diabetes 37(12): 1595-1607.
- Sasaoka T, Ishiki M, Sawa T, Ishihara H, Takata Y, et al. (1996) Comparison of the insulin and insulin-like growth factor 1 mitogenic intracellular signaling pathways. Endocrinology 137(10): 4427-4434.
- Pfeifle B, Ditschuneit H (1981) Effect of insulin on growth of cultured human arterial smooth muscle cells. Diabetologia 20(2): 155-158.
- Leitner JW, Kline T, Carel K, Goalstone M, Draznin B, et al. (1997) Hyperinsulinemia potentiates activation of p21Ras by growth factors. Endocrinology 138(5): 2211-2214.
- Ferrannini E, Natali A, Muscelli E, Nilsson PM, Golay A, et al. (2011) Natural history and physiological determinants of changes in glucose tolerance in a non-diabetic population: the RISC study. Diabetologia 54(6): 1507-1516.
- DeFronzo RA, Ferrannini E (1991) Insulin resistance. A multifaceted syndrome responsible for NIDDM, obesity, hypertension, dyslipidemia, and atherosclerotic cardiovascular disease. Diabetes Care 14(3): 173-194.
- Ferrannini E, Balkau B, Coppack SW, Dekker JM, Mari A, et al. (2007) Insulin resistance, insulin response, and obesity as indicators of metabolic risk. J Clin Endocrinol Metab 92(8): 2885-2892.
- Rawshani A, Rawshani A, Franzén S, Eliasson B, Svensson AM, et al. (2017) Mortality and cardiovascular disease in type 1 and type 2 diabetes. N Engl J Med 376(15): 1407-1418.
- Sung KC, Choi JH, Gwon HC, Choi SH, Kim BS, et al. (2013) Relationship between insulin resistance and coronary artery calcium in young men and women. PLoS One 8(1): e53316.
- Gu A, Yue Y, Desai RP, Argulian E (2017) Racial and ethnic differences in antihypertensive medication use and blood pressure control among us adults with hypertension: the national health and nutrition examination survey, 2003 to 2012. Circ Cardiovasc Qual Outcomes 10: e003166.
- DeFronzo RA (1981) The effect of insulin on renal sodium metabolism. A review with clinical implications. Diabetologia 21(3): 165-171.
- Skott P, Hother NO, Bruun NE, Giese J, Nielsen MD, et al. (1989) Effects of insulin on kidney function and sodium excretion in healthy subjects. Diabetologia 32(9): 694-699.
- Fossum E, Høieggen A, Reims HM, Moan A, Rostrup M, et al. (2004) High screening blood pressure is related to sympathetic nervous system activity and insulin resistance in healthy young men. Blood Press 13(2): 89-94.
- Zhou MS, Wang A, Yu H (2014) Link between insulin resistance and hypertension: What is the evidence from evolutionary biology? Diabetol Metab Syndr 6(1): 12.
- Cooper SA, Whaley CA, Habibi J, Wei Y, Lastra G, et al. (2007) Renin-angiotensin-aldosterone system and oxidative stress in cardiovascular insulin resistance. Am J Physiol Heart Circ Physiol 293(4): H2009-H2023.
- Bansilal S, Farkouh ME, Fuster V (2007) Role of insulin resistance and hyperglycemia in the development of atherosclerosis. Am J Cardiol 99(4): 6-14.
- Franklin SS, Larson MG, Khan SA, Wong ND, Leip EP, et al. (2001) Does the relation of blood pressure to coronary heart disease risk change with aging? The Framingham Heart Study. Circulation 103(9): 1245-1249.
- Khattar RS, Swales JD, Dore C, Senior R, Lahiri A, et al. (2001) Effect of aging on the prognostic significance of ambulatory systolic, diastolic, and pulse pressure in essential hypertension. Circulation 104(7): 783-789.
- Brunt EM (2005) Nonalcoholic steatohepatitis: pathologic features and differential diagnosis. Semin Diagn Pathol 22(4): 330-338.
- McCullough AJ (2006) Pathophysiology of nonalcoholic steatohepatitis. J Clin Gastroenterol 40(S1): S17-S29.
- Ekstedt M, Hagström H, Nasr P, Fredrikson M, Stål P, et al. (2015) Fibrosis stage is the strongest predictor for disease-specific mortality in NAFLD after up to 33 years of follow-up. Hepatology 61: 1547-1554.
- Bugianesi E, McCullough AJ, Marchesini G (2005) Insulin resistance: a metabolic pathway to chronic liver disease. Hepatology 42(5): 987-1000.
- Fracanzani AL, Valenti L, Bugianesi E, Andreoletti M, Colli A, et al. (2008) Risk of severe liver disease in nonalcoholic fatty liver disease with normal aminotransferase levels: a role for insulin resistance and diabetes. Hepatology 48(3): 792-798.
- Bugianesi E, Gastaldelli A, Vanni E, Gambino R, Cassader M, et al. (2005) Insulin resistance in non-diabetic patients with non-alcoholic fatty liver disease: sites and mechanisms. Diabetologia 48(4): 634-642.
- Paradis V, Perlemuter G, Bonvoust F, Dargere D, Parfait B, et al. (2001) High glucose and hyperinsulinemia stimulate connective tissue growth factor expression: a potential mechanism involved in progression to fibrosis in nonalcoholic steatohepatitis. Hepatology 34(4): 738-744.
- Hanley AJ, Williams K, Festa A, Wagenknecht LE, D’Agostino RB, et al. (2005) Liver markers and development of the metabolic syndrome: the insulin resistance atherosclerosis study. Diabetes 54(11): 3140-3147.
- Park SH, Kim BI, Yun JW, Kim JW, Park DI, et al. (2004) Insulin resistance and C-reactive protein as independent risk factors for non-alcoholic fatty liver disease in non-obese Asian men. J Gastroenterol Hepatol 19(6): 694-698.
- Angulo P, Keach JC, Batts KP, Lindor KD (1999) Independent predictors of liver fibrosis in patients with nonalcoholic steatohepatitis. Hepatology 30(6): 1356-1362.
- Rosso C, Mezzabotta L, Gaggini M, Salomone F, Gambino R, et al. (2016) Peripheral insulin resistance predicts liver damage in nondiabetic subjects with nonalcoholic fatty liver disease. Hepatology 63(1): 107-116.
- Svegliati BG, Bugianesi E, Bouserhal T, Marini F, Ridolfi F, et al. (2007) post-load insulin resistance is an independent predictor of hepatic fibrosis in virus C chronic hepatitis and in non-alcoholic fatty liver disease. Gut 56(9): 1296-1301.
- Bugianesi E, Manzini P, D’Antico S, Vanni E, Longo F, et al. (2004) Relative contribution of iron burden, HFE mutations, and insulin resistance to fibrosis in nonalcoholic fatty liver. Hepatology 39(1): 179-187.
- Korkmaz H, Unler GK, Gokturk HS, Schmidt WE, Kebapcilar L, et al. (2015) Noninvasive estimation of disease activity and liver fibrosis in nonalcoholic fatty liver disease using anthropometric and biochemical characteristics, including insulin, insulin resistance, and 13C-methionine breath test. Eur J Gastroenterol Hepatol 27(10): 1137-1143.
- Kahn BB, Flier JS (2000) Obesity and insulin resistance. J Clin Invest 106(4): 473-481.
- Younossi ZM (2019) Non-alcoholic fatty liver disease: a global public health perspective. J Hepatol 70(3): 531-544.
- Marchesini G, Bugianesi E, Forlani G, Cerrelli F, Lenzi M, et al. (2003) Nonalcoholic fatty liver, steatohepatitis, and the metabolic syndrome. Hepatology 37(4): 917-923.
- Tada T, Toyoda H, Sone Y, Yasuda S, Miyake N, et al. (2019) Type 2 diabetes mellitus: a risk factor for progression of liver fibrosis in middle-aged patients with non-alcoholic fatty liver disease. J Gastroenterol Hepatol 34(11): 2011-2018.
- Bjorkstrom K, Franzen S, Eliasson B, Miftaraj M, Gudbjornsdottir S, et al. (2019) Risk factors for severe liver disease in patients with type 2 diabetes. Clin Gastroenterol Hepatol 17(13): 2769-2775.
- Kim Y, Chang Y, Cho YK, Ahn J, Shin H, et al. (2019) Obesity and weight gain are associated with progression of fibrosis in patients with nonalcoholic fatty liver disease. Clin Gastroenterol Hepatol 17(3): 543-550.
- Wallace IR, McKinley MC, Bell PM, Hunter SJ (2013) Sex hormone binding globulin and insulin resistance. Clin Endocrinol (Oxf) 78(3): 321-329.
- Moghetti P, Castello R, Negri C, Tosi F, Perrone F, et al. (2000) Metformin effects on clinical features, endocrine and metabolic profiles, and insulin sensitivity in polycystic ovary syndrome: a randomized, double-blind, placebo-controlled 6-month trial, followed by open, long-term clinical evaluation. J Clin Endocrinol Metab 85(1): 139-146.
- Jayagopal V, Kilpatrick ES, Jennings PE, Hepburn DA, Atkin SL, et al. (2003) The biological variation of testosterone and sex hormone-binding globulin (SHBG) in polycystic ovarian syndrome: implications for SHBG as a surrogate marker of insulin resistance. J Clin Endocrinol Metab 88(4): 1528-1533.
- Teede HJ, Misso ML, Deeks AA, Moran LJ, Stuckey BG, et al. (2011) Assessment and management of polycystic ovary syndrome: summary of an evidence-based guideline. Med J Aust 195(6): S65-112.
- Diamanti KE, Dunaif A (2012) Insulin resistance and the polycystic ovary syndrome revisited: an update on mechanisms and implications. Endocr Rev 33(6): 981-1030.
- Handelsman DJ, Wartofsky L (2013) Requirement for mass spectrometry sex steroid assays in the Journal of Clinical Endocrinology and Metabolism. J Clin Endocrinol Metab 98(10): 3971-3973.
- Kaiser N, Sasson S, Feener EP, Boukobza VN, Higashi S, et al. (1993) Differential regulation of glucose transport and transporters by glucose in vascular endothelial and smooth muscle cells. Diabetes 42(1): 80-89.
- Zhou MS, Schulman IH, Zeng Q (2012) Link between the renin-angiotensin system and insulin resistance: implications for cardiovascular disease. Vasc Med 17(5): 330-341.
- Le TN, Nestler JE, Strauss JF III, Wickham EP III (2012) Sex hormone-binding globulin and type 2 diabetes mellitus. Trends Endocrinol Metab 23(1): 32-40.






























