Cardio-Ankle Vascular Index (CAVI) Measurement of Arterial Stiffness Associated with Cardiovascular Disease Risk Factors in Type 2 Diabetes Patients
Kanyarat Kanokphichayakrai1, Watchara Kaewmahanin2, Orathai Tangvarasittichai3 and Surapon Tangvarasittichai3*
1Division of Echocardiogram, Buddhachinaraj Hospital, Thailand
2Department of Cadio-Thoracic Technology, Faculty of Allied Health Sciences, Naresuan University, Thailand
3Department of Medical Technology, Faculty of Allied Health Sciences, Naresuan University, Thailand
Submission: November 06, 2017; Published: November 22, 2017
*Corresponding author: Surapon Tangvarasittichai, Department of Medical Technology, Faculty of Allied Health Sciences, Naresuan University, Phitsanulok, 65000, Thailand, Tel: +66-08-9638-8382; Fax: +66-0-5596-6300; Email: surapon14t@yahoo.com
How to cite this article: Kanyarat K, Watchara K, Orathai T, Surapon T. Cardio-Ankle Vascular Index (CAVI) Measurement of Arterial Stiffness Associated with Cardiovascular Disease Risk Factors in Type 2 Diabetes Patients. Curre Res Diabetes & Obes J. 2017; 5(1): 555651. DOI: 10.19080/CRDOJ.2017.04.555651
Abstract
Cardiovascular disease (CVD) is common occur in type 2 diabetes mellitus (T2DM) patients. We aim to evaluate the association of cardio-ankle vascular index (CAVI) with the CVD risk factors. Anthropometric and CAVI measurements, blood pressure (BP) and biochemical variables were measured. One hundred and eighty seven women were allocated to 2 groups. Group (Gr)-1; 56T2DM patients with CAVI>0.9 were significantly higher in age, BP, mean arterial pressure (MAP), fasting blood sugar (Glu), total cholesterol (TC), triglyceride (TG), low density lipoprotein (LDL-C), hs-CRP, insulin, insulin resistance (HOMA-IR) and significantly lower in BMI, high density lipoprotein (HDL-C)levels than Gr-2; 131 non-T2DM subjects with CAVI<0.9. In bivariate correlation, CAVI was significantly correlated with Age, BMI, MAP, TC, HDL-C and hs-CRP (p<0.05). Multiple forward stepwise linear regression analyses of the significant variables showed that the independent predictors of CAVI were Age (b=0.448, R2=0.406, p<0.001), hs-CRP (b=0.220, R2 =0.507, p<0.001), BMI (b=-0.383, R2=0.545, p<0.001), HDL-C (b=-0.350, R2=0.484, p<0.001), TC (b=0.256, R2=0.508, p<0.001) and MAP (b=0.248, R2 =0.524, p<0.001). CAVI was associated with CVD risk factors and can be used as a CVD risk factor in T2DM patients. CAVI measurement is a useful tool to assess arterial stiffness and evaluate for subclinical coronary atherosclerosis in asymptomatic patients.
Keywords: Cardio-ankle vascular index (CAVI); Arterial stiffness; Hs-CRP; HDL-C; Mean arterial pressure
Introduction
Cardiovascular disease (CVD) is common occur in type 2 diabetes mellitus (T2DM) patients and higher in morbidity and mortality rate than non-diabetic subjects [1]. Cardiovascular disease morbidity is a consequence of the progression of vascular disease. Atherosclerosis is asymptomatic in the early stages and can be caused from either injury or defect of the protective function of the arterial endothelia. Atherosclerosis is commonly caused by plaque and may follow arteriosclerosis (the hardening and loss of elasticity of arterioles). The progression of atherosclerosis is an irreversible, lipid accumulation concomitant with calcification of the arteries and remodeling in microcirculation structure. Plaque formation is the further progression and plaque rupture will cause an acute morbid event [2]. Atherosclerosis is well correlated with arterial stiffness and the progression of coronary artery thickening [3,4]. Thus, early diagnosis of atherosclerosis is important for prevention and intervention of coronary artery disease.
Cardio-ankle vascular index (CAVI) was developed to assess arterial stiffness. The CAVI measurement is noninvasive and is not dependents on the blood pressure during measurement. It is measured between the brachial and ankle arteries the same as the brachial ankle pulse wave velocity (baPWV) measurement and used the conventional baPWV for calculation [5]. Horinaka et al. [6] demonstrated that CAVI has increased performance over brachial ankle pulse wave velocity in coronary artery disease prediction and has high-level of reproducibility [7,8]. Many studies have examined the association of CAVI with atherosclerosis [9,10]. In general, changing in mean arterial pressure (MAP) may cause by the structural degradation and the change in composition of the arterial wall which are the main determinants of arterial stiffness. When elastin fibers within the blood vessels are lost, they become thinner and fragmented and the stiffer collagen fibers become load bearing [11]. Additionally, in the balance of the elastin synthesis and breakdown by the matrix metalloproteinases contributes the arterial stiffness progression [12]. Atherosclerosis is an inflammatory disease, and inflammation also affects the stiffness of the large arteries [13]. Further changes in the arterial wall composition occur due to inflammatory cell infiltration and endothelial dysfunction [14,15]. We aim to evaluate CAVI (CAVI>0.9, arterial stiffness) as CVD risk factors and the association of CAVI with CVDrisk factors in T2DM patients.
Methods
Study population
Two hundred and five of female participants from the Cardiovascular Diseases in Diabetes Patients Project during October 2012-December 2013were used in the present study. Eighteen participants didn’t participate in CAVI measurement section. The eligible of participants were one hundred and eighty seven in the present study. We categorized these participants into 2 groups: Group (Gr)-1 as 56 subjects with CAVI measurement>0.9 and T2DM; Gr-2 as control Gr consist with 131 subjects with CAVI measurement<0.9 and without T2DM. Volunteers were excluded, if they had an ongoing febrile illness, history of a connective tissue disorders, nonatherosclerotic arterial disease, history of lower extremity bypass or percutaneous angioplasty in the preceding year. The research protocol was approved by the Ethics Committee of the Naresuan University. All participants gave informed consent before their provided blood samples and underwent assessment for CAVI measurement.
Anthropometric, blood pressure and CAVI measurements
Questionnaires were used to record clinical characteristics including diagnosis of hypertension, diabetes, a history of MI or stroke, smoking, alcohol use, and medications of each participant at the study visit. Anthropometric measurements of the study included height, weight and waist circumference (WC).The body mass index (BMI) was calculated from height and weight as kg/ m2. Blood pressure (BP) was measured by using Omron HEM- 7080 (Omron Health care, Tokyo, Japan). Pulse pressure (PP) was determined by subtracting the diastolic from the systolic blood pressure (Syst), and mean arterial pressure (MAP) was calculated by using the formula: [(systolic blood pressure)+(2 x diastolic blood pressure)]/3 [16].
The CAVI measurement is made in the supine position after 5 min of rest by using a VS-1500N (Vasera, VS-1500N ver. 04; Fukuda Denshi, Tokyo, Japan). Electrocardiography electrodes were placed on the both wrists. A heart sounds detector microphone was placed on the sternum and the cuffs were wrapped around both arms and ankles. Blood pressure and pulse wave were measured by pressurizing four points at the same time. The phonocardiogram was measured at the same time on two left sternal borders, and the time from the two sounds (close to the aortic valve) to the attainment of the pulse wave was measured [5,6]. The averages of the right and left CAVI values were used for the analyses. The cut-off value of CAVI >9.0 was demonstrated for estimating the coronary artery stenos is using an ROC curve [6, 9].The coefficient of variation of CAVI measurement is less than 5%, indicates that CAVI has good reproducibility [7].
Blood sample collection and biochemical determination
All participants were fasted for 8-12 h for the blood test. Plasma glucose (Glu), blood urea nitrogen (BUN), total cholesterol (TC), triglycerides (TG) and high density lipoprotein cholesterol (HDL-C) were measured by enzymatic method (Roche diagnostic, Switzerland). Serum creatinine (CT) level was determined based on the Jaffe reaction. LDL-C level was calculated with Friedewald’s formula in specimens with TG level<400 mg/dl.
Highly sensitive C-reactive protein (hs-CRP) assay
Highly sensitive-CRP concentrations were determined by using latex particle enhanced immunoturbidometric assay on the Hitachi 912 auto-analyzer (Roche Diagnostic, Switzerland)that has been standardized against the World Health Organization reference. The normal range of hs-CRP was<3.0 mg/l (<0.03 g/l).
Insulin assay
Fasting insulin levels were measured based on microparticle enzyme immunoassay (MEIA) technology using Abbott reagents with Axsym system (Abbott laboratories, Illinois, USA). All participants underwent evaluation of insulin resistance index (IR) by using the Homeostasis model assessment (HOMA)- formula [17]. HOMA of insulin resistance (IR) was defined using the following formula: fasting glucose (mmol/l)x fasting insulin (μU/ml)/22.5 [17].
Statistical analysis
All variables are expressed as median and interquartile range. The Mann-Whitney U test was used to estimate difference between groups. Spearman rank correlation was used to assess the bivariate correlation of all clinical markers in the study participants. Clinical variables that correlated with CAVI (arterial stiffness) in the present study were tested as independent variables by using multivariate forward stepwise linear regression analysis. Tests were two-tailed, and a p-value <0.05 was considered significant by using the SPSS version 13.0 (SPSS, Chicago, IL).
Results
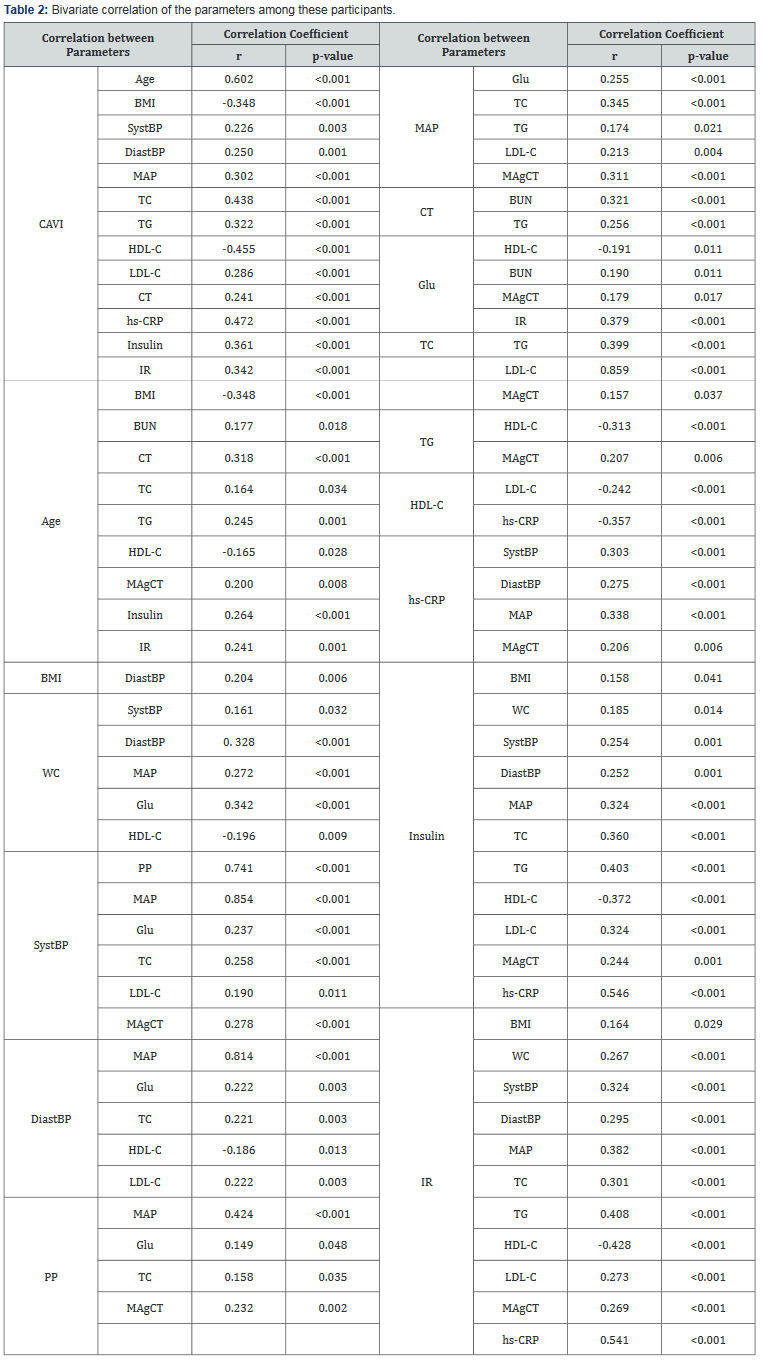
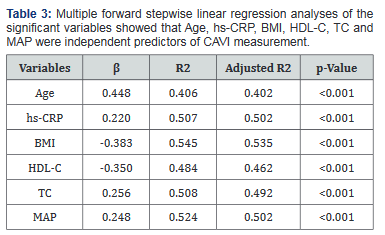
The comparison of general clinical characteristics of Gr-1 and Gr-2 was demonstrated in Table 1.Gr-1demonstrated physically inactive, older, decreased BMI, and significantly greater SystBP and DiastBP, MAP, BUN, CT, TC, TG,LDL-C, hs- CRP, insulin, IR levels and lower HDL-C. Bivariate correlations demonstrated that CAVI was significantly correlated with Age (r=0.602, p<0.001), BMI (r= -0.348, p<0.001), SystBP (r=0.226, p=0.003), DiastBP (r=0.250, p=0.001), MAP (r=0.302, p<0.001), CT (r=0.241, p<0.001), TC (r=0.438, p<0.001), TG (r=0.322, p<0.001), LDL-C (r=0.286, p<0.001), HDL-C (r= -0.455, p<0.001), hs-CRP (r=0.472, p<0.001), insulin (r=0.361, p<0.001)and IR (r=0.342, p<0.001).The correlation of the other clinical variables was shown in Table 2. We used multiple forward stepwise linear regression analysis to examine effects of variables in the association of these variables with CAVI. Statistics were listed in Table 3. Age, hs-CRP, BMI, HDL-C, TC and MAP showed the association with CAVI, which remained highly significant after adjusting for any clinical or laboratory confounding variables [Age (b =0.448, R2 =0.406, p<0.001), hs-CRP (b =0.220, R2 =0.507, p<0.001),BMI (b =-0.383, R2 =0.545, p<0.001), HDL-C (b =-0.350, R2 =0.484, p<0.001), TC (b =0.256, R2 =0.508, p<0.001) and MAP (b =0.248, R2 =0.524, p<0.001).

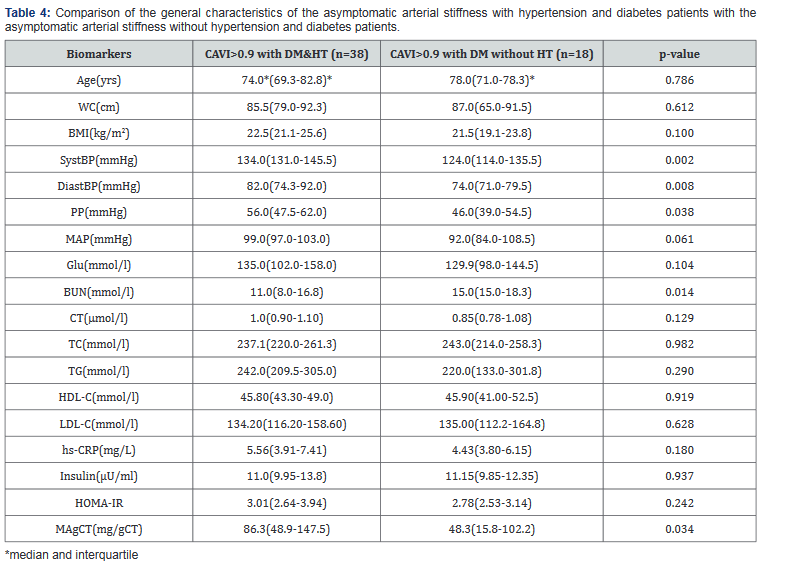
Of the 56 subjects of CAVI>0.9 and T2DM group, 38 had CAVI>0.9 with T2DM and HT and 18 had CAVI>0.9 with T2DM and without HT. The CAVI>0.9 with T2DM and HT group had significantly higher SystBP, DiastBP, PP, BUN and MA than the CAVI>0.9 withT2DM without HT group (p<0.05) as in Table 4. However, both groups had increased Age, BMI, WC, MAP, TC, TG, LDL-C, hs-CRP, insulin and IR levels. The subjects of CAVI>0.9 and T2DM with HT group may have more severe symptoms than the arterial stiffness without HT and DM group, but they all have the same risk for cardiovascular events.
Discussion
Our finding suggests that atherogenic factors such as hypertension, history of diabetes, dyslipidemia, inflammation and insulin resistance, may contribute to the acceleration of arterial stiffening associated with visceral adiposity or obesity. Diabetes, hypertension and aging are associated with accelerated the risk of atherosclerosis disease [11,18-20], which can cause vessel wall structural changes [21] as early initiators of atherosclerosis and manifested as increased arterial stiffness [18]. Arterial stiffness measurement can use as a marker of prognostic and diagnostic of coronary heart disease [18,19]. The CAVI can demonstrate the reflection of arterial compliance. Atherosclerosis may cause from the atheroma formation in the tunica intima (elastin and collagenous tissue) and/or wall hardening in the tunica media (smooth muscles of the artery). Intimal deformation induces plaque rupture and is closely associated with CVD development. Medial hardening reduces arterial compliance, has effects on hemodynamics and atherosclerosis progression. The vascular endothelium cells play the pivotal role in the vascular tone regulation by vasoactive factors releasing to regulate and response for the composition maintenance of the normal arterial wall. Vascular endothelium cells dysfunction plays an important role in arterial stiffness and atherosclerosis. In the present study, subjects with arterial stiffness demonstrated significant increase of all risk factors of atherosclerosis and correlated with the atherosclerosis risk factors as in Table 1& Table 2. Data from the interview, over 90% of the study subjects have ever obese at the middle age.
Inflammation plays the major role and major component of atherosclerosis [22] and also has the important role in the large arteries stiffness [13] by changing in the arterial wall composition due to inflammatory cell infiltration or endothelial dysfunction [14]. Inflammation is a major contributor in atheroma formation, as the initiation step and proceeds with the progression and propagation of the disease [22]. Several cytokines in circulation such as IL-6 and TNF-a are elevated in patient with atherosclerosis, and correlated with disease burden. C-reactive protein levels also predict the risk of future cardiovascular events and CVD [23], and individuals with apparently healthy [24]. Many studies demonstrated the association between arterial stiffness and hs-CRP levels in healthy individuals and isolated systolic hypertension patients [24,25].
Overweight and obesity are associated with increased CVD risk factors such as dyslipidemia, hypertension and inflammation. It’s the adverse effect on blood vessels leading to arterial stiffness and premature vascular aging [26]. Many studies have demonstrated the association of obesity with impaired function of the large arteries, which might be the consequence of meta¬bolic dysregulation, inflammation, obstructive sleep apnea and other metabolic disorders [27]. The present study demonstrated the association between arterial stiffness and total cholesterol and HDL cholesterol levels. Patients with familial combined hyperlipidemia tend to have more arterial stiffness than normal controls [28]. Ferrier et al. [29] demonstrated that in patients with isolated systolic hypertension, arterial stiffness may be reduced by cholesterollowering treatment accompanied by increased HDL-C and reduced TC and LDL-C [30]. In our study, the significant association of TC, HDL-C with arterial stiffness was observed not only in bivariate but also in multivariate regression analysis. Thus research evidence supports the utility of arterial stiffness as a surrogate marker of end-organ damage in hypertension [3]. Age and MAP are the principal physiological contributors to increased arterial stiffness [31]. The MAP probably reflects to vascular changes independent of age and impact on the size of the smooth muscular arteries (small-to-medium), cause the higher peripheral vascular resistance, which can be analyzed by pulse wave reflections [32]. Despite the association of MAP levels [33], therapeutic with antihypertensive [34] and trends of the changing in blood pressure [35], there is still the greater association with arterial stiffness from HT in these subjects [36].
Many studies demonstrated that stiffening in elastic medium and large arteries are associated with cardiovascular risk factors such as HT, dyslipidemia, obesity, smoking, diabetes, and aging. These cardiovascular risk factors have also promoted atherosclerosis development [37,38]. Many studies have shown that endothelial dysfunction [39] and the risk of vascular events, (myocardial infarction, cerebral stroke, and peripheral arterial insufficiency) increases with increased arterial intima-media thickness (IMT) [40] and show good correlation with IMT and atherosclerosis [41].
Conclusion
CAVI was associated with the CVD risk factors and can be used as a CVD risk factor in T2DM patients. CAVI measurements a useful tool to assess arterial stiffness and evaluate for subclinical coronary atherosclerosis in asymptomatic patients.
Acknowledgement
We sincerely thank the Cardiovascular and Diabetes Prevention in Elderly Project of Faculty of Allied Health Sciences for financial support to this study. We wish to thank all coworkers for their blood collection and technical assistance. We particularly thank the patients who participated in this study and Asst. Prof. Dr. Ronald A. Markwardt, Burapha University, for his critical reading and correcting of the manuscript..
References
- Gu K, Cowie CC, Harris MI (1999) Diabetes and decline in heart disease mortality in US adults. JAMA 281(14): 1291-1297.
- Buja LM (2000) Atherosclerosis: pathologic anatomy. In: Willerson JT, Cohn JN (Eds.), Cardiovascular Medicine. Churchill Livingstone, New York, USA, pp. 1325-1335.
- Boutouyrie P, Tropeano AI, Asmar R, Gautier I, Benetos A, et al. (2002) Aortic stiffness is an independent predictor of primary coronary events in hypertensive patients: a longitudinal study. Hypertension 39(1): 10-15.
- Weber T, Auer J, O’Rourke MF, Kvas E, Lassnig E, et al. (2004) Arterial stiffness, wave reflections, and the risk of coronary artery disease. Circulation 109(2): 184-189.
- Shirai K, Utino J, Otsuka K, Takata M (2006) A novel blood pressure independent arterial wall stiffness parameter; Cardio-Ankle Vascular Index (CAVI). J Atheroscler Thromb 13(2): 101-107.
- Horinaka S, Yabe A, Yagi H, Ishimura K, Hara H, et al. (2009) Comparison of atherosclerotic indicators between cardio ankle vascular index and brachial ankle pulse wave velocity. Angiology 60(4): 468-476.
- Yambe T, Yoshizawa M, Saijo Y, Yamaguchi T, Shibata M, et al. (2004) Brachio-ankle pulse wave velocity and Cardio-Ankle Vascular Index (CAVI). Biomed Pharmacother 58(Suppl 1): S95-S98.
- Takaki A, Ogawa H, Wakeyama T, Iwami T, Kimura M, et al. (2008) Cardio-Ankle Vascular Index is superior to brachial-ankle pulse wave velocity as an index of arterial stiffness. Hypertens Res 31(7): 1347- 1355.
- Nakamura K, Tomaru T, Yamamura S, Miyashita Y, Shirai K, et al. (2008) Cardio-Ankle Vascular Index is a candidate predictor of coronary atherosclerosis. Circ J 72(4): 598-604.
- Kadota K, Takamura N, Aoyagi K, Yamasaki H, Usa T, et al. (2008) Availability of Cardio-Ankle Vascular Index (CAVI) as a screening tool for atherosclerosis. Circ J 72(2): 304-308.
- O’Rourke MF, Nichols WW (2005) Aortic diameter, aortic stiffness, and wave reflection increase with age and isolated systolic hypertension. Hypertension 45(4): 652-658.
- Zieman SJ, Melenovsky V, Kass DA (2005) Mechanisms, pathophysiology, and therapy of arterial stiffness. Arterioscler Thromb Vasc Biol 25(5): 932-943.
- McEniery CM, Spratt M, Munnery M, Yarnell J, Lowe GD, et al. (2010) An analysis of prospective risk factors for aortic stiffness in men: 20-year follow-up from the Caerphilly prospective study. Hypertension 56(1): 36-43.
- Wilkinson IB, McEniery CM (2004) Arterial stiffness, endothelial function and novel pharmacological approaches. Clin Exp Pharmacol Physiol 31(11): 795-799.
- Laurent S, Cockcroft J, Van Bortel L, Boutouyrie P, Giannattasio C, et al. (2006) Expert consensus document on arterial stiffness: methodological issues and clinical applications. Eur Heart J 27(21): 2588-2605.
- Domanski MJ, Mitchell GF, Norman JE, Exner DV, Pitt B, et al. (1999) Independent prognostic information provided by sphygmomanometrically determined pulse pressure and mean arterial pressure in patients with left ventricular dysfunction. J Am Coll Cardiol 33(4): 951-958.
- Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, et al. (1985) Homeostasis model assessment: insulin resistance and betacell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28(7): 412-419.
- O’Rourke MF, ManciaG (1999) Arterial stiffness. J Hypertens 17(1): 1-4.
- Laurent S, Boutouyrie P, Asmar R, Gautier I, Laloux B, et al. (2001) Aortic stiffness is an independent predictor of all-cause and cardiovascular mortality in hypertensive patients. Hypertension 37(5): 1236-1241.
- Beckman JA, Creager MA, Libby P (2002) Diabetes and atherosclerosis. Epidemiology, pathophysiology and management. JAMA 287(19): 2570-2581.
- Oliver JJ, Webb DJ (2003) Noninvasive assessment of arterial stiffness and risk of atherosclerotic events. Arterioscler Thromb Vasc Biol 23(4): 554-566.
- Libby P (2002) Inflammation in atherosclerosis. Nature 420(6917): 868-874.
- Liuzzo G, Biasucci LM, Gallimore JR, Grillo RL, Rebuzzi AG, et al. (1994) The prognostic value of C-reactive protein and serum amyloid a protein in severe unstable angina. N Engl J Med 331(7): 417-424.
- Ridker PM, Rifai N, Rose L, Buring JE, Cook NR (2002) Comparison of C-reactive protein and low-density lipoprotein cholesterol levels in the prediction of first cardiovascular events. N Engl J Med 347(20): 1557- 1565.
- Kampus P, Kals J, Ristimae T, Muda P, Ulst K, et al. (2007) Augmentation index and carotid intima-media thickness are differently related to age, C-reactive protein and oxidized low-density lipoprotein. J Hypertens 25(4): 819-825.
- Safar ME, Czernichow S, Blacher J (2006) Obesity, arterial stiffness, and cardiovascular risk. J Am Soc Nephrol 17(4Suppl 2): S109‑S111.
- Jelic S, Bartels MN, Mateika JH, Ngai P, DeMeersman RE, et al. (2002) Arterial stiffness increases during obstructive sleep apneas. Sleep 25(8): 850-855.
- Brouwers MC, Reesink KD, van Greevenbroek MM, Meinders JM, Schaper N, et al. (2009) Increased arterial stiffness in familial combined hyperlipidemia. J Hypertens 27(5):1009-1016.
- Ferrier KE, Muhlmann MH, Baguet JP, Cameron JD, Jennings GL, et al. (2002) Intensive cholesterol reduction lowers blood pressure and large artery stiffness in isolated systolic hypertension. J Am Coll Cardiol 39(6): 1020-1025.
- Yokoyama H, Kawasaki M, Ito Y, Minatoguchi S, Fujiwara H (2005) Effects of fluvastatin on the carotid arterial media as assessed by integrated backscatter ultrasound compared with pulse-wave velocity. J Am Coll Cardiol 46(11): 2031-2037.
- Armentano R, Megnien JL, Simon A, Bellenfant F, Barra J, et al. (1995) Effects of hypertension on viscoelasticity of carotid and femoral arteriesin humans. Hypertension 26(1): 48-54.
- McEniery CM, Wilkinson IB, Avolio AP (2007) Age, hypertension and arterial function. Clin Exp Pharmacol Physiol 34: 665-671.
- Khattar RS, Swales JD, Senior R, Lahiri A (2000) Racial variation in cardiovascular morbidity and mortality in essential hypertension. Heart 83(3): 267-271.
- Bhopal R, Hayes L, White M, Unwin N, Harland J, et al. (2002) Ethnic and socio-economic inequalities in coronary heart disease, diabetes and risk factors in Europeans and South Asians. J Public Health Med 24(2): 95-105.
- Lyratzopoulos G, McElduff P, Heller RF, Hanily M, Lewis PS (2005) Comparative levels and time trends in blood pressure, total cholesterol, body mass index and smoking among Caucasian and South-Asian participants of a UK primary-care based cardiovascular risk factor screening programme. BMC Public Health 5: 125.
- Patel JV, Lim HS, Gunarathne A, Tracey I, Durrington PN, et al. (2008) Ethnic differences in myocardial infarction in patients with hypertension: effects of diabetes mellitus. QJM 101(3): 231-236.
- Scuteri A, Najjar SS, Muller DC, Andres R, Hougaku H, et al. (2004) Metabolic syndrome amplifies the age-associated increases in vascular thickness and stiffness. J Am Coll Cardiol 43(8): 1388-1395.
- Sharrett AR, Ding J, Criqui MH, Saad MF, Liu K, et al. (2006) Smoking, diabetes, and blood cholesterol differ in their associations with subclinical atherosclerosis: the Multiethnic Study of Atherosclerosis (MESA). Atherosclerosis 186(2): 441-447.
- Ja¨rvisalo MJ, Lehtima¨ki T, Raitakari OT (2004) Determinants of arterial nitrate-mediated dilatation in children: role of oxidized lowdensity lipoprotein, endothelial function, and carotid intima-media thickness. Circulation 109: 2885-2889.
- Lorenz MW, Markus HS, Bots ML, Rosvall M, Sitzer M (2007) Prediction of clinical cardiovascular events with carotid intima-media thickness: a systematic review and meta-analysis. Circulation 115(4): 459-467.
- Tanriverdi H, Evrengul H, Kara CO, Kuru O, Tanriverdi S, et al. (2006) Aortic stiffness, flow-mediated dilatation and carotid intima-media thickness in obstructive sleep apnea: non-invasive indicators of atherosclerosis. Respiration 73(6): 741-750.






























