Reversal of Diabetes Mellitus in Obese Individuals following Bariatric Surgery-Single Center Experience at a Tertiary Care Hospital in Sri-Lanka
Dilini Abeyratne1, Udai Akalanka Wijethunga2, Umesha Wijenayake3, Chaminda Alahakoon3, Kavinga Gunawardane4, Niranjala Meegodawidanage1, Chaminda Garusinghe1, Thejana Kamil Wijeratne1, Uditha Sirimewan Bulugahapitiya3 and Gowri Malka Ratnayake5*
1Colombo South Teaching Hospital-Kalubowila, Sri Lanka
2GV Health, Shepparton VIC, Australia
3National Hospital of Sri-Lanka, Colombo
4District General Hospital-Negambo, Sri-Lanka
5Medway NHS Foundation Trust, UK
Submission: November 22, 2022; Published: December 12, 2022
*Corresponding author: Gowri Malka Ratnayake, Medway NHS Foundation Trust, UK
How to cite this article: Dilini A, Udai Akalanka W, Umesha W, Chaminda A, Kavinga G, et al. Reversal of Diabetes Mellitus in Obese Individuals following Bariatric Surgery-Single Center Experience at a Tertiary Care Hospital in Sri-Lanka. Curre Res Diabetes & Obes J 2022; 16(2): 555933.DOI: 10.19080/CRDOJ.2022.16.555933
Abstract
Obesity is a chronic progressive disease characterized by an increase in body fat stores. Diabetes mellitus has become another major pandemic causing huge cost to the healthcare system. There is a well-established link between the development of T2DM and being overweight or obese. Bariatric surgery is a well-recognized, effective treatment option for patients with obesity and T2DM where advantages of bariatric surgery go beyond the simple weight loss. In order to evaluate the short-term (at 1year) and long term (at 5years) remission of diabetes following bariatric surgery, we conducted a retrospective analysis among the patients who underwent the procedure at Colombo South Teaching Hospital, Sri Lanka between 2013-2020. Diagnosis and remission of diabetes were defined by ADA criteria. Among the 245 patients who underwent bariatric surgery, 109 (44.4%) had pre-operative T2DM, 44 (17.9%) had IFG, and 92 (37.7%) had complete normoglycemia with FBS <100mg/dl and HbA1C <5.6%. Pre-operatively 75 (68.9%) were females and 34 (31.1%) were males. Only the cohort of patients with pre-operative T2DM (n=109) was included in the outcome analysis. The pre-operative population with diabetes had following mean age -40±10years, females-71.6% (n=78), mean weight - 115.8±24.4kg (range = 75-204), mean BMI -45.4±7.4kg/m2 (range = 31.2-68.1), mean HbA1c -8.5±1.6%, mean FPG-140±47mg/dL and average duration of diabetes -3.5 years. The averages of FPG and HbA1C for the IFG group were 106±11mg/dl and 5.8±0.6% respectively. The pre-operative non-diabetic population had a mean weight of 121.5±35kg (range=74-181), mean BMI of 45.1±6.5kg/m2, mean FPG of 95.8±18mg/dl and mean HbA1c of 5.5±0.6%. Post-operative 1 year analysis of pre-operative diabetes group revealed a mean weight of 81.1kg±16.9 (range-52-140), mean BMI of 32.2kg/m2±5.8 with a mean weight loss of 32.1±14.8kg. Mean HbA1c and FPG improved to 5.5%±0.9 and 99±23mg /dl. 87.1% achieved diabetes remission with 58.7% reaching FPG of <100mg/dl and 28.4% reaching IFG. 15 (13.9%) had persistent diabetes. 95.4% (n-44) of pre-operative IFG patients achieved FPG of <100mg/dl while 2 (4.6%) remained in the IFG category. Mean FPG and HbA1c declined to 90±16mg/dl and 5.2±0.5% respectively.
1 year Diabetes Remission rate males and females were 90.3% and 84.6% respectively and the long-term diabetes remission rate was 80% among males and 91% among females. Separate analysis of 55 patients for 5year long term data revealed a mean weight, BMI and weight loss of 84 kg±13.6, 34.1 kg/m2±4.9 and 41.0kg±19.2 respectively. Mean FPG (102mg/dl) and HbA1C (5.7%±0.89) improved by 29.2% and 32.9% respectively. However, there was an upward trend of HbA1c, mean weight and mean BMI when compared to 1year value. Diabetes remission at 5 years was seen in 41 (74.5%) achieving FPG of <100mg/dl in 25 (45.4%) and IFG in 16 (29%) with 14 (25.5%) having diabetes including new occurrence of diabetes in 1 patient. Age of onset of diabetes, gender, pre-operative weight, and duration of diabetes had an impact on diabetes reversal. Pre-operative BMI had no impact on diabetes remission. (p=0.35). RYGB had better reversal of diabetes where LSG and LMGB had similar outcome. In conclusion, bariatric surgery is associated with both short- and long-term diabetes remission.
Keywords: Bariatric surgery; Obesity; Diabetes reversal; Diabetes remission criteria
Abbreviations: LAGB: Laparoscopic Gastric Banding; IDF: International Diabetes Federation; T2DM: Type 2 Diabetes; WHO: World Health Organization
Introduction
Obesity is a chronic progressive disease characterized by an increase of body fat stores. The World Health Organization (WHO) has declared obesity the largest global chronic health problem leading to many disabilities and death, affecting adults and children [1]. It has been projected that 60% of the world’s population could be overweight (2.2 billion) or obese (1.1 billion) by 2030 if the current trends continue to rise [2]. In adults (age over 18 years), a BMI of 30 kg/m2 or above is defined as obesity and a BMI of 25- 29.9 kg/m2 is defined as overweight. Obesity can be further classified as grade I (BMI of 30.0–34.9), grade II (BMI of 35.0–39.9) and grade III (BMI of >40). Southeast Asians have lower BMI cut-off points (23-27.5 as over-weight and >27.5 as obesity) [3]. Rather than a disease of its own, obesity is often associated with comorbidities such as cardiovascular disease, stroke, type 2 diabetes mellitus (T2DM), hypertension, sleep apnea, cholelithiasis, osteoarthritis, infertility, and an increased risk of cancer [4].
Diabetes mellitus has become another major pandemic causing huge cost to the healthcare system. The incidence of diabetes mellitus among people aged 20 to 79 was 6.4% globally in 2010 and is expected to increase to 7.7% by 2030 [5]. In developing nations, the prevalence of type 2 diabetes (T2DM) is expected to increase by 69%, compared to 20% in developed nations. According to International Diabetes Federation (IDF) s from 2017, there are 425 million diabetics globally [6].
Moreover, the link between type 2 diabetes mellitus (T2DM) and obesity has become a major public health concern in recent years, posing a significant socioeconomic burden on society. Unhealthy eating patterns, sedentary lifestyles, and genetic predisposition all contribute to the unresolved burden of diabetes and obesity. Diagnosis of diabetes mellitus is made when the fasting plasma glucose (FPG) is ≥126mg/dL (7.0 mmol/L)/ HbA1C ≥6.5% (48mmol/mol). Impaired fasting plasma glucose is diagnosed when FPG ranging from 100 to 125mg/dL (from 5.6 to 6.9mmol/L)/HbA1C 5.7%-6.4% [7].
There is a well-established link between the development of T2DM and being overweight or obese. The data suggests a 7-fold increase in the possibility of T2DM in those with obesity. Current models predict 9.5% of adults will have diabetes by 2030, where one-third of this would be directly attributable to obesity [8]. There is a large number of research supporting the link between rising BMI and increased morbidity and mortality in those who are affected. Each 5 units increment of BMI, lead to rise in total mortality, chronic kidney disease and diabetes mellitus by 30%, 60% and 120% respectively [9].
During the recent past, significant emphasis was given for diabetes remission. Although T2DM was historically thought to be an incurable and progressive disease, the WHO global report on diabetes first highlighted the reversal of T2DM in 2016 [10]. The American Diabetes Association (ADA) favoured HbA1c below the level currently used for initial diagnosis of diabetes, 6.5% (48mmol/mol), and remaining at that level for at least 3months without continuation of the usual anti-hyperglycemic agents as the main defining measurement of diabetes remission [11,12]. An FPG lower than 126mg/dL (7.0 mmol/L) can in some settings be used as an alternate criterion for remission, even though it has certain disadvantages like requiring sample collection while fasting overnight, together with significant variation between repeated measurements [12].
Bariatric surgery is a well-recognized, effective treatment option for patients with obesity and for those with T2DM. Studies reported that the advantages of bariatric surgery go beyond the simple weight loss. Sri-Lankan experience suggests that bariatric surgery leads to loss of 10kg in the first month, another 20kg by 9–12months, and this rapid weight loss may result in massive cardiovascular and other health benefits [13]. Available studies clearly demonstrate the benefit of bariatric surgery in achieving cardiovascular health in association with improvement of blood pressure, plasma glucose, lipid and fatty liver [14,15]. One Sri Lankan study published recently has shown a clinically meaningful improvement of 10-year Framingham cardiovascular risk score following bariatric surgery [16].
Bariatric surgery by decreasing the chronic inflammation in obesity and altering the biomarkers and the gut microbiota leads to long-term remission of T2DM along with cardiovascular benefits. Systematic reviews on how bariatric surgery affects diabetes remission show an 80% success rate for diabetic remission [17]. A study comparing surgical vs non-surgical management revealed that 73% of the surgical group has achieved diabetes remission compared to 13% in intensive medical treatment arm [18]. In order to evaluate the remission of diabetes following bariatric surgery, we conducted a retrospective analysis among the patients who underwent the procedure at a renowned bariatric surgical facility in Sri Lanka. This is the first instance of T2DM remission following bariatric surgery in Sri Lanka.
Methodology
The obesity services clinic at the Colombo South Teaching Hospital, Sri Lanka, the country’s leading tertiary care facility for bariatric surgery, was the site of this study. A retrospective analysis of clinical follow-up data of the clinic attendees was carried out for this study. The indications for bariatric surgery in Sri Lanka is BMI≥35kg/m2 (even when there are no medical problems) or BMI≥30kg/m2 with ≥1 obesity related comorbidities such as: T2DM, hypertension, dyslipidemia, obstructive sleep apnoea/obesity Hypoventilation syndrome, non-alcoholic fatty liver disease /nonalcoholic steatohepatitis, debilitating arthritis, impaired quality of life particularly among who have failed to lose weight or to maintain long term weight loss, despite appropriate comprehensive medical care [19].
The objective of the study was to assess the short (1year) and long (5years) term effects of bariatric surgery on glycaemic control and to determine the proportion of patients who had undergone T2DM remission following bariatric surgery. A database was designed to collect demographic, pre-operative, and post-operative clinical data in patients with obesity undergoing bariatric surgery at the above center. Completely anonymized details were collected from previously consented patients. All patients subjected to Bariatric surgery from 2013 to 2020 with a background history of T2DM prior to surgery, who have completed at least 12months of post-operative follow-up visits were included in the first analysis of short-term outcomes at 1year. Patients who had the surgery during the above time period and who completed a 5-year follow-up were included for assessment of long-term outcomes. All patients with T2DM who had bariatric surgery fulfilling the above criteria were included.
The data on the demography, anthropometry, duration of diabetes prior to surgery, anti-diabetic medications and laboratory test results on FPG, and HbA1c done within 2 weeks prior to surgery were collected as pre-operative baseline data. Anthropometry, HbA1c and FPG results, and data on antiglycaemia medications were collected at post-operative 1 and 5-year follow-ups. In this study, impaired fasting glucose was defined as FBS between 100 and 125mg/dl or HbA1C between 5.7% and 6.4%, and diabetes mellitus as FBS equal to or greater than 126mg/dl or HbA1C equal to or greater than 6.5% [11,12]. Moreover, diabetes remission was diagnosed when the FPG is <126mg/dL without any antidiabetic medications [12].
Institutional ethics review board approval for retrospective analysis of above clinical data was obtained. Data were analyzed using SPSS version 25 and the findings were presented using median, mean (standard deviations) and percentages. Improvement of glycaemia status compared to the baseline was assessed by analysis of variance (means), medians, inter ranges and chi-squared tests as needed. P value of <0.05 was considered as the level of statistical significance.
Results
Among the 245 patients who underwent bariatric surgery between 2013 and 2020, 109 (44.4%) had pre-operative T2DM, 44 (17.9%) had IFG, and 92 (37.7%) had complete normoglycemia with FBS below 100mg/dl and HbA1C below 5.6%. At the time of the baseline survey, there were 75 (68.9%) females and 34 (31.1%) men among those with diabetes. Only the cohort of patients with pre-operative T2DM (n=109) was included in the outcome analysis. The pre-operative population with diabetes ranged in age from 13 years to 65years. The mean age of the population was 40±10 years. Majority (71.6%, n=78) of the patients with T2DM awaiting bariatric surgery were females. Over 50% of the population had a BMI between 40.1-50kg/m2. Diabetes remission rate at 1 year among males (90.3%) were higher than the females (84.6%). And the long-term diabetes remission rate was 80% among males and 91% among females.
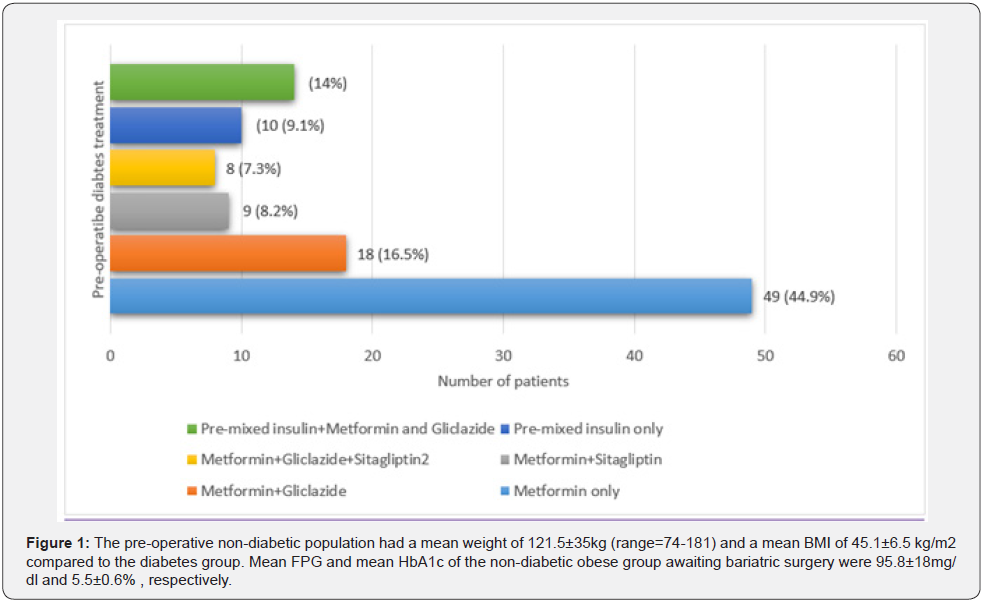
Analysis of pre-operative data
The diabetes group had a mean weight of 115.8±24.4 kg (range = 75-204) and a mean pre-operative BMI of 45.4±7.4 kg/m2 (range = 31.2-68.1) (Table 1). Their mean HbA1c level was 8.5±1.6 % and mean FPG was 140± 47mg/dL. The average duration of diabetes prior to bariatric surgery was 3.5years. The averages of FPG and HbA1C for the IFG group were 106±11 mg/dl and 5.8± 0.6% respectively (Figure 1).
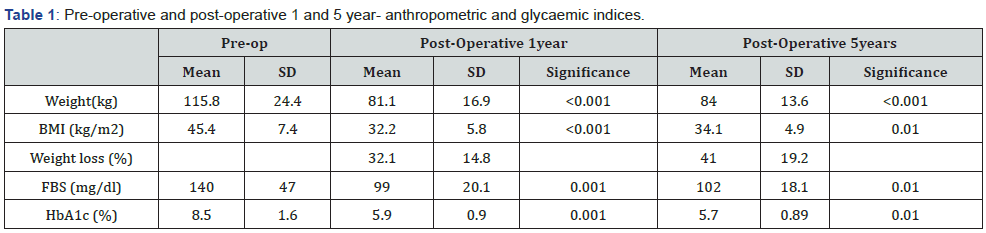
Short term improvement of glycaemic status- postoperative 1 year follow up
The group who had pre-operative diabetes had a mean weight of 81.1kg (SD=± 16.9) (IOR= 52-140) and a mean BMI of 32.2kg/ m2±5.8 with a mean weight loss of 32.1±14.8kg at 1 year follows up. At one year’s follow-up, the pre-operative diabetes group’s mean HbA1c, which was 8.5% at baseline, had significantly decreased to 5.5%±0.9 (range=4.4%-6.5%) and their mean FPG was reduced from 140mg/dl to 99±23mg/dl. At 1-year follow-up, 94 patients (87.1%) who had pre-operative diabetes improved blood sugar levels achieved diabetes remission according to the criteria we discussed above (FPG to non-diabetic range of less than 126mg/dl along with the ability to completely omit all diabetes medications, including oral medications and insulin).
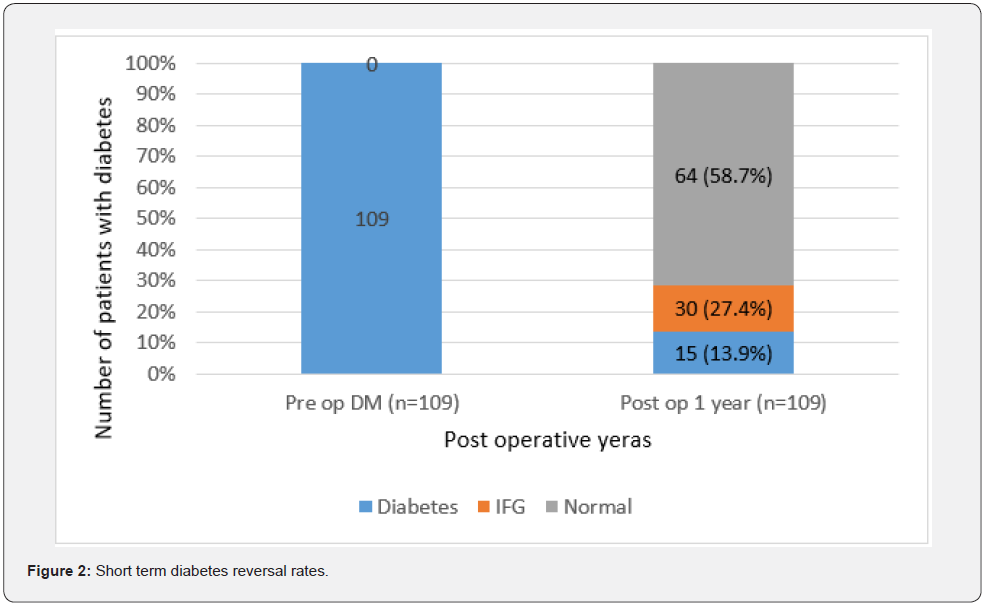
Among those who achieved diabetes remission at 1year, 64 patients (58.7%) reached complete normoglycemia with fasting blood glucose values of less than 100mg/dl whereas 30 patients (28.4%) were in impaired fasting glucose category. There were 15 patients with persistent diabetes at 1year follow up which was 13.9% of initial diabetic population (Figure 2). Among the 15 with persistent diabetes at 1 year, 8 patients were treated solely with oral hypoglycemic medications (Metformin alone in 3, Gliclazide and Metformin in 2, Sitagliptin and Metformin in 2, Metformin, Gliclazide, and Sitagliptin in 1 patient), and 7 were treated with pre-mixed insulin in conjunction with Metformin. Only three of the patients with persistent diabetes who were taking a combination of two oral medications continued to take them at the same pre-operative doses; all the other patients with persistent diabetes had the option of reducing the quantity of oral hypoglycemic medications or drug doses, or both. Pre-operatively, 24 patients (all on premixed insulin) required insulin; however, after the 1-year follow-up 70% of them achieved remission of diabetes while only 7 patients were still using pre-mixed insulin. In addition, all the individuals who needed insulin at 1 year had reduced requirement for daily dose of insulin.
Among the pre-operative IFG patients (n=44), 95.4% of majority (n=42) achieved completely normal blood glucose level of less than 100mg/dl while 2 (4.6%) remained in the IFG category. Pre-operative mean fasting glucose of 106mg/dl declined to 90mg/dl whereas mean HbA1C improved to 5.2% from the preoperative value of 5.8% which was statistically significant (Figure 3). Pre-operative and post-operative blood glucose distribution and changes in weight, BMI, FBS and HbA1C are summarized in table 1.
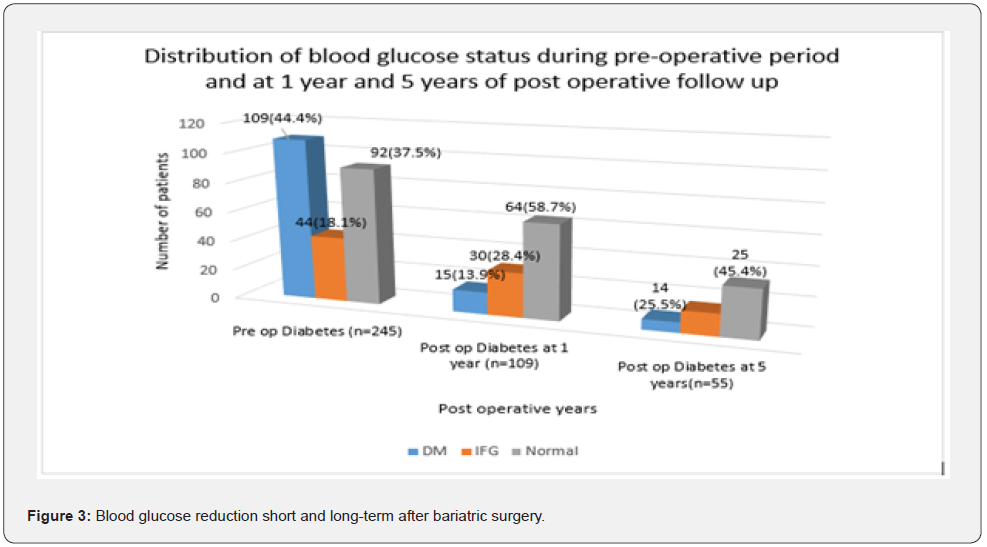
Long-term effects of Bariatric surgery on improvement of blood glucose level-Analysis of data at 5years. 57 patients with preoperative diabetes from the original 109 population were eligible to complete the post-operative 5-year follow-up after the surgery and 55 had continuous follow up where 2 were defaulted. Further analysis revealed a mean weight of 84kg± 13.6 (range= 50-107) at 5years and a mean BMI of 34.1 kg/m2±4.9 (range=22-45). Mean weight loss was 41.0kg±19.2 (range=29-63). Mean fasting blood glucose was 102mg/dl±18.1 (range=78-116) at 5 year follow up which showed a 29.2% reduction from the pre-operative value, though there was a slight increment of mean value from 99 to 102 when comparing with the baseline at 1 year. Mean HbA1c of 5.7%±0.89 was seen at 5year follow up with a 32.9% improvement compared to pre-operative value. However, there was an upward trend of HbA1c, mean weight and mean BMI when compared to 1 year value.
Separate analysis of the initial pre-operative population who were eligible for 5years follow up (n=55) revealed a diabetes remission among 41 (74.5%). Among them, 25 (45.4%) achieved normoglycemia with blood glucose level of <100mg/dl and 16 (29%) who had diabetes reversal were in the impaired plasma glucose range. 14 patients (25.5%) were having diabetes at 5years follow up with persistence of pre-operative diabetes in 13 patients with the new occurrence of diabetes in 1 patient.
Does the diabetes reversal depend on the baseline characteristics of the study population?
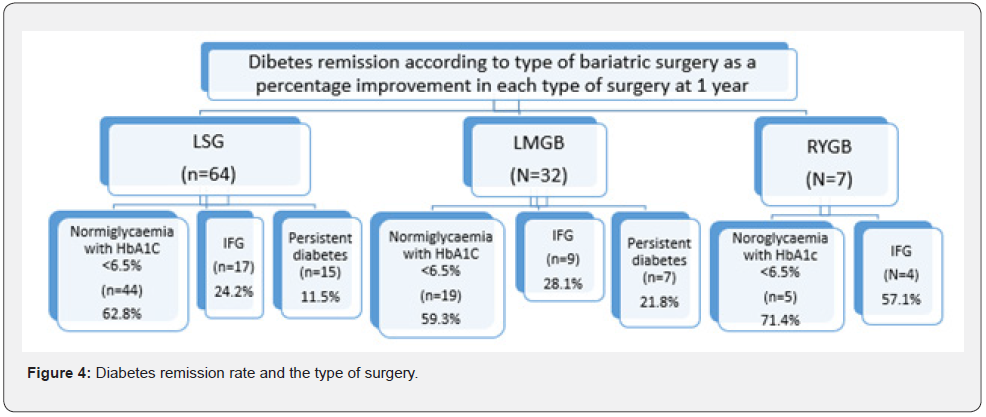
Patients who were diagnosed with DM before the age of 40years had higher improvement of mean HbA1C, mean FPG, and diabetes remission rates compared to the group who first developed the disease after the age of 40 (p=<0.05). The longterm diabetes remission rate was 100% among those who were <30years of age at the time of surgery. Pre-operative BMI had no impact on diabetes remission. (p=0.35). Of the 55 patients who completed their 5years follow up 45 had LSG, 9 had LMGB and only 1 had RYGB. In the LSG cohort, 32 (71.1%) achieved diabetes remission, improvement to the state of impaired glucose was seen in 7(15.5%) and persistent diabetes was seen in 5 (11.1%). There was one patient who had new onset of diabetes in this group. Among those who underwent LMGB 55.5% achieved long-term diabetes remission. Only 1 had undergone RYGB and that patient achieved improvement of initial diabetes up to a level of IFG. After 5 years from surgery both LSG and LMGB both had a similar rate of persistence of diabetes (Figure 4).
Discussion
Research on remission of Type 2 DM has drawn a great deal of attention over recent years. It has indeed revolutionized the treatment approaches in managing Type 2 DM. Bariatric surgery is a great advancement in contemporary medicine that provides outstanding results in terms of remission of diabetes mellitus.
Table 2 summarizes studies which reported diabetes remission upon bariatric surgery. Our study aimed to assess both short-term (1-year follow-up) and long-term (5-years follow-up) remission rates and associated patterns and predictors of Type 2 DM remission among the largest cohort of bariatric surgery patients in the country with island-wide participation. Bariatric surgery leads to rapid improvement in glycemic parameters, possibly within hours to days even before it causes significant loss of weight [20]. This may be related to a novel hormonal milieu in response to structural changes in the gastrointestinal tract and changes in the gut microbiota [21]. Several-fold greater concentrations of GLP-1 levels are noticed in blood in response to meals. This may impact peripheral metabolism, central appetite regulation, and food intake. As changes in physiology and anatomy are permeant, glucose homeostasis achieved by these mechanisms is long-lasting [20,22]. Systematic reviews on the effects of bariatric surgery on diabetes remission demonstrate a reversal of diabetes among 80% [17]. A study comparing surgical vs non-surgical management revealed that 73% of the surgical group has achieved diabetes remission compared to 13% in the intensive medical treatment arm [18].
Studies have shown that bariatric surgery is associated with a diabetes remission rate of 72% after 2years of surgery [23]. Five years after surgery, complete remission as indicated by FPG of less than 100mg/dl was achieved in 46.6% whereas partial remission as indicated by FPG 100-126mg/dl was achieved among 12.0% in the same study [23]. Long-term remission rates of diabetes in bariatric surgery rely on multiple factors, such as type of surgery, patient adherence to lifelong dietary changes, and continuing medical surveillance [23]. However, current data suggest a less durable long-term resolution of diabetes. SLEEVEPASS randomized controlled trial showed a remission of T2DM in 37% of sleeve gastrectomies and 45% of RYGB patients, which accounts for 30-50% of long-term diabetes remission [24]. Shorter T2DM duration, younger age, better pre-operative glycemic control, absence of pre-operative insulin treatment, baseline BMI, baseline C-peptide level and degree of physical activities are some of the suggested factors affecting remission of diabetes following Bariatric surgery [25].
In our study, the mean weight of 115.8kg before surgery decreased to 81.1 kg after one year and remained at 84kg after surgery for five years. Mean weight loss at 1 year was 32.1kg±14.8 and at 5years was 41.9kg±19.2. Mean HbA1c of 8.5% at baseline has reduced up to 5.5%±0.9 at 1year and mean HbA1C at 5 years was 5.7%±0.89 which showed a rise in comparison to 1-year post-surgery values. The literature suggests that the remission of diabetes was related to the degree of weight loss achieved and higher BMI before the surgery is associated with greater diabetes remission rates [23]. Our study showed 84% and 90% diabetes reversal rates for BMI categories of 40.1-50kg/m2 and 50.1-60kg/ m2 respectively. However extreme BMI category of above 60 kg/ m2 had a less remission rate (40%).
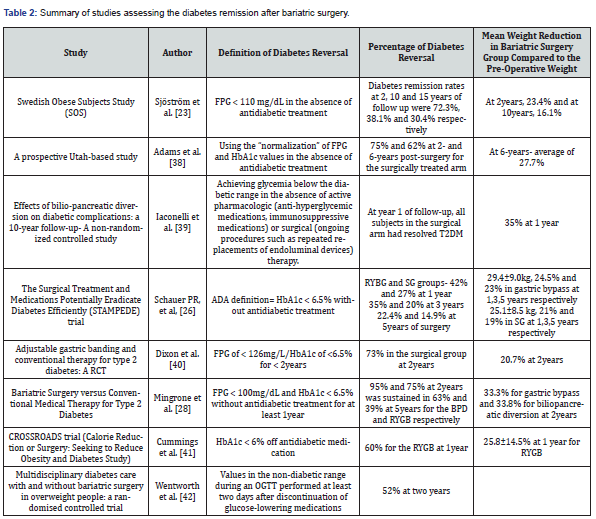
Consistent with published data, gastric bypass is associated with greater diabetes remission compared to sleeve gastrectomy [26]. 5-year follow-up data of a study, comparing diabetes remission in 61 patients with T2DM who had RYGB or laparoscopic gastric banding (LAGB), or lifestyle modification showed 60% in the RYGB group and 50% of the LAGB group had diabetes remission at 5 years. Also, there was a reduction in the long-term maintenance rate from 60% in 1st year to 30% in 5 years though the weight loss was maintained nearly at the same rate (27% vs 25%) [27]. A landmark study by Mingrone et al. compared remission rates of diabetes among three groups (RYGB, biliopancreatic bypass (BPD) and conservatively treated arm). This study has considered remission of diabetes when they achieved FPG<100mg/dL and HbA1c<6.5% without the need for insulin or oral hypoglycemic drugs for at least 1 year. Remission rates were reported as 95%, 75% and 0% at 2years for the BPD, RYGB and conservative arms, [28] respectively with a sustained remission of 63% and 39% at 5 years for BPD and RYGB, respectively [29].
Our data reveal a >70% remission rate of diabetes in all types of surgeries performed, however, the highest improvement rates were seen in the RYGB group. Longitudinal Assessment of Bariatric Surgery 2 (LABS-2), a large prospective cohort study on long-term results of RYGB) demonstrated a significant glycemic improvement that was beyond the usually expected weight loss. In most patients, RYGB affects the improvement of glycemic status even before a significant weight loss compared to other surgical procedures [30,31]. There has been no major difference in the outcome between laparoscopic RYGB vs open RYGB [31,32]. Our study reveals that the majority in the baseline population was in 30-40- and 40-50-year groups and persistence of diabetes at 5 years was mostly seen above the 40-year population. The sustainability of diabetes remission and lack of diabetes recurrence were mostly seen among younger patients and in patients treated with dietary measures or oral drugs compared to insulin therapy [33]. Age was defined as a negative factor for diabetes remission by Aung et al suggesting the late onset of diabetes after the age of 40 is associated with less sustainability of diabetes remission after bariatric surgery [34]. This may be further reinforced by the finding of lesser weight loss in older individuals after bariatric surgery [35]. Similarly, our study also demonstrated a higher T2DM remission rate in the less than 50 age group when compared to people more than 50 years (88% vs 64%). (p=<0.05).
The relationship between gender and diabetes remission lacks clarity [36]. However, according to our study, the majority of the people with diabetes awaiting Bariatric surgery were females 71.6% (n=78). Males had a 90.4% reversal of diabetes at 1 year whereas females had a rate of 84.6%. The higher diabetes remission rates in males observed in our study could be due to the lower number of male participants in the study. Pre-operative insulin therapy was negatively associated with diabetes remission [37]. Even though some suggest pre-operative BMI is a marker of diabetes remission, our study did not demonstrate any such association.
Strengths and limitations
This was the first study on diabetes remission following Bariatric surgery in Sri Lanka. As the country’s leading centre for Bariatric surgery, we provide care for people with obesity islandwide, increasing the generalizability of the data. The successful follow-up rate of the patients and the good quality of data are other positive aspects of our study. Our study focused on shortterm remission of diabetes at 1-year follow-up and long-term remission of diabetes by considering the blood sugar results at post-operative 5 years adhering to the defined remission criteria, particularly the prolonged remission. A major limitation of our study was the retrospective analysis of existing data. However, most of the findings were compatible with available regional and global data with a significantly higher number (>85%) achieving diabetes remission in 1 year.
Conclusion
Bariatric surgery is associated with both short- and long-term diabetes remission.
Author Contribution
DA did data collection, manuscript writing, organizing and reviewing, UAW, UA, CA did data collection and analysing, KA did manuscript writing, CG did organize and analysing, NMW did proof reading, UB did critical revision of manuscript, GMR did data collection, organizing, coordinating and reviewing.
Ethical Approval
Ethical approval was obtained from the institutional ethics committee.
Acknowledgement
WE acknowledge the work of research assistant and in charge medical officer of bariatric clinic at Colombo South Teaching Hospital for their enormous help.
References
- Frühbeck G, Toplak H, Woodward E, Yumuk V, Maislos M, et al. (2013) Obesity: the gateway to ill health - an EASO position statement on a rising public health, clinical and scientific challenge in Europe. Obes Facts 6(2): 117-120.
- Kelly T, Yang W, Chen CS, Reynolds K, He J (2008) Global burden of obesity in 2005 and projections to 2030. Int J Obes (Lond) 32: 1431-1437.
- Park Y, Wang S, Kitahara CM, Steven C. Moore, Amy Berrington de Gonzalez, et al. (2014) Body mass index and risk of death in Asian Americans. Am J Public Health 104(3): 520-525.
- Djalalinia Shirin, Mostafa Qorbani, Niloofar Peykari, Roya Kelishadi (2015) Health impacts of Obesity. Pak J Med Sci 31(1): 239-2342.
- Lozano R, Naghavi M, Foreman K (2013) Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 380: 2095-2128.
- Shaw JE, Sicree RA, Zimmet PZ (2010) Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res Clin Pract 87(1): 4-14.
- (2022) American Diabetes Association Professional Practice Committee. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes—2022. Diabetes Care 45(Suppl 1): S17-S38.
- Gatineau M, Hancock C, Holman N (2014) Adult obesity and type 2 diabetes. Public Health England.
- Berringtonde Gonzalez A, Hartge P, Cerhan JR, Alan J Flint, Lindsay Hannan, et al. (2010) Body-mass index and mortality among 1.46 million white adults. N Engl J Med 363(23): 2211-2219.
- (2016) World Health Organization Global Report on Diabetes.
- Ramos-Levi AM, Cabrerizo L, Matia P, Sanchez-Pernaute A, Torres AJ, et al. (2013) Which criteria should be used to define type 2 diabetes remission after bariatric surgery. BMC Surgery 13: 8.
- Matthew C Riddle, William T Cefalu, Philip H Evans, Hertzel C Gerstein, Michael A Nauck, et al. (2021) Consensus Report: Definition and Interpretation of Remission in Type 2 Diabetes. Diabetes Care 1 October 44(10): 2438-2444.
- Uditha Bulugahapitiya, Thejana Wijeratne, Ranil Jayawardena, Sarath Lekamwasam, Asela Gunawardena, et al. (2021) The First Sri Lankan Experience on Laparoscopic Bariatric Surgery. Obesity Surgery 44(10): 2438-2444.
- Gunatilake S, Weerakoon D, Subasinghe C, Bulugahapitya U, Wijerathna T, et al. (2019) Short term metabolic outcome following bariatric surgery; A single center experience from Asia. Endocrine Abstracts, 63: P903.
- Wijetunga Udai, Bulugahapitiya Uditha, Wijerathne T, Jayasuriya Anuradha, Ratnayake Gowri et al. (2019) SAT-104 Reversal of Nonalcoholic Fatty Liver Disease with Bariatric Surgery in South Asians: Has the Cure Been Finally Found? Real World Data from a Sri Lankan Tertiary Care Setting. J Endocr Soc 3(Suppl 1): SAT-104.
- Abeyratne D Wellala, V Wijethunga, U Alahakoon, C Abeysinghe, G Ratnayake, et al. (2022) Improvement of Framingham 10-year risk score in an obese population undergoing bariatric surgery: single centre experience in a Tertiary care Hospital-Sri Lanka: [Abstract]. ZoomForward2022: European Congress on Obesity. Obes Facts 15(Suppl 1): 1-240.
- Purnell JQ, Selzer F, Wahed AS, Pender J, Pories W, et al. (2016) Type 2 Diabetes Remission Rates After Laparoscopic Gastric Bypass and Gastric Banding: Results of the Longitudinal Assessment of Bariatric Surgery Study. Diabetes Care 39(7): 1101-1107.
- Ferrannini E, Mingrone G (2009) Impact of different bariatric surgical procedures on insulin action and beta-cell function in type 2 diabetes. Diabetes Care 32(3): 514-520
- (2018) Guidelines for management of overweight and obesity, Guideline for Health care Providers, non-communicable disease unit, Ministry of Health, Nutrition and Indigenous Medicine. (1st), Sri-Lanka, India, p. 21
- Federico A, Dallio M, Tolone S, Gravina AG, Patrone V, et al. (2016) Gastrointestinal Hormones, Intestinal Microbiota and Metabolic Homeostasis in Obese Patients: Effect of Bariatric Surgery. In Vivo 30(3): 321-330.
- Medina DA, Pedreros JP, Turiel D, Quezada N, Pimentel F, et al. (2017) Distinct patterns in the gut microbiota after surgical or medical therapy in obese patients. PeerJ 5: e3443.
- Hutch CR, Sandoval D (2017) The Role of GLP-1 in the Metabolic Success of Bariatric Surgery. Endocrinology 158(12): 4139-4151.
- Sjöström L, Lindroos AK, Peltonen M, Torgerson J, Bouchard C, et al. (2004) Swedish Obese Subjects Study Scientific Group. Lifestyle, diabetes, and cardiovascular risk factors 10 years after bariatric surgery. N Engl J Med 351(26): 2683-2693.
- Jans Anders, Näslund Ingmar, Ottosson Johan, Szabo Eva, Näslund Erik, et al. (2019) Duration of type 2 diabetes and remission rates after bariatric surgery in Sweden 2007-2015: A registry-based cohort study. PLOS Medicine 16(11): e1002985.
- Adams TD, Gress RE, Smith SC, Halverson RC, Simper SC, et al. (2007) Long-term mortality after gastric bypass surgery. N Engl J Med 357(8): 753-761.
- Salminen P, Helmio M, Ovaska J, Juuti A, Leivonen M, et al. (2018) Effect of laparoscopic sleeve gastrectomy vs. laparoscopic Roux-en-Y gastric bypass on weight loss at 5 years among patients with morbid obesity: The SLEEVEPASS randomized clinical trial. JAMA 319(3): 241-254.
- Tsilingiris D, Koliaki C, Kokkinos A (2019) Remission of Type 2 Diabetes Mellitus after Bariatric Surgery: Fact or Fiction? Int J Environ Res Public Health 16(17): 3171.
- Schauer PR, Bhatt DL, Kirwan JP, Wolski K, Aminian A, et al. (2017) Bariatric surgery versus intensive medical therapy for diabetes—5-year outcomes. N Engl J Med 376(7): 641-651.
- Courcoulas AP, Gallagher JW, Neiberg RH, Emily B Eagleton 1, James P DeLany, et al. (2020) Bariatric surgery vs lifestyle intervention for diabetes treatment: 5-year outcomes from a randomized trial. J Clin Endocrinol Metab 105(3): 866-876.
- Mingrone G, Panunzi S, De Gaetano A, Guidone C, Iaconelli A, et al. (2012) Bariatric surgery versus conventional medical therapy for type 2 diabetes. N Engl J Med 366(17): 1577-1585.
- Mingrone G, Panunzi S, De Gaetano A, Guidone C, Iaconelli A, et al. (2015) Bariatric-metabolic surgery versus conventional medical treatment in obese patients with type 2 diabetes: 5 year follow-up of an open-label, single-centre, randomised controlled trial. Lancet 386(9997): 964-973.
- Mario Kratz (2020) Long-Term Diabetes Remission Rates after Bariatric Surgery: Surprisingly Low in Spite of Sustained Weight Loss. The Journal of Clinical Endocrinology & Metabolism 105(6): e2291-e2293,
- Belle SH, Chapman W, Courcoulas AP, Flum DR, Gagner M, et al. (2008) Relationship of body mass index with demographic and clinical characteristics in the Longitudinal Assessment of Bariatric Surgery (LABS). Surg Obes Relat Dis 4(4): 474-480.
- Flum DR, Belle SH, King WC, Wahed AS, Berk P, et al. (2009) Perioperative safety in the longitudinal assessment of bariatric surgery. N Engl J Med 361(5): 445-454.
- Chikunguwo SM, Wolfe LG, Dodson P, Meador JG, Baugh N, et al. (2010) Analysis of factors associated with durable remission of diabetes after Roux-en-Y gastric bypass. Surg Obes Relat Dis 6(3): 254-259.
- Aung L, Lee WJ, Chen SC, Ser KH, Wu CC, et al. (2016) Bariatric surgery for patients with early-onset vs late-onset type 2 diabetes. JAMA Surg 151(9): 798-805.
- Chang WW, Hawkins DN, Brockmeyer JR, Faler BJ, Hoppe SW, et al. (2019) Factors influencing long-term weight loss after bariatric surgery. Surg Obes Relat Dis 15(3): 456-461.
- Jensen MD (2008) Role of body fat distribution and the metabolic complications of obesity. J Clin Endocrinol Metab 93(11 Suppl 1): S57-63.
- Blackstone R, Bunt JC, Cortes MC, Sugerman HJ (2012) Type 2 diabetes after gastric bypass: remission in five models using HbA1c, fasting blood glucose, and medication status. Surg Obes Relat Dis 8(5): 548-555.
- Adams Ted D, Gress Richard E, Smith Sherman C, Halverson R Chad, Simper Steven C, et al. (2007) Long-Term Mortality after Gastric Bypass Surgery. New England Journal of Medicine 357(8): 753-761.
- Iaconelli A, Panunzi S, De Gaetano A, Manco M, Guidone C, et al. (2011) Effects of bilio-pancreatic diversion on diabetic complications: a 10-year follow-up. Diabetes Care 34(3): 561-567.
- Dixon JB, O'Brien PE, Playfair J, Chapman L, Schachter LM, et al. (2008) Adjustable gastric banding and conventional therapy for type 2 diabetes: a randomized controlled trial. JAMA 299(3): 316-323.
- Cummings DE, Arterburn DE, Westbrook EO, Kuzma JN, Stewart SD, et al. (2016) Gastric bypass surgery vs intensive lifestyle and medical intervention for type 2 diabetes: the CROSSROADS randomised controlled trial. Diabetologia 59(5): 945-953.
- Wentworth JM, Playfair J, Laurie C, Ritchie ME, Brown WA, et al. (2014) Multidisciplinary diabetes care with and without bariatric surgery in overweight people: a randomised controlled trial. Lancet Diabetes Endocrinol 2(7): 545-552.






























