The Effects of Dietary Fibers on Improvement of Glucose Metabolism
Yo Tsuchiya*
School of Nutrition, College of Nursing and Nutrition, Shukutoku University, Japan
Submission: March 17, 2022; Published: March 29, 2022
*Corresponding author: Yo Tsuchiya, School of Nutrition, College of Nursing and Nutrition, Shukutoku University, Japan
How to cite this article: Yo Tsuchiya. The Effects of Dietary Fibers on Improvement of Glucose Metabolism. Curre Res Diabetes & Obes J 2022; 15(4): 555917.DOI: 10.19080/CRDOJ.2022.15.555917
Abstract
Dietary fiber (indigestible polysaccharides) intake is widely recommended as a nutritional therapy to improve hyperglycemia and diabetes. Many researchers have reported the beneficial effects of dietary fibers such as cellulose, pectin, inulin, and guar as regulators of glucose metabolism. In addition, new types of indigestible polysaccharides have been shown to be novel dietary fibers that attenuate postprandial glycemia. This review summarizes the understanding and progress of traditional and recent studies on the effects of dietary fiber on glucose metabolism.
Keywords: Dietary fiber; Glycemia; Diabetes; Glucose absorption; Intestinal microbiota; Proteoglycan
Abbreviations: SCFA: Short-chain fatty acids; PG: Proteoglycans; IMD: Isomaltodextrin
Introduction
Various medical and nutritional therapies have been developed worldwide to improve glucose metabolism disorders, especially hyperglycemia and diabetes. As a nutritional therapy for these disorders, dietary fiber intake suppresses postprandial increases in plasma glucose levels. It has been reported that intake of vegetables containing abundant dietary fiber before intake of carbohydrates attenuated postprandial glycemia compared to single intake of carbohydrates in normal healthy subjects and patients with type 2 diabetes [1,2].
Since the 1970s, dietary fibers, such as cellulose, pectin, guar, and inulin, have been reported to improve glucose metabolism [3-5]. Cellulose is a structural component of the cell wall of plants and a homopolysaccharide consisting of a linear chain of several hundred to thousands of β1-4 glycosidic bound D-glucose. Pectin is a heteropolysaccharide present in apples and citrus fruits. Its main component was galacturonic acid. Guar, also known as guar gum, is a galactomannan polysaccharide found in guar beans. It has been reported that these three polysaccharides attenuate the increase in postprandial plasma glucose levels quickly by decreasing glucose absorption in the small intestine [4-7]. To understand these mechanisms, researchers have suggested that the presence of dietary fibers in the intestine increases the viscosity of the luminal fluid and the thickening of the rate-limiting unstirred layer. Thus, these processes attenuate glucose absorption from the luminal to the serosal side. Inulin is a heteropolysaccharide mainly found in Jerusalem artichokes and chicory. It is composed mainly of fructose units and fructans with terminal glucose. Inulin intake has been reported to reduce postprandial glycemia over a long period, such as 1-2 months [5].
Discussion
Recently, novel mechanisms in dietary fiber’s effects on improving glucose metabolism disorders have been clarified. The intestinal microbiota is considered as indicator for managing type 2 diabetes [8,9]. It has been reported that dietary fibers and short-chain fatty acids (SCFA) produced from dietary fibers by bacterial fermentation in the intestine affect changes in microbiota composition and relative populations of bacterial species [8-10]. These articles also reported that SCFA improves insulin sensitivity in patients with type 2 diabetes. In addition, Shao et al. [11] found that inulin attenuates hyperglycemia in diabetic mice by improving the intestinal microbiota [11]. These studies suggest that SCFA is a key regulator of the dietary fiber-mediated improvement of diabetes via the intestinal microbiota. In the future, we expect to investigate the detailed mechanisms, including the molecular level, between SCFT and the improvement of diabetes.
Several new types of dietary fibers have been reported to regulate glucose metabolism. One of them is isomaltodextrin (IMD). IMD is a highly branched α-glucan that is produced artificially and enzymatically from starch. In 2017, Sadakiyo et al. [12] found that IMD attenuated the postprandial increase of plasma glucose level and that it occurred via suppressing both the intestinal glucose transport from mucosal side to serosal side and the activities of carbohydrate-digestive enzymes [12].
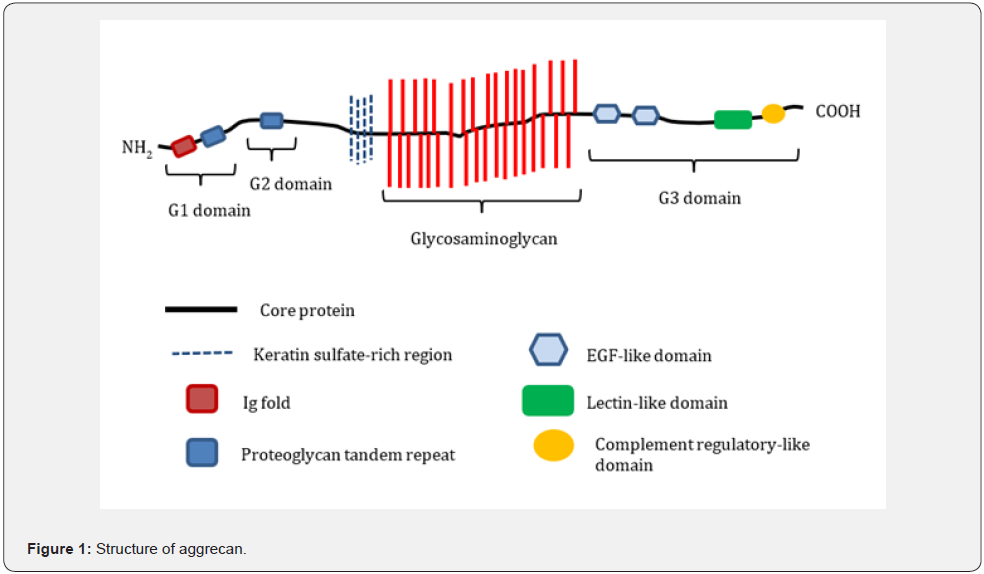
In 2015, Tsuchiya et al. [13] found that proteoglycans (PG) are new ingestible polysaccharide-like materials that regulate glucose metabolism [13]. PG is the major non-collagenous extracellular matrix component, particularly in cartilage and skin. Although it was not food originally, PG was purified abundantly from the nasal cartilage of salmon, not only as a therapeutic agent but also as a food product [13-15]. Tsuchiya et al. [13] found that oral administration of salmon PG decreased glucose absorption in the jejunum, thereby attenuating the increase in plasma glucose levels in rats. As shown in Figure 1, salmon PG belongs to the aggrecan family and is composed of a core protein section attached to glycosaminoglycan (GAG) chains which is the polysaccharide part [16]. These GAG chains primarily contain chondroitins A and C. The GAG chains of aggrecans, such as salmon PG, are negatively charged and highly hydrophilic [17]. It is possible that these chains conjugate glucose and subsequently prevent glucose absorption.
Conclusion
Although many beneficial dietary fibers and polysaccharides have improved hyperglycemia and diabetes, the detailed molecular mechanisms underlying their effects are not clear. Further studies are needed to understand this. In addition, which kind of food containing dietary fibers and polysaccharides is the most useful for improving glucose metabolism? The answer is not the only one. We may select available food depending on the dietary habits and conditions of food production in each country and/or their local areas.
References
- Sun L, Goh HJ, Govindharajulu P, Leow MK, Henry CJ (2020) Postprandial glucose, insulin and incretin responses differ by test meal macronutrient ingestion sequence (PATTERN study). Clin Nutr 39(3): 950-957.
- Imai S, Matsuda M, Hasegawa G, Fukui M, Obayashi H, et al. (2011) A simple meal plan of 'eating vegetables before carbohydrate' was more effective for achieving glycemic control than an exchange-based meal plan in Japanese patients with type 2 diabetes. Asia Pac J Clin Nutr 20(2): 161-168.
- Jenkins DJ, Wolever TM, Leeds AR, Gassull MA, Haisman P, et al. (1978) Dietary fibres, fibre analogues, and glucose tolerance: importance of viscosity. Br Med J 1(6124): 1392-1394.
- Johnson IT, Gee JM (1981) Effect of gel-forming gums on the intestinal unstirred layer and sugar transport in vitro. Gut 22(5): 398-403.
- Kaur N, Gupta AK (2002) Applications of inulin and oligofructose in health and nutrition. J Biosci 27(7): 703-714.
- Kay RM (1982) Dietary fiber. J Lipid Res 23(2):221-242.
- Flourie B, Vidon N, Florent CH, Bernier JJ (1984) Effect of pectin on jejunal glucose absorption and unstirred layer thickness in normal man. Gut 25(9): 936-941.
- Hills RD Jr, Pontefract BA, Mishcon HR, Black CA, Sutton SC, et al. (2019) Gut Microbiome: Profound Implications for Diet and Disease. Nutrients 11(7): 1613.
- Beam A, Clinger E, Hao L (2021) Effect of Diet and Dietary Components on the Composition of the Gut Microbiota. Nutrients 13(8): 2795.
- Birkeland E, Gharagozlian S, Birkeland KI, Valeur J, Måge I, et al. (2020) Prebiotic effect of inulin-type fructans on faecal microbiota and short-chain fatty acids in type 2 diabetes: a randomised controlled trial. Eur J Nutr 59(7): 3325-3338.
- Shao T, Yu Q, Zhu T, Liu A, Gao X, et al. (2020) Inulin from Jerusalem artichoke tubers alleviates hyperglycaemia in high-fat-diet-induced diabetes mice through the intestinal microflora improvement. Br J Nutr 123(3): 308-318.
- Sadakiyo T, Ishida Y, Inoue SI, Taniguchi Y, Sakurai T, et al. (2017) Attenuation of postprandial blood glucose in humans consuming isomaltodextrin: carbohydrate loading studies. Food Nutr Res 61(1): 1325306.
- Tsuchiya Y, Kawamata K, Tomita M, Tsuboi M, Takahashi T, et al. (2015) Effects of Salmon Nasal Cartilage Proteoglycan on Plasma Glucose Concentration and Active Glucose Transport in the Small Intestine. J Nutr Sci Vitaminol (Tokyo) 61(6): 502-505.
- Majima M, Takagaki K, Sudo S, Yoshihara S, Kudo Y, et al. (2001) Effect of proteoglycan on experimental colitis. Int Congr Ser 1223: 221-224.
- Sashinami H, Takagaki K, Nakane A (2006) Salmon cartilage proteoglycan modulates cytokine responses to Escherichia coli in mouse macrophages. Biochem Biophys Res Commun 351(4): 1005-1010.
- Aspberg A (2012) The different roles of aggrecan interaction domains. J Histochem Cytochem 60(12): 987-996.
- Prydz K, Dalen KT (2000) Synthesis and sorting of proteoglycans. J Cell Sci 113(2): 193-205.






























