Role of Resistin in Type 2 Diabetes Mellitus and Obesity in HHcy-Patients. Opposite Effects of Intermedin in Experimental Models
Federico Cacciapuoti1* and Fulvio Cacciapuoti2
1Department of Internal Medicine, “L. Vanvitelli” University, Italy
2Cardiology and ICU, “V. Monaldi” Hospital, Italy
Submission: July 03, 2022; Published: July 15, 2022
*Corresponding author: Federico Cacciapuoti, Department of Internal Medicine, L. Vanvitelli” University, Naples, Italy
How to cite this article: Federico C, Fulvio C. Role of Resistin in Type 2 Diabetes Mellitus and Obesity in HHcy-Patients. Opposite Effects of Intermedin in Experimental Models. Curre Res Diabetes & Obes J 2022; 16(1): 555926.DOI: 10.19080/CRDOJ.2022.16.555926
Abstract
Increased homocysteine serum levels (HHcy) cause the over-expression of Resistin, that yields the inflammation of adipocytes by some inflammatory cytokines, responsible of obesity, and insulin resistance often evolving towards type 2 diabetes mellitus and some cardiovascular diseases. Contrarily to Resistin, Intermedin of the calcitonin family, ameliorates HHcy-induced inflammation of adipose tissue, favoring the weight loss and the insulin sensitivity. The anti-inflammatory effects on adipocytes developed by this peptide depend by a novel mechanism that balances the pro-inflammatory/anti-inflammatory cytokines ratio, synthesized such as M1/M2 ratio. Insulin sensitivity and weight loss were obtained in mice treated with Intermedin. In the future, Intermedin administration could be used for type 2 diabetes mellitus and overweight treatment in humans too. But further studies are requested to evaluate that.
Keywords: Hyperhomocysteinemia; Type 2 diabetes mellitus; Adipocytes; Obesity; Resistin; Intermedin
Abbreviations: HHcy: HyperHomocysteinemia; T2DM: Type 2 Diabetes Mellitus; ERS: Endothelial Reticulum Stress; TNF: Tumor Necrosis Factor; IL-6: InterLeukin-6P; JNK: c-Jun-N-Kinase; Akt: Activity-Threonine Kinase; Hcy: Homocysteine; Met: Methionine; MTHFR: Methilene- Tetra-Hydro-Folate Reductase; CBS: Cystationine-Beta-Synthase; CSE: Cystationine-Gamma-Lyase; ROS: Reactive Oxygen Species; LDL: Low Density Lipoprotein; IMD: Intermedin; AM2: Adreno Medullin 2; AMP: Adenyl Mono Phosfate; AMPK: Activation Protein Kinase; PKA: Protein Kinase Activationue; WAT: White Adipose Tissue
Introduction
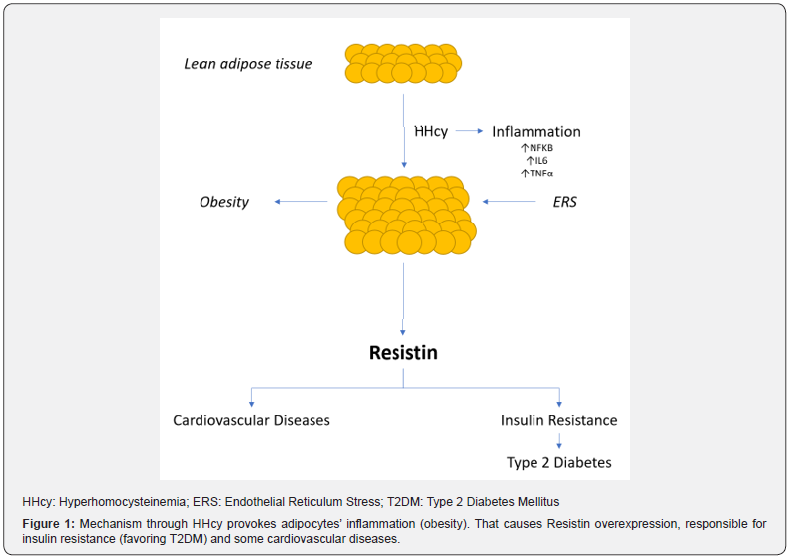
Some evidence suggest that hyperhomocysteinemia (HHcy) can favor insulin resistance, responsible for increased glycemic serum levels. That can provoke pre-diabetes or type 2 diabetes mellitus (T2DM) and/or other numerous pathological conditions, such as obesity and some cardiovascular diseases é [1-4]. Li et al. [4] hypothesized that, in experimental hyper-homocysteinemicmodels, insulin resistance was obtained through the expression of Resistin from adipose tissue via endothelial reticulum stress (ERS). In detail, HHcy causes an inflammatory reaction of adipocytes, responsible to produce some pro-inflammatory cytokines, such as tumor necrosis factor-alfa (TNF-alfa), and interleukin (IL-6). In turn, these activate c-jun N terminal-kinase (JNK) which inhibits serine/threonine protein-kinase (Akt) activity that impairs insulin signaling pathway, causing the over-expression of Resistin. On the other hand, Resistin overexpression, with consequent insulin resistance, represents a frequent cause of both T2DM and obesity [4-7].
Homocysteine
Homocysteine (Hcy) is an amino acid formed during the metabolism of Methionine (MET). Its storage (HHcy) depends on the defective re-methylation and/or trans-sulfuration pathways [8]. Particularly, re-methylation route favors Met re-synthesis. On the contrary, trans-sulfuration pathway concerns the further metabolism of Hcy until its final product (cysteine). Both pathways require some enzymes, respectively for Met synthesis (MTHFR) and cysteine production (CBS, CSE) [9]. But these enzymes require, to operate, some coenzymes, as folic acid, vitamin B12 (MTHFR) and vitamin B6 (CBS, CSE). Their deficiency induces HHcy [10], that besides acts as a potent inflammatory factor of adipocytes [11]. Previously, it was demonstrated that Hcy is able to induce expression of proinflammatory factors by the mediation of oxidative stress [12]. Thus, HHcy can be considered a chronic inflammatory condition [13]. On the other hand, this inflammation of adipocytes causes the Resistin expression through some proinflammatory cytokines [2].
Resistin
HHcy, a cysteine rich peptide hormone, may promote insulin resistance, through the Resistin expression from adipocytes in rodents, via the activation of the inflammatory pathways. In humans, contrarily to the rodents, the source of Resistin is mainly represented by macrophages, that are similar to adipocytes [14]. Resistin over-expression can be obtained in HHcy-patients through the generation of Reactive Oxygen Species (ROS), that weaken the function of insulin secreting cells. Resistin works by helping the body’s cells to absorb glucose. In turn, the body converts this into fat, inducing obesity, especially of the visceral type [15].
Resistin is a hormone involved in the pathogenesis of insulin resistance and may link T2DM with obesity through some inflammatory markers [16] (Figure 1). But, Resistin is also associated to atherosclerosis, endothelial dysfunction, some cardiovascular disease, alcoholic fatty liver disease and inflammation [17-19]. In addition, Resistin is responsible for high levels of low-density lipoproteins (LDL) and favors the accumulation of these in the arteries, with consequent atherosclerosis [20]. Its role in pro-inflammatory processes has been demonstrated in several studies [21,22]. High levels of Resistin have also been associated with other chronic inflammatory diseases, such as rheumatoid arthritis, but its role in systemic lupus erythematous is controversial [23,24]. In addition, Resistin, and the derived insulin resistance, can act as anti-apoptoic, pro-angiogenic molecule with pro-metastatic activity and regulate metabolic activities in cancer’s cells [25-27].
Intermedin
Contrarily to Resistin, Intermedin (IMD), also known as adrenomedullin 2 (AM2), a calcitonin family peptide, acts with opposite effects of Resistin on insulin resistance and obesity [28,29]. Referring to HHcy, it was demonstrated that IMD ameliorates HHcy-induced chronic inflammation in adipose tissue and improves insulin sensitivity in experimental models. In addition, IMD protects the myocardium from ischemia-reperfusion injury, by inhibiting oxidative stress [30]. As previously affirmed HHcy promotes the inflammatory state of adipocytes, through the Resistin expression. A novel mechanism was proposed by Pang et al. for Hcy-induced macrophages’ inflammation. That consists in the prevalence of M1/M2 ratio (M1 consists of pro-inflammatory cytokines, as IL-1, IL-6, TNF-alfa; M2 includes anti-inflammatory cytokines, such as IL-1, IL-4, IL-6, IL-10, IL-11, IL-13). The impaired M1/M2 ratio favors the inflammation of macrophages causing Resistin expression [31]. Contrarily to this mechanism, the over-expression of IMD balances the M1/M2 ratio, reducing the inflammatory reaction of adipocytes and inhibits insulin resistance [32] (Figure 2). This effect happens by the activation of AMP-activated protein kinase (AMPK). That stimulates glucose uptake in skeletal muscle and reduces hepatic glucose production [33]. In addition, IMD can attenuate inflammation in adipose tissue of obese rats, through receptor-mediated cAMP-dependent protein kinase (PKA) activation receptor [34].
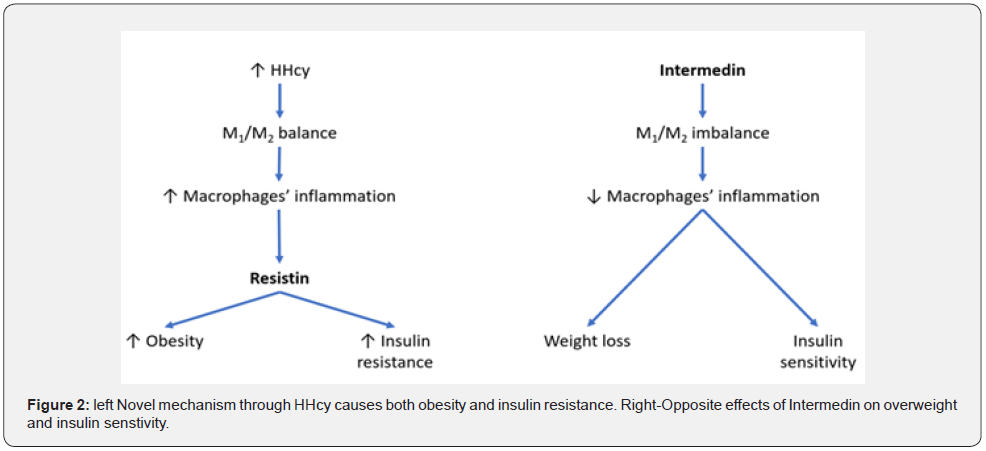
In summary, whereas HHcy favors the adipocytesinflammation for Resistin expression, in turn, induces insulin resistance and obesity. The M1/M2 ratio imbalance happening in macrophages through IMD, favours that [35]. For this novel mechanism, IMD administration could likely represent a new therapeutic strategy to antagonize insulin resistance, T2DM, obesity and other cardio-metabolic diseases [36]. Concerning the obesity, it was observed that, the IMD infusion (300 ng/Kg x hour) in obese rats attenuates inflammation in white adipose tissue (WAT). This effect was obtained through the mitogen-activated protein kinase (MAPK) and nuclear factor kB (NF-kB) activation by the receptor PKA pathway. The WAT activation of obese rats decreased body weight and results to be associated with anorexigenic effects [37] and inhibition of gastric emptying [38]. Consequentially, IMD could be interesting as a potential target for the treatment of insulin resistance and obesity in humans too, even if several other investigations both in animal models and in humans are requested [34].
Funding
The Author received no financial support for the research and/or publication of the article.
References
- Platt DE, Harin E, Salameh P, Merhi M, Sabbah N, et al. (2017) Type 2 diabetes mellitus and hyperhomocysteinemia: a complex interation. Diabetol Metab Syndr 9: 19.
- Buyschaert M, Dramais AS, Wallemacq PE, Hermans P (2000) Hyperhomocysteinemia in type 2 diabetes: Relationship to macroangipathy, nephropathy and insulin resistance. Diab Care 23(12): 1816-1822.
- Cacciapuoti F (2013) The possible link between homocysteine levels and insulin resistance: Effects of folic acid and vitamin supplementation in enhancing vascular damage. Commentaries I.A.S.
- Li Y, Zhang H, Jiang C, Xu M, Pang Y, et al. (2013) Hyperhomocysteinemia promotes insulin resistance by inducing endoplasmic reticulum stress in adipose tissue. J Biol Chem 288(14): 9583-9592.
- Li Y, Jiang C, Zu G, Wang N, Zhu Y, et al. (2008) Homocysteine upregulates resistin production from adypocites in vivo and in vitro. Diabetes 57(4): 817-827.
- Aquilante CL, Kasmiki L, Knutsen SD, Zine I (2008) Relationship between plasma resistin concentrations: inflammatory kemokynes and components of the metabolic syndrome in adults. Metabolism 57(4): 498-501.
- Hirosumi J, Tuncman G, Chang L, Gorgun CZ, Uysal KT, et al. (2002) A central role of JNK in obesity and insulin resistance. Nature 420(6913): 333-336.
- Riedijk MA, Stoll B, Chako S, Scherbeek H, Sunehag A, et al. (2007) Methionine transmethylation and transsulfuration in the piglet gastrointestinal tract. Proc Natl Sci USA 104(9): 3408-3413.
- Selhub J (1999) Homocysteine metabolism. Annu Rev Nutr 19: 217-246.
- Fodinger M, Budmayer H, Sunder-Plassmann G (1999) Molecular genetics of homocysteine metabolism. Miner Electrolyte Metabol 25: 268-278.
- Geisel J, Zimbelmann I, Schorr H, Knapp JP, Bodis M, et al. (2001) Genetic defects as important factors for moderate hyperhomoc: steinemia. Clin Chem Lab Med 39: 698-704.
- Zeng X, Dai J, Remick DG, Wang X (2003) Homocysteine-mediated expression and secretion of monocyte chemoactant protein-1 and interleukin-8 in human monocytes. Circ Res 93(4): 311-320.
- Meigs JH, Jacques PF, Selhub J, Singer DE, Nathan DM, et al. (2001) Fasting plasma homocysteine levels in the insulin resistance syndrome: the Framingham offspring study. Diabetes Care 24(8): 1403-1410.
- Parel I, Buckels AC, Kinghom IJ, Murdock PR, Holbrook JD, et al. (2003) Resistin is expressed in human macrophages and directly regulated by PPAR gamma activators. Biochem Biophys Res Commun 300(2): 472-476.
- Li Y, Jiang Xu G, Wang N, Zhu Y, Tang C, et al. (2008) Homocysteine upregulates Resistin production from adipocytes in vivo and in vitro. Diabetes 57(4): 817-827.
- Steppan CM, Lazar MA (2002) Resistin and obesity-associated insulin resistance. Trends Endocr Metabol 13(1): 18-23.
- Muse ED, Feldman DI, Blaha MJ, Dardari Z, Blumenthal RS, et al. (2015) The association of Resistin with cardiovascular disease in the multi-ethnic study of atherosclerosis: Atherosclerosis 239(1): 101-108.
- Ntaios G, Gatsellis NK, Dalekos GN (2013) Adipokynes as mediators of endothelial function and atherosclerosis: Atherosclerosis 227(2): 216-221.
- Filova M, Haluzik M, Gay S, Senolt L (2009) The role of resistin as a regulator of inflammation: implications for various human pathologies. Clin Immunol 133(2): 157-170.
- Kunathoor VV, Febbraio M, Podrez EA, Moore HJ, Andersson L, et al. (2002) Scavenger receptors class I/II and CD 36 are the principal receptors responsible for the uptake of modified low density lipoprotein leading the lipid loading in macrophages. J Biol Chem 277(2): 49982-49988.
- Kaser S, Kaser A, Sandhrofer A, Ebenbichler CF, Filg H, et al. (2003) Resistin messenger-RNA expression is increased by pro-inflammatory cytokines in vitro: Biochem Biophys Res Commun 309(2): 286-290.
- Bokarewa M, Nagaev I, Dalhberg L, Smith U, Tarkowshi A (2005) Resistin an adipokine with potent pro-inflammatory properties: J Immunol 174(9): 5789-5795.
- Baker JF, Morales M, Qatanani M, Cucchiara A, Nakos E, et al. (2011) Resistin levels in lupus and associations with diverse-specific measures, insulin resistance and coronary calcification. J Rheumatol 38(11): 2369-2375.
- Li HM, Zheng TP, Leng RX (2016) Emerging role of adipokines in systemic lupus erythematous. Immunol Res 64(4): 820-830.
- Hernandez ASM, Rodriguez MSM, Rodriguez ASM, Armentia SM, Pachon J (2018) Clinical significance of the Resistin in clinical practice. Clin Med Rep.
- Delamaga M (2014) Resistin as a biomarker linking obesity and inflammation to cancer: potential clinical perspectives. Biomark Med 8(1): 107-118.
- Deb A, Deshmukh B, Rameteke P, Bathi FK, Bhati MK (2021) Resist in a journey from metabolism to cancer. Transl Oncology 14(10): 101178.
- Li Y, Jiang C, Wang X, Zhang Y, Shibahara S, et al. (2007) Adrenomedullin is a novel adipokine: adrenomedullin in adipocytes and adipose tissues. Peptides 28(5): 1129-1143.
- Shimosawa T, Ogihara T, Matsui H, Asano T, Ando K, et al. (2003) Deficiency of adrenomedullin induces insulin resistance by increasing oxidative stress. Hypertension 41(5): 1080-1085.
- Li H, Bian Y, Zhang N, Guo J, Wang C, et al. (2013) Intermedin protects against myocardial ischemia-reperfusion injury in diabetic rats. Cardiovasc Diabetol 12: 91.
- Pang Y, Li Y, Ying L, Sun L, Zhang S, et al. (2016) Intermedin restores hyperhomocysteinemia-induced macrophage polarization and improves insulin resistance in mice. Journ Biol Chem 291(23): 12336-12345.
- Fujisaka S, Usui L, Bukhari A, Ikutani M, Oya T, et al. (2009) Regulatory mechanisms for adipose tissue M1 and M2 macrophages in diet-induced obese mice. Diabetes 58(11): 2574-2582.
- Steinberg GR, Kemp BE (2009) AMPK in health and disease. Physiol Rev 89(3): 1025-1078.
- Dai HB, Wang FZ, Kang Y, Sun J, Zhou H, et al. (2021) Adrenomedullin attenuates inflammation in white adipose tissue of obese rats through receptor-mediated pathway. Obesity 29(1): 86-97.
- Lu Y, Weng L (2022) Editorial: new therapeutic approaches against inflammation and immune regulator in metabolic related disease. Front Pharmacol 13: 878608.
- Zhang SY, Xu MJ, Wang X (2018) Adrenomedullin 2/Intermedin: a putative drug candidate for treatment of cardio-metabolic diseases. Br J Pharmacol 175(8): 1230-1240.
- Martinez V, Wang L, Tachè Y (2006) Peripheral adrenomedullin inhibits gastric emptying through CGRP8-37 sensitive receptors and prostaglandinds pathways in rats. Peptides 27(6): 1376-1382.
- Hirsch AB, McCuen RW, Arimura A, Schubert (2003) Adrenomedullin stimulates somatostatin and thus inhibits histamine and acid secretion in the fundus of the stomach. Regul Rept 110(3): 189-195.






























