Evaluation of Non-Alcoholic Fatty Liver Disease (NAFLD) in Obese Adult Volunteers According to Increasing Insulin Resistance and Loss of Glucose Control
Thomas Forst1,2*, Isabel Botz1, Matthias Berse3, Stephan Voswinkel4, Mares-Elaine Strempler3, Sybille Baumann3 and Armin Schultz1
1CRS Clinical Research Services Mannheim GmbH, Germany
2Johannes Gutenberg University Mainz, Germany
3CRS Clinical Research Services Berlin GmbH, Germany
4MLM Medical Labs, Mönchengladbach, Germany
Submission: January 28, 2022; Published: February 21, 2022
*Corresponding author: Thomas Forst, CRS Clinical Research Services Mannheim GmbH, Grenadierstrasse 1, 68167 Mannheim, Germany
How to cite this article: Thomas F, Isabel B, Matthias B, Stephan V, Mares-Elaine S, et al. Evaluation of Non-Alcoholic Fatty Liver Disease (NAFLD) in Obese Adult Volunteers According to Increasing Insulin Resistance and Loss of Glucose Control. Curre Res Diabetes & Obes J 2022; 15(3): 555914.DOI: 10.19080/CRDOJ.2022.15.555914
Abstract
Our study investigated non-alcoholic fatty liver disease in obese adult volunteers in accordance to glucose metabolism and insulin resistance. This observational study was conducted in obese adult volunteers (body mass index of at least 28 kg/m2), stratified in 3 groups: diagnosis of type 2 diabetes mellitus, normal glucose tolerance, and impaired glucose tolerance. Liver fat content and stiffness were measured using transient elastography. Indices for fatty liver disease and insulin resistance were calculated based on clinical and laboratory measures. A total 301 volunteers were recruited, 109 of which had a diagnosis of type 2 diabetes mellitus. Based on the HOMAIR score for insulin resistance, 21% participants were categorized as insulin sensitive and 79% as insulin resistant. Overall, 84% had a liver fat content above 5%. The proportion of participants with a liver fat content of more than 33%, indicating severe fatty liver disease, was significantly higher in the participants with insulin resistance (73.7%) than in insulin-sensitive participants (56.3%, p<0.01). Liver fat content correlated with the HOMAIR (r=0.3958; p<0.0001). In line with published data, our results confirm a prevalence of around 80% for non-alcoholic fatty liver disease in overweight adult subjects. An increase in liver fat and stiffness was found in line with deteriorating glucose control and increasing insulin resistance, even in the pre-diabetic stage.
Keywords: Obesity; Insulin resistance; Non-alcoholic fatty liver disease
Abbreviations: NAFLD: Non-Alcoholic Fatty Liver Disease; PDFF: Proton-Derived Fat Fraction; ANOVA: Analysis of Variance; NFS: NAFLD Fibrosis Score; kPA: Kilopascal; CAP: Controlled Attenuation Parameter; AST: Aspartate Aminotransferase; ALT: Alanine Aminotransferase; gamma-GT: Gamma Glutamyl Transpeptidase
Introduction
Obesity, non-alcoholic fatty liver disease (NAFLD), insulin resistance, and the metabolic syndrome share many causal pathways in their genesis and represent important risk factors in the development of cardiometabolic complications. NAFLD is characterized by excessive triglyceride accumulation in the liver tissue, exceeding 5% of the total liver mass. NAFLD is also considered as the hepatic manifestation of the metabolic syndrome, covering central obesity, insulin resistance, hypertension, dyslipidemia, and inflammation [1-3]. Notably, compared to overall visceral adipose tissue, liver fat is suggested to be even a stronger driver of cardiovascular comorbidities [4,5].
Moreover, NAFLD was found to be associated with endothelial dysfunction [6] and unstable coronary plaques [7]. Not all obese subjects develop NAFLD, but obesity is considered an important risk factor for NAFLD [8].
In the western world, NAFLD has become the most common cause of liver injury and is recognized as a predictor of hepatic and cardiovascular morbidity and mortality [9]. The prevalence of NAFLD consistently increases in parallel to the spread of obesity and type 2 diabetes mellitus [2]. It is estimated that in western societies, one in three adults and one in ten children is affected by NAFLD [10]. In specific risk populations, such as severely obese persons or patients with type 2 diabetes mellitus, the prevalence of NAFLD is as high as 90% and 70%, respectively [11,12]. In case of aggravating hepatic inflammation and fibrosis, 30%-40% of the cases with NAFLD progress to non-alcoholic steatosis hepatis (NASH), which further increases the risk of liver cirrhosis or the development of hepatic cellular carcinoma. The aim of our study is to investigate the incidence and progression of fatty liver disease in overweight and obese adult volunteers related to increasing insulin resistance and deteriorating glucose control. The data presented here are baseline data of an ongoing prospective study.
Methods
Patients and study design
The data presented here are baseline data from overweight and obese individuals of an ongoing epidemiological study performed at the CRS study sites in Mannheim and in Berlin, Germany. The study was performed in accordance with the declaration of Helsinki. The study was reviewed by the local ethical review board and was registered in the German registry of clinical studies (DKRS, registration number DRKS00017516).
All participants had given written informed consent prior to any procedures. Main inclusion criteria were age 18 to 80 years and BMI above 28 kg/m2. Main exclusion criteria involved diabetes mellitus type 1, maltose-malabsorption, acute gastrointestinal disease, gastrointestinal resection, and systemic treatment with corticoids. Study participants arrived at the study sites in the morning after being fasted overnight for at least ten hours. A fasting blood sample was taken from all study participants for the measurement of glucose, insulin, aspartate aminotransferase (AST), alanine aminotransferase (ALT), gamma glutamyl transpeptidase (gamma-GT), triglycerides, thrombocytes, and albumin. Insulin, aspartate aminotransferase (AST), alanine aminotransferase (ALT), gamma glutamyl transpeptidase (gamma-GT), triglycerides, and albumin were in serum and fasted glucose in sodium fluoride (NaF) plasma were analyzed with Cobas® 6000 analyzers (Roche, Germany). Thrombocytes were analyzed in EDTA blood using a Sysmex XN-1000 blood counter (Sysmex Deutschland GmbH, Germany).
Liver fat and stiffness was measured in all participants using transient elastography (FibroScan®, Echosens, Paris, France). The device transmits a mechanical vibration to the tissue and induces elastical shear-wave propagation which is tracked by pulse echo ultrasound signals at a measuring depth of 2.5 to 6.5 cm. The FibroScan® device evaluates the liver fat content given by the controlled attenuation parameter (CAP) and expressed as decibel per meter (dB/m) and liver stiffness given as the as the Young’s modulus (E) and expressed as kilopascal (kPA) [13,14]. At least ten reliable and valid measurements were taken with either the M or XL probe by specially trained staff members.
Clinical and biomarker scores
Several clinical/laboratory scores were calculated as predictive markers for the evaluation of liver function and integrity. The fatty liver index (FLI) is predominantly reflects liver steatosis, the AST/ ALT ratio, the BARD score, and the NAFLD fibrosis score are more indicative for liver inflammation and fibrosis [2].
The FLI is calculated based on the laboratory markers triglycerides and gamma-GT and the body composition markers BMI and waist circumference [15] according to the formula
FLI = ey divided by (1 + ey) × 100,
where e is the Euler’s number 2.71828 and y is 0.953 × ln (triglycerides, mg/dL) + 0.139 × BMI, kg/m2 + 0.718 × ln (GGT, U/L) + 0.053 × waist circumference, cm – 15.745) [2].
An FLI <30 is considered as a low, 30 to 59 as an intermediate, and ≥ 60 as a high indicator for steatosis hepatis. The simplest score to predict liver fibrosis is the AST to ALT ratio (AST/ALT), with a value ≥ 0.8 considered indicative of liver fibrosis.
The NAFLD fibrosis score (NFS) is a composite score for the prediction of liver fibrosis including the variables age, hyperglycemia/diabetes, BMI, platelet count (thrombocytes), the AST/ALT ratio (AAR), and albumin [16,17]. It is calculated according to the formula
NFS = -1.675 + (0.037 × Age [years]) + (0.094 × BMI [kg/m2]) + [1.13× IFG/diabetes [yes= 1, no=0]) + (0.99× AST/ALT ratio) – (0.013 × thrombocytes [x109/L]) – (0.66 × albumin [g/dL]).
A NAFLD score below -1.455 is considered to represent no or mild fibrosis, a score between -1.455 and 0675 moderate, and a score above 0.675, severe fibrosis. A further score for the prediction of liver fibrosis in patients with NAFLD is the BARD score [18]. It is a composite score combining the risk factors BMI, ratio of liver transaminases (AAR), and diabetes. A BARD score of 0 – 1 indicates a low risk of advanced fibrosis, scores of 2 – 4 indicate a high risk.
For the assessment of insulin resistance, the HOMAIR was calculated from the fasting glucose and insulin concentration using following the formula
HOMAIR = fasting insulin [μU/mL] × fasting glucose [mg/dL] divided by 405 (19).
Participants without a diagnosis of type 2 diabetes mellitus underwent a standardized oral glucose tolerance test. After a 10-hour fasting period, they drank 300 mL glucose solution containing 75 g glucose (Accu-Chek® Dextrose O.G-T.) within 5 minutes. Blood samples were collected before and 60 and 120 min after intake of the solution. Participants were stratified according to their results in normal glucose tolerance (NGT, 2-h glucose below 140 mg/dL/7.8 mmol/L) or impaired glucose tolerance (IGT, glucose ranging from 140 mg/dL (7.8 mmol/L) to 199 mg/ dL (11.0 mmol/L). A value of 200 mg/dL or higher would have diagnosed diabetes mellitus.
Statistical analysis
For statistical comparisons between study groups and other variables of interest (e.g., HOMAIR and Fibrosis categories), the type of analysis was chosen based on the type of data (numerical / categorical) and number of groups to be compared. If more than two groups were compared regarding a numerical variable, 95% confidence intervals of the numerical variables by group were added to the summary statistics. An overall influence of the grouping variable was assessed by the p-value of the F-test of a simple analysis of variance (ANOVA) model with the grouping variable as the single independent variable. Pairwise differences were assessed by calculating the least square mean difference including 95% confidence interval. Statistical significance of differences was assessed by using the p-value of the corresponding t-test.
If two groups were compared regarding a numerical variable, 95% confidence intervals of the numerical variables by group were added to the summary statistics. Groups were compared by calculating arithmetic mean difference, parametric 95%-confidence interval, and the p-value of the corresponding t-test. If two or more groups were compared regarding a binary variable, groups were compared overall and pairwise by using a Chi-square test.
Furthermore, two linear regressions were performed with liver stiffness and liver elasticity as dependent variables and HOMAIR, AST/ALT ratio, FLI and NFS as independent variables. Pearson correlation coefficients were calculated for all variables with a p-value < 0.05 in the regression analyses. For group comparisons, raw p-values are given. A p-value < 0.05 was considered as statistically significant. P-values were not adjusted for multiplicity.
Results
Demographic data and baseline characteristics
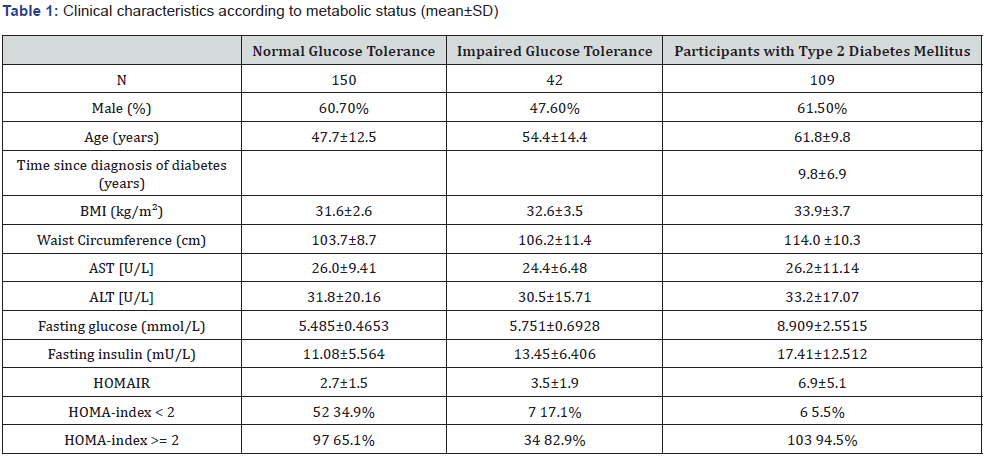
In total, 301 men and women participated in this crosssectional study. Altogether 109 had a prior diagnosis of T2DM, average time since diagnosis was 9.8 years. The 192 non-diabetic participants were stratified as NGT (150) and IGT (42) according to the result of an oral glucose tolerance test. Between the groups, there were significant differences in age. Participants with T2DM were older than participants in the NGT (14 years, p < 0.0001) and IGT (7 years, p=0.008) groups. Participants in the NGT group were in average 7 years younger than participants in the IGT (p=0.0013). Participants with T2DM also had higher BMI and larger waist circumference compared to NGT and IGT. The clinical characteristics of the study participants according to their metabolic categorization are given in Table 1.
Glucose metabolism characteristics
Of the total study population, 21% participants were categorized as insulin sensitive (HOMAIR < 2) and 79% as insulin resistant (HOMAIR ≥ 2.0). In T2DM, the proportion of participants with HOMAIR equal or above 2 was highest (94.5%), compared to NGT (65. %) and IGT (82.9%).
Analysis based on glucose tolerance
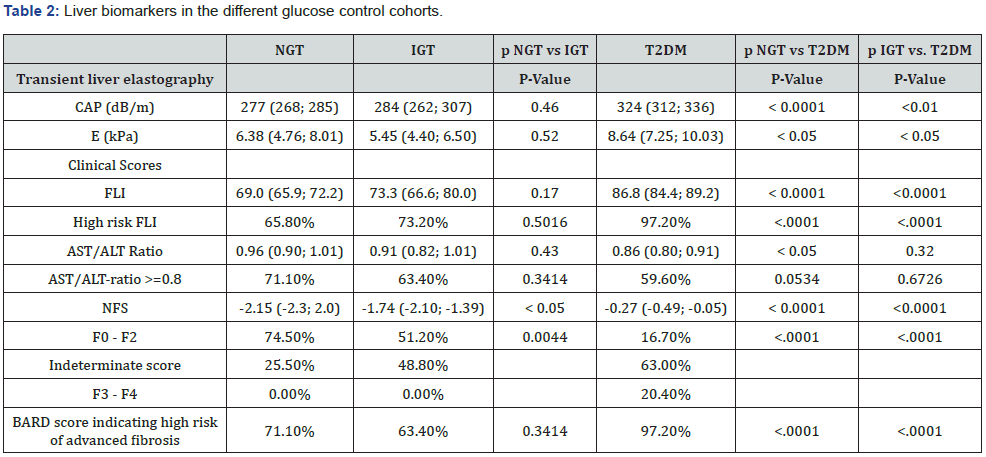
Transient liver elastography parameter CAP and of the composite index FLI both show marked increase of fatty liver in participants with T2DM. Mean values for CAP increased with decreasing glucose tolerance, T2DM presented with significantly higher values than the two non-diabetic groups. Consistent with the increase in CAP, the average FLI score was also significantly higher (p<0.0001) in the T2DM group. Participants with T2DM had significantly highest risk for fatty liver (p<0.0001), with 97.2% having FLI indices high risk of fatty liver. The risk was not significantly different between NGT and IGT (Table 2).
Data presented as: mean (95% confidence intervals) / percentage of participants
NGT: Normal Glucose Tolerance; IGT: Impaired Glucose Tolerance; T2DM: Type 2 Diabetes mellitus; FLI: Fatty Liver Index; NFS: Non-Alcoholic Fatty Liver Disease Fibrosis Score
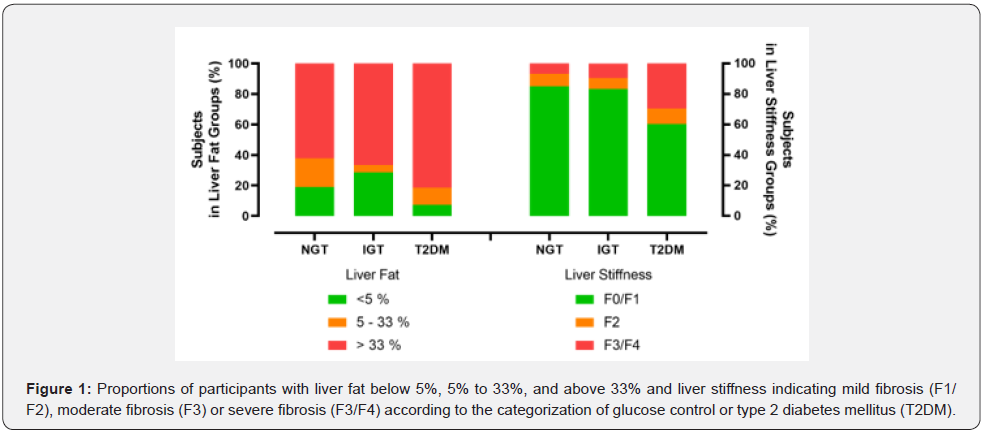
A larger proportion of participants with a diagnosis of T2DM had liver fat content greater than 33% or liver stiffness indicating fibrosis (F3/F4; Figure 1). The percentage of participants with a liver fat content of more than 33% according to the transient liver elastography [20] was significantly higher in the T2DM group (82%) compared to the NGT group (62%, p<0.0001) and in the IGT group (67%, p<0.0001). Transient liver elastography parameter E showed highest liver stiffness in the T2DM group and was significantly (p < 0.05) higher than in the NGT or IGT group (Table 2). The percentage of participants with a severe increase in liver stiffness categorized as F3 or F4 based on elastography measurement [21] was significantly higher in the T2DM group (29.6%) compared to the NGT group (6.8%, p<0.0001) and the IGT group (9.5% in IGT, p<0.0001) (Figure 1). Applying the BARD score, the proportions of participants at high risk for advanced liver fibrosis was highest in the T2DM group (97%) compared to the NGT (71%, p<0.0001) and to the IGT (63%, p<0.0001).
The AST/ALT ratio was significantly higher in T2DM group compared to NGT and IGT (p <0.0001), whereas the proportion of participants with an AST/ALT ratio equal or greater than 0.8 indicating high risk of fibrosis was not significantly different between the groups. NFS scores showed significantly higher mean values in the T2DM group compared to both NGT and IGT (p <0.0001), and in the IGT group compared to the NGT group (p <0.05). The NFS score indicated severe fibrosis (F3) or cirrhosis (F4) in 20. % of the participants in the T2DM group.
Analysis based on insulin resistance
Results of transient elastography were also analyzed applying the categories of insulin resistance (HOMAIR). In participants with insulin resistance the proportion with liver fat content greater than 33% or increased liver stiffness was visibly larger than in participants sensitive to insulin (Figure 2). The CAPvalue as a marker for liver fat content was significantly higher in insulin-resistant individuals with 304.0 dB/m (95% CI: 295.8; 312.2) compared to 260.5 dB/m (95% CI: 247.4; 273.5) in insulinsensitive individuals (p<0.0001). The proportion of participants with a liver fat content of more than 33% was significantly larger in insulin-resistant participants (73.7%) than in insulin-sensitive participants (56.3%, p<0.01).
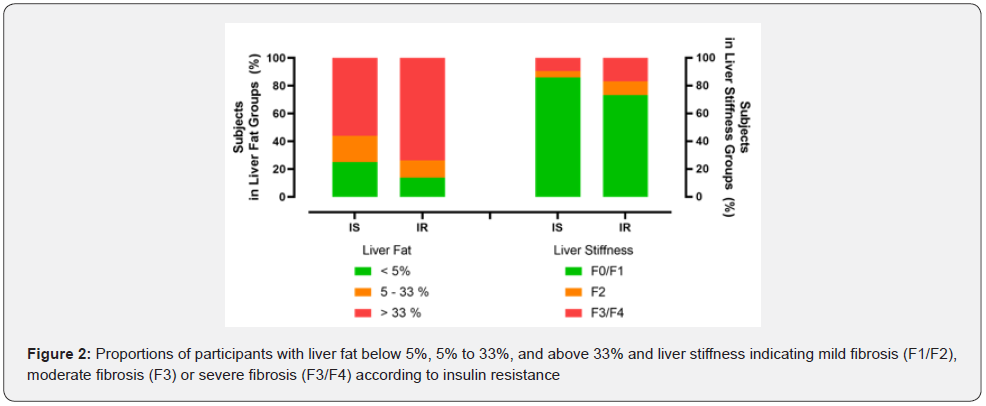
In insulin-resistant participants, liver stiffness was significantly higher (7.60 kPa [95% CI: 6.39; 8.81]) compared to 5.13 kPa (95% CI: 4.28; 5.98) in insulin-sensitive participants (p=0.04). Also, pronounced liver stiffness (F3 or F4) was significantly more frequent in insulin-resistant (16.8%) than in insulin-sensitive participants (9.4%; p<0.05). Multivariate regression analysis revealed correlation between liver fat (CAP) with HOMA (r=0.3958; p<0.0001) and the FLI (r=0.4133; P<0.0001).
Discussion
In this cross-sectional analysis from 301 overweight or obese participants transient elastography identified 83.9% of the participants with a fat content above 5% as indicative for NAFLD. In line with published data, our results confirm a prevalence of around 80% for NAFLD in participants with a BMI > 28 kg/m2 [22-24].
Prevalence of NAFLD was highest in obese participants with manifest T2DM. Categorizing the participants included in the study according to their glucose control, the incidence of NAFLD increased from 81.1% of participants with NGT or 71.4% in participants with IGT to 92.6% of participants with a diagnosis of T2DM. Severe steatosis hepatis (defined as a fat content above 33%) was found in 62.2% of participants with NGT, 66.7% of participants with IGT and in 69.8 % in T2DM.
Liver stiffness significantly increased in T2DM compared with the NGT and the IGT group. No difference in mean liver stiffness could be observed between the NGT and the IGT group. The proportion of patients with elevated liver stiffness increased from 14.9% in NGT, to 16.6% in IGT, and to 24.1% in T2DM. Because common hepatic laboratory markers are weak for detection of liver steatosis, a couple of scores combining clinical and laboratory measures have been developed and validated for NAFLD screening in larger patient populations. The FLI is a score developed the prediction of liver steatosis comprising triglycerides, BMI, and the waist circumference [25]. In our patients with T2DM, the FLI was significantly higher compared to the NGT and IGT group. Linear regression analysis revealed an association between the FLI and liver fat content as expressed by liver elastography. In contrast to the FLI used as an indicator for steatosis, the AST/ALT ratio, the NFS, and the BARD score are predictive for liver fibrosis [18,22]. In this cross-sectional study, the AST/ ALT ratio was significant lower in T2DM compared to NGT, but not compared to the IGT group. The NFS and the BARD score as indices for liver fibrosis significantly worsened from the NGT over the IGT and the T2DM group.
Insulin resistance appears to play an essential role in the development and progression of NAFLD, and vice versa [3,23]. In obese NAFLD individuals without known T2DM, an overall downregulation of insulin signaling genes have been shown compared to lean and obese controls without liver steatosis [24]. In liver biopsies from individuals with NAFLD, gene expression analysis revealed a shift in the ratio of insulin receptor subtype A and B with important implications on intracellular insulin signaling [26,27]. Despite most overweight participants included in our study were categorized as insulin resistant according to the HOMAIR score, 22% of the participants were overweight or obese with sustained insulin sensitivity. Liver steatosis and liver stiffness was significantly higher in insulin resistant compared to insulin sensitive individuals and the HOMAIR score showed a close correlation to the liver fat fraction as measured with transient elastography.
There are some important limitations which need to be considered. Liver fat content and liver stiffness were measured with transient elastography and not with MRI or liver biopsies. In the recent years, transient liver elastography has become a widely used and established method for the assessment of NAFLD and NASH [14,20,28]. The comparability with the histological assessment of liver biopsies or the quantification of liver fat and fibrosis using magnetic resonance tomographic based methods like proton-derived fat fraction (PDFF) is still matter of dispute. The participants with T2DM and the participants with IGT were older compared with the participants in the NGT group. Even we cannot rule out an effect of aging on liver fat or liver stiffness in our study population, previous studies did not show a linear relationship between NAFLD and age. Some previous studies even reported a decrease in the prevalence of NAFLD with increasing age [29,30].
The data included in this manuscript represent markers from a cross-sectional approach that do not allow conclusions on the course of the different liver indices over time. The participants included in this observational survey will be followed on an annual basis, which might allow us to get a better understanding in the progress of these biomarkers over time.
Conclusion
In conclusion, our data have shown that more than three out of four people with a BMI above 28 kg/m2 have an increased liver fat content as defined of more than 5% of liver tissue and 1 participant out of 4 had increased liver stiffness as an indicator for liver fibrosis. Liver fat content and liver stiffness in overweight individuals increase over a wide continuum of insulin resistance and declining glucose control.
Acknowledgments
The authors thank Dr. Manfred Wargenau of M.A.R.C.O GmbH & Co. KG, Düsseldorf for data management and statistics, and Nadja Faißt of CRS Mannheim for medical writing assistance.
Author Contributions
Thomas Forst was involved in creating study design, performance of the study, and the preparation of the manuscript.
Sybille Baumann, Matthias Berse, Isabel Botz, Armin Schultz, and Mares-Elaine Strempler were involved in conducting the study and the manuscript preparation.
Stephan Voswinkel was involved in the analysis of the laboratory samples and the preparation of the manuscript.
References
- Manne V, Handa P, Kowdley KV (2018) Pathophysiology of Nonalcoholic Fatty Liver Disease/Nonalcoholic Steatohepatitis. Clinics in liver disease 22(1): 23-37.
- Gerges SH, Wahdan SA, Elsherbiny DA, El-Demerdash E (2021) Non-alcoholic fatty liver disease: An overview of risk factors, pathophysiological mechanisms, diagnostic procedures, and therapeutic interventions. Life Sci 271: 119220.
- London A, Lundsgaard AM, Kiens B, Bojsen-Møller KN (2021) The Role of Hepatic Fat Accumulation in Glucose and Insulin Homeostasis-Dysregulation by the Liver. J Clin Med 10(3): 390.
- Fabbrini E, Magkos F, Mohammed BS, Pietka T, Abumrad NA, et al. (2009) Intrahepatic fat, not visceral fat, is linked with metabolic complications of obesity. Proceedings of the National Academy of Sciences of the United States of America 106(36): 15430-15435.
- Cioffi CE, Narayan KMV, Liu K, Uppal K, Jones DP, et al. (2020) Hepatic fat is a stronger correlate of key clinical and molecular abnormalities than visceral and abdominal subcutaneous fat in youth. BMJ open diabetes research & care 8(1): e001126.
- Narayan J, Das HS, Nath P, Singh A, Mishra D, et al. (2020) Endothelial Dysfunction, a Marker of Atherosclerosis, Is Independent of Metabolic Syndrome in NAFLD Patients. International journal of hepatology 2020: 1825142.
- Akabame S, Hamaguchi M, Tomiyasu K, Tanaka M, Kobayashi-Takenaka Y, et al. (2008) Evaluation of vulnerable coronary plaques and non-alcoholic fatty liver disease (NAFLD) by 64-detector multislice computed tomography (MSCT). Circulation journal 72(4): 618-625.
- Lonardo A, Mantovani A, Lugari S, Targher G (2020) Epidemiology and pathophysiology of the association between NAFLD and metabolically healthy or metabolically unhealthy obesity. Annals of hepatology 19: 359-366.
- Chiriac S, Stanciu C, Girleanu I, Cojocariu C, Sfarti C, et al. (2021) Nonalcoholic Fatty Liver Disease and Cardiovascular Diseases: The Heart of the Matter. Canadian journal of gastroenterology & hepatology 2021: 6696857.
- Alkhouri N, Feldstein AE (2016) Noninvasive diagnosis of nonalcoholic fatty liver disease: Are we there yet? Metabolism 65: 1087-1095.
- Boza C, Riquelme A, Ibanez L, Duarte I, Norero E, et al. (2005) Predictors of nonalcoholic steatohepatitis (NASH) in obese patients undergoing gastric bypass. Obesity surgery 15(8): 1148-1153.
- Vernon G, Baranova A, Younossi ZM (2011) Systematic review: the epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment pharmacol ther 34(3): 274-285.
- Mueller S, Sandrin L (2010) Liver stiffness: a novel parameter for the diagnosis of liver disease. Hepatic medicine: evidence and research 2: 49-67.
- Zhang X, Wong GL, Wong VW (2020) Application of transient elastography in nonalcoholic fatty liver disease. Clinical and molecular hepatology 26(2): 128-141.
- Bedogni G, Bellentani S, Miglioli L, Masutti F, Passalacqua M, et al. (2006) The Fatty Liver Index: a simple and accurate predictor of hepatic steatosis in the general population. BMC gastroenterology 6: 33.
- Angulo P, Hui JM, Marchesini G, Bugianesi E, George J, et al. (2007) The NAFLD fibrosis score: a noninvasive system that identifies liver fibrosis in patients with NAFLD. Hepatology 45(4): 846-854.
- Treeprasertsuk S, Björnsson E, Enders F, Suwanwalaikorn S, Lindor KD (2013) NAFLD fibrosis score: a prognostic predictor for mortality and liver complications among NAFLD patients. World J Gastroenterol 19(8): 1219-1229.
- Harrison SA, Oliver D, Arnold HL, Gogia S, Neuschwander-Tetri BA (2008) Development and validation of a simple NAFLD clinical scoring system for identifying patients without advanced disease. Gut 57(10): 1441-1447.
- Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, et al. (1985) Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28: 412-419.
- Karlas T, Petroff D, Garnov N, Böhm S, Tenckhoff H, et al. (2014) Non-invasive assessment of hepatic steatosis in patients with NAFLD using controlled attenuation parameter and 1H-MR spectroscopy. PloS one 9: e91987.
- Wong VW, Vergniol J, Wong GL, Foucher J, Chan HL, et al. (2010) Diagnosis of fibrosis and cirrhosis using liver stiffness measurement in nonalcoholic fatty liver disease. Hepatology 51(2): 454-462.
- McPherson S, Stewart SF, Henderson E, Burt AD, Day CP (2010) Simple non-invasive fibrosis scoring systems can reliably exclude advanced fibrosis in patients with non-alcoholic fatty liver disease. Gut 59(9): 1265-1269.
- Khan RS, Bril F, Cusi K, Newsome PN (2019) Modulation of Insulin Resistance in Nonalcoholic Fatty Liver Disease. Hepatology 70(2): 711-724.
- Suppli MP, Rigbolt KTG, Veidal SS, Heebøll S, Eriksen PL, et al. (2019) Hepatic transcriptome signatures in patients with varying degrees of nonalcoholic fatty liver disease compared with healthy normal-weight individuals. Am J Physiol Gastrointest Liver Physiol 316(4): G462-g472.
- Koehler EM, Schouten JN, Hansen BE, Hofman A, Stricker BH, et al. (2013) External validation of the fatty liver index for identifying nonalcoholic fatty liver disease in a population-based study. Clinical gastroenterology and hepatology: the official clinical practice journal of the American Gastroenterological Association 11(9): 1201-1204.
- Krause C, Geißler C, Tackenberg H, El Gammal AT, Wolter S, et al. (2020) Multi-layered epigenetic regulation of IRS2 expression in the liver of obese individuals with type 2 diabetes. Diabetologia 63(0): 2182-2193.
- Honma M, Sawada S, Ueno Y, Murakami K, Yamada T, et al. (2018) Selective insulin resistance with differential expressions of IRS-1 and IRS-2 in human NAFLD livers. International journal of obesity 42(9): 1544-1555.
- Wong VW, Adams LA, de Ledinghen V, Wong GL, Sookoian S (2018) Noninvasive biomarkers in NAFLD and NASH - current progress and future promise. Nature reviews Gastroenterology & hepatology 15(8): 461-478.
- Golabi P, Paik J, Reddy R, Bugianesi E, Trimble G, et al. (2019) Prevalence and long-term outcomes of non-alcoholic fatty liver disease among elderly individuals from the United States. BMC gastroenterology 19(1): 56.
- Younossi ZM, Koenig AB, Abdelatif D, Fazel Y, Henry L, et al. (2016) Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 64(1): 73-84.






























