Copazan Herbal gel and Wound Healing in vitro: Assessment of the Functional Biomaterial for Veterinary Application
Victoria Tamara Perchyonok*
VTPCHEM PTY LTD, Australia
Submission: December 20, 2017; Published: February 16, 2018
*Corresponding author: JVictoria Tamara Perchyonok, VTPCHEM PTY LTD, Glenhuntly, 3163, Australia.
How to cite this article: Victoria Tamara Perchyonok. Copazan Herbal gel and Wound Healing in vitro: Assessment of the Functional Biomaterial for Veterinary Application. Adv Biotech & Micro. 2018; 8(4): 555744. DOI: 10.19080/AIBM.2018.08.555744
Abstract
Objective: To evaluate performance of Copazan Herbal Gel in wound healing process in vitro as well as to evaluate the free radical scavenging properties of the new materials, microbiological and bioadhesive capacity of the Copazan Herbal Gel and the individual components of the gel.
Methods: Bio-adhesive studies, microbiology and free radical defense capacity were investigated in order to assess the suitability of these designer materials.
Results:The Copazan Herbal Gel showed a high adhesive force and were only slightly swollen in the aqueous medium. All the test samples gave an average inhibition zone against Staphylococcus aureus larger than the chlorhexidine gluconate control disc. The hydrogels also had significant free radical defense capability.
Conclusion:In this study we demonstrated that the Copazan Herbal Gel are suitable bio-active material capable of efficiently counteracting potential free radical damage generated during the wound healing treatment in vitro.
Keywords: Copazan herbal gel; Topical application; Chitosan hydrogels; Antimicrobial systems; Wound healing; Free radical
Introduction
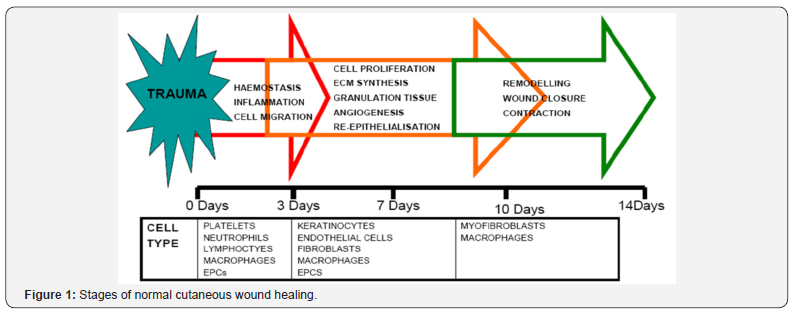
Wound healing is a specific biological process related to the general phenomenon of growth and tissue regeneration [1]. Wound healing progresses through a series of interdependent and overlapping stages in which a variety of cellular and matrix components act together to reestablish the integrity of damaged tissue and replacement of lost tissue [2](Figure 1). Recently, much attention has been given to the use of chitosan in veterinary applications, as a wound healing agent, antimicrobial agent, bandage material, skin grafting template, hemostatic agent and drug delivery vehicle. In this section, various investigations carried out recently on veterinary applications of chitosan are discussed as well as their potential uses in veterinary medicine. The chemical structure of chitosan, which is similar to that of hyaluronic acid, suggested that it would act as a wound healing agent [3]. Mechanisms of acceleration of wound healing have been investigated in vitro and in vivo by several groups [4-6]. Since 1985, Minami and his group have been developing chitin and chitosan products for veterinary use and they have reported their clinical effects on wound healing and their biological activities [7]. They found these polymers and products to accelerate wound healing, decrease treatment frequency, and give comfortable and painless wound surface protection. In addition to accelerating wound healing, chitosan is also capable of activating host defenses to prevent infection, thereby offering an alternative to the use of antibiotics. Chitosan enhances the functions of inflammatory cells such as polymorphonuclear leukocytes (PMN) (phagocytosis, production of osteopontin and leukotriene B4), macrophages (phagocytosis, production of interleukin (IL-1), transforming growth factor h1 and platelet-derived growth factor). As a result, chitosan promotes granulation and organization. Therefore, it is beneficial for healing of large open wounds in animals [8]. One of the most interesting effects of chitin and chitosan on wound healing is formation of granulation tissue with angiogenesis. Chitin and chitosan induce fibroblasts to release interleukin-8, which is involved in migration and proliferation of fibroblasts and vascular endothelial cells.
Copaiba oleoresin is widely used as a popular medicine, through topical and oral administration. It has various ethnopharmacological indications, including: wounds, asthma, as an antiseptic for wounds, skin ulcers, aching joints, ovarian cysts, uterine myoma, weak uterus, vaginal discharge, ovarian problem, ulcers, sore throat, uterine infections, general inflammations, as a tonic and to treat ulcers and other digestive diseases, and cancer, and leishmanioses [9]. Copazan HerbalGelR is a medical grade isotonic hydrogel made from high molecular– weight biopolymer that promotes wound healing in oral cavity, dry mouth management and to help reinforce your mouth’s own defense system. Additional benefits of the natural oils such as oleo di copaiba, calendula oil and aloe vera gel provide the additional anti-inflammatory, pain-management and wound healing properties to address all aspects of healing and wound management as well as providing additional benefits of natural oils. The main objective is to evaluate the Copazan HerbalGelR as a non-adhesive wound dressing through evaluation of evaluation of bioadhesion, inhibition zone and assessment of free radical capacity defense properties in vitro.
Methods
Microbiology investigation of the Copazan Herbal gel and individual components of the gel
A type strain of Staphylococcus aureus (ATCC 12600), obtained from the American Type Culture Collection (Manassas, USA) was used as test bacterium for estimating the antibacterial activity of the hydrogels. The antibacterial activity of the prepared chitosan hydrogels were tested using the standard Kirby-Bauer agar disc diffusion method [10]. Five to 6mm deep Muller-Hinton agar (Oxoid, Basingstoke, UK) plates were inoculated by streaking a standardized inoculum suspension that match a 0.5 McFarland standard and containing 107-108 colony forming units/ml with a throat cotton swab. For each test sample 500μg of hydrogel was applied to a 6mm diameter paper disc. The paper discs were placed on the inoculated Muller- Hinton agar medium and incubated at 37 °C for 24 hours. The diameter of the zones of growth inhibition was measured with a caliper. Each measurement was done in triplicate and the testing of each sample was repeated 3 times. The antibacterial efficacy of the prepared gels were compared to antibiotic sensitivity discs (Mast Laboratories, Merseyside UL) containing 25μg of chlohexadine gluconate per disc.
Bioadhesive investigation
Bioadhesion studies were done using Chatillon apparatus for force measurement [11]. This method determines the maximum force and work needed to separate two surfaces in intimate contact. The Copazan Herbal Gel and combination of the starting materials (0.1g) were homogeneously spread on a 1cm2 glass disk and then the disks were fixed to the support of the tensile strength tester using double side adhesive. The gel was brought into contact with the commercially available band aid, in order to simulate the skin attachment or the contact with slice of pork skin was established in order to imitate adhesion of the gel to the oral mucosa structure, after a preset contact time (1min) under contact strength (0.5N) the 2 surfaces were separated at a constant rate of displacement (1mm/s). The strength was recorded as a function of the displacement, which allowed to determine the maximal detachment force, Fmax, and the work of adhesion, W, which was calculated from the area under the strength-displacement curve.
Free radical scavenging ability
We adopted the method of Kyselovaet al to test for radical scavenging ability of the new hydrogels. This method records changes in water solubility of the model protein bovine serum albumin (BSA) exposed to free radicals generated by the Fenton reaction system [12]. The incubation mixtures contained the following reagents: bovine serum albumin (0.8mg/ml), phosphate buffer, pH 7.4 (10mM), EDTA (4.8mM), Fe(NH4)2(SO4)2 (4mM), ascorbate (4mM) and H2O2 (0.2%) in water to reach a total volume of 2.5ml. The hydrogels were added and the reaction mixture was incubated for 20min at room temperature.After completion of the reaction, the mixture was centrifuged at 3500rpm for 10min. The supernatant was precipitated with an equal volume of trichloroacetic acid (10%) at 0 °C. The precipitate thus obtained was re-dissolved in 1ml of Na2CO3 (10%) in NaOH (0.5M) and the final volume made up to 2.5ml by adding distilled water. An aliquot of the solution was used for protein determination using the method of Lowry et al. [13].
Statistics: The Student’s T-test was used to analyze the data.
Free radical scavenging ability
We adopted the method of Kyselova et al. [14] to test for radical scavenging ability of the new hydrogels. This method records changes in water solubility of the model protein bovine serum albumin (BSA) exposed to free radicals generated by the Fenton reaction system [14]. The incubation mixtures contained the following reagents: bovine serum albumin (0.8mg/ml), phosphate buffer, pH 7.4 (10mM), EDTA (4.8mM), Fe(NH4)2(SO4)2 (4mM), ascorbate (4mM) and H2O2 (0.2%) in water to reach a total volume of 2.5ml. The hydrogels were added and the reaction mixture was incubated for 20min at room temperature. After completion of the reaction, the mixture was centrifuged at 3500rpm for 10min. The supernatant was precipitated with an equal volume of trichloroacetic acid (10%) at 0 °C. The precipitate thus obtained was redissolved in 1ml of Na2CO3 (10%) in NaOH (0.5M) and the final volume made up to 2.5ml by adding distilled water. An aliquot of the solution was used for protein determination using the method of Lowry et al.
Statistics: The Student’s T-test was used to analyze the data.
Results and Discussion
Bio-adhesion in vitro model and Copazan herbal gel
High adhesiveness of the gels is desired to maintain an intimate contact with the tooth structure. The chitosan hydrogels showed a high adhesive force and work of adhesion. This can be expected due to the well-known intrinsic bio-adhesive properties of chitosan. The adequate water absorption capacity together with the cationic nature [15] which promotes binding to the negative surface of the dentin structure can also explain these results. According to Caffaggi, hydration of the polymer causes mobilization of the polymer chains and hence influences polymeric adhesion [16]. Appropriate swelling is important to guarantee adhesiveness; however, over hydration can form slippery non-adhesive hydrogels [3]. The correlation between the force and work of adhesion is noticeable in all the hydrogels (Figure 2).
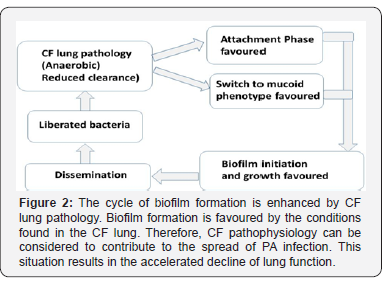
Microbiology of Copazan herbal gel and individual components
All the test samples gave an average inhibition zone larger than the chlorhexidine gluconatecontrol disc, thereby confirming the antibacterial activity of the Copazan Herbal Gel and individual combinations against Staphylococcus aureus (Figure 3). There was a significant difference between the rest of the samples when compared to each other and the positive control. Chlorhexidine is a broad-spectrum biocide effective against Gram-positive bacteria, Gram-negative bacteria and fungi [4]. Chlorhexidine inactivates microorganisms with a broader spectrum than other antimicrobials (e.g. antibiotics) and has a quicker kill rate than other antimicrobials (e.g. povidone-iodine) [5]. It has both bacteriostatic (inhibits bacterial growth) and bactericidal (kills bacteria) mechanisms of action, depending on its concentration. Chlorhexidine kills by disrupting the cell membrane [6]. Upon application in vitro, chlorhexidine can kill nearly 100% of Gram-positive and Gram-negative bacteria within 30 seconds [7]. Since chlorhexidine formulations can destroy the majority of categories of microbes, there is limited risk for the development of an opportunistic infections [8].
A number of mechanisms explaining the antimicrobial activity of chitosan have been postulated [17]. One of the proposed mechanisms (is that the cross-linker moieties incorporated into hydrophilic chitosan increase their solubility and ease of penetration of the hydrogels into the cells of microorganisms. The chitosan then binds to microbial DNA, inhibits the transformation of mRNA and protein synthesis, and thereby inhibits metabolism [18]. Another suggested antibacterial mechanism of chitosan is the interaction between positively charged protonated NH3+ groups of the chitosan molecules and negatively charged microbial cell surfaces. The electrostatic interaction results in changes in the properties of the cell wall permeability with leakage of the intracellular electrolytes causing internal osmotic imbalance that inhibit the growth of the microorganisms [19]. The antimicrobial activity of copaiba oils was tested against Gram-positive and Gramnegative bacteria, yeast, and dermatophytes. Oils obtained from Copaifera martii, Copaifera officinalis, and Copaifera reticulata (collected in the state of Acre) were active against Gram-positive species (Staphylococcus aureus, methicillin-resistant S. aureus, Staphylococcus epidermidis, Bacillus subtilis, and Enterococcus faecalis) with minimum inhibitory concentrations ranging from 31.3-62.5μg/ml. The oils showed bactericidal activity, decreasing the viability of these Gram-positive bacteria within 3h [20]. The antibacterial and antimicrobial properties of Calendula oil are well documented the and support the traditional use of the plant in the therapy of bacterial infection [20].
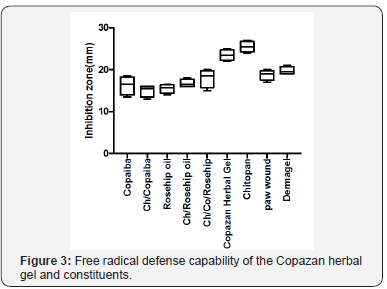
Free radical defense capability of the Copazan herbal gel and constituents
When wound occurs, it is generally accompanied by classical symptoms of inflammation, such as pain, redness and edema. In the inflammation stage, the main aim is the removal of debris, damage tissue, and bacteria by neutrophils and macrophages, which have a role in antimicrobial defense and debridement of devitalized tissue by production of proteolytic enzyme and reactive oxygen species. The amount of uncontrolled ROS is the main cause of the inability of healing process to continue and therefor it would be ideal to utilize the antioxidant capacity of the “designer hydrogels“ to detect and able to “fight the free radical excess” have been assessed using previously described model using well-established that HO radical can be generated from a reaction known as the biologic Fenton reaction and this reaction requires the presence of H2O2.
Chlorhexidine gluconate is an effective antimicrobial agent with potent antimicrobial and anti-inflammatory properties and has been widely used as an antiseptic agent for the irrigation of root canals as well as a medicament during root-canal treatment [1]. It has also been found that in combination with Ca(OH)2 and H2O2, CHX it is more effective against Streptococcus species and in eliminating Enterococcus faecalis from root canal infections [6,7]. However, recent reports suggested CHX as a potential genotoxic agent toward leucocytes, oral mucosal cells and lymphocytes and the use of CHX on its own, or in combination with these products may be potentially genotoxic and cause tissue damage [8]. The amount of uncontrolled ROS is the main cause of the inability of the healing process to continue and therefore it would be ideal to utilize the antioxidant capacity of the “designer hydrogels” to detect and fight the free radical excess.
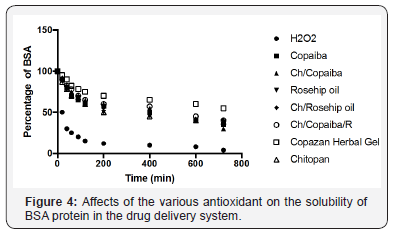
It is well established that HO• can be generated from a reaction known as the Fenton reactionin the presence of H2O2 [17,18] and the generation of HO• has been shown to be a critical factor in various ROS-induced oxidative stresses [19,20]. Protein cross-linking can be used as a model for detection of free radical activity [8]. Water soluble Bovine Serum Albumin (BSA) can be polymerized by hydroxyl radicals generated by the Fenton reaction system of Fe2+/EDTA/ H2O2/ascorbate [1]. As a result, the protein loses its water solubility and the polymerized product precipitates. The decrease in the concentration of the water-soluble protein can then be detected [9]. We reported earlier that protein cross-linking as a model for detection of free radical activity and activation of “molecular defense forces”. Bovine serum albumin (BSA), a completely water-soluble protein, was polymerized by hydroxyl radicals generated by the Fenton reaction system of Fe2+/EDTA/H2O2/ ascorbate [6-8]. As a result, the protein loses its water-solubility and the polymerized product precipitates. The decrease in the concentration of the water-soluble protein can easily be detected. We considered worthwhile to study the chitosan as a “build in defense mechanism” for the in-vitro generated free radical production and “site specific” in vitro model counter reaction of the hydrogel.
Therefore we adopted the method for recording changes in water solubility of the model protein bovine serum albumin (BSA) exposed to free radicals generated by an inorganic chemical system. As clearly demonstrated by the Figure 4, upon exposure to standard H2O2 in the form of Fe2+/EDTA/ H2O2/ascorbate solution as a base line determinate free radical generation under “prototype in-vitro free radical damage”, upon incorporation of the chitosan substituted hydrogels, the build in antioxidant capacity and there for free radical defense of the in-vitro model has been activated and are of significant value to take notice. This model represents the practical approach of insitu monitoring and test the amount of free radical production and synergistic antioxidant defense of the system (Figure 4) [21- 27].
Conclusion
Copazan Herbal GelR is a medical grade isotonic hydrogel made from high molecular-weight biopolymer that promotes wound healing in vitro. As part of in vitro evaluation of the Copazan HerbalGelR the the parameters such as bio adhesion, inhibition zone and assessment of free radical capacity defense properties of the materials were measure and potential clinical applications of this promising material are discussed.
References
- Spangberg L, Haapasalo M (2002) Rationale and efficacy of root canal medicaments and root filling materials with emphasis on treatment outcome. Endodontic Topics 2(1): 35-58.
- Portenier I, Waltimo T, Haapsalo M (2003) Enterococcus faecalis-the root canal survivor and ‘star’ in post treatment disease. Endodontic Topics 6(1): 135-159.
- Wehling J, Dringen R, Zare RN, Maas M, Rezwan K, et al. (2014) Bactericidal activity of partially oxidized nanodiamonds. ACS Nano 8(6): 6475-6483.
- Alacam A, Tulunoglu O, Oygur T, Bilici S (2000) Effects of topical Catalase application on dental pulp tissue: a histopathological evaluation. J Dent 28(5): 333-339.
- Chapple IL (1997) Reactive oxygen species and antioxidants in inflammatory diseases. J Clin Periodontol 24(5): 287-296.
- Steinberg D, Heling I, Daniel I, Ginsburg I (1999) Antibacterial synergistic effect of chlorhexidine and hydrogen peroxide against Streptococcus sobrinus, Streptococcus faecalis and Staphylococcus aureus. J Oral Rehabil 26(2): 151-156.
- Zerella JA, Fouad AF, Spangberg LS (2005) Effectiveness of a calcium hydroxide and chlorhexidine digluconate mixture as disinfectant during retreatment of failed endodontic cases. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 100(6): 756-761.
- Yeung SY, Huang CS, Chan CP, Lin CP, Lin HN, et al. (2007) Antioxidant and pro-oxidant properties of chlorhexidine and its interaction with calcium hydroxide solutions. Int Endod J 40(11): 837-844.
- KyselovaZ, Rackova L, Stefek M (2003) Pyridoindole antioxidant stobadine protected bovine serum albumin against the hydroxyl radical mediated cross-linking in vitro. Arch Gerontol Geriatr 36(3) 221-229.
- Cafaggi S, Leardi R, Parodi B, Caviglioli G, Russo E, et al. (2005) Preparation and evaluation of chitosan-poloxamer 407 based matrix for buccal drug delivery. J Control Release 102: 159-169
- Patel VF, Liu F, Brown MB (2011) Advances in oral transmucosal drug delivery. J Control Release 153(2): 106-116.
- Todar K (2012) Antimicrobial agents in the treatment of infectious disease. Online textbook of bacteriology, USA, pp.1-6.
- Barras A, Martin FA, Bande O, Baumann JS, Ghigo JM, et al. (2013) Glycan-functionalized diamond nanoparticles as potent E. coli antiadhesives. Nanoscale 5(6): 2307-2316.
- Kremer M (1999) Mechanism of the Fenton reaction. Evidence for a new intermediate. Phys ChemChem Phys 1(15): 3595-3605.
- Fenton H (1894) Oxidation of tartaric acid in presence of iron. J Chem Soc Trans 65: 899-911
- Yoshino F, Yoshida A, Okada E, Okada Y, Maehata Y et al. (2012) Dental resin curing blue light induced oxidative stress with reactive oxygen species production. Journal of Photochemistry and Photobiology B: Biology 114: 73-78.
- Halliwell B (1989) Free radicals, reactive oxygen species and human disease: a critical evaluation with special reference to atherosclerosis. Br J Exp Pathol 70(6): 737-757.
- Skucaite N, Peciuliene V, Vitkauskiene A, Machiulskiene V (2010) Susceptibility of endodontic pathogens to antibiotics in patients with symptomatic apical periodontitis. J Endod 36(10): 1611-1616.
- Hermann B (1920) Calcium hydroxidals mittelzurn, behandeln und fullen von Wurzelkanalen. Thesis, Wurzburg.
- Ashby M (2014) Reactive oxygen species and dental health. Systems Biology of Free Radicals and Antioxidants pp. 3873-3897.
- Battino M, Bullon P, Wilson M, Newman H (1999) Oxidative injury and inflammatory periodontal diseases: the challenge of anti-oxidants to free radicals and reactive oxygen species. Crit Rev Oral Biol Med 10(4): 458-476.
- Perchyonok V, Souza T, Felliti R, Zhang S, Grobler S. et al. (2015) Bio-Functional Nanodiamond Restorative Materials Containing Bio- Additives: In vitro Approach. OJST 5(5): 117-126.
- Park JH, SaravanakumarG, Kim K, Kwon IC (2010) Targeted delivery of low molecular drugs using chitosan and its derivatives. Adv Drug Deliv Rev 62(1): 28-41.
- Wu QX, Lin DQ, Yao SJ (2014) Design of chitosan and its water soluble derivatives-based drug carriers with polyelectrolyte complexes. Mar Drugs 12(12): 6236-3253.
- Bauer A, Kirby W, Sherris J, Turck M (1966) Antibiotic susceptibility testing by a standardized single disc method. Am J Clin Pathol 45(5): 493-496.
- Perchyonok V, Zhang S, Basson N, Grobler S, Oberholzer T, et al. (2014) Insights into functional tetracycline/antioxidant containing chitosan hydrogels as potential bio-active restorative materials: Structure, function and antimicrobial activity. OJST 4: 99-108.
- Mohamed N, Fahmy M (2012) Synthesis and antimicrobial activity of some novel cross-linked chitosan hydrogels. Int J Mol Sci 13(9): 11194- 11209.






























