Glycaemia During COVID-19: A Primary Care Perspective
Eleanor Gettinby1, John Scott2 and Moulinath Banerjee3,4*
1Hull York Medical School, UK
2Tonge Fold Health Centre, UK
3Royal Bolton Hospital, UK
4Edge Hill University Medical School, UK
Submission: December 01, 2022; Published: January 05, 2023
*Corresponding author: Moulinath Banerjee, Royal Bolton Hospital, BL4 0JR, Bolton, UK
How to cite this article: E Gettinby, J Scott, M Banerjee. Glycaemia During COVID-19: A Primary Care Perspective. Curre Res Diabetes & Obes J 2023; 16(3): 555936.DOI: 10.19080/CRDOJ.2023.16.555936
Abstract
Background & Aims: Patients with diabetes fare poorly when infected with COVID-19 but there is a paucity of data about how COVID-19 infection impacts glycaemic control in the general population. This study aimed to assess the change in glycaemia in patients in relation to COVID-19 symptoms during the pandemic.
Materials and Method: This was a retrospective study of 226 patients with Diabetes and Non-Diabetic Hyperglycaemia (NDH) from a single urban General Practice in the UK. An audit of those patients who had undergone measurement of HbA1c both pre-pandemic and peri-pandemic and any change in HbA1c was recorded followed by a telephone survey for any COVID-19 symptoms during the previous 12 months was undertaken.
Results: 117 patients had diabetes and 109 patients had NDH. 45.3% of patients with diabetes included in the study were shown to have a significant change in HbA1c over the course of the pandemic. 13.7% of patients with diabetes were found to have COVID-19 symptoms according to the King’s College criteria. 3.6% of patients with NDH were found to have a significant change in HbA1c. 23.9% of patients with NDH were found to have COVID symptoms. Genders including age, gender, ethnicity, pre-pandemic HbA1c, and Townsend’s social deprivation score were adjusted for but no single deterministic factor for the change in HbA1c was observed.
Conclusion: There was a significant change in glycaemia in a group of patients with diabetes and in a small minority of patients with NDH, but no relationship with King’s College symptoms was seen. In this study, the overall numbers were small and derived from a single practice so a larger study would help us to understand the factors influencing glycaemia better.
Keywords: COVID-19; Pandemic; Diabetes; HbA1c; Symptoms
Introduction
In January 2020, the World Health Organisation (WHO) declared the COVID-19 outbreak as a global health emergency [1]. Since then, many people have experienced varying degrees of symptoms of COVID-19 from being asymptomatic to death. It is well known that patients with diabetes fare poorly when infected with COVID-19 and there is growing evidence suggesting that COVID-19 may trigger new-onset diabetes in some patients [2]. Mortality amongst patients with diabetes who contracted COVID-19 infection is higher than the general population [3,4], but it is not yet known why people with diabetes fare so poorly. HbA1c has been identified as a predictor of the severity of COVID-19 as patients with diabetes who had poorer glycaemic control face worse outcomes following COVID-19 infection [5]. Some clinicians have noticed a pattern of patients with previously well-controlled diabetes experiencing a significant rise in glycaemia following what the patients described as a bad flu-like illness. This led to questions about whether this was COVID and if so, how COVID-19 affects glycaemia.
There is increasing evidence to suggest that the pathogenesis of SARS-COV-2 interacts with glucose metabolism which may potentially lead to the changes in HbA1c levels seen in some diabetic patients [6]. Currently, there is a paucity of data about how COVID-19 infection impacts glycaemic control in the general population. This study aimed to assess the change in glycaemia in patients who had their HbA1c measured before the pandemic came to the UK (between June 2019-January 2020) and during the pandemic (January 2020- June 2020). We also related the change in glycaemia with symptoms suggestive of COVID-19 during the pandemic.
Methods
In this real-world study, an audit of patients who underwent measurement of HbA1c both before and during the pandemic as part of their routine clinical care. Change in HbA1c of 0.5% or 4mmol/mol was considered clinically significant as per current clinical practice [7]. The patients were deemed to have either no significant change in HbA1c, significant rise or significant fall in HbA1c according to table 1. Those patients who consented verbally to be included were then asked about any COVID-19 related symptoms based on King’s College 14 symptoms scale [8].

Results
A total of 226 patients of Tonge Fold Health Centre consented to the study; 117 of these had Diabetes and 109 had Non-Diabetic Hyperglycaemia (NDH). Table 2 summarises the demography of the patients included in the study, their pre-pandemic and peripandemic HbA1c levels and the proportion with COVID symptoms.
Patients with diabetes who were involved in the study, had a mean change in HbA1c from 60.7mmol/mol (pre-pandemic) to 62.2mmol/mol (peri-pandemic); giving an average rise in HbA1c of 1.47mmol/mol. 86.3% of this group of patients showed no COVID symptoms according to the King’s college criteria and 88% showed no COVID symptoms according to the NHS criteria (Table 3).
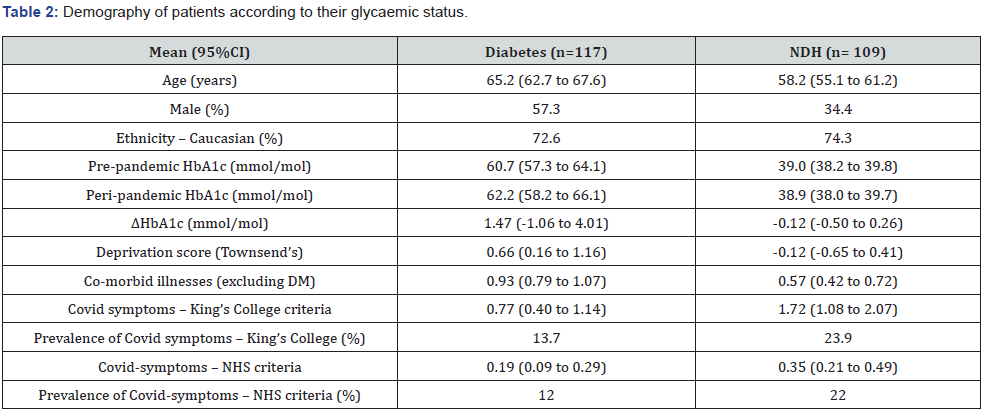
Patients with NDH had a mean change in HbA1c from 39.0mmol/mol pre-pandemic to 38.9 mmol/mol peri-pandemic; giving a mean change in HbA1c of –0.12mmol/mol. 76.1% of this group of patients showed no COVID symptoms according to the King’s college criteria and 78% showed no COVID symptoms according to the NHS criteria. In both groups, the average change in HbA1c was within the boundary of ± 4mmol/mol.
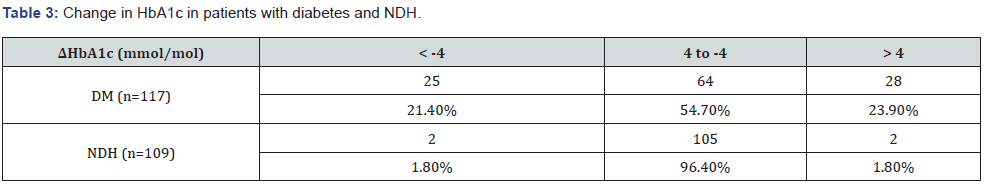
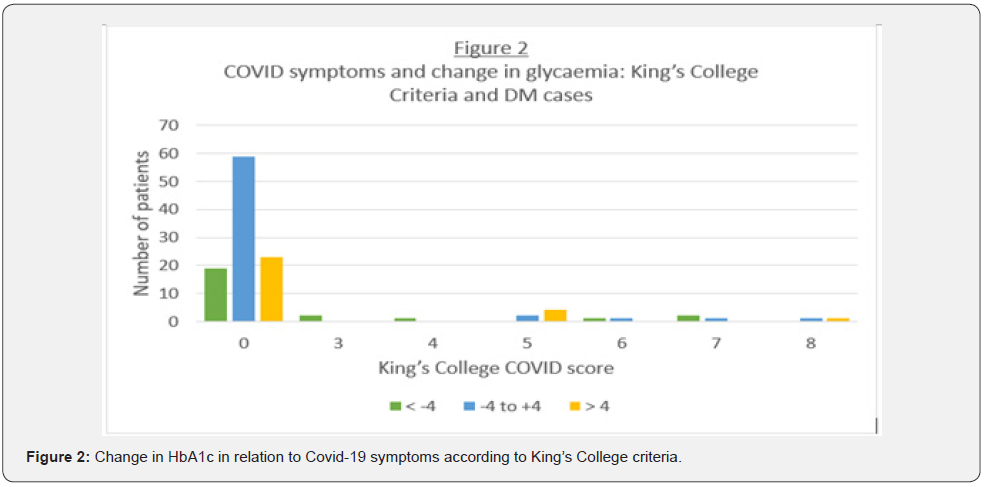
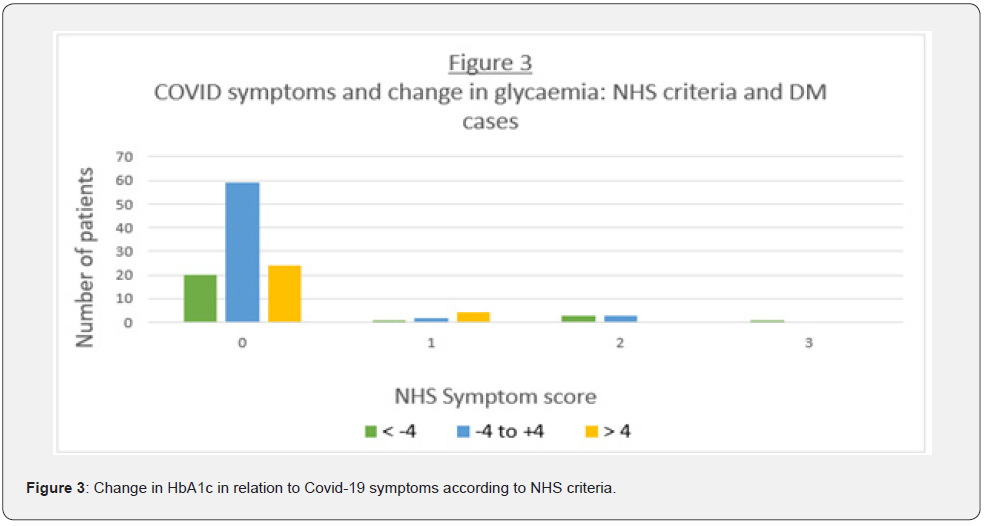
54.7% of patients with diabetes did not have any significant change in glycaemia across the study period. 23.9% of patients with diabetes were shown to have a significant rise in HbA1c and 21.4% of patients with diabetes were shown to have a significant fall in HbA1c. This shows that 45.3% of patients with diabetes were shown to have a significant change in their HbA1c levels over the study period. 96.4% of patients with NDH were shown to have no significant change in glycaemia. 1.8% of patients with NDH were shown to have a significant rise in HbA1c and 1.8% of patients were shown to have a significant fall in HbA1c. There were no patients with pre-existing NDH, whose HbA1c had worsened (>48 mmol/mol) to suggest progression to Type 2 diabetes. Out of the 117 patients with diabetes, the majority had a King’s College COVID score of zero, the majority of whom had no change in their HbA1c and a small minority had a significant rise or fall in HbA1c (Figure 2). A similar distribution was seen when the NHS COVID symptom criteria were used (Figure 3).
There were significant correlations between the presence of diabetes and age (r=0.235, p<0.0001), gender (r=0.206, r=0.002) – favouring male gender and social deprivation according to Townsend’s index (r=0.141, p=0.035). The number of comorbidities that our patients had was related to age (r=0.433, p<0.0001), gender (r=0.162, p=0.015) – favouring male gender, ethnicity (r=-0.165, p=0.013) favouring Caucasian ethnicity, presence of diabetes (r=0.658, p<0.001). Pre-pandemic HbA1c was higher amongst men (r=0.201, p=0.002) and was correlated with the number of comorbidities (r=0.398, p<0.0001). Both the King’s College score and the NHS scores were correlated inversely with age (r=-0.17, p=0.01 and r= -0.174, p=0.009 respectively), gender (r=-0.212, p=0.001 and r= -0.221, p=0.001 respectively) – being higher amongst women and presence of diabetes (r=- 0.182, p=0.006 and r= -0.136, p=0.04 respectively). There was no correlation observed between either the King’s College or NHS symptom scores and the change in HbA1c observed during the study period. Regression analysis adjusting for age, gender, pre-pandemic HbA1c and Social Deprivation index according to Townsend’s score did not reveal any single deterministic factor for the change in HbA1c as observed in this cohort.
Discussion
Our study was a retrospective analysis of real-world data collected from primary care – which looked at the impact of the COVID-19 pandemic on the change in glycaemic control of patients who were known to have diabetes and those who were at high risk of developing diabetes (NDH). There are several studies that have delineated a relationship between diabetes and higher glycaemic state with the severity of COVID-19 infection [9] and even mortality following it [10,11]. There is no study in the literature which has described the effect of the pandemic on glycaemic control, with or without the patients being affected with COVID-19 infection.
A systematic review and meta-analysis by Chen et al looked at three studies; the combined results of these studies showed that severe COVID-19 infection was associated with higher blood glucose [12]. They also showed that HbA1c was higher in patients with severe COVID-19 infection than those with mild COVID-19 infection, however, the difference in the HbA1c was not statistically significant. This study included patients who have had a positive COVID-19 test whereas our study used symptoms as a measure of potential COVID-19 infection. However, only two of the studies included in the systematic review by Chen et al explored the relationship between COVID-19 and HbA1c. Hence, the relationship between COVID-19 infection and change in HbA1c and the factors affecting it is still not very clear.
Our study demonstrates that amongst the patients with NDH, there was no significant change in HbA1c. Though 54.7% of patients with diabetes had no significant changes seen during the study period, 21.4% of patients had a significant reduction in their HbA1c, while 23.9% of patients had a significant rise in their HbA1c. This dichotomous response of change in HbA1c can be related to patients’ response towards the pandemic and its associated sociocultural changes that were seen. In India, among 312 children and young people (Mean age 12.4 +/- 4.2 years, 46.2% males) with Type 1 diabetes, from a single Paediatric Endocrine Unit in Pune, India, who faced a significant reduction in their physical activities (mean 270 mins /week – 123-480 mins pre-pandemic and 120-420 mins peri-pandemic) during the lockdown. This resulted in increased BMI (17.6 +/- 3.9 to 18.1 +/- 3.7, p=0.006), body fat content (17.9 +/- 9.3% to 19.7 +/- 9.8%, p<0.001) and a paradoxical statistically significant reduction in their HbA1c 10.0% (86 mmol/mol) to 9.8% (84mmol/mol), p= 0.028 [13]. In a cohort of 207 Italian adults with Type 1 diabetes using continuous glucose monitoring devices and cared for via a hospital diabetes clinic, data were compared before and during the pandemic. With comparable use of CGM (91.2+/-9.1% vs 90.1 +/- 8.6%, p=0.081), there was no significant change in mean glucose (9.6+/- 2.0 vs 9.5+/- 1.9mmol/l, p=0.165) or estimated HbA1c (7.7+/-1.3 vs 7.6+/- 1.1%, p=0.098), but there was significant improvement of time in range (55.6 +/- 17.6 vs 58.2 +/- 18.1, p=0.002) and time in severe hypoglycemia defined as < 54 mg/dl or 3.0 mmol/l (1.42+/- 2.39 vs 0.58+/- 1.17, p<0.001). The improvement in glycaemia was thought to be due to a more regulated lifestyle, increased time for self-care and also reduction in glucose variability, which is often seen with exercise [14].
Amongst 128 patients with Type 2 diabetes from Italy, within the first 3 months of lockdown, there was a significant rise in adiposity (BMI 29.5+/-6 to 30.1+/-6.3, p<0.001) and HbA1c 7.0+/- 0.8 to 7.3 +/- 0.9%, p<0.001). There was a clear correlation between an increase in BMI and a rise in HbA1c (β 0.246, p<0.001) in this cohort [15]. Amongst a cohort of 1009 patients with Type 2 diabetes from Japan, there was an increase in HbA1c from 7.45% to 7.53% despite having no official lockdown during the COVID-19 pandemic. Women, patients above the age of 65 years, those with a BMI greater than 25 kg/m2 and patients who did not use insulin had worsening of their glycaemic control. The authors proposed psychological stress to be the major driver for this worsening of glycaemic control observed among these patients [16]. Sankar et al. [17] reported a cohort of 110 patients with Type 2 diabetes from India, where they found a small (8.2 +/- 1.3% vs 8.12 +/- 1.6%) but statistically insignificant reduction in HbA1c during the pandemic. This was associated with similar body weight (71.5+/- 14.8 vs 71.8+/-13.6kg) over the study period. The benefit of an improved diet with increased consumption of fruit and vegetables as well as a reduction in the use of calorie-dense snacks during the pandemic were important factors for these observations.
Out of a mixed cohort of 369 patients being treated at 3 clinics in London, of whom 70.5% had Type 1 diabetes and 52.9% were women and the mean duration of diabetes of 17.0+/-13.7 years, paired results of HbA1c to compare prepandemic and peri-pandemic results showed an improvement from 62.13 to 60.16 mmol/mol (p=0.0033) [18]. The completed Diabetes Self-management Questionnaire among the participants suggested that COVID-19-related anxiety may have resulted in improvement in diabetes self-care, which was reflected in the improved glycaemic control. It seems that in patients with Type 1 diabetes, the COVID-19 pandemic might have had a favourable effect on glycaemic control mainly due to increased time for selfmanagement and a more regulated lifestyle. On the contrary, the patients with Type 2 diabetes seemed to have a neutral or worsening of their glycaemic control during the pandemic. Our cohort of patients with Type 2 diabetes showed results, where there were rise, fall and static HbA1c levels, suggesting that the patients had different behavioural responses during the pandemic.
Our study did not find any relationship between the change in glycaemia and COVID-19 symptoms. Though such an association has not been reported before, it would be logical to expect such a relationship to exist as a higher HbA1c has been associated with poorer outcomes following a COVID-19 infection [4,19,20]. All previous studies recruited patients with Type 1 and Type 2 diabetes from dedicated Diabetes and Endocrine clinics. Our study is the first to report on patients with diabetes whose diabetes management was provided by Primary or Community care. This study is also the first to include patients who were at risk of developing Type 2 diabetes (NDH) and report their glycaemic response during the COVID-19 pandemic.
Study Limitations
The main limitations of this study are first, that patients did not have COVID-19 tests. Therefore, the results seen may not be attributable to COVID-19 as the patients who did report COVID-19 symptoms according to the King’s College or NHS symptom scores may not have been COVID-related. Secondly, the King’s College and NHS symptom scores are subjective measures of illness. So, this may be a poor measure of the severity of the illness. Thirdly, the large impact of the pandemic on people’s lives may have caused an increased number of lifestyle-related factors which may have impacted HbA1c levels. These factors have not been accounted for in this study. Lastly, our study did not include any of the patients, who may have had serious COVID-19 infection and had a poor outcome or were not able to participate in the study. Hence, there is a strong possibility that there was an inadvertent selection bias towards cases, who were not seriously affected by COVID-19 infection.
Conclusion
The glycaemic change was significant in a group of patients with diabetes and in a small minority with NDH. The symptoms, either by King’s College Score or NHS symptom score did not influence the change in HbA1c observed. There was no correlation observed between the symptom scores and a change in HbA1c. This might suggest that other lifestyle factors such as diet, exercise, or compliance with medications, could have influenced the change in HbA1c. The assessment of these factors was beyond the scope of this project. As the overall numbers were small and derived from a single practice, a cohort of larger numbers would help us to understand the factors influencing the change in HbA1c as seen here.
References
- Jee Y (2020) WHO International Health Regulations Emergency Committee for the COVID-19 outbreak. Epidemiol Health 42: e2020013.
- Accili D (2021) Can COVID-19 cause diabetes? Nature Metabolism 3(2): 123-125.
- de Jong M, Woodward M, Peters SAE (2020) Diabetes and COVID-19–Related Mortality in Women and Men in the UK Biobank: Comparisons with Influenza/Pneumonia and Coronary Heart Disease. Diabetes Care 44(2): e22-e24.
- Holman N, Knighton P, Kar P, et al. (2020) Risk factors for COVID-19-related mortality in people with type 1 and type 2 diabetes in England: a population-based cohort study. The lancet Diabetes & endocrinology 8(10): 823-833.
- Singh AK, Khunti K (2020) Assessment of risk, severity, mortality, glycemic control and antidiabetic agents in patients with diabetes and COVID-19: A narrative review. Diabetes Research and Clinical Practice 165: 108266.
- Lim S, Bae JH, Kwon HS (2021) COVID-19 and diabetes mellitus: from pathophysiology to clinical management. Nature Reviews Endocrinology 17(1): 11-30.
- Kaiafa G, Veneti S, Polychronopoulos G, Dimitrios Pilalas, Stylianos Daios, et al. (2021) Is HbA1c an ideal biomarker of well-controlled diabetes? Postgraduate Medical Journal 97(1148): 380-383.
- Crouch H (2020) King’s College London launches Covid-19 symptom reporting app London.
- Gregory JM, Slaughter JC, Duffus SH, T Jordan Smith, Lauren M LeStourgeon, et al. (2020) COVID-19 Severity Is Tripled in the Diabetes Community: A Prospective Analysis of the Pandemic’s Impact in Type 1 and Type 2 Diabetes. Diabetes Care 44(2): 526-532.
- Barron E, Bakhai C, Kar P, Andy Weaver, Dominique Bradley, et al. (2020) Associations of type 1 and type 2 diabetes with COVID-19-related mortality in England: a whole-population study. The Lancet Diabetes & Endocrinology 8(10): 813-822.
- Zhu Z, Mao Y, Chen G (2021) Predictive value of HbA1c for in-hospital adverse prognosis in COVID-19: A systematic review and meta-analysis. Primary Care Diabetes 15(6): 910-917.
- Chen J, Wu C, Wang X, Jiangyi Yu, Zilin Sun (2020) The Impact of COVID-19 on Blood Glucose: A Systematic Review and Meta-Analysis. Front Endocrinol (Lausanne) 11: 574541.
- Shah N, Khadilkar V, Oza C, Madhura Karguppikar, Shital Bhor, et al. (2022) Impact of decreased physical activity due to COVID restrictions on cardio-metabolic risk parameters in Indian children and youth with type 1 diabetes. Diabetes Metab Syndr 16(7): 102564.
- Capaldo B, Annuzzi G, Creanza A, Clemente Giglio, Raffaele De Angelis, et al. (2020) Blood Glucose Control During Lockdown for COVID-19: CGM Metrics in Italian Adults with Type 1 Diabetes. Diabetes Care 43(8): e88-e89.
- Biamonte E, Pegoraro F, Carrone F (2021) Weight change and glycemic control in type 2 diabetes patients during COVID-19 pandemic: the lockdown effect. Endocrine 72(3): 604-610.
- Tanji Y, Sawada S, Watanabe T, Takashi Mita, Yasutaka Kobayashi, et al. (2021) Impact of COVID-19 pandemic on glycemic control among outpatients with type 2 diabetes in Japan: A hospital-based survey from a country without lockdown. Diabetes Research and Clinical Practice 176: 108840.
- Sankar P, Ahmed WN, Mariam Koshy V, Rittin Jacob, Saranya Sasidharan (2020) Effects of COVID-19 lockdown on type 2 diabetes, lifestyle and psychosocial health: A hospital-based cross-sectional survey from South India. Diabetes & Metabolic Syndrome: Clinical Research & Reviews 14(6): 1815-1819.
- Distaso W, Malik MMAH, Semere S, Amal AlHakami, Emma C Alexander, et al. (2022) Diabetes self-management during the COVID-19 pandemic and its associations with COVID-19 anxiety syndrome, depression and health anxiety. Diabetic Medicine 39(10): e14911.
- Smith SM, Boppana A, Traupman JA, Enrique Unson, Daniel A Maddock, et al. (2021) Impaired glucose metabolism in patients with diabetes, prediabetes, and obesity is associated with severe COVID-19. J Med Virol 93(1): 409-415.
- Lazarus G, Audrey J, Wangsaputra VK, Alice Tamara 3, Dicky L Tahapary (2021) High admission blood glucose independently predicts poor prognosis in COVID-19 patients: A systematic review and dose-response meta-analysis. Diabetes Res Clin Pract 171: 108561.






























