An in vitro cytotoxic activity of Bees’ honey in Huh-7 cell line
Arnab Banerjee1*, Debasmita Das2, Bithin Kumar Maji1 and Sandip Mukherjee1
1Department of Physiology, Serampore College, Serampore, Hooghly, West Bengal, India
2Bengal Institute of Pharmaceutical Sciences (BIPS), Kalyani, Nadia, West Bengal, India
Submission: December 24, 2017; Published: April 10, 2018
*Corresponding author: Arnab Banerjee, Research scholar, Department of Physiology, Serampore College, Serampore, Hooghly-712201, West Bengal, India.
How to cite this article: Arnab Banerjee, Debasmita Das, Bithin Kumar Maji, Sandip Mukherjee. An in vitro cytotoxic activity of Bees’ honey in Huh-7 cell line. Adv Biotech & Micro. 2018; 9(1): 555751. DOI: 10.19080/AIBM.2018.09.555751
Abstract
Hepatocellular carcinoma (HCC) is one of the major health problem and deadliest disease worldwide. The cure rate is very low especially among patients who are ineligible for surgical or percutaneous methods. HCC is generally viewed as a chemotherapy-resistant blight. The major phytoconstitutent chrysin present in Bee’s honey (BH), which is in charge of number of exercises and one of them is anticancer and antiproliferative effects against various tumors. The present study was planned to assess the anticancer potential of Bee’s honey against Huh-7, human liver cancer cells. The cytotoxic impact of BH on Huh-7 cells was tried by MTT examine. Cells were tested with 15%, 20%, 25% concentrate of BH for 48 hours. The apoptotic genes Bax and Bcl-2 expression were assessed by utilizing RT-PCR, and after that decide the protein expression of caspase 3 and 9 by western blot. Comet assay was performed in Huh-7 cells for the DNA damage and apoptosis. Cell viability investigation uncovered the cytotoxic impact of BH on Huh-7cell line. RT-PCR of anti- apoptotic Bcl-2 and pro-apoptotic Bax demonstrated that there is down and up control, individually, of these two qualities with an unfriendly impact. Additionally, BH treatment set off the actuation of caspase-9 followed by caspase-3 protein expression. Also, BH incited DNA damage and apoptosis on Huh-7 cells as uncovered by Comet tail length. Bees’ honey has antiproliferative and cytotoxic impression on Huh-7 cells, presumably through apoptosis induction.
Keywords: honey; Hepatocellular carcinoma; Huh-7; Cytotoxicity; Apoptosis
Abbrevations: HCC: Hepatocellular carcinoma; BH: Bee’s Honey; PHC: Primary Hepatic Carcinoma; GAPDH: Glyceraldehyde- 3-Phosphate Dehydrogenase; SCGE: Single Cell Gel Electrophoresis; PT: Permeability Transition; ApAF-1: Apoptosis-Activating Factor 1
Introduction
Hepatocellular carcinoma (HCC) is one of the major health problem and deadliest disease worldwide. The cure rate is very low especially among patients who are ineligible for surgical or percutaneous methods. HCC is generally viewed as a chemotherapy-resistant blight with abdominal mass, abdominal pain, emesis, anemia, back pain, jaundice, itching, weight loss and fever [1]. Primary hepatic carcinoma (PHC) is very common cancers in the five malignant tumors of the world, which has unobvious pathogenesis, rapid progression and is mostly in late stage at clinical diagnosis [2]. Recent evidences have confirmed that tumor development is closely related to the abnormal cell apoptosis of tumor, the broken balance between cell proliferation and apoptosis is an important factor in tumorigenesis [3-5]. The effective and established chemotherapeutic agents for HCC are currently unavailable and its recurrence rate is high, the prognosis of HCC is still poor. Surgical elimination is often considered as a treatment of choice in patients with non-cirrhotic livers. As liver is often detected in late stage, less than 30-40% of patients only are eligible for surgery and transplant. Percutaneous removal is just the non-surgical system that can offer cure. Liver is not tolerant to radiation and in this way radiotherapy is not in the slightest degree a possibility for hepatocellular carcinoma quiet. Globally, the rates of death are higher in males than in females. East and South-East Asia and Middle and Western Africa have higher number of liver cancer cases, whereas South-Central and Western Asia, as well as Northern and Eastern Europe have comparatively lower number of such cases. Among primary liver cancers, hepatocellular carcinoma (HCC) speaks to the major histological subtype, accounting for 70% to 85% of the total liver cancer burden worldwide.
Cholangiocarcinomas that arise primarily from the epithelial lining of the bile duct (intra and extra hepatic bile duct) are relatively rare, but high incidence rates are found in Thailand and other parts of Eastern Asia largely due to the elevated prevalence of liver fluke infection [6]. Cirrhosis due to either hepatitis B, hepatitis C, or alcohol is the significant reason of hepatocellular carcinoma. Currently two types of apoptosisrelated genes are found, which are promoting apoptosis and inhibition of apoptosis genes [7]. In these genes Bcl-2 and Bax have the most closely related to apoptosis [8]. Natural product as Bee’s honey with antioxidant, chemopreventive, antiatherogenic, immunoregulatory, antimicrobial and wound healing properties [9-16]. There are verious signaling pathways such as stimulation of TNF- release, inhibition of cell proliferation, induction of apoptosis and cell cycle arrest, as well as inhibition of lipoprotein oxidation, mediates the beneficial effects exerted by honey and its major components such as chrysin and other flavonoids [17]. Photochemical screening revealed that BH contains a variety of biologically active compounds such as flavonoids, vitamins, anti-oxidants as well as hydrogen peroxides (H2O2) [18]. In cell culture accumulation of H2O2 as free radicals, results in oxidative stress, which is followed by lipid peroxidation in the cellular membrane and ultimately leads to cellular damage and can promote peroxide-induced cell death [19]. It is reported that Tualang honey is able to inhibit the growth of oral squamous cell carcinoma and osteosarcoma cells [20].
Greek honeys with high amount of phenolic compounds modulate oestrogenic activity whereas a thyme honey-enriched diet may prevent cancer-related processes in breast, and prostate and endometrial cancer cells [21]. BH has hepatoprotective effect on acute liver injuries induced by metanil-yellow in vivo, and the effect of BH against metanil yellow induced liver damage is related to its antioxidant/anti-inflammatory properties which attenuate the activation of NF-𝜅B and its controlled genes like TNF-𝛼 and IL-1𝛽 [22]. Since there are very limited studies on the effects of Honey of Bee on Liver cancer protection and the side effects of current cancer therapies are major.In this stage, the present study was proposed to evaluate the apoptosis induction potential of BH with three different concentrations in Huh-7 cell line and also to find its possible mechanism of action. So, the present study may be our initiation for the future drug against human hepatocellular carcinoma.
Material and Methods
Preparation of bee honey
There are two reasons for selecting BH, first is the synergism between the different chemical constituents in honey has been reported; and there are a number of volatile compounds reported to be abundant in honey products that might be lost during simple fractionation methods. Bees’ honey (BH) was purchased from the local market and diluted with DMEM to obtain 15%, 20% and 25% solution concentrations. Artificial honey was prepared to exclude the effect of honey osmolarity and acidity on the Huh7 cells [15].
Cell cultures
The Huh-7 cells were grown in Dulbecco’s Modified Eagle medium (Gibco, USA) supplemented with 10% of fetal bovine serum, 100 IU/ ml penicillin and 100μg/ml of streptomycin (Gibco, USA) using 25 cm2 flasks (Nunc, Denmark) in a CO2 incubator (Thermo scientific) at 37 °C.
MTT assay
Trypan blue was utilized to determine the viability of cells. Exponentially growing cells were harvested, counted by hemocytometer and diluted with medium, yielding a concentration of 1×105 cells/ml. From this cell suspension, 100μl was pipetted into 96-well microtiter plates (Nunc, Denmark) and incubated for 24 hours in a 5% CO2 incubator (Heal Force) at 37 °C. Cells were then treated with 15%, 20% and 25% solution concentrations. After adding the extract samples, new medium was added to make the final volume of 200μl per well. The plate was then incubated in the 5% CO2 incubator at 37 °C for 48 hours. At the end of treatment time, the medium was replaced by 100μl MTT (Sigma, St Louis, MO) per well and incubated for another 4 hours at 37 °C. The reaction was stopped by adding 100μl DMSO, AR grade (Sigma) to each well to dissolve the purple-blue MTT formazan precipitate. Finally, the absorbance was read with the ELISA reader (LX-800) and calculates the percentage of viable and non-viable cells.
RNA extraction and reverse transcriptase polymerase chain reaction (RT-PCR)
Total RNA was extracted using TriZol reagent (Invitrogen, Carlsbad, CA). Oligo (dT)-primed RNA (1μg) was reverse transcribed using the Super Script II transcriptase kit (RR047A, Takara, Shiga, Japan) according to the manufacturer’s instructions. cDNA obtained was amplified by PCR with Taq DNA polymerase (Fermentas, Burlington, Canada). The presence of possible target genes Bax and Bcl-2 were determined using the obtained cDNA and glyceraldehyde- 3-phosphate dehydrogenase (GAPDH) as the internal control (Table 1). The PCR products were then resolved on 1% agarose gel. The bands were identified based on the product size using DNA ladder. Gel images were analyzed by scanning densitometry (Image J, NIH) and values were normalized to quantity of GAPDH and presented as percentage mRNA relative to control.

Western blot analysis
Protein was extracted by Minute TM Total Protein Extraction Kit (For Animal Cultured Cells and Tissues). To detect changes in apoptosis-related proteins (Caspase 3 and Caspase 9), Huh 7 cells (1 × 106 cells) seeded on a 10cm2 dish were treated with BH at different concentrations. The cells were harvested and lysed using a lysis buffer, and the protein concentrations of the cell lysates were quantified with a protein assay kit. The equivalent of 20-50μg of total proteins were separated on 10% SDS-PAGE, then electroblotted on PVDF membrane; the membranes were blocked with 10% non-fat dry milk in TBS-T overnight at 4 °C, then incubated with primary antibodies against with caspase 3 and caspase 9, β-actin (1:1000) for 1.5 h at room temperature. After a wash with TBS-T, membranes were incubated with HRPconjugated secondary antibodies (dilution 1:1,000) for 1 h at room temperature. Finally, the protein-antibody complexes were visualized by chemiluminescence (ECL system, Pierce). Protein expression was normalized to that of β-actin.
Comet assay
0.025% Trypsin was added to the cells after removing media. At that time point, the cells were kept at 37 °C for 5 minutes to detach cells. Clear window frosted microscopic slide was coated with 1% normal melting agarose. The well was punched at one side of the slide the aliquot of 10μl cell suspensions (~10,000 cells in 10μl or less volume) was added and to this 20μl of lysis solution was added followed by another coat of 0.5% of low melting agarose at 37 °C the slide was placed in moisture chamber for overnight. After lysis, the slide was immersed in the freshly prepared electrophoresis buffer (10 mM Tris, 0.08 mM Boric acid, 0.5 M EDTA, pH 8.2) and electrophoresed for 1h at 50V after electrophoresis slide was dehydrated by immersing in absolute alcohol for 3-5 times. Then slide was stained with ethidium bromide followed by observation under transilluminator for comet formation and image was photographed and analyzed.
Statistical analysis
Data were expressed as Mean±SE. Kruskal-Wallis non parametric one-way analysis of variance (ANOVA) test was performed to find whether or not scores of different groups differ significantly and to test intergroup significant difference, Mann-Whitney U multiple comparison tests was performed by using Stat-Direct Software (UK). Differences were considered significant if P<0.05.
Results
Effects of BH on the viability of Huh-7 cells were examined by the MTT assay. The results revealed that various concentrations of BH could strongly control cell growth. MTT assay showed a significant dose dependent decrease in cell viability of Huh-7 cell line. Cell viability diminished from 100% to 74.6%, 58.6%, and 28.4% as the dose of BH was increased from 0% to 15% (P<0.05), 20% (P<0.01) to 25% mg/ml (P<0.01), respectively trace the fact that the cellular structure was gradually reduced with the increasing concentration of the BH (Figure 1). With contrast to the control, the number of living cells decreased over incubation time and with increasing BH concentrations. Therefore, treatment with low concentrations for longer treatment times could result in cytotoxic effect on Huh-7 cells with contrast to higher concentrations for shorter treatment times. This upshot clearly shows that the inhibitory effect of BH on Huh-7 cell growth was exerted in a time and concentration dependent manner.

According to various genotoxicologist comet assay is a very common and also well utilized system for the estimation of apoptosis, DNA fragmentation and also DNA damage. In the present study, BH prompted DNA damage of Huh-7 cells with enlarge length of comet tail were likewise examined and analyzed (Figures 2 & 3). Exceptionally huge dose dependent DNA damage was observed after treatment of Huh-7 with BH. After effects of this study detected that Huh-7 cells demonstrated most surprising comet tail length when these cells were treated with 25% BH. To resolve the molecular mechanisms of BH induced apoptosis in Huh-7 cells, the expression levels of several apoptosis-related genes and proteins were inspected. Results revealed that increasing concentration of BH down regulated the anti-apoptotic Bcl-2 gene as evidenced by dose dependent decreased mRNA expression (Figure 4). Whereas, dose dependent up regulation of mRNA expression of pro-apoptotic Bax gene (Figure 5) was observed when Huh-7 cells are treated with the increasing concentration of BH (15%, 20%, 25%). The endogenous control GAPDH gave strong positive bands. Further, the protein expression levels of caspase 3 and caspase 9 were verified by western blotting. Both, caspase-3 and caspases-9 were found to be activated in Huh 7 cells when they were treated with BH. This activation were also found to be dose dependent as protein expression of caspases-3 and caspases-9 were increased consistently with the increasing dose of BH (15%, 20%, 25%) (Figure 6 & 7).
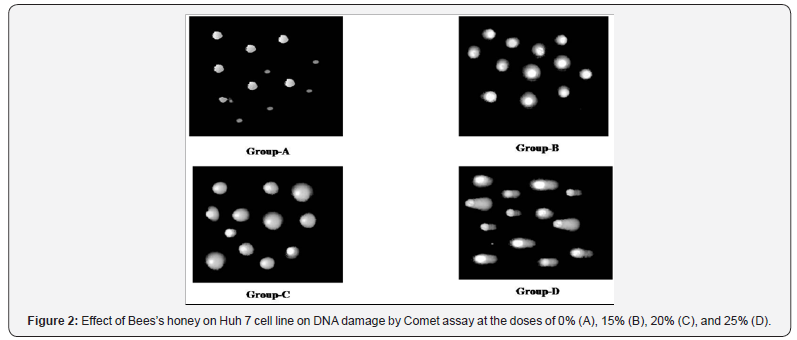
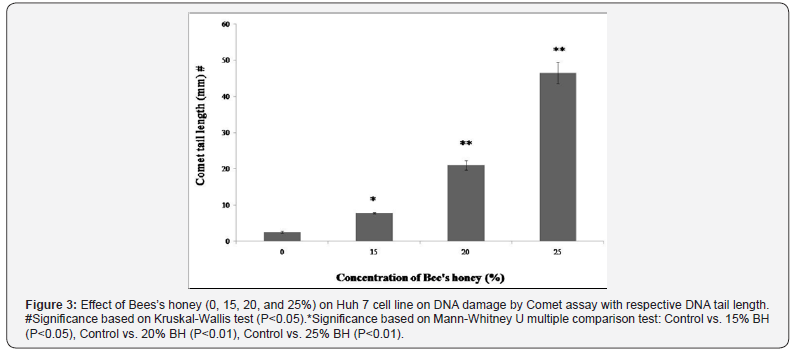


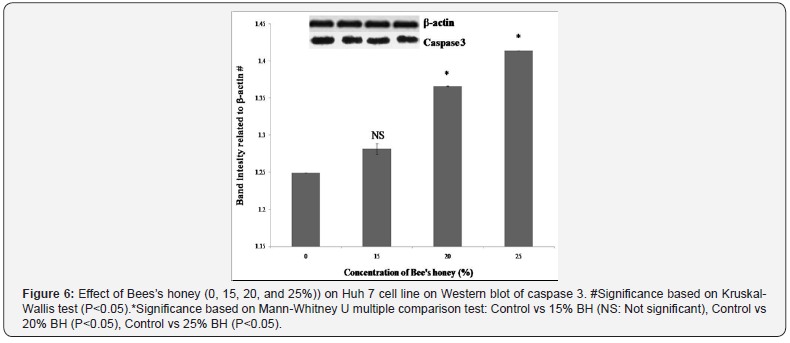
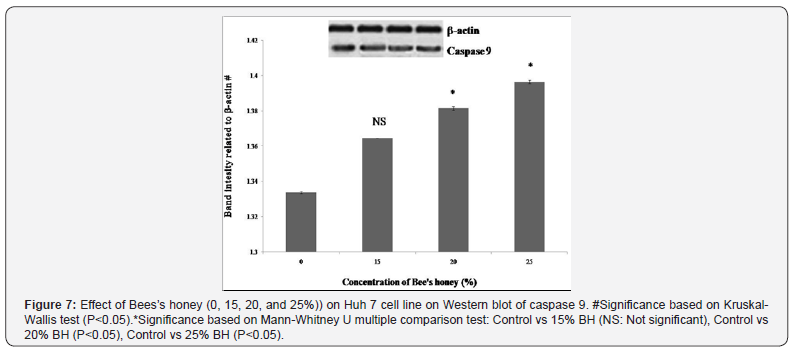
Discussion
In the present era traditional medicines are used for its multidrug resistance property, have taken a key role in the treatment of various diseases. Bioactive compounds present in BH have the potential to be used as medications by providing novel and new products for the prevention and treatment of various diseases. Such medications are now emerging as target for the researchers to find a cure for diseases including various cancers [23]. The present study limns the possibility of BH as a source of anti-cancer drug. BH was endeavored for its cytotoxic and apoptotic properties in Huh-7 cells in vitro. Previous studies have reported that BH is selectively toxic against various types of cancerous cells without harming healthy cells [24]. Results of MTT assay gives the confirmation of cytotoxic effects of BH on the Huh-7 human liver cancer cells in the present study. Percentage cell viability of Huh-7 cell lines was carried out by using Trypan blue dye exclusion technique. The viable cells at concentration of 15% BH are 74.6%; 58.6% viable cells at 20% BH concentration and at concentration of 25% BH, the viability of the cells are 28.4%.
It indicates that BH has cytotoxic activity in Huh-7 cancer cell lines in a dose dependent manner. Apoptosis surmises a very important role in the cancer prevention. A cell fails to undergo apoptosis by means of mutation or biochemical hindrance, can keep dividing and develop to become a tumor [25]. In this way, we primarily chose to test the anti-cancer effects of BH by the mechanism of apoptosis in Huh-7 cells. The single cell gel electrophoresis (SCGE) or comet assay combines for detecting DNA single strand breaks, alkali-labile sites and cross linking with the single cell approach typical of cytogenetic assays of DNA damage. The present study revealed that, treatment of Huh-7 cells with BH was resulted in increased comet parameters like comet tail length, thereby indicating that level of the DNA damage increased in a dose-dependent manner.
Bcl-2 is astonishing for promulgate its anti-apoptotic role in various cell types. It has been found that in radiation or chemotherapy Bcl2 play a crucial role in resistance of cancer cells [26]. Antiapoptotic Bcl2 is now a focal point for novel anticancer therapeutics as a result of its commitment to cancer development and progression by promoting cell survival. On the other hand, Bax is pro-apoptotic in nature and is scattered over many tissues of human body. Bax can proffer apoptotic signal to a cell by the formation homodimer. It seems to be, by the competing way Bcl-2 form heterodimers with Bax and can inhibit the development of apoptosis [27]. In the present study, the average band intensity of Bcl-2 was lower in Huh-7 cells boosted with 15% BH than the cells without supplementation. Conversely, BH dose dependently promotes Bax expression in Huh-7 cells. The molecular mechanism of proliferation and apoptosis has been found to be related with the development of hepatocellular cancer, highlighting the reduction of apoptosis as a major underpinning factor. In the present study, it is evident that BH has encouraged the Bax upregulation concurrently with Bcl-2 down regulation in a dose dependent manner in Huh-7 cells. Therefore, these results are well in line with the earlier report that there are upregulation of pro-apoptotic proteins Bax and Bak and down regulation of anti-apoptotic protein Bcl2 in cell lines receiving apoptosis [28]. In this way, we postulate that BH regulate balance between Bcl2 and Bax expression and induces apoptosis in Huh-7 human liver cancer cells in the present study.
Caspases are considered for to be act as a key mediator of apoptosis. Caspase-3 is a frequently activated death protease, catalyzing the specific cleavage of many key cellular proteins. Activation of caspase-3 may occur by both mitochondrial cytochrome c release and caspase-9 dependent or independent pathway. Hitherto, two significant groups of caspases have been distinguished and are thought to be involved in apoptosis pathways, including executioners or effectors (caspase-3, -6, -7) and initiators (caspase-2, -8, -9, -10) [29].
Among the executioner caspases, caspase-3 is considered to be the most crucial one which is activated by any of the initiator caspases (caspase-8, caspase-9, or caspase-10) [30]. Further, caspase-3 is accounted for to be essential for apoptotic chromatin condensation and DNA fragmentation in all cell types examined. These makes the caspases-3 a significant controller of certain processes associated with disassembles of the cell and the formation of apoptotic bodies. But, it may also function before or at the stage when allegiance to loss of cell viability is made [31]. Magnificent proof is there that disturbance of mitochondrial functions as evident by loss of trans membrane potential, permeability transition (PT) and release of cytochrome c leading to impaired electron transport are considered as the important event in many apoptotic cell deaths [32]. The release of cytochrome c actuated by a variety of death stimuli results in the activation of a complex of apoptosis-activating factor 1 (Apaf-1) and caspase-9, leading to the cytochrome c-dependent processing of pro-caspase-3 and apoptosis in a number of scenarios. Intrinsic and extrinsic pathways are activated by their own initiator caspases, namely caspase -9 and-8 respectively. The activation of initiator caspases will, in turn, leads to activation of executioner caspases. Caspase-3 is considered as the most denigrate of killer caspases. The execution pathway leads to cellular structural changes, such as chromatin condensation, cell shrinkage, and formation of apoptotic bodies followed by phagocytosis of the apoptotic cell [33]. In the present study, we investigated whether different concentrations of BH can activate the caspases in Huh-7 human liver cancer cells. The activity of both caspase-9 and caspase-3 were radically lifted when Huh- 7 cells were treated 15%, 20%, 25% of BH, suggesting that the BH has induced apoptosis typically via mitochondrial-mediated intrinsic pathway.
The western blot analysis of caspases likewise demonstrated that the protein expression of cleaved caspase-3 and -9 were dose-dependently increased upon treatment with the BH. In the present study, the caspase 3 and caspase 9 expressions were increased significantly in Huh-7 cells treated with graded increasing doses of the BH. This result proposes the involvement of caspase cascade in BH mediated apoptosis in Huh-7 cells. Taking everything into account, results of this study demonstrate that the Bee’s honey have good potential for use as cancer chemotherapeutic agent. Furthermore, our data indicate that the BH specifically dilute viability of hepatocellular carcinoma cell lines possibly through G0/G1 or S phase arrest or possibly via induction of sub-G0/G1 DNA fragmentation. It seems to be, the mechanism of the action is still ongoing. From the present study we can conclude that Bee’s honey is an effective agent, inhibiting the growth of Huh-7 cells through several possible mechanisms. These results are promising and also challenging in the field of cancer prevention and treatment. But, further investigations are needed to elucidate the exact biochemical and molecular mechanisms of cell death by BH and to confirm its role as candidates for human liver cancer prevention and here is our initiation for the future drug.
Acknowledgement
Authors are really grateful to Prof. Dhrubajyoti Chattopadhyay, Vice Chancellor of Amity University, Kolkata, for his continuous encouragement and valuable suggestions. We are thankful to Department of Physiology (UG & PG), Department of Zoology (UG & PG), Serampore College, Bengal Institute of Pharmaceutical Sciences (BIPS), Kalyani for their help during this study.
References
- Bishayee A, Politis T, Darvesh AS (2010) Resveratrol in the chemoprevention and treatment of hepatocellular carcinoma. Cancer Treat Rev 36(1): 43-53.
- Zubaidah RM, Tan GS, Tan SB, Lim SG, Lin Q, et al. (2008) 2-D DIGE profiling of hepatocellular carcino-ma tissues identified isoforms of far upstream binding protein (FUBP) as novel candidates in liver carcinogenesis. Proteomics 8(23-24): 5086-5096.
- Chen GG, Lai PB, Chak EC, Xu H, Lee KM, et al. (2001) Immunohistochemical analysis of pro-apoptotic Bid level in chronic hepatitis, hepatocellular carcinoma and liver metastases. Cancer Lett 172(1): 75-82.
- Kitamura T, Watanabe S, Sato N (1998) Liver regeneration, liver cancers and cyclins. J Gastroenterol Hepatol 13(S1): S96-S99.
- Cheng EH, Kirsch DG, Clem RJ, Ravi R, Kastan MB, et al. (1997) Conversion of Bcl-2 to a Bax-like death effector by caspases. Science 278(5345): 1966-1968.
- Jemal A, Bray F, Center MM, Ferlay J, Ward E, et al. (2011) Global cancer statistics. CA Cancer J Clin 61(2): 69-90.
- Cleveland JL, Ihle JN (1995) Contenders in FasL/TNF death signaling. Cell 81(4): 479-82.
- Wyllie AH (1980) Glucocorticoid-induced thymocyte apoptosis is associated with endogenous endonuclease activation. Nature 284(5756): 555-556.
- Al-Waili NS, Haq A (2004) Effect of honey on antibody production against thymus-dependent and thymus-independent antigens in primary and secondary immune responses. J Med Food 7(4): 491-494.
- French VM, Cooper RA, Molan PC (2005) The antibacterial activity of honey against coagulase-negative Staphylococci. J Antimicrob Chemother 56(1): 228-231.
- Gheldof N, Engeseth NJ (2002) Antioxidant capacity of honeys from various floral sources based on the determination of oxygen radical absorbance capacity and inhibition of in vitro lipoprotein oxidation in human serum samples. J Agric Food Chem 50(10): 3050-3055.
- Mabrouk GM, Moselhy SS, Zohny SF, Ali EM, Helal TE, et al. (2002) Inhibition of methylnitrosourea (MNU) induced oxidative stress and carcinogenesis by orally administered bee honey and Nigella grains in Sprague Dawely rats. J Exp Clin Cancer Res 21(3): 341–346.
- Molan PC (2006) The evidence supporting the use of honey as a wound dressing. Int J Low Extrem Wounds 5(2):122.
- Schramm DD, Karim M, Schrader HR, Holt RR, Cardetti M, et al. (2003) Honey with high levels of antioxidants can provide protection to healthy human subjects. J Agric Food Chem 51(6): 1732-1735.
- Swellam T, Miyanaga N, Onozawa M, Hattori K, Kawai K, et al. (2003) Antineoplastic activity of honey in an experimental bladder cancer implantation model: In vivo and in vitro studies. Int J Urol 10(4): 213- 219.
- Tonks A, Cooper RA, Price AJ, Olan PC, Jones KP (2001) Stimulation of TNF-alpha release in monocytes by honey. Cytokine 14(4): 240-242.
- Woo KJ, Jeong YJ, Park JW, Kwon TK (2004) Chrysin-induced apoptosis is mediated through caspase activation and Akt inactivation in U937 leukemia cells. Biochem Biophys Res Commun 325: 1215-1222.
- Mohammadzadeh S, Sharriatpanahi M, Hamedi M, Amanzadeh Y, Ebrahimi SSE, et al. (2007) Antioxidant power of Iranian propolis extract. Food Chem 103(3): 729-733.
- Linden A, Gulden M, Martin HJ, Maser E, Seibert H (2008) Peroxideinduced cell death and lipid peroxidation in C6 glioma cells. Toxicol In Vitro 22(5):1371-1376.
- Ghashm AA, Othman NH, Khattak MN, Ismail NM, Saini R (2010) Antiproliferative effect of Tualang honey on oral squamous cell carcinoma and osteosarcoma cell lines. BMC Complementary and Alternative Medicine 10(49): 1-8.
- Tsiapara AV, Jaakkola M, Chinou I, Graikou K, Tolonen T, et al. (2009) Bioactivity of greek honey extracts on breast cancer (MCF-7), prostate cancer (PC-3) and endometrial cancer (Ishikawa) cells: Profile analysis of extracts. Food Chemistry 116(3): 702-708.
- Al-Malki AL, Sayed AAR (2013) Bees’ honey attenuation of metanilyellow- induced hepatotoxicity in rats. Evid Based Complement Alternat Med 2013: 614580.
- Sharmila K, Padma PR (2013) Anticancer activity of Artemisia vulgaris on hepatocellular carcinoma (HepG2) cells. Int J Pharmacy and Pharmaceutical Sci 5: 479-483.
- Pongsathon Premratanachai, Chanpen Chanchao (2014) Review of the anticancer activities of bee products. Asian Pac J Trop Biomed 4(5): 337-344.
- Williams NG, Roberts TM (1994) Signal transduction pathways involving the Raf proto-oncogene. Cancer Metastasis Rev 13(1): 105- 116.
- Terada T, Nakanuma Y (1996) Expression of apoptosis, proliferating cell nuclear antigen, and apoptosis-related antigens (bcl-2, cmyc, Fas, Lewis(y) and p53) in human cholangiocarcinomas and hepatocellular carcinomas. Pathol Int 46(10): 764-770.
- Naseri MH, Mahdavi M, Davoodi J, Tackallou HS, Goudarzvand M, et al. (2015) Up regulation of Bax and down regulation of Bcl2 during 3-NC mediated apoptosis in human cancer cells. Cancer Cell Int 15: 55.
- Hill MM, Adrain C, Duriez PJ, Creagh EM, Martin SJ (2004) Analysis of the composition, assembly kinetics and activity of native Apaf-1 apoptosomes. EMBO J 23(10): 2134-2145.
- Elmore S (2007) Apoptosis: A review of programmed cell death. Toxicol Pathol 35(4): 495-516.
- Porter A, Janlcke R (1999) Emerging roles of caspase-3 in apoptosis. Cell Death Differ 6(2): 99-104.
- Kluck RM, Martin SJ, Hoffman BM, Zho JS, Green DR, et al. (1997) Cytochrome c activation of CPP32-like proteolysis plays a critical role in a Xenopus cell-free apoptosis system. EMBO J 16(15): 4639-4649.
- Delgado ME, Olsson M, Lincoln FA, Zhivotovsky B, Rehm M (2013) Determining the contributions of caspase-2, caspase-8 and effector caspases to intracellular VDVADase activities during apoptosis initiation and execution. Biochim Biophys Acta 1833(10): 2279-2292.
- Banerjee A, Sengupta A, Maji B, Nandi A, Pal S, et al. (2017) Possible cytotoxic activity of Annona muricata Leaves in Huh-7 Human Liver Cancer Cells. Hepatol Pancreat Sci 1: 104.






























