Useful Measures to Assess Glucose Dynamics from Continuous Glucose Monitoring Data
Kohnert KD* and Salzsieder E
Division of Clinical Research, Institute of Diabetes Gerhardt Katsch, Germany
Submission: March 28, 2017; Published: April 12, 2017
*Corresponding author: Kohnert KD, Division of Clinical Research, Institute of Diabetes Gerhardt Katsch, Karlsburg, Germany, Tel: 49 -0- 38355-68406; Email: kohnert@diabetes-karlsburg.de
How to cite this article: Kohnert K, Salzsieder E. Useful Measures to Assess Glucose Dynamics from Continuous Glucose Monitoring Data. Curre Res Diabetes & Obes J. 2017; 1(5): 555574. DOI: 10.19080/CRDOJ.2017.01.555574
Abstract
Blood glucose control is a fundamental element in preventing micro- and macrovascular complications of diabetes. Many patients with diabetes can improve glycemic control by use of continuous glucose monitoring. Analysis of continuously monitored data further allows glucose dysregulation to reveal early in the development of diabetes or the metabolic syndrome and response to therapeutic intervention. But traditional measures of glycemia currently used in clinical practice are insufficient to characterize the various aspects of glucose profile complexity that may be important in different states of dysglycemia and the design of optimal antidiabetes therapy. This article discusses several dynamical complexity measures, their association with metabolic characteristics, and possible implications for glucose control.
Abbreviations: CGM: Continuous Glucose Monitoring; DFA: Detrended Fluctuation Analysis; PCP: Poincare Plots; SFE: Shape of the Fitting Ellipse; AFE: New Metrics Area; ApEn: Approximate Entropy; SampEn: Sample Entropy; MSE: Multiscale Entropy
Introduction
Although useful in clinical practice, metrics proposed for continuous glucose monitoring (CGM) in diabetes control [1] such as mean glucose, SD of glucose, and time or percentage of time spent in hyper- and hypoglycemia, are not appropriate to reveal the internal dynamics of CGM time series. These metrics are derived from linear models of glucose analysis and mostly fail to characterize glucose dynamics [2]. As a matter of fact, the variation of blood glucose levels is not linear, and glucose profiles contain nonlinear, non-stationary components [3]. Several nonlinear analytical methods have been recently applied to quantify the complexity of blood glucose signals, including detrended fluctuation analysis (DFA) [4-11], Poincare plots (PCP) and various entropy measures [12-15].
These studies show that the complexity of blood glucose variations is lower in patients with diabetes as compared with nondiabetic subjects. Beyond traditional estimates of glycemia and glycemic variability, complexity measures target the glucoregulatory system and have the potential to assess how treatment modalities can modify the dynamics of glucose. The clinical importance of the current dynamical measures, however, it is not yet clear. And it is neither known whether the complexity measures are affected by the diabetes therapy nor have correlations been examined between various dynamical indices.
Quantification of Glucose Dynamics and Association with Glycemic Control Measures
Table1 summarizes the measures of glucose dynamics recently used in studies on hyperglycemia.
Detrended fluctuation analysis
The Detrended fluctuation analysis (DFA) is useful to assess long-range correlations in time series. The use of this method yields the DFA scaling exponent α that reflects the degree of complexity. Alpha values <1.5 indicate long-range negatively and α>1.5 positively correlated fluctuation. Churruca et al. [4] and Ogata et al. [5] reported a loss of complexity in glucose profiles of patients with the metabolic syndrome and with diabetes, e.g., and the scaling exponents α were higher than in healthy persons. An exploratory study conducted by Khovanova et al. [6] and Thomas et al. [7] provided a dynamical definition of glycemic stability and supported the potential of DFA for time series analysis in diabetic patients.
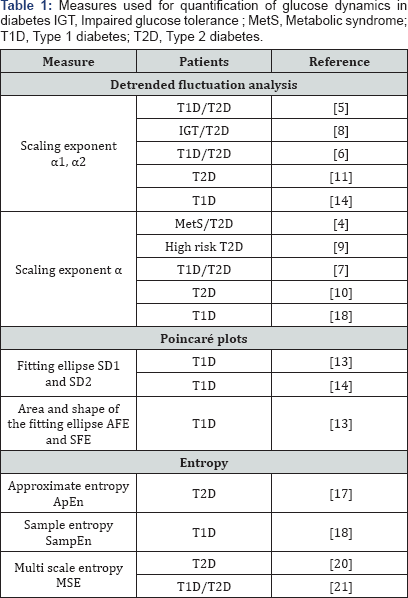
Yamamoto et al. [8] showed that the loss of glucose profile complexity stretched from the short-range (α1) to the long-range scaling exponent (α2) with the worsening glycemia. The results of these studies led to the conclusion that changes in glucose dynamics may already occur before full-blown hyperglycemia develops. Indeed, Varela's group [9,10] has recently demonstrated that DFA α was capable of indicating the risk of developing T2D. But it remains unknown whether dynamical alterations are primarily dependent on exogenous factors, e.g., antihyperglycemics, or endogenous factors such as the residual β-cell function and insulin resistance. A retrospective analysis by Kohnert et al. [11] in patients with T2D found that the loss of dynamical complexity relates to the decline in β-cell reserve and increasing glycemic variability.
Poincaré plot analysis
The standard oincaré plot (PCP) used to visualize the non-linear pattern of glucose dynamics is a scattergram constructed by locating data points from the CGM time series [12,13]. Quantification of the plots is done using SD1 and SD2 statistics, where the minor axis (SD1) comprises the data points perpendicular to the line of identity and SD2 those dispersed along the major axis of the fitting ellipse. Crenier [13] has validated these standard measures for the geometry of PCP in T1D and introduced the new metrics area (AFE) and shape of the fitting ellipse (SFE). As he reported, all the metrics of the PCP geometry were higher in diabetic subjects than in the healthy control group. Worthy of note, these parameters decreased upon continuous subcutaneous insulin infusion therapy, indicating that they were modifiable by exogenous factors. More recently, Garcia Maset et al. [14] have reported that the loss of complexity in glucose time series of pediatric patients with T1D, measured as DFA α and PCP parameters, was correlated with increased glycemic variability.
Entropy measures
In addition to the previous algorithms the complexity of glucose time series can be accessed through several other methods, including approximate entropy (ApEn) and sample entropy (SampEn) [15,16]. Both require for computation the three parameters length of the data segment (m), similarity criterion (r) and length of the data (N). However, a consensus is still lacking about the proper selection of the parameters. Using ApEn, Lytrivi & Crenier [17] found an increase of glucose profile complexity in T1D upon switching therapy from multiple daily insulin injections to continuous subcutaneous insulin infusion. The ApEn increase was inversely related with the DFA exponent α and reduced glycemic variability, indicating that therapy closer to physiological insulin secretion can improve glycemic complexity. SampEn is a modification of ApEn but has the advantage of being less sensitive to the step length within the time series [15]. Regarding the analysis of glucose complexity, studies on patients with T1D indicated that SampEn was associated with insulin resistance and with time in hypoglycemia (<3.9 mol/l) [18].
Since its introduction [19] multiscale entropy (MSE) represents the predominating method to characterize the complexity of physiological signals [19]. The MSE approach is based on sample entropy computation over a range of timescales and has been proposed for assessment of glucose dynamics. Costa et al. [20] introduced the term "dynamical geometry" to set the framework for the analysis of blood glucose time series using computation methods. Using the MSE approach, recent studies have shown that the temporal structure of glucose fluctuations is more complex in nondiabetic subjects than in those with T2D. One study reported a significant correlation between the MSE index with conventional measures of glycemia [20], i.e., glycated hemoglobin A1c (HbA1c) and mean blood glucose. Chen et al. [21] identified no such correlations in their study, including a mixed cohort of T1D and T2D patients. In a preliminary investigation, we observed no significant association of the MSE index with mean glucose and merely a weak correlation with HbA1c (Kohnert et al. unpublished).
Modifiable and Non-Modifiable Factors Associated with Glucose Dynamics
The internal structures of CGM time series imply a complex dynamic process regulated by a set of interactions between various hormones and metabolic components. And an important question is whether individuals within a subgroup of diabetes have their dynamic structure. As shown by Rahaghi et al. [22] and confirmed in several studies, time-scale analysis does not reveal major differences between diabetes types, even though they do show lower glucose complexity compared with nondiabetic subjects. Insulin resistance is expected to lower glucose dynamics, especially in T2D patients not treated with insulin injections, and significant correlation between insulin resistance and body mass index has been reported [18] However, insulin sensitivity is modifiable either by physical exercise or administration of insulin sensitizers. Other modifiable factors include meal intake and enteral feeding, which alter the individual dynamic signature [22]. Presumably, any therapy, approaching the physiological glucose regulation, is capable of shifting the glucose dynamics toward more healthy conditions, as demonstrated in patients with T1D when switching the therapy from daily insulin injections to insulin infusion [17]. Assessing the effects of non-modifiable factors such as age and diabetes duration is complicated by the fact that close correlations exist between aging, obesity, and insulin resistance.
Implications for Diabetes Control
Whereas the glycemic variability characterizes the magnitude of the time series, the structural variability defines the complexity of the time series. Both parameters represent two complementary categories [23]. Despite the paucity of clinical data, the use of measures of blood glucose dynamics appears to have a considerable potential in diabetes control. Current study results consistently show that glucose complexity decreases during progression from early glucoregulatory dysfunctions to established diabetes. Thus, glucose series complexity measures may be both beneficial in detecting the malfunctions in the glucoregulatory system before the blood glucose reaches pathological levels [9] and in characterizing the diabetes stability after therapeutic intervention. The influence of antidiabetic agents is, however, as yet unknown.
Clinical studies will need to be conducted to clarify whether therapy modalities, beyond merely normalizing blood glucose levels can reverse multiscale dynamics toward those of nondiabetic subjects. Consequently, the glucoregulatory system could be the future target for individualized diabetes therapy to restore healthy multiscale dynamics. Furthermore, it remains an important goal to find out whether glucose complexity measures are useful as markers for the predisposition or the development of late diabetes complications. Dynamic indices, measuring glucose dynamics on different time scales, e.g., multiscale entropy, appear to be preferable for diabetes control. There is no doubt that blood glucose dynamics is a fundamental concept in the future management of diabetes.
References
- Bergenstal RM, Ahman AJ, Bailey T, Beck RW, Bissen J, et al. (2013) Recommendations for standardizing glucose reporting and analysis to optimize clinical decision making in diabetes: the Ambulatory Glucose Profile (AGP) Diabetes Technol Ther 15(3): 198-211.
- Bremer T, Gough DA (1999) Is blood glucose predictable from previous values? A solicitation for data. Diabetes 48(3): 445-451.
- Wilson T, Holt T, Greenhalgh T (2001) Complexity and clinical care BMJ 323(7314): 685-688.
- Churruca J, Virgil L, Luna E, Ruiz-GJ, Varela M (2008) The route to diabetes: Loss of complexity in the glycemic profile from health through the metabolic syndrome to type 2 Diabetes Metab Syndr Obes 1: 3-11.
- Ogata H, Tokuyama K, Nagasaka S, Ando A, Kusaka I et al. (2006) Long- range negative correlations of glucose dynamics in humans and its breakdown in diabetes mellitus. Metabolism Am J Physiol Regul Integr Comp Physiol 291(6): R1638-R1643.
- Khovanova NA, Khovanov IA, Shabno L, Griffiths F, Holt TA (2013) Characterisation of linear predictability and non-stationarity of subcutaneous glucose profiles. Comput Methods Programs Biomed 110(3): 260-267.
- Thomas F, Signal M, Chase JG (2015) Using continuous glucose monitoring data and detrended fluctuation analysis to determine patient condition: a Review J Diabetes Sci Technol 9(6): 1327-1335.
- Yamamoto N, Kubo Y, Ishizawa K, Kim G, Moriya T, et al. (2010) Detrended fluctuation analysis is considered to be useful as a new indicator for short-term glucose complexity. Diabetes Technol Ther 12(10): 775-783.
- Varela M, Vigil L, Rodriguez C, Vargas B, Garcia-Carretero (2016) Delay in the detrended fluctuation analysis crossover point as a risk factor for type 2 diabetes mellitus. J Diab Res 2016.
- Rodriguez de Castro C, Virgil L, Vargas B, Garcia Delgado E, Garcia Carretero R, et al. (2017) Glucose time series complexity as a predictor of type 2 diabetes. Diabetes Metab Res Rev 33(2): e2831.
- Kohnert KD, Heinke P, Vogt L, Augstein P, Salzsieder E (2014) Declining R-cell function is associated with the lack of long-range negative correlation in glucose dynamics and increased glycemic variability: A retrospective analysis in patients with type 2 diabetes. J Clin Transl Endocrinol 1(4): 192-199.
- Kovatchev BP, Clarke WL, Breton M, Brayman K, McCall A (2005) Quantifying temporal glucose variability in diabetes via continuous glucose monitoring: mathematical methods and clinical application. Diabetes Technol Ther 7(6): 849-862.
- Crenier L (2014) Poincare plot quantification for assessing glucose variability from continuous glucose monitoring systems and a new risk marker for hypoglycemia: application to type 1 diabetes patients switching to continuous subcutaneous insulin infusion. Diabetes Technol Ther 16(4): 247-254.
- Garcia Maset L, Gonzales LB, FurquetGl, Suay FM, Marco RH (2016) Study of glycemic variability through time series analyses (detrended fluctuation analysis and Poincare plotI in children and adolescents with type 1 diabetes. Diabetes Technol Ther 18(11) 719-724.
- Yentes M, Hunt N, Schmidt KK, Kaipust JP, McGrath D et al. (2013) Sample entropy outperforms approximate entropy with small data sets. www.asbweb.org/conference/2013/abstracts/39.
- Humeau-HA (2015) The multiscale entropy algorithm and its variants: a review. Entropy 17(5): 3110-3123.
- Lytrivi M, Crenier L (2014) Glucose variability outcome for type 1 diabetic patients switching to CSII: improved complexity patterns beyond glucose dispersion reduction. European Association for the Study of Diabetes (EASD) 50th Ann Meeting, September 15-119, 2014, Vienna Austria, Abstract t 1004.
- Crenier L, Lytrivi M, Van Dalem A, Keymeulen B, Corvilain B (2016) Glucose complexity estimates insulin resistance in either nondiabetic individuals or in type 1 diabetes. J Clin Endocrinol Metab 101(4): 1490-1497.
- Costa M, Goldberger AL, Peng CK (2002) Multiscale analysis of complex physiologic time series. Phys Rev Lett. 89(6): 068102.
- Costa MD, Henriques T, Munshi MN, Segal AR, Goldberger A (2014) Dynamical glucometry: use of multiscale entropy analysis in diabetes. Chaos 24(3): 033139.
- Chen JL, Chen PF, Wang HM (2014) Decreased complexity of glucose dynamics in diabetes: evidence from multiscale entropy analysis of continuous glucose monitoring system data. Am J Physiol Regul Integr Comp Physiol 307(2): R179-R183.
- Rahghi FN (2007) Human blood glucose dynamics. UC San Diego: Bioengeneering.b 6635674.
- Schubert P (2013) The application of nonlinear methods to characterize human variability from time series. German J Sports Med 64(5): 132 -140.






























