Intracranial Extracerebral Neuroglial Heterotopia Herniating to Cleft Lip Through Large Frontoethmoidal Defect Associated with Periventricular Nodular Heterotopia: A Constellation of Abnormalities in a Child with FLNA mutation
Sara T Alharbi1, Kalthoum Tlili Graiess1* and Raniah Alqawahmed2
1Neuroradiology Section, Department of Radiology, Prince Sultan Military Medical City, Saudi Arabia
2Department of pathology, Prince Sultan Military Medical City, Saudi Arabia
Submission: April 01, 2023;Published: April 17, 2023
*Corresponding author: Kalthoum Tlili-Graiess, Neuroradiology Section, Department of Radiology, Prince Sultan Military Medical City Riyadh, Saudi Arabia
How to cite this article: Sara T A, Kalthoum Tlili G, Raniah A. Intracranial Extracerebral Neuroglial Heterotopia Herniating to Cleft Lip Through Large Frontoethmoidal Defect Associated with Periventricular Nodular Heterotopia: A Constellation of Abnormalities in a Child with FLNA Mutation. Theranostics Brain, Spine & Neural Disord. 2023; 4(4): 555644. DOI: 10.19080/JOJS.2023.04.555644
Abstract
Glioneuronal Heterotopias are rare congenital masses composed of disorganized neuroectodermal tissue of variable differentiation and found outside the central nervous system (CNS). They are frequently reported in the head and neck, commonly in the nasal region [1,2]. However, intracranial extracerebral glioneuronal heterotopia (IEGH) is extremely rare [3]. We report a case of IEGH at the anterior cranial fossa extending to cleft lip through a large frontoethmoidal defect and associated with FLNA related-periventricular nodular heterotopia (PVNH).
Keywords: Central nervous system; Neurons; Axons; Magnetic resonance imaging
Abbrevations: CNS: Central Nervous System; MRI: Magnetic Resonance Imaging; PVNH: Periventricular Nodular Heterotopia; IEGH: Intracranial Extracerebral Glioneuronal Heterotopia; GFAP: Glial Fibrillary Acid Protein
Case Description
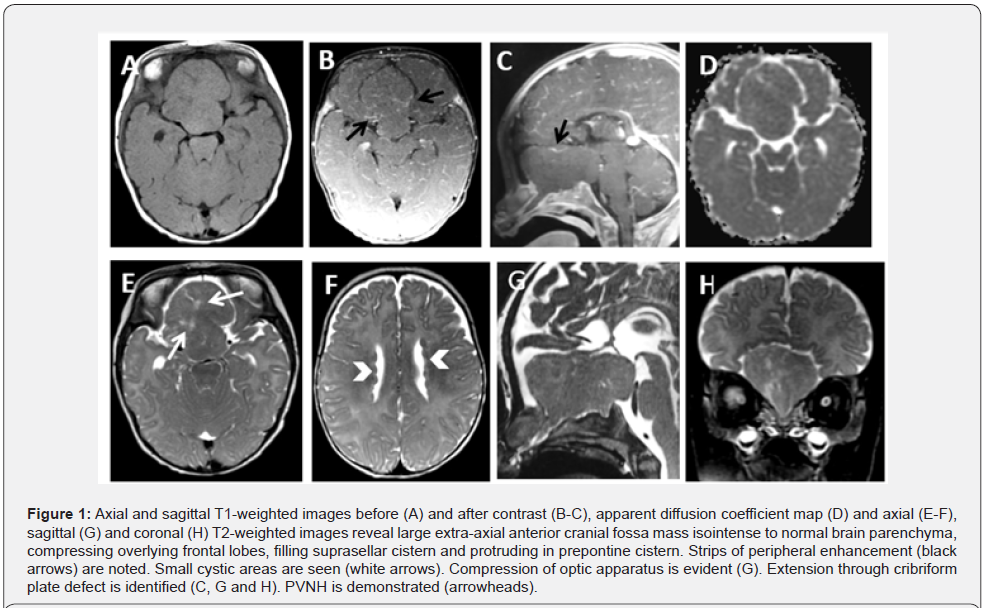
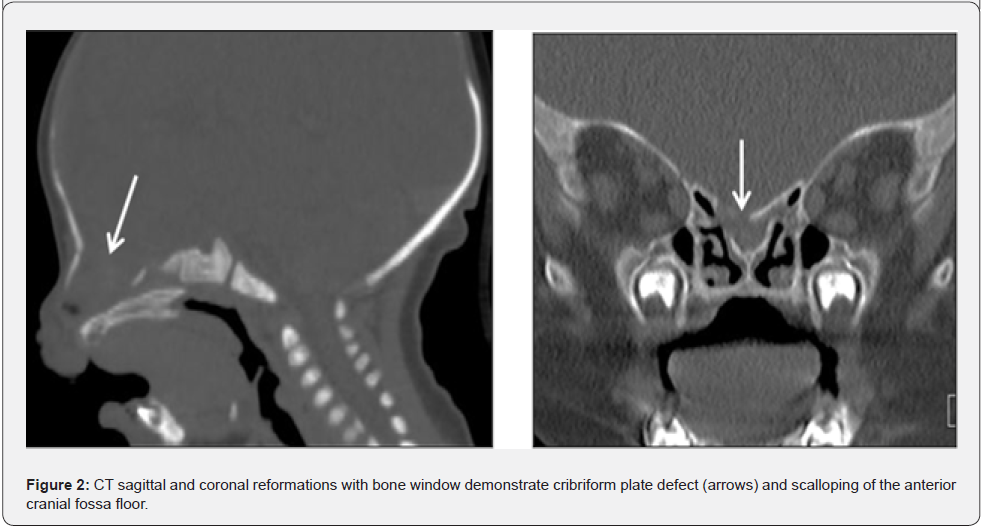
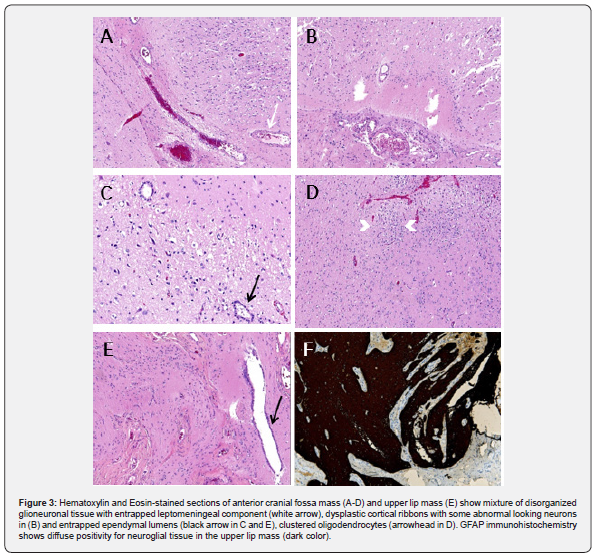
A 6-month-old girl was referred to the imaging department with the diagnosis of intracranial tumor and cleft lip. MRI (Figure 1) revealed a large well-circumscribed extra-axial mass at the floor of anterior cranial fossa herniating into cleft lip through cribriform plate defect. The mass was filling the suprasellar cistern. It showed signal intensity like brain parenchyma in all sequences with some internal linear T2-hyperintensities and strips of peripheral enhancement. Optic chiasm and cisternal segments of optic nerves were superiorly displaced and markedly thinned. Internal carotid arteries and anterior cerebral arteries were displaced. Subependymal nodules were identified along both lateral ventricles isointense to grey matter represented PVNH. Cribriform plate bone defect, cleft palate and scalloping of anterior cranial fossa floor were demonstrated on CT scan (Figure 2). At surgery, a whitish mass was found looking like brain tissue but with malformed configuration. The left optic nerve was displaced superolateral. Right optic nerve and optic chiasma were encased in some areas, difficult to be differentiated from the mass leading to residual part of the mass. Encased olfactory nerves were not saved. Histology (Figure 3) of the intracranial mass revealed mixture of glioneuronal tissues including mostly disorganized and occasionally frankly dysplastic cortical ribbons, some abnormal looking neurons, clustered oligodendrocytes, heterotopic neuronal clusters in white matter, trapped ependymal lumens and leptomeninges occupied by the neuroglial tissue with few microcalcifications. Immunostaining showed positivity for glial fibrillary acid protein (GFAP), low Synaptophysin. Ki-67 score and negative BRAF mutation testing. The upper lip mass displayed focally ulcerated squamosa mucosa with underlying fibrovascular stroma and mixture of glioneuronal tissue including ependymal lumens. Whole Exome Sequencing revealed mutation in the FLNA gene at chromosome Xq28.
Discussion
IEGHs are non-hereditary congenital masses composed of disorganized but mature glioneuronal elements. Choristoma, neuroglial hamartoma, neurogenic hamartoma, and accessory brain are other terms that have been applied to such masses [1,2]. IEGHs are extremely rare. To date, only 20 cases are found in the English literature [2]. Anterior cranial fossa, suprasellar cistern and middle cranial fossa are the most frequent reported locations [3]. Extension into extracranial sites through skull bases foramina or destructed skull base was reported in 70% of IEGH in middle and anterior cranial fossae [2]. Extension to nasopharynx through craniopharyngeal cleft was also reported. Craniofacial anomalies including clef lip and palate are commonly associated with IEGH [4].
To the best of our knowledge, we report the first case of IEGH herniating into cleft lip through a large cribriform plate defect and associated with FLNA- related PVNH. These multiple abnormalities signify early insult of fetal development and raises the query about the implication of FLNA mutation in IEGH development. FLNA gene likely acts early during development and is required for initial attachment of neurons onto the radial glial scaffolding before migration from the ventricular zone. Failure of migrating neurons to attach to radial glia is one mechanism leading to PVNH [5].
The widely known theories to explain IEGH pathogenesis are protrusion, detachment, and outpouching theories [6]. Moreover, migration theory states that embryonic neuroepithelial tissue aberrantly migrates into the subarachnoid space, differentiates at the 5th or 6th week of embryogenesis, and continues to grow resulting in heterotopia [7]. Because most IEGH are large at presentation, it is likely that the etiological event occurs early in gestation. Furthermore, due to frequent association with polymicrogyria or intracerebral neuronal heterotopia, IEGH may be closely related to migration disorder. However, the mechanism by which migrated tissue becomes well organized is unknown. Our findings support the migration theory and early development of IEGH. Differentiation of IEGH from congenital brain tumors is important and largely determines the extent of surgical resection. MRI is fundamental for accurate preoperative diagnosis, while CT is useful to detect bony scalloping and defects. IEGH is characteristically extra-axial and isointense to brain parenchyma. Unlike most brain tumors, IEGH shows minimal or absent enhancement and lacks aggressive features [5,6]. Interestingly, the herniated portion of IEGH in our case shows heterogenous and slightly higher T2 signal than the intracranial portion, like the nasopharyngeal portion in Kau et al. [4] cases.
Histologically, IEGH is composed of neuroectodermal tissue comprising variably differentiated neuroglial cells and features reminiscent of ependyma and choroid plexus. As noted in our case, IEGH is composed of mainly glial elements such as astrocytes and oligodendrocytes, with occasional neurons, axons, ependymal cells, and choroids plexus tissues [7]. Disorganized or wellorganized cerebellar tissue with Purkinje and granule cell layers, associated cyst, adipose tissue, and dilated vessels was reported in some cases [2]. Significant mitotic activity or atypical tumor-like vasculature are not seen.
Combination of multiple tissue components such as mature neuroectodermal tissue with adipose tissue in some cases can lead to misdiagnoses of teratoma. Absence of tissues derived from other germ layers is the clue to exclude teratoma [2]. Nevertheless, some cases of IEGH are accompanied by nasopharyngeal teratoma [1]. Low MIB-1 labeling index confirms IEGH benign nature. However, rare are the previous reports of MIB-1 labeling index employed for IEGH diagnosis and no previous report of Ki-67 score and BRAF mutation testing were performed [3]. Present consensus for IEGH management is early surgical resection to prevent compression of surrounding structures and bone deformity. No recurrence is reported in the literature; however, total resection cannot be achieved in some cases due adhesion to vital structures. Longterm prognosis is favorable compared to other congenital brain masses [3].
Conclusion
Our case raises the query about the implication of FLNA mutation in IEGH development. Further reports with genetic work-up would improve our comprehension of IEGH pathogenesis and may expand our understanding of CNS maldevelopment. Radiologists should be familiar with this rare entity as it is clinically and pathologically benign. Extra-axial location is intensity to brain tissue and minimal enhancement on MRI allow for accurate diagnosis.
References
- Gyure KA, Morrison AL, Jones RV (1999) Intracranial extracerebral neuroglial heterotopia: A case report and review of the literature. Ann Diagn Pathol 3(3): 182-186.
- Cho HJ, Kim HN, Kim KJ, Lee KS, Myung JK, et al. (2014) Intracranial extracerebral glioneuronal heterotopia with adipose tissue and a glioependymal cyst: a case report and review of the literature. Korean J Pathol 48(3): 254-257.
- Park JE, Park MC, Yoon SH, Kim JH (2008) Intradural extracerebral choristoma. Case report and review of the literature. Pediatr Neurosurg 44(4): 318-323.
- Kau T, Gysin C, Dohmen-Scheufler H, Brotschi B, Schiegl H, et al. (2011) Transsphenoidal extension of heterotopic glioneuronal tissue: pathoanatomic considerations in symptomatic neonates. Childs Nerv Syst 27(5): 771-778.
- Severino M, Schwartz ES, Thurnher MM, Rydland J, Nikas I, et al. (2010) Congenital tumors of the central nervous system. Neuroradiology 52(6): 531-548.
- Parmar H, Hawkins C, Bouffet E, Rutka J, Shroff M, et al. (2006) Imaging findings in primary intracranial atypical teratoid/rhabdoid tumors. Pediatr Radiol 36(2): 126-132.
- Harris CP, Townsend JJ, Klatt EC (1994) Accessory brains (extracerebral heterotopias): unusual prenatal intracranial mass lesions. J Child Neurol 9(4): 386-389.






























