Efficacy and Safety of Teneligliptin as Add on Therapy in Indian Type 2 Diabetes Mellitus T2DM Patients Having Dyslipidemia
Parmar Vinendra M* and Goswami Sunita S
Department of pharmacology, Gujarat Technological University, India
Submission: April 10, 2020; Published: August 12, 2020
*Corresponding author: Parmar Vinendra M, Ph. D scholar, Department of pharmacology, L. M. College of pharmacy, Gujarat Technological University, Ahmedabad, Gujarat, India
How to cite this article: Parmar V M, Goswami S S. Efficacy and Safety of Teneligliptin as Add on Therapy in Indian Type 2 Diabetes Mellitus T2DM Patients Having Dyslipidemia. Curre Res Diabetes & Obes J 2020; 13(4): 555869. DOI: 10.19080/CRDOJ.2020.13.555869
Abstract
Objectives:The purpose of this study was to investigate the efficacy and safety of teneligliptin, a completely unique and highly selective DPP-4 inhibitor in type 2 diabetes mellitus (T2DM) patients having dyslipidemia who are inadequately controlled by relevant conventional therapy in India.
Methods: Study protocol was approved by Institutional Ethics Committee. Diabetic patients having dyslipidemia (male/female) were randomized to receive treatments in two groups, namely conventional therapy [treatment (A)] and add on teneligliptin 20 mg with conventional therapy [treatment (B)] for 24 weeks. Predesigned case report form (CRF) was used to collect information from the prescribing physicians regarding the efficacy and safety of teneligliptin. Efficacy variables included change in serum glycaemic, lipid, and cytokines (IL-6, TNF- α and adiponectin) levels from baseline to week 24. Treatment-emergent adverse events (TEAEs) were also assessed.
Results: A complete of 120 T2DM patient having dyslipidemia were analysed using graph pad prism. Teneligliptin, as add on therapy to conventional therapy significantly reduced serum lipid profile (TC, TG, and LDL) as well as glycaemic parameters (HbA1c, FBG, and PPBG) along with significant rise in serum adiponectin levels as compared to conventional therapy
Conclusion: Add- on therapy with teneligliptin was found superior over convetional therapy in term of significantly reduced glycemic as well as lipid profile. Further, it was found safe and well tolerated in T2DM patients having dyslipidemia.
Keywords: Teneligliptin; DDP-4 inhibitor; Metformin; Total Cholesterol; Glycated haemoglobin; Adiponectin; Type 2 diabetes mellitus; Dyslipidemia
Abbreviations: NCD: Non-Communicable Disease; IDF: International Diabetes Federation; T2DM: Type 2 Diabetes Mellitus; DPP-4: Dipeptidyl Peptidase-4; GLP-1: Glucagon-like Peptide-1; HbA1c: Glycated Haemoglobin; IEC: Institutional Ethics Committee; DCGI: Drugs Controller General of India; BMI: Body Mass Index; FBG: Fasting Plasma Glucose; PPBG: Post Prandial Blood Glucose; AE: Adverse Event; SAP: Sample Analysis Plan; SD: Standard Deviation
Introduction
Diabetes mellitus (DM) is found to be related to alterations in lipid metabolism in term of high total cholesterol (TC), triglycerides (TG), low density lipoprotein (LDL) and low high density lipoprotein (HDL), that ends up in dyslipidemia and worsens the prognosis of diabetic patients having atherosclerosis and Cardiovascular disorder(CVD) [1]. The International Diabetes Federation (IDF) estimates the whole number of diabetic subjects is to rise to 69.9 million by the year 2025 [2]. The population of diabetes is rapidly growing because of the expansion of population, urbanisation, ageing, and increasing prevalence of obesity and morbidity [3].
Dyslipidemia, a long-time established risk factor for CVD has effect on 50% of diabetes patients as compared to non-diabetic population [4]. In DM patient’s insulin deficiency or resistance activates intracellular hormone-sensitive lipase which increases the discharge of non-esterified fatty acids (NEFA) from triglycerides that’s stored in centrally distributed adipose tissue. High circulating levels of NEFA also increase hepatic triglyceride production [5,6]. Lower levels of adiponectin are considered as an independent risk factor for developing Type 2 DM, dyslipidemia, and cardiovascular diseases [7].
A recent statement from one amongst the meta analysis report for the standards of medical care in Diabetes by the American Diabetes Association (ADA) has recommended that initially treatment with metformin as monotherapy after inadequate life style modification, followed by sulfonylurea, thiazolidinedione, Dipeptidyl peptidase-4 (DPP-4) inhibitors, sodium-glucose cotransporter 2 inhibitors (SGLT2-i), glucagon like peptide 1 (GLP 1) receptor agonist, or insulin alone or in combination [8]. However, it’s still difficult to search out an antidiabetic agent with long-term glucose control, minimal hypoglycaemia, no weight gain with affordable price. The optimum treatment with antidiabetic drugs to get fair glycaemic control should go hand-in-hand with lipidlowering drugs [9,4]. In diabetic patients having dyslipidemia, in keeping with current guideline, statins are preferred as they are well tolerated, efficacious and don’t adversely affect glycaemic control [10].
DPP-4 inhibitors are considered as a cornerstone within the management of T2DM due to their robust efficacy and favourable tolerability profile [11]. Our previous study has demonstrated teneligliptin as add on therapy showing better glycemic control as compared to conventional therapy in Indian patients having T2DM [12]. As there is no future study conducted on add on therapy of teneligliptin in diabetic dyslipidemic patients, our study was designed to evaluate efficacy, safety, and tolerability treatment which can led to improvement in the effectiveness of standard therapy in diabetic patients having dyslipidemia in India. Currently only few reports are available on the role of inflammatory biomarkers in type 2 diabetes and folks with impaired lipid metabolism in Indian population. During this manuscript, we have also tried to incorporate the role of inflammatory regulators (IL 6, TNF ά and adiponectin) with respect to add on teneligliptin therapy in type 2 diabetic patients having dyslipidemia in Indian population.
Materials and Methods
Ethics approval
The protocol of clinical study, informed consent form and relevant essential documents were approved by Institutional Ethics Committee (IEC); Safety, Health and welfare Ethics committee, registered under Drug Controller General of India (DCGI). The study was conducted according to the Ethical principles of Declaration of Helsinki; Good Clinical Practices guidelines issued by the Central Drugs Standard Control Organization (CDSCO), Indian Councils of Medical Research (ICMR).
Study design and procedure
A prospective, randomized, open label, study to assess the efficacy and safety of teneligliptin as an add-on therapy to conventional treatment in T2DM patients having dyslipidemia. This study was conducted at Jivraj Mehta Hospital and Bakeri Medical Research Centre, Ahmedabad.
Eligibility criteria
Study protocol was clearly defined for the patients and informed consent was obtained from all patients before to participation. The study included male and female patients with T2DM having dyslipidemia, aged >18 years, HbA1c levels of >7.0%, and body mass index (BMI) of 20.0–35.0 kg/m2 (both inclusive). Patients were excluded if they had serious disease such as kidney, liver, and cerebral stroke, history of severe heart disease or arrhythmias, taking DPP-4 inhibitor other than teneligliptin, taking statin other than atorvastatin and on insulin therapy, pregnant, and history of alcohol and tobacco use.
Intervention
Eligible patients were randomized in 1:1 ratio to receive either metformin/glimepiride and atorvastatin (treatment A) or metformin/glimepiride, atorvastatin, and add on teneligliptin (Treatment B). Treatment for both the groups remained stable and it included: teneligliptin 20 mg/day, metformin 500 mg/day, glimepiride 2 mg/day and atorvastatin 20 mg/day for 24 weeks. Patient’s demographics data, physical and clinical examination, laboratory assessments were documented in predesigned case report form (CRF).
Biochemical parameters
Lipid profile including serum Total cholesterol (TC), Triglyceride (TG), Low density lipoprotein (LDL), High density lipoprotein (HDL) and Glycemic parameters including serum glycated haemoglobin (HbA1c), fasting blood glucose (FBG) and post prandial blood glucose (PPBG) levels and Inflammatory cytokine levels IL6, TNF-α, and adiponectine levels were measured at baseline and at the end of 24 weeks in both the treatment groups.
Methodology
Serum cholesterol was estimated by cholesterol oxidase method from ROCHE on COBAS INTEGRA 400 WITH Exteranal quality control (EQAS) from BIO-RAD laboratories (USA) internal quality control from ROCHE diagnostic. Serum LDL and HDL were tested by direct non-immunological method on COBAS INTEGRA 400PLUS. Serum Triglyceride was tested by lipase glycerol method. Values of serum lipids were entered into the computer and computer analysis of the data was obtained
Cytokines estimation
From each subject, serum were collected and stored at -80ºC until further analysis. Serum IL-6 and TNF-α levels were measured using the enzyme-linked immunosorbent assay (ELISA) according to the manufacturer’s instructions (KRISHGEN Biosytems, Mumbai). The assay sensitivity ranges of KRISHGEN Biosytems kits were 3.12-200 pg/ml for IL-6 and 6.8-500 pg/ml for TNF-α in serum samples. The ELISA kits were validated with inter- and intra-assay precision. For adiponectin level was measured using the enzyme-linked immunosorbent assay (ELISA) according to the manufacturer’s instructions (KINESIS Dx). The assay sensitivity ranges of KINESIS Dx kit was 2-34 μg /ml adiponectine in serum samples. The ELISA kit was validated with inter- and intraassay precision. The (IL-6, TNF- α, and adiponectin) cytokines absorbance measured at 450nm.
Efficacy and safety end points
The primary efficacy end point was the change in glycemic and lipid parameter from baseline to 24 weeks. Secondary efficacy endpoints include change in inflammatory (IL-6, TNF- α, and adiponectin) cytokine levels from baseline to 24 weeks. During the clinical study period, we monitored possible adverse events (AEs), laboratory values, vital sign and physical examination results. Safety aspects were measured by recording AEs including symptomatic assessment by Naranjo causality scale for adverse events [13]. The incidence of AE in terms of number per patient was calculated based on the number of events, the number of patients and the total observation period.
Sample size and statistical analysis
The primary end point, difference in mean HbA1c from baseline to 24 week was assumed 0.5% and also the standard deviation (SD) of 0.9% for each treatment group. Based on a power of 80% and a type I error rate of alpha= 0.05 (2-tailed), a sample size of at least 60 patients per group was required to detect a clinically significant difference between both the groups [14]. Categorical data was presented as absolute number/ percentage of patients while quantitative data was presented as mean ± standard deviation (SD). Within group comparison was performed using paired t-test based on the distribution of data. Unpaired t-test was used to analyse the quantitative data for between group comparisons. Missing data was handled using Last observation carried forward (LOCF) method. P value of less than 0.05 was considered as statistical significant difference. Data were calculated using Graph Pad prism version 5.0.
Results
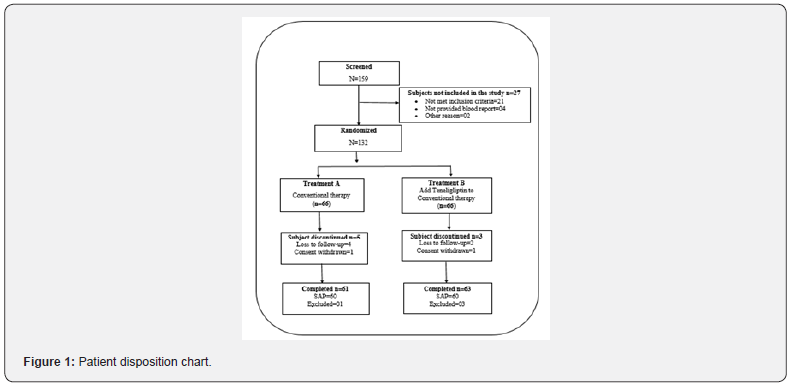
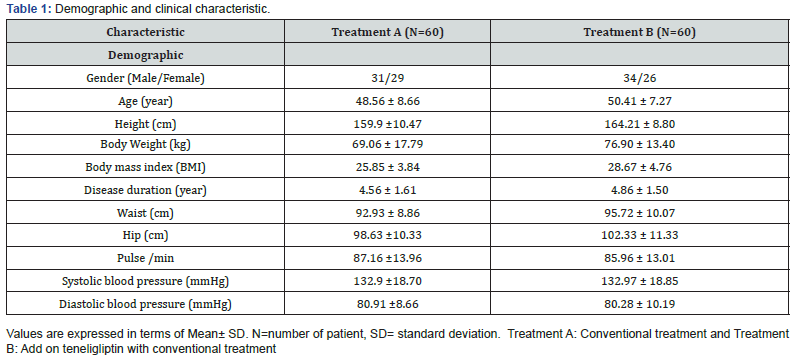
A flow chart is presented showing the disposition of participants throughout the study (Figure 1). Out of 159 screened patients, 132 eligible patients were randomized during this clinical study. Treatment A included 61 patients and treatment B included 63 patients. As per sample analysis plan (SAP), sixty patients in each group were analysed to detect a clinically significant difference between both the groups. Both the groups had similar demographic and clinical characteristic parameters at baseline (Table 1).
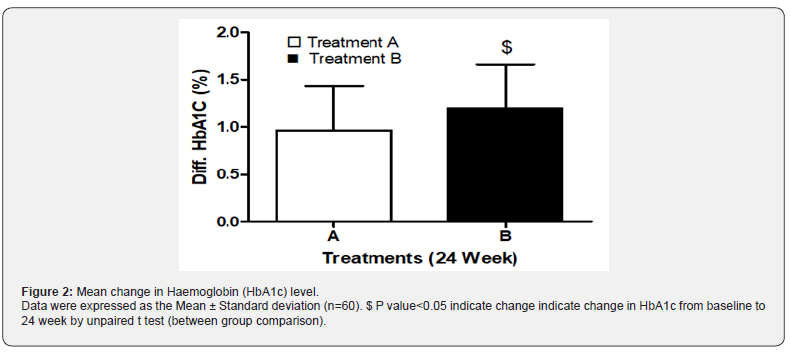
Glycaemic parameters
HbA1c level was found comparable in both the treatment groups at the baseline. However, there was gradual reduction in HbA1c over the period of 24 weeks in both the treatment groups. Between groups comparison showed significant reduction (p<0.05) in HbA1c in treatment B as compared to treatment A (Figure 2).
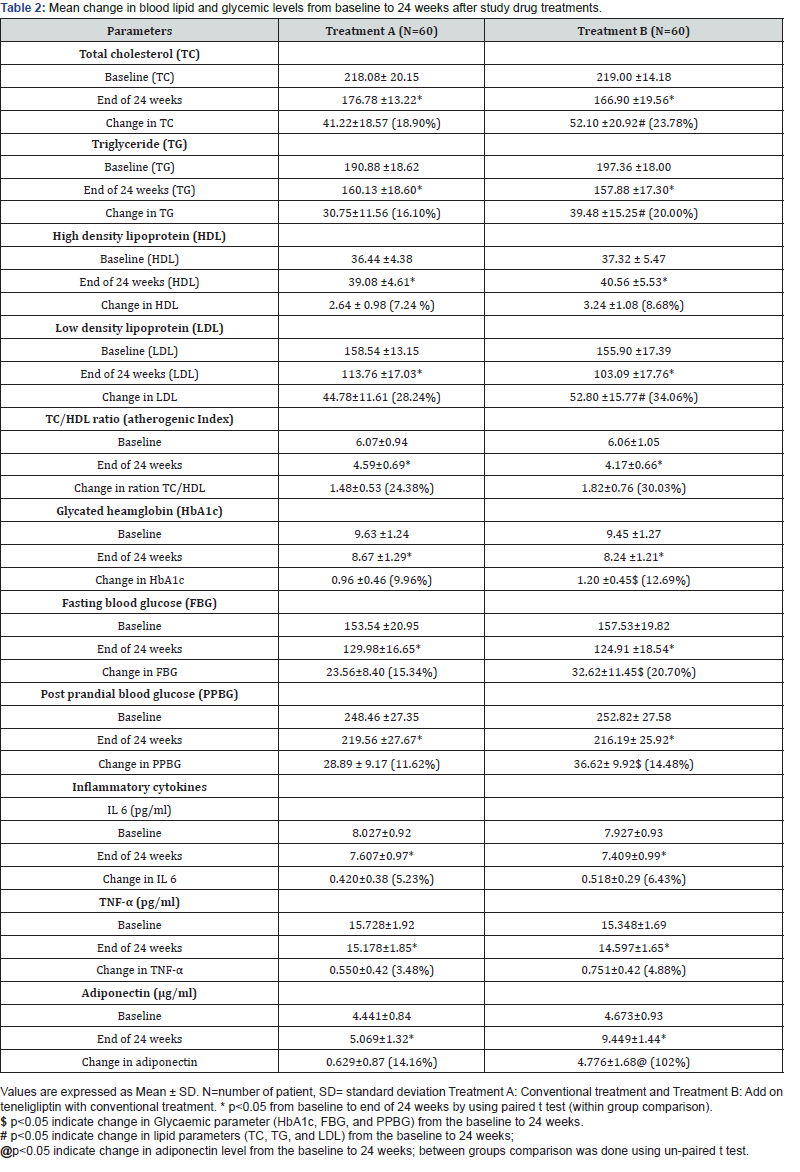
Blood glucose levels (FBG and PPBG) were found comparable in both the treatment groups at the baseline. However, there were significant reduction in FBG and PPBG levels over a period of 24 weeks in both the treatment groups. However, reduction in glycaemic parameters (HbA1c, FBG and PPBG) was statistically significant in treatment B as compared to treatment A after 24 weeks (Table 2). Reduction in mean change of HbA1c was 0.96 ± 0.46 and 1.20 ±0.45 in in treatment A & B respectively. Reduction in mean changes in FBG and PPBG levels were 23.56 ± 8.40 and 28.89 ± 9.17 in treatment A as compared to 32.62±11.45 and 36.62± 9.92in treatment B respectively.
Lipid parameters
Lipid parameters were found comparable in both the treatment groups at the baseline. However, there were gradual reduction in Total cholesterol (TC), Triglyceride (TG), atherogenic index (TC/HDL ratio) and Low density lipoprotein (LDL) over the period of 24 weeks in both the treatment groups (paired student t-test). The improvement in lipid profile was significant in treatment B as compared to treatment A at the end of 24 weeks. Mean changes of these parameters have been shown in table 2.
Total cholesterol levels
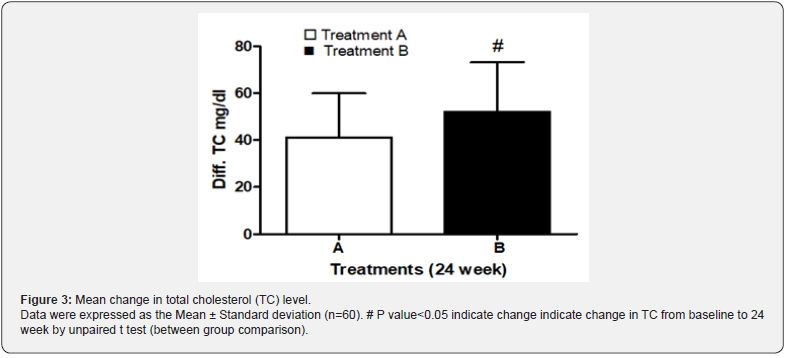
There was significant decrease in serum TC levels after 24 weeks from baseline in both treatment groups. The treatment A reduced the serum concentration of TC from 218.08± 20.15 mg/dL to 176.78 ±13.22 mg/dL and in the treatment B 219.00 ±14.18 mg/dL to 166.90 ±19.56 mg/dL. Further, between groups comparison showed significant reduction in TC in treatment B as compared to treatment A (Figure 3).
Serum triglyceride levels
Reduction in mean change of TG level was 30.75±11.56 mg/ dL and 39.48 ±15.25 mg/dL in treatment A & B. The mean change in TG levels significantly increased in treatment B as compared to treatment A at the end of 24 week. Between groups comparison showed significant reduction in TG in treatment B as compared to treatment A (Figure 4).

Serum HDL cholesterol
At the end of 24 weeks, add on teneligliptin group (treatment B) showed tendency to increase HDL level. Improvement in mean change of HDL in treatment A was 7.24% and 8.68 % in treatment B.
Serum LDL cholesterol levels
At the end of 24 weeks, add on teneligliptin group (treatment B) showed tendency to reduce in serum LDL levels. Reduction in mean change of LDL in treatment B was 34.06% as compared to 28.24 mg/dL in treatment A. The mean change in LDL levels significantly increased in treatment B as compared to treatment A at the end of 24 week. Between groups comparison showed significant reduction in LDL in treatment B as compared to treatment A.
Cytokines levels
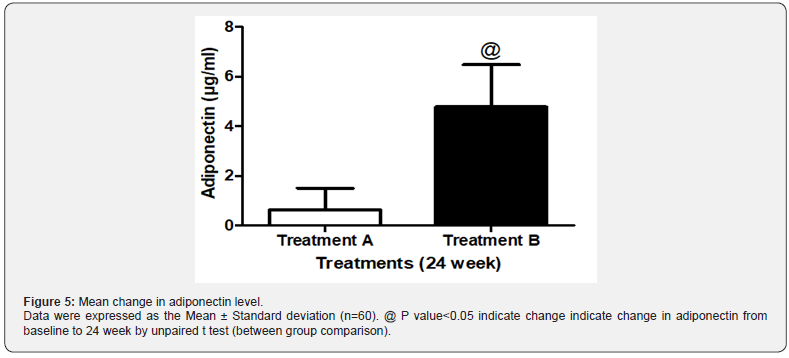
Serum Adiponectin levels: At the end of 24 weeks, add on teneligliptin group (treatment B) showed tendency to increase adiponectin level. Mean change of adiponectin in treatment B was 4.776±1.68 μg/ml as compared to 0.629±0.87 μg/ml in treatment A. The mean change in adiponectin levels significantly improved in treatment B as compared to treatment A at the end of 24 week. Between groups comparison showed better improvement in HDL in treatment B as compared to treatment A (Figure 5).
In addition to above, reduction in inflammatory cytokine levels (IL 6 and TNF-α &) was also observed in treatment B as compared to treatment A at the end of 24 weeks but was not found statistically significant (Table 2).
Safety assessment
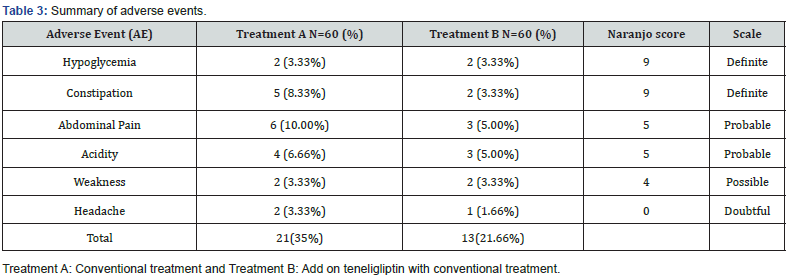
The most common AEs experienced in both the treatment groups were hypoglycaemia, constipation, abdominal pain, acidity, weakness and headache. The incidence of AE was 35% in treatment A and 21.66% in treatment B group (Table 3). Revealing better tolerability and safety of teneligliptin under the add on therapy.
Discussion
Diabetes is progressive disease which include both microvascular and macrovascular complications [15]. The relation between DM and lipid profile has been established during the past decades [16]. Both lipid profile and diabetes are shown to be the important predictors for metabolic disturbances including dyslipidaemia, hypertension, and cardiovascular diseases [4]. Diabetic dyslipidemia accounts for around 80 percent diabetic deaths due to cardiovascular complications [17]. Diabetes is typically managed employing a stepwise approach involving diet and lifestyle modification followed by addition of oral as well as IV antidiabetic drugs [18]. Although international and native guideline recommended lifestyle management as the mainstay of treatment for T2DM, with metformin as the preferred initial oral antihyperglycemic agent in most of the patients, but there is need for extra additional approach synergistic [19].
In the present study, a trial has been made to evaluate the efficacy and safety of newly developed DPP-4 inhibitor, teneligliptin 20 mg in T2DM patients having dyslipidemia who are not adequately controlled by ongoing conventional therapy. The potential effect of teneligliptin in the management of hyperlipidemia and obesity has been established in preclinical studies [20,21]. Dipeptidyl peptidase 4 inhibitors which inhibit the endogenous glucagon like peptide 1 (GLP) metabolism and thereby increases GLP-1 level in the physiological range [22]. They act by regulating insulin and glucagon secretion [23]. Rise in new beta-cells and inhibition of their apoptosis is seen with DPP- 4 which might be potentially improve the disease pathogenesis [24]. Teneligliptin suppresses proinflammatory activation of macrophages and adipocytes [25]. Therefore, it is a possible target for cardio protective effect.
Present work clearly demonstrated that teneligliptin addition to glimepiride/ metformin stable dose significantly reduced HbA1c level as compared to conventional therapy at 24 weeks from the baseline. Further, we also observed significant reduction in blood glucose levels (FBG and PPBG) in both the treatment groups. Results of the present study are found consistent with the previous clinical reports from japan, where efficacy of teneligliptin as add on treatment decreased HbA1c, FBG and PPBG levels at 12 weeks and the same study was expanded to 52 weeks [26]. Our study suggested that, the addition of teneligliptin 20 mg to conventional therapy (metformin/glimepiride) significantly improved the efficacy of conventional therapy. Our study results support the previous clinical study wherein, combination of teneligliptin with that of insulin reduced HbA1c level and showed synergistic effect [14]. HbA1c levels in blood are one of key marker to know glycemic control [27]. In our study, tenligliptin as add on treatment significantly decreased HbA1c in patients with T2DM which might be possibly due to its synergistic action. DM is often related to with alterations in lipid metabolism and abnormalities in serum lipid profile. Dyslipidemia is common in T2DM patients with poorly controlled glycemia [28]. We observed significant decrease in HbA1c and lipid profile at the end of 24 weeks in teneligliptin treated patients. Addition of teneligliptin showed significant reduction in TC at the end of 24 weeks. These findings are supported by one in all the meta-analysis report where in lipid lowering effect of DPP-4 inhibitors revealed a decrease in TC [29].
Beneficial effects of metformin on lipids could be because of inhibition of fatty acids released from adipose tissues, its direct effect on VLDL-C metabolism and/or secondary to enhance insulin sensitivity [30]. Elevated TG levels have been recognized as a risk factor for progression of CVD [31]. In this study, addition of teneliglptin reduced TG levels by 20.02% at the end of 24 week. Reduction in TG rich lipoprotein may include enhanced expression of the LDL receptors, increased expression of lipoprotein lipase, and reduced expression of apo C-III and very low-density lipoproteins [32]. Thus, teneligliptin might act through either of these mechanism to reduce this.
Besides, add on therapy of teneligliptin showed reduction in LDL-C levels (34.06 %) in treatment B. Meta-analysis report by Wulffle et al., showed efficacy of metformin in reducing TC, TG and LDL-C in their study with a minimum of 6 weeks of treatment in T2DM patients [33]. So, we conclude that add on therapy of teneligliptin together with metformin might give better reduction in serum LDL levels. Further, mean atherogenic index (TC/HDL ratio) was found comparable from baseline to week 24 for both the treatment groups. The mean atherogenic index decreased (30.03%) in treatment B when put next with the treatment A (28.24%) from baseline data. There is a report of significant decrease in TC, TG and LDL-C with increase in levels of HDL-C after 3 months of treatment with metformin in T2DM patients [34]. Pravin Kumar and Gokul reported a decrease of 16%, 12% and 10% of TC, TG and LDL-C respectively and a 15% increase of HDL-C; and achieving the lipid control goals with metformin - glimepiride combination therapy of 26 weeks in T2DM in their study [1].
Thus, our results were also parallel with the findings of various reports. Therefore, it will be suggested that teneligliptin addition with conventional antidiabetic therapy can be useful in controlling diabetes with dyslipidemia. Thus our results shown promising data of teneligliptin in T2DM patients with dyslipidemia.
Increased in adipose tissue mass occurs by the expansion of pre-existing adipocytes or by generating new small adipocytes. Chronic overfeeding leads to adipocyte hypertrophy and is related to decrease adiponectin levels and increased IL-6, and TNF-α production. In addition, the abnormal functioning of adipocytes like lipodystrophy and inability to store triglycerides and fatty acids may lead to ectopic fat storage in liver, skeletal muscle etc., thus causing dyslipidemia and insulin resistance. This condition can also play an crucial role in the development of a chronic low-grade pro-inflammatory state related to adipose tissue dysfunction and diabetes [35].
Tumor necrosis factor-α and IL-6 are important mediators of inflammation and will provide a possible link between visceral fat and systemic inflammation. They are both known to promote lipolysis and therefore the secretion of free fatty acids, which contribute to a rise in hepatic glucose output and IR, impair adipocyte differentiation, and promote inflammation [36]. Experimental studies and cross-sectional analysis have shown that circulating IL-6 is related to hyperglycemia and insulin resistance. It’s also been shown that circulating IL-6 increases with the degree of insulin resistance [37]. The protective effects of adiponectin in the prevention of progression of insulin resistance and in cardiovascular events, and its potent influence in components of the metabolic syndrome, have made it a highly promising therapeutic target [38]. These markers though well understood in terms of their regulation in diabetes population are still lacking acceptance as clinical markers because of the variation of levels among various ethnic groups [39]. Currently limited information is available on inflammatory cytokines in type 2 diabetes and people with impaired glucose and lipid metabolism in Indian population. Our study has demonstrated significantly raised adiponectine levels in treatment B as compared to treatment A. Also, an improvement in adiponectin level found associated with reduction(p>0.005) of inflammatory cytokines (IL-6 & TNF-α) which was clearly demonstrated in our study.
Safety assessment
Clinical symptomatic assessment was done for AEs like hypoglycemia and constipation, which were considered as definite; abdominal pain, and acidity were considered as probable; weakness and headache considered as possible and doubtful respectively by Naranjo AE assessment scale. It is also to be noted that, the incidence of hypoglycemic symptoms was similar 3.33% in both the groups. For selection of antihyperglycemic agents for add on therapy it include, individual patient characteristic, glucose-lowering efficacy, risk of hypoglycemia, body weight gain, and cardiovascular benefits associated with the drugs are preferentially considered [40].
In summary, results of our study showed that, add on therapy of teneligliptin with standard therapy of anti-diabetic class of drug significantly improved lipid levels by increasing level of good cholesterol as compared to the conventional therapy, this was possibly due to synergistic action of teneligliptin. Hence, during this scenario, the present study supports the initiation of treatment of T2DM with dyslipidemia using teneligliptin, which is affordable and effective in decreasing the glycaemic as well as lipid levels together with lifestyle modifications. Further add on therapy should be initiated if treatment is not satisfactory because optimum glycaemic level is must for the reduction of elevated lipid levels and thereby preventing atherosclerosis and its complications.
Conclusion
In our study teneligliptin, as add on therapy was found well tolerated and effective in T2DM with dyslipidemic patient population. Tenligliptin add on treatment with atorvastatin was found better alongwith conventional-therapy in reducing glycemic and lipid parameters as well as improvement in adiponectin levels in diabetic patients with dyslipidemia.
Acknowledgment
The authors acknowledge the guidance of Dr.Parag Shah, Dr. Krishna Shah, Dr. Alka Makad, Dr. Chirag Vaghela, Dr. Premal Thakor and Dr. Shubha Desai without their support this work would not have been possible. We also thankful to Krishna shah for providing scientific evaluation and assessment of cytokines. We are also thankful to staff of research department Jivraj Mehta Hospital and Bakeri medical research centre, Ahmedabad for extending required help and research facility for the present study.
Author’s Contribution
The corrospending author has designed the work, data collection, data analysis, statistical analysis, and prepared manuscript. Both authors discussed and provide critical feedback on manuscript.
References
- Qin J, Li R, Raes J, Arumugam M, Burgdorf KS, et al. (2010) A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464: 59-65.
- Tang WH, Kitai T, Hazen SL (2017) Gut microbiota in cardiovascular health and disease. Circ Res 120(1): 1183-1196.
- Emoto T, Yamashita T, Kobayashi T, Sasaki N, Hirota Y, et al. (2017) Characterization of gut microbiota profiles in coronary artery disease patients using data mining analysis of terminal restriction fragment length polymorphism: gut microbiota could be a diagnostic marker of coronary artery disease. Heart Vessels 32(1): 39-46.
- Ley RE, Turnbaugh PJ, Klein S, Gordon JI (2006) Microbial ecology: human gut microbes associated with obesity. Nature 444(7122): 1022-1023.
- Henao-Mejia J, Elinav E, Jin C (2012) Inflammasome-mediated dysbiosis regulates progression of NAFLD and obesity. Nature 482: 179-185.
- Pedersen HK, Gudmundsdottir V, Nielsen HB (2016) Human gut microbes impact host serum metabolome and insulin sensitivity. Nature 535: 376-381.
- Khan MT, Nieuwdorp M, Backhed F (2014) Microbial modulation of insulin sensitivity. Cell Metab 20(5): 753-760.
- Mouzaki M, Comelli EM, Arendt BM (2013) Intestinal microbiota in patients with non-alcoholic fatty liver disease. Hepatology 58(1): 120-127.
- Zhu L, Baker SS, Gill C, Liu W, Alkhouri R, et al. (2013) Characterization of gut microbiomes in non-alcoholic steatohepatitis (NASH) patients: a connection between endogenous alcohol and NASH. Hepatology 57(2): 601-609.
- Gopalakrishnan V, Helmink BA, Spencer CN, Reuben A, Wargo JA, et al. (2018) The influence of the gut microbiome on cancer, immunity, and cancer immunotherapy. Cancer Cell 33(4): 570-580.
- Tilg H, Adolph TE, Gerner RR, Moschen AR (2018) The intestinal microbiota in colorectal cancer. Cancer Cell 33(6): 954-964.
- Kamo T, Akazawa H, Suda W, Saga-Kamo A, Shimizu Y, et al. (2017) Dysbiosis and compositional alterations with aging in the gut microbiota of patients with heart failure. PLoS One 12(3): e0174099.
- Lau K, Srivatsav V, Rizwan A, Nashed A, Liu R, et al. (2017) Bridging the gap between gut microbial dysbiosis and cardiovascular diseases. Nutrients 9(8): E859.
- Ma J, Li H (2018) The Role of Gut Microbiota in Atherosclerosis and Hypertension. Front Pharmacol 9: 1082.
- Bäckhed F, Ding H, Wang T, Hooper LV, Koh GY, et al. (2004) The gut microbiota as an environmental factor that regulates fat storage. Proc Natl Acad Sci USA 101(4): 15718-15723.
- Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, et al. (2006) An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444(7122): 1027-1031.
- Samuel BS, Shaito A, Motoike T, Rey FE, Backhed F, et al. (2008) Effects of the gut microbiota on host adiposity are modulated by the short-chain fatty-acid binding G protein-coupled receptor, Gpr41. Proc Natl Acad Sci USA 105(43): 16767-16772.
- Tolhurst G, Heffron H, Lam YS, Parker HE, Habib AM, et al. (2012) Short-chain fatty acids stimulate glucagon-like peptide-1 secretion via the G-protein-coupled receptor FFAR2. Diabetes 61(2): 364-371.
- Bäckhed F, Manchester JK, Semenkovich CF, Gordon JI (2007) Mechanisms underlying the resistance to diet-induced obesity in germ-free mice. Proc Natl Acad Sci USA 104(3): 979-984.
- Ley RE, Bäckhed F, Turnbaugh P, Lozupone CA, Knight RD, et al. (2005) Obesity alters gut microbial ecology. Proc Natl Acad Sci USA 102: 11070-11075.
- Cani PD, Amar J, Iglesias MA, Poggi M, Knauf C, et al. (2007) Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 56(7): 1761-1772.
- de La Serre CB, Ellis CL, Lee J, Hartman AL, Rutledge JC, et al. (2010) Propensity to high-fat diet-induced obesity in rats is associated with changes in the gut microbiota and gut inflammation. Am J Physiol Gastrointest Liver Physiol 299(4): G440-G448.
- Muccioli GG, Naslain D, Bäckhed F, Reigstad CS, Lambert DM, et al. (2010) The endocannabinoid system links gut microbiota to adipogenesis. Mol Syst Biol 6: 392.
- Cani PD, Bibiloni R, Knauf C, Waget A, Neyrinck AM, et al. (2008) Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet-induced obesity and diabetes in mice. Diabetes 57(6): 1470-1481.
- Belzer C, de Vos WM (2012) Microbes inside--from diversity to function: the case of Akkermansia. ISME J 6(8): 1449-1458.
- Wall R, Ross RP, Shanahan F, O'Mahony L, O Mahony C, et al. (2009) Metabolic activity of the enteric microbiota influences the fatty acid composition of murine and porcine liver and adipose tissues. Am J Clin Nutr 89(5): 1393-1401.
- Creely SJ, McTernan PG, Kusminski CM, Fisher fM, Da Silva NF, et al. (2007) Lipopolysaccharide activates an innate immune system response in human adipose tissue in obesity and type 2 diabetes. Am J Physiol Endocrinol Metab 292(3): E740-E747.
- Amar J, Serino M, Lange C, Chabo C, Iacovoni J, et al. (2011) Involvement of tissue bacteria in the onset of diabetes in humans: evidence for a concept. Diabetologia 54(12): 3055-3061.
- Lewis K, Lutgendorff F, Phan V, Söderholm JD, Sherman PM, et al. (2010) Enhanced translocation of bacteria across metabolically stressed epithelia is reduced by butyrate. Inflamm Bowel Dis 16(7): 1138-1148.
- Cope K, Risby T, Diehl AM (2000) Increased gastrointestinal ethanol production in obese mice: implications for fatty liver disease pathogenesis. Gastroenterology 119(5): 1340-1347.
- Spruss A, Kanuri G, Wagnerberger S, Haub S, Bischoff SC, et al. (2009) Toll-like receptor 4 is involved in the development of fructose-induced hepatic steatosis in mice. Hepatology 50(4): 1094-1104.
- Henao-Mejia J, Elinav E, Jin C, Hao L, Mehal WZ, et al. (2012) Inflammasome-mediated dysbiosis regulates progression of NAFLD and obesity. Nature 482(7384): 179-185.
- Shanab AA, Scully P, Crosbie O, Buckley M, O Mahony L, et al. (2011) Small intestinal bacterial overgrowth in non-alcoholic steatohepatitis: association with toll-like receptor 4 expression and plasma levels of interleukin 8. Dig Dis Sci 56(5): 1524-1534.
- Drosos I, Tavridou A, Kolios G (2015) New aspects on the metabolic role of intestinal microbiota in the development of atherosclerosis. Metabolism 64(4): 476-481.
- Jie Z, Xia H, Zhong SL (2017) The gut microbiome in atherosclerotic cardiovascular disease. Nat Commun 8: 845.
- Kasahara K, Tanoue T, Yamashita T, Yodoi K, Matsumoto T, et al. (2017) Commensal bacteria at the crossroad between cholesterol homeostasis and chronic inflammation in atherosclerosis. J Lipid Res 58(3): 519-528.
- Karlsson FH, Fak F, Nookaew I, Tremaroli V, Fagerberg B, et al. (2012) Symptomatic atherosclerosis is associated with an altered gut metagenome. Nat Commun 3: 1245.
- Li J, Lin S, Vanhoutte PM, Woo CW, Xu A (2016) Akkermansia muciniphila protects against atherosclerosis by preventing metabolic endotoxemia-induced inflammation in apoe-/- mice. Circulation 133(24): 2434-2446.
- Chen WY, Wang M, Zhang J, Barve SS, McClain CJ, et al. (2017) Acrolein disrupts tight junction proteins and causes endoplasmic reticulum stress-mediated epithelial cell death leading to intestinal barrier dysfunction and permeability. Am J Pathol 187(12): 2686-2697.
- Gui T, Shimokado A, Sun Y (2012) Diverse roles of macrophages in atherosclerosis: from inflammatory biology to biomarker discovery. Mediat Inflamm 2012: 693083.
- Harris K, Kassis A, Major G, Chou CJ (2012) Is the gut microbiota a new factor contributing to obesity and its metabolic disorders?. J Obes 2012: 879151.
- Akira S, Takeda K (2004) Toll-like receptor signalling. Nat Rev Immunol 4: 499-511.
- Xu H, Barnes GT, Yang Q, Tan G, Yang D, et al. (2003) Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J Clin Investig 112(12): 1821-1830.
- Edfeldt K, Swedenborg J, Hansson GK, Yan ZQ (2002) Expression of toll-like receptors in human atherosclerotic lesions: a possible pathway for plaque activation. Circulation 105(10): 1158-1161.
- Barton GM, Kagan JC (2009) A cell biological view of Toll-like receptor function: regulation through compartmentalization. Nat Rev Immunol 9(8): 535-542.
- Guzzo C, Ayer A, Basta S, Banfield BW, Gee K (2012) IL-27 enhances LPS-induced proinflammatory cytokine production via upregulation of TLR4 expression and signaling in human monocytes. J Immunol 188(2): 864-873.
- Laman JD, Schoneveld AH, Moll FL, van Meurs M, Pasterkamp G, et al. (2002) Significance of peptidoglycan, a proinflammatory bacterial antigen in atherosclerotic arteries and its association with vulnerable plaques. Am J Cardiol 90(2): 119-123.
- Kanno S, Nishio H, Tanaka T, et al. (2015) Activation of an innate immune receptor, Nod1, accelerates atherogenesis in Apoe-/- mice. J. Immunol 194(2): 773-780.
- Li X, Shimizu Y, Kimura I (2017) Gut microbial metabolite short-chain fatty acids and obesity. Biosci Microbiota Food Health 36(4): 135-140.
- Bergeron N, Williams PT, Lamendella R, Faghihnia N, Grube A, et al. (2016) Diets high in resistant starch increase plasma levels of trimethylamine-N-oxide, a gut microbiome metabolite associated with CVD risk. Br J Nutr 116(12): 2020-2029.
- Brown JM, Hazen SL (2018) Microbial modulation of cardiovascular disease. Nat Rev Microbiol 16(3): 171-181.
- Brown JM, Hazen SL (2015) The gut microbial endocrine organ: bacterially derived signals driving cardiometabolic diseases. Annu Rev Med 66: 343-359.
- Wang Z, Tang WH, Buffa JA, Fu X, Britt EB, et al. (2014) Prognostic value of choline and betaine depends on intestinal microbiota-generated metabolite trimethylamine-N-oxide. Eur Heart J 35(14): 904-910.
- Shih DM, Wang Z, Lee R, Meng Y, Che N, et al. (2015) Flavin containing monooxygenase 3 exerts broad effects on glucose and lipid metabolism and atherosclerosis. J Lipid Res 56(1): 22-37.
- Warrier M, Shih DM, Burrows AC, Ferguson D, Gromovsky AD, et al. (2015) The TMAO-generating enzyme flavin monooxygenase 3 is a central regulator of cholesterol balance. Cell Rep 10(3): 326-338.
- Chen K, Zheng X, Feng M, Li D, Zhang H (2017) Gut microbiota-dependent metabolite trimethylamine n-oxide contributes to cardiac dysfunction in western diet-induced obese mice. Front Physiol 8: 139.
- Guasch-Ferre M, Hu FB, Ruiz-Canela M, Bulló M, Toledo E, et al. (2017) Plasma metabolites from choline pathway and risk of cardiovascular disease in the PREDIMED (Prevention with Mediterranean Diet) Study. J Am Heart Assoc 6(11): e006524.
- Troseid M, Ueland T, Hov JR, Svardal A, Gregersen I, et al. (2015) Microbiota-dependent metabolite trimethylamine-N-oxide is associated with disease severity and survival of patients with chronic heart failure. J Int Med 277(6): 717-726.
- Senthong V, Li XS, Hudec T, Coughlin J, Wu Y, et al. (2016a) Plasma trimethylamine N-Oxide, a gut microbe–generated phosphatidylcholine metabolite, is associated with atherosclerotic burden. J Am Coll Cardiol 67(22): 2620-2628.
- Senthong V, Wang Z, Li XS, Fan Y, Wu Y, et al. (2016b) Intestinal microbiota-generated metabolite trimethylamine-N-Oxide and 5-year mortality risk in stable coronary artery disease: the contributory role of intestinal microbiota in a COURAGE-like patient cohort. J Am Heart Assoc 5(6): e002816.
- Zhu W, Gregory JC, Org E, Buffa JA, Gupta N, et al. (2016) Gut microbial metabolite TMAO enhances platelet hyperreactivity and thrombosis risk. Cell 165(1): 111-124.
- Zheng Y, Li Y, Rimm EB, Hu FB, Albert CM, et al. (2016) Dietary phosphatidylcholine and risk of all-cause and cardiovascular-specific mortality among US women and men. Am J Clin Nutr 104(1): 173-180.
- Nagata C, Wada K, Tamura T, Konishi K, Kawachi T, et al. (2015) Choline and betaine intakes are not associated with cardiovascular disease mortality risk in Japanese men and women. J Nutr 145(8): 1787-1792.
- Collins HL, Drazul-Schrader D, Sulpizio AC, Koster PD, Williamson Y, et al. (2016) L-Carnitine intake and high trimethylamine N-oxide plasma levels correlate with low aortic lesions in ApoE (-/-) transgenic mice expressing CETP. Atherosclerosis 244: 29-37.
- Kuipers F, Bloks VW, Groen AK (2014) Beyond intestinal soap–bile acids in metabolic control. Nat Rev Endocrinol 10(8): 488-498.
- Jones ML, Martoni CJ, Ganopolsky JG, Labbé A, Prakash S (2014) The human microbiome and bile acid metabolism: dysbiosis, dysmetabolism, disease and intervention. Exp Opin Biol Ther 14(4): 467-482.
- Zheng X, Huang F, Zhao A, Lei S, Zhang Y, et al. (2017) Bile acid is a significant host factor shaping the gut microbiome of diet-induced obese mice. BMC Biol 15(1): 120.
- Joyce SA, Gahan CG (2017) Disease-associated changes in bile acid profiles and links to altered gut microbiota. Dig Dis 35(3): 169-177.
- Ridlon JM, Harris SC, Bhowmik S, Kang DJ, Hylemon PB (2016) Consequences of bile salt bio-transformations by intestinal bacteria. Gut Microbes 7(1): 22-39.
- Hansson GK, Robertson AK, Soderberg-Naucler C (2006) Inflammation and atherosclerosis. Annu Rev Pathol 1: 297-329.
- Tremaroli V, Backhed F (2012) Functional interactions between the gut microbiota and host metabolism. Nature 489(7415): 242-249.
- Townsend MK, Aschard H, De Vivo I, Michels KB, Kraft P (2016) Genomics, telomere length, epigenetics, and metabolomics in the nurses’ health studies. Am J Public Health 106(9): 1663-1668.
- Yamashiro K, Tanaka R, Urabe T, Ueno Y, Yamashiro Y, et al. (2017) Gut dysbiosis is associated with metabolism and systemic inflammation in patients with ischemic stroke. PLoS One 12(2): e0171521.
- Honour J (1982) The possible involvement of intestinal bacteria in steroidal hypertension. Endocrinology 110(1): 285-287.
- Yang T, Santisteban MM, Rodriguez V, Li E, Ahmari N, et al. (2015) Gut dysbiosis is linked to hypertension. Hypertension 65(6): 1331-1340.
- Karbach SH, Schonfelder T, Brandao I, Wilms E, Hörmann N, et al. (2016) Gut microbiota promotes angiotensin ii-induced arterial hypertension and vascular dysfunction. J Am Heart Assoc 5(9): e003698.
- Koh A, De Vadder F, Kovatcheva-Datchary P, Backhed F (2016) From dietary fiber to host physiology: short-chain fatty acids as key bacterial metabolites. Cell 165(6): 1332-1345.
- Rey FE, Faith JJ, Bain J, Muehlbauer MJ, Stevens RD, et al. (2010) Dissecting the in vivo metabolic potential of two human gut acetogens. J Biol Chem 285(29): 22082-22090.
- Louis P, Flint HJ (2017) Formation of propionate and butyrate by the human colonic microbiota. Environ Microbiol 19(1): 29-41.
- Duncan SH, Barcenilla A, Stewart CS, Pryde SE, Flint HJ (2002) Acetate utilization and butyryl coenzyme A (CoA): acetate-CoA transferase in butyrate-producing bacteria from the human large intestine. Appl Environ Microbiol 68(10): 5186-5190.
- Gomez-Arango LF, Barrett HL, McIntyre HD, Callaway LK, Morrison M, et al. (2016) Increased systolic and diastolic blood pressure is associated with altered gut microbiota composition and butyrate production in early pregnancy. Hypertension 68(4): 974-981.
- Marques FZ, Nelson E, Chu PY, Horlock D, Fiedler A, et al. (2017) High-fiber diet and acetate supplementation change the gut microbiota and prevent the development of hypertension and heart failure in hypertensive mice. Circulation 135(10): 964-977.
- Tan JK, McKenzie C, Marino E, Macia L, Mackay CR (2017) Metabolite-sensing g protein-coupled receptors-facilitators of diet-related immune regulation. Annu Rev Immunol 35: 371-402.
- Pluznick JL, Protzko RJ, Gevorgyan H, Peterlin Z, Sipos A, et al. (2013) Olfactory receptor responding to gut microbiota-derived signals plays a role in renin secretion and blood pressure regulation. Proc Natl Acad Sci U.S.A 110(11): 4410-4415.
- Natarajan N, Hori D, Flavahan S, Steppan J, Flavahan NA, et al. (2016) Microbial short chain fatty acid metabolites lower blood pressure via endothelial G protein-coupled receptor 41. Physiol Genomics 48(11): 826-834.
- Miyamoto J, Kasubuchi M, Nakajima A, Irie J, Itoh H, et al. (2016) The role of short-chain fatty acid on blood pressure regulation. Curr Opin Nephrol Hypertens 25(5): 379-383.
- Packer CS, Rice AE, Johnson TC (2014) Oxidized low density lipoprotein (OX-LDL) induced arterial muscle contraction signaling mechanisms. Open Hyperten J 6: 20-26.
- Peluso I, Morabito G, Urban L, Ioannone F, Serafini M (2012) Oxidative stress in atherosclerosis development: the central role of LDL and oxidative burst. Endocr Metab Immune Disord Drug Targets 12(4): 351-360.
- Koopen AM, Groen AK, Nieuwdorp M (2016) Human microbiome as therapeutic intervention target to reduce cardiovascular disease risk. Curr Opin Lipidol 27(6): 615-622.
- Daliri EB, Lee BH, Oh DH (2017) Current perspectives on antihypertensive probiotics. Probiot Antimicrob Proteins 9(2): 91-101.
- Anbazhagan AN, Priyamvada S, Priyadarshini M (2017) Gut microbiota in vascular disease: therapeutic target? Curr Vasc Pharmacol 15(14): 291-295.
- Santisteban MM, Qi Y, Zubcevic J, Kim S, Yang T, et al. (2017) Hypertension-linked pathophysiological alterations in the gut. Circ Res 120(2): 312-323.
- Ettinger G, MacDonald K, Reid G, Burton JP (2014) The influence of the human microbiome and probiotics on cardiovascular health. Gut Microbes 5(6): 719-728.
- He M, Shi B (2017) Gut microbiota as a potential target of metabolic syndrome: the role of probiotics and prebiotics. Cell Biosci 7: 54.
- Fuentes MC, Lajo T, Carrion JM, Cune J (2013) Cholesterol-lowering efficacy of Lactobacillus plantarum CECT 7527, 7528 and 7529 in hypercholesterolaemic adults. Br J Nutr 109: 1866-1872.
- Jones ML, Martoni CJ, Prakash S (2012) Cholesterol lowering and inhibition of sterol absorption by Lactobacillus reuteri NCIMB 30242: a randomized controlled trial. Eur J Clin Nutr 66(11): 1234-1241.
- Gomez-Guzman M, Toral M, Romero M, Jiménez R, Galindo P, et al. (2015) Antihypertensive effects of probiotics Lactobacillus strains in spontaneously hypertensive rats. Mol Nutr Food Res 59(11): 2326-2336.
- Costabile A, Buttarazzi I, Kolida S, Quercia S, Baldini J, et al. (2017) An in vivo assessment of the cholesterol-lowering efficacy of Lactobacillus plantarum ECGC 13110402 in normal to mildly hypercholesterolaemic adults. PLoS One 12(12): e0187964.
- Yoo JY, Kim SS (2016) Probiotics and prebiotics: present status and future perspectives on metabolic disorders. Nutrients 8(3): 173.
- Catry E, Bindels LB, Tailleux A, Lestavel S, Neyrinck AM, et al. (2018) Targeting the gut microbiota with inulin-type fructans: preclinical demonstration of a novel approach in the management of endothelial dysfunction. Gut 67(2): 271-283.
- Rault-Nania MH, Gueux E, Demougeot C, Demigné C, Rock E, et al. (2006) Inulin attenuates atherosclerosis in apolipoprotein E-deficient mice. Br J Nutr 96(5): 840-844.
- Hoving LR, Katiraei S, Heijink M, Pronk A, van der Wee-Pals L, et al. (2018) Dietary mannan oligosaccharides modulate gut microbiota, increase fecal bile acid excretion, and decrease plasma cholesterol and atherosclerosis development. Mol Nutr Food Res 62(10): e1700942.
- Zhu L, Zhang D, Zhu H, Zhu J, Weng S, et al. (2018) Berberine treatment increases Akkermansia in the gut and improves high-fat diet-induced atherosclerosis in Apoe (-/-) mice. Atherosclerosis 268: 117-126.
- Bird JK, Raederstorff D, Weber P, Steinert RE (2017) Cardiovascular and anti-obesity effects of resveratrol mediated through the gut microbiota. Adv Nutr 8(6): 839-849.
- Chen ML, Yi L, Zhang Y, Zhou X, Ran L et al. (2016) Resveratrol attenuates trimethylamine-N-Oxide (TMAO)-induced atherosclerosis by regulating TMAO synthesis and bile acid metabolism via remodeling of the gut microbiota. MBio 7(2): e02210-15.
- Colman RJ, Rubin DT (2014) Fecal microbiota transplantation as therapy for inflammatory bowel disease: a systematic review and meta-analysis. J Crohns Colitis 8(12): 1569-1581.
- Vrieze A, Van Nood E, Holleman F, Salojärvi J, Kootte RS, et al. (2012) Transfer of intestinal microbiota from lean donors increases insulin sensitivity in individuals with metabolic syndrome. Gastroenterology 143(4): 913-916.
- (2014) Draft Guidance for Industry: Enforcement Policy Regarding Investigational New Drug Requirements for Use of Fecal Microbiota for Transplantation to Treat Clostridium difficile Infection Not Responsive to Standard Therapies. Silver Spring, MD: US Food and Drug Administration.
- Petrof E, Gloor G, Vanner S, Weese SJ, Carter D, et al. (2013) Stool substitute transplant therapy for the eradication of Clostridium difficile infection: ‘repopulating’ the gut. Microbiome 1(1): 3.






























