A Case of Transient Neonatal Diabetes Mellitus Successfully Managed with Basal Rate only Insulin Pump
Michael M Henry MD1^, Sumeet Arora MD2^, Vatcharapan Umpaichitra MD3, Elna Kochummen MD4, Tong Wooi Ch’ng MD5, Dawn Hagerty RN3, Zachary Ibrahim MD6, Sheila Perez-Colon MD7, Vivian L Chin MD3 and Lawrence Fordjour MD8*
1Division of Neonatology, Department of Pediatrics, Pediatrix of Maryland, P.A. Adventist HealthCare Shady Grove Medical Center, Rockville, Maryland
2Division of Pediatric Endocrinology, Department of Pediatrics, Artemis Hospital, Gurgaon, HR, India
3Division of Pediatric Endocrinology, Department of Pediatrics, SUNY Downstate Health Sciences University, Brooklyn, New York
4Division of Pediatric Endocrinology, Department of Pediatrics, Pediatric Endocrinology and Diabetes Associates, Richmond, Virginia
5Department of Pediatrics, Sunway Medical Center, Malaysia
6Division of Neonatal-Perinatal Medicine, Cohen Children’s Medical Center, Northwell Health, New Hyde Park, New York
7Elite Endocrine MD, Doctors’ Center Hospital, Puerto Rico
8Division of Neonatology, Department of Pediatrics, SUNY Downstate Health Sciences University, Brooklyn, New York
^equal contributors
Submission: February 13, 2023; Published: March 07, 2023
*Corresponding author: Lawrence Fordjour, Division of Neonatology, Department of Pediatrics, 450 Clarkson Ave. Box 49, Brooklyn, NY 11203, USA
How to cite this article: Michael M Henry, Sumeet Arora, Vatcharapan Umpaichitra, Elna Kochummen, Tong Wooi Ch’ng, et al. A Case of Transient Neonatal Diabetes Mellitus Successfully Managed with Basal Rate only Insulin Pump. Curre Res Diabetes & Obes J 2023; 16(3): 555938.DOI: 10.19080/CRDOJ.2023.16.555938
Abstract
Introduction: Neonatal diabetes mellitus is a rare condition that presents in the first few weeks to months of life. The neonate presents with low birth weight, dehydration, and hyperglycemia with or without ketoacidosis. Management using insulin drip, subcutaneous insulin injection and continuous subcutaneous insulin infusion (CSII) each present their own challenges. There is risk of hypoglycemia even with tiny doses of insulin.
Case: We present a case of transient neonatal diabetes mellitus (TNDM) due to paternal uniparental disomy at the 6q24 region, initially diagnosed late at DOL 29 due to the intermittent nature of hyperglycemia. The patient was born with low birth weight, macroglossia, umbilical hernia, and required repair of bilateral inguinal hernia. The other challenges involve management of insulin administration due to the limited amount of subcutaneous tissue in neonates as well as the minute doses of insulin required. Our patient was successfully managed with the most advanced insulin pump at the time with diluted insulin, using a basal rate-only regimen at total daily dose of 0.01-0.2u/kg/day, which is much lower than reported in the current literature. Additionally, this report shows that by titrating to pre-feed glucose, no episodes of hypoglycemia were noted. She had much less blood sugar variability and remained euglycemic. A 90-degree steel catheter was used successfully.
Conclusion: TNDM due to paternal uniparental disomy at the 6q24 region can successfully be treated using basal rate-only CSII regimen, titrated to pre-feed glucoses, and 90-degree steel catheter.
Keywords: transient neonatal diabetes; insulin pump; chromosome 6q24
Background
Neonatal diabetes mellitus (NDM) is an extremely rare condition which affects about 1 in 200,000 live births [1]. In contrast to infants born to diabetic mothers, these babies are typically growth restricted and present in the first 6 months of life with a wide range of symptoms, including failure to thrive (FTT), dehydration, ketoacidosis, seizures or coma. Two types of NDM are described, transient and permanent. Each is associated with a distinct clinical course and corresponding mutations have been described. Early detection of genetic mutations may help determine optimal therapy, prognosis and genetic counseling of the family. The management of hyperglycemia is challenging due to miniscule amounts of insulin required as well as the risk of significant blood glucose oscillations. In this case report we describe a patient with transient neonatal diabetes mellitus (TNDM) due to paternal uniparental disomy at the 6q24 region who was successfully treated using basal rate-only CSII regimen, titrated to pre-feed glucoses, and 90-degree steel catheter, in an under reported population. The patient’s parents have given their written informed consent to publish this case.
Case Description
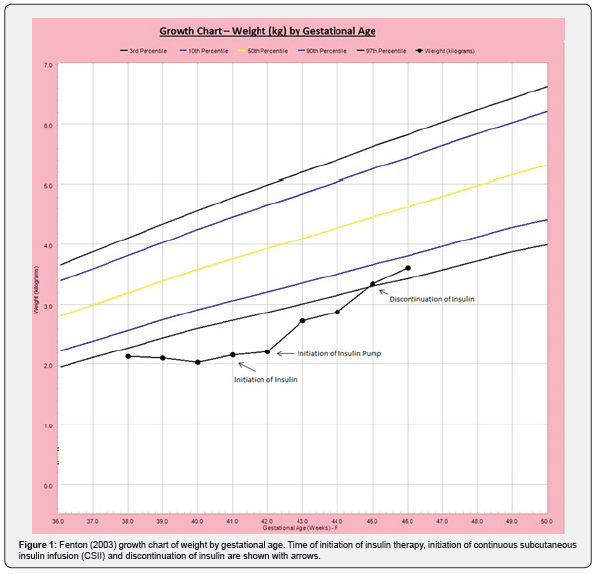
A female infant born at 38 weeks gestation, to a 29-yearold G4P2012 mother via spontaneous vaginal delivery with Apgar scores of 9 and 9 at 1 minute and 5 minutes, respectively. Maternal and paternal ethnicities were African American, with no maternal history of hypertension, pre-eclampsia, smoking or diabetes mellitus. Family history was significant for maternal great grandmother with diabetes mellitus, unknown type. Birth weight was 2125 grams (3rd percentile), length was 46 cm (10th percentile), and head circumference was 32 cm (10th percentile) (Figure 1). The infant was admitted to neonatal intensive care unit for low birth weight. Newborn screening along with evaluation for inborn errors of metabolism was negative. The physical exam was unremarkable except for decreased subcutaneous fat, small umbilical hernia and macroglossia. Upon admission, the patient was normothermic at 36.8°C but had hypoglycemia of 2.64mmol/l (48mg/dl) and 1.65 mmol/l (39mg/dl) which improved with feeding. At approximately 12 hours of life, the patient was noted to be hypothermic at 34.5°C, along with lethargy and vomiting. At that time, hyperglycemia was noted on bedside glucose of 11.2-12.2mmol/l (205-222mg/ dL), confirmed with serum glucose. A sepsis evaluation was performed, and empiric antibiotics were started. The next day, feed was resumed and blood glucose turned to normal. Antibiotics were discontinued after negative cultures.
On day of life (DOL) 8, the infant had a recurrence of emesis and hyperglycemia of 10.5mmol/l (190mg/dl) and hypothermia of 36.2°C for the second time and underwent a repeat partialsepsis work-up. The patient was kept NPO, given D5W solution with a GIR of 3.5mg/kg/min to maintain euglycemia, which resolved the next day. On DOL 14, while continuing to receive antibiotics, she developed vesicular lesions on both palms of her hands for which a herpes simplex virus work-up was done and acyclovir was started and subsequently discontinued after negative culture and PCR. Anti-fungal treatment was started for a similar appearing diaper rash and oral thrush all of which resolved by discharge.
On DOL 16 due to anemia, the patient received a transfusion of packed red blood cells in anticipation of bilateral inguinal hernia repair. During the blood transfusion, hyperglycemia up to 22mmol/l (400mg/dl) was noted on routine testing. The hyperglycemia resolved and was attributed to administration of transfusion. The infant tolerated the surgery well. Hypercaloric formula was introduced, leading her to regain birth weight. However, on DOL 20, repeat blood glucose testing was done but hyperglycemia (fourth episode) of 9.9mmol/l (180mg/ dl) prefeed and 15.2mmol/l (276mg/dl) post-feed were noted again. Pediatric endocrinology was consulted, and diagnostic testing showed an inappropriately low insulin level (2.47pmol/l) relative to the plasma glucose level (15.2mmol/l or 276mg/dl). Islet cell, GAD-65 and insulin autoantibodies were negative. These antibodies were done at the request of the genetics laboratory. Abdominal ultrasound confirmed the presence of the pancreas. TSH and Free T4 were normal. Recurrent hyperglycemia, low insulin level, clinical features of IUGR, macroglossia, umbilical hernia and low subcutaneous fat were consistent with the clinical diagnosis of neonatal diabetes mellitus.
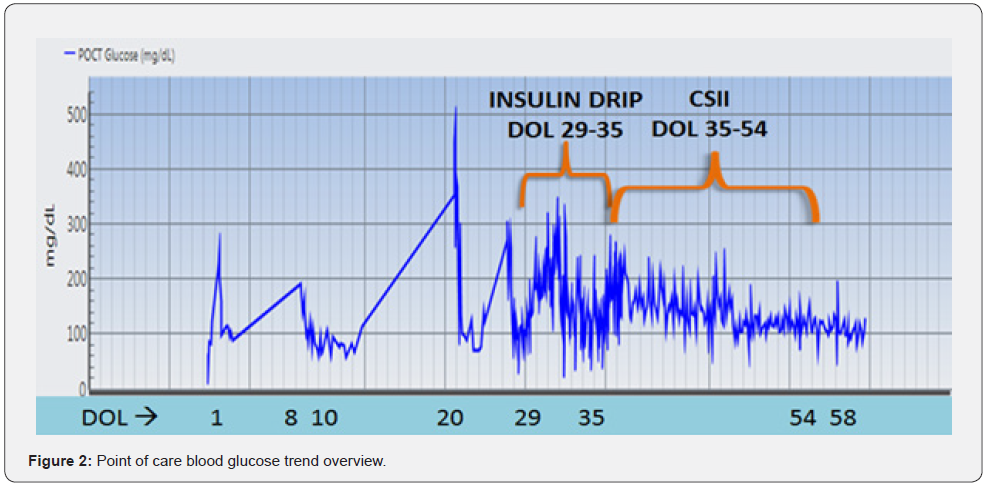
Hyperglycemia was managed initially by intravenous insulin drip therapy with regular insulin on DOL 29 at 0.01u/kg/hr (ranging up to 0.06u/kg/hr by titration). On DOL 35, the patient was transitioned to continuous subcutaneous insulin infusion (CSII) using the newest available pump at the time, Minimed® Medtronic TM 630G with the Sure T® infusion set, a fine 29-gauge perpendicular metallic needle. The infusion sets were changed every third day and the insertion sites used primarily were upper outer thigh, gluteal region and occasionally abdomen. A basal rate-only regimen was started using diluted lispro (1:10 using diluent). Multiple basal rates were set up in the pump for ease of use; the patient’s insulin requirement was 0.0025u/hr to 0.025u/hr (total daily dose 0.01 to 0.2 u/kg/day). Pre-feed blood glucose levels were monitored with a goal to maintain blood glucose between 5.51 and 11mmol/l (100 and 200mg/dl) by adjusting the basal rates up or down by 0.0025u/hr with a minimum of two readings before rate adjustments were made and pump was suspended if blood glucose was less than 3.85 mmol/l (70mg/dl). No hypoglycemia was detected with pump use. The patient was completely weaned off CSII on DOL 54 when hyperglycemia resolved (Figure 2). Upon discontinuation of subcutaneous insulin, post prandial blood glucose (2 hours post-feed) was monitored for a duration of 48 hours to ensure continued euglycemia. Mild self-resolving induration was noted at the infusion site despite site change every 2-3 days. Genetic testing revealed paternal uniparental disomy at the 6q24 region, the most common cause of TNDM (2). Study of the methylation pattern of the promoter of ZAC gene and PCR, H 11-F, and H-11 D2 sites revealed abnormal exclusive paternal contribution. Four weeks after initiation of insulin therapy, she showed remarkable catch-up weight gain and was discharged on DOL 57 with weight at 6th percentile for gestational age with continued weight gain on follow up.
Discussion
The natural history of TNDM is its usual presentation from birth to four weeks of age with IUGR and hyperglycemia requiring insulin therapy and typically resolve after a median of 3 months with complete resolution by 18 months of age but can recur later in adolescence or during pregnancy. Other clinical features include macroglossia (seen in one third of paternal uniparental disomy of 6q24 gene) and umbilical hernia. The more severe the genetic mutation, the earlier the presentation and higher the likelihood of ketoacidosis [2]. TNDM occurs due to overexpression of two genes, ZAC and HYMAI at chromosome 6q24 [2]. It has been hypothesized that the mutation at this gene locus leads to reduction in beta cell mass, affecting insulin secretion [3-5].
The 6q24 region is normally expressed by the paternal allele, and the maternal allele is imprinted by methylation [2,3]. Of note, we want to highlight that in the case of FTT, with episodic hyperglycemia (without ketoacidosis) neonatal diabetes should be considered. She had two early self-resolving episodes of hyperglycemia, with appropriate sepsis evaluations. The third episode of hyperglycemia inappropriately attributed to an adverse blood transfusion reaction, was another missed opportunity for NDM work up. Fortunately, hyperglycemia was detected on routine glucose monitoring and with appropriate insulin therapy, baby showed good catch-up weight gain. Upon close examination, she had subtle features such as macroglossia and umbilical hernia, present in TNDM. She was born IUGR, typical for most infants with TNDM [6], due to the lack of insulin that acts as a fetal growth factor [3]. The FTT along with hyperglycemic episodes warranted a workup for NDM. She did not exhibit timely catch-up weight prior to diagnosis of NDM. This case highlights the importance of making the diagnosis of NDM and having a high index of suspicion.
Our patient has a benign family history. In this case, the risk to offspring and siblings is low. In familial cases, the mutation found was paternal duplication of 6q24 chromosome. There is a 50% chance of inheriting the mutation from the father and a lower chance if the mutation is passed from mother, with subsequent generations still at risk [7]. Typically, hyperglycemia in NDM is treated with insulin however oral sulfonylureas can be occasionally used in cases caused by activating ABCC8 or KCNJ11 mutations of the potassium ATP channel [8]. Management of NDM has been described but there is no consensus on a single optimal therapy. Most authors report initial management with intravenous insulin used as a bridge followed by subcutaneous (SC) therapy, with only few reports using SC injections as initial therapy [9]. The prolonged use of IV therapy is limited by the need of a patent IV line for an extended duration and continued hospitalization.
There has been no reported clinical trial due to the rare nature of NDM, however there are multiple case reports on the use of CSII for the management of NDM. Rabonne et al. [9] reviewed 40 papers of transient and permanent NDM treated with insulin from 1994 to 2016. They identified 29 case reports of CSII and 38 case reports of non-pump therapy. Non-pump therapy included initial treatment with IV insulin followed by subcutaneous injections with different types of insulin being used. NPH insulin was used at total daily doses (TDD) of 0.1- 1.5u/kg/day in 9 cases, long-acting insulin at 0.27-1u/kg/day in 6 cases, short acting insulin at doses of 0.4-0.7u/kg/day in 3 cases, and rapid acting in 1 case at <0.03-0.95u/kg/day. Different regimens that were used for CSII included basal rate with mealtime boluses or basal rate adjustments for meals [9]. Only 1 case reported using basal rate only, without any meal boluses. Lispro insulin was used with these pumps due to its approval for use by most licensing authorities in different countries and with no age limit. In most reports, lispro was diluted either 1:10 or 1:40. There were 2 case reports that used regular insulin in their pump. The German Working Group for Pediatric Diabetes reported an average TDD on the pump at onset of NDM was 0.75u/kg/day and TDD for basal rates was 0.56u/kg/day [10].
Our patient required a TDD of 0.01-0.2u/kg/day, which is a much lower dose compared to most case reports mentioned previously. This may possibly be due to a milder presentation of TNDM. Compared to CSII, non-pump therapy was associated with severe hypoglycemia, difficulty in stabilizing glucose levels, as well as difficulty in administering tiny doses of insulin. Overall, CSII provided better glycemic control and fewer hypoglycemia and no episodes of severe hypoglycemia [9]. Consistent with these findings, our patient did not experience any hypoglycemia while on CSII but did have hypoglycemia while on IV insulin drip.
There are also different types of infusion sets available on the market. Some authors report that infusion sets with angled cannulas were better than perpendicular cannulas due to concern for dislodgement [11]. With the availability of newer metallic needles that have a finer gauge and shorter in length, we chose to use the metallic needle to minimize the risk of catheter kinking. Despite the paucity of subcutaneous fat, the straight metallic needle was used successfully, and other than a minor induration which self-resolved, the patient tolerated it well.
Conclusion
We highlight a case of a neonate with transient neonatal diabetes mellitus successfully managed using the newest pump at the time, Minimed® Medtronic TM 630G insulin pump, which delivered diluted insulin as low as basal rate of 0.0025units/hr (patient required basal 0.0025u/hr to 0.025u/hr). Our approach allowed for less blood glucose variability and no hypoglycemia compared to the IV insulin drip. Our patient did well by using a steel catheter for infusion, adjusting basal rates only to pre-feed glucose values, and using diluted insulin lispro 1:10, to allow delivery of even more miniscule amounts of insulin during the weaning period.
Conflicts of Interest
The authors have no conflicts of interest to declare.
Author Contributions
Both MH and SA contributed equally to writing the manuscript and should be considered first authors. VU, EK, TWC, DH, ZI, SPC, VLC and LF contributed to revisions and acceptance of the final manuscript.
References
- Rachel E J Besser, Sarah E Flanagan, Deborah G J Mackay, I K Temple, Maggie H Shepherd, et al. (2016) Prematurity and Genetic Testing for Neonatal Diabetes. Pediatrics 138(3).
- Temple IK, JP Shield (2002) Transient neonatal diabetes, a disorder of imprinting. J Med Genet 39(12): 872-875.
- Aguilar-Bryan L, J Bryan (2008) Neonatal diabetes mellitus. Endocr Rev 29(3): 265-291.
- I K Temple, R S James, J A Crolla, F L Sitch, P A Jacobs, et al. (1995) An imprinted gene(s) for diabetes? Nat Genet 9(2): 110-112.
- I K Temple, R J Gardner, D J Mackay, J C Barber, D O Robinson, et al. (2000) Transient neonatal diabetes: widening the understanding of the etiopathogenesis of diabetes. Diabetes 49(8): 1359-1366.
- Siri Atma W Greeley, Susan E Tucker, Rochelle N Naylor, Graeme I Bell, Louis H Philipson (2010) Neonatal diabetes mellitus: a model for personalized medicine. Trends Endocrinol Metab 21(8): 464-472.
- Polak M, H Cave (2007) Neonatal diabetes mellitus: a disease linked to multiple mechanisms. Orphanet J Rare Dis 2: 12.
- Ewan R Pearson, Isabelle Flechtner, Pål R Njølstad, Maciej T Malecki, Sarah E Flanagan, et al. (2006) Switching from insulin to oral sulfonylureas in patients with diabetes due to Kir6.2 mutations. N Engl J Med 355(5): 467-477.
- Ivana Rabbone, Fabrizio Barbetti, Raffaella Gentilella, Gilberto Mossetto, Riccardo Bonfanti (2017) Insulin therapy in neonatal diabetes mellitus: a review of the literature. Diabetes Res Clin Pract 129: 126-135.
- Thomas M Kapellen, Bettina Heidtmann, Eggert Lilienthal, Birgit Rami-Merhar, Charlotte Engler-Schmidt, et al. (2015) Continuous Subcutaneous Insulin Infusion in Neonates and Infants Below 1 Year: Analysis of Initial Bolus and Basal Rate Based on the Experiences from the German Working Group for Pediatric Pump Treatment. Diabetes Technol Ther 17(12): 872-879.
- Tara Bharucha, Justin Brown, Ciara McDonnell, Robecca Gebert, Peter McDougall, et al. (2005) Neonatal diabetes mellitus: Insulin pump as an alternative management strategy. J Paediatr Child Health 41(9-10): 522-526.






























