Imaging in Neurodegenerative Syndromes: How to Recognize Dementia Spectrum Disorders?
Felipe Velasquez-Botero1*, Maria Alejandra Nieto-Salazar2, Adriana Carolina Toro-Velandia2, Radhika Bassi3, Luis Alejandro Morales-Ojeda4, Marlon Reyes5, Mariangel Teresa Gonzalez Hernandez6, Jessica Mariela Amaya-Alvarez7, Krupa Ann Mathew8, Coralvia Yaroslangna Villanueva-Perez9, Harendra Kumar10, Rabindra Oli11 and Flor Alcocer12
1CES University, Colombia. Larkin Community Hospital, USA
2Juan N. Corpas University, Colombia. Larkin Community Hospital, USA
3Ross University School of Medicine, Barbados, Larkin Community Hospital, USA
4UC University, Venezuela. University of Southern California, USA
5American University of Antigua College of Medicine, Antigua and Barbuda
6University of Carabobo, Venezuela
7Universidad Salvadorena Alberto Masferrer, El Salvador. Larkin Community Hospital, USA
8Tbilisi State Medical University, Georgia
9Francisco of Miranda National Experimental University, Venezuela
10Dow University of Health Sciences, Pakistan
11Nepal Medical College, Kathmandu University, Nepal
12Universidad De Oriente, Venezuela
Submission: October 12, 2022;Published: October 26, 2022
*Corresponding author: Felipe Velasquez Botero, CES University, Colombia, Larkin Community Hospital, USA
How to cite this article: Felipe Velasquez B, Maria Alejandra N S, Adriana C T V, Radhika B, Luis Alejandro M O, et al. Imaging in Neurodegenerative Syndromes: How to Recognize Dementia Spectrum Disorders?. Theranostics Brain, Spine & Neural Disord. 2022; 4(3): 555639. DOI: 10.19080/TBSND.2022.04.555639
Abstract
Dementia is a neurological disorder characterized by a decline in cognition that is significant enough to interfere with the activities of daily living. According to the DSM-5, this condition is best represented as a syndrome rather than one disease. The complexity of its clinical manifestations and diagnostic approaches presents several challenges for the medical community. In this review article, we discuss the role of imaging findings in dementia spectrum disorders in order to improve the comprehensive approach to this multifaceted condition. All the imaging modalities and radiologic findings described in this article pertain to the most prevalent forms of dementia. Dementia brain scanning techniques include structural imaging (MRI, CT) and functional imaging (DTI, MR spectroscopy, PET, and SPECT). Our review found that structural and functional imaging methods are essential when evaluating the dementia spectrum. A substantial decline in cognitive function characterizes this neurological disorder, and every imaging study has its indications, specific findings, and limitations. Due to the similarity in anatomical and functional changes of these disorders, neuroimaging is anticipated to play a significant role in the elucidation and precise diagnosis and management of these patients.
Keywords: Dementia; Neurodegenerative disorder; Imaging; Neuroimaging
Keywords: AD: Alzheimer’s Disease, FTD: Frontotemporal Dementia, LBD: Lewy Body Dementia, PDD: Parkinson’s Disease Dementia, WKS: Wernicke-Korsakoff Syndrome, CSF: Cerebrospinal Fluid, MRI: Magnetic Resonance Imaging, CT: Computed Tomography, EEG: Electroencephalogram, FDG: Fluorodeoxyglucose, PET: Positron Emission Tomography, SPECT: Single-Photon Emission Computerized Tomography, DCE: Dynamic Contrast-Enhanced, DTI: Diffusion Tensor Imaging, H-MRS: Proton Magnetic Resonance Spectroscopy, MSA-P: Multiple System Atrophy-Parkinsonian Type Dementia, SWI: Susceptibility Weighted Imaging, PD: Parkinson Disease, DAT: Dopamine Transporter, DWI: Diffusion-Weighted Imaging, ADC: Apparent Diffusion Coefficient.
Introduction
The dementia spectrum encloses a wide range of disorders characterized by a substantial decline in cognitive function with an inability to perform activities of daily living independently compared to age-matched controls [1]. According to the DSM-5, this neurological condition can be classified into two main categories: neurodegenerative (formerly known as “irreversible”) and nonneurodegenerative (formerly known as “reversible”) dementias [2]. Among the most common forms of neurodegenerative dementias are Alzheimer’s disease (AD), Frontotemporal dementia (FD), Lewy body dementia (LBD), Parkinson’s disease dementia (PDD), and Wernicke-Korsakoff syndrome (WKS). Nonneurodegenerative dementias are those compelled by etiologies that can be prevented or reversed, such as vascular dementia and normal pressure hydrocephalus [2]. Approximately 44 million people worldwide have dementia, which is predicted to triple by 2050. Around 60% of all cases of dementia are caused by Alzheimer’s disease, while vascular dementia accounts for about 20% of cases worldwide [3,4]. In developed nations, dementia prevalence rates are higher than in developing countries [5].
Dementia syndrome clinical features include progressive memory impairment, behavioral disinhibition, loss of empathy, headache, malaise, behavioral changes, and psychiatric episodes [2,6]. This neurological condition is diagnosed primarily by evaluating the clinical manifestations, physical examination, and laboratory tests. In addition, radiological input is frequently used to assist in the diagnosis. However, there are several instances in which imaging results may not be specific or misinterpreted by the attending physician. This review study aims to comprehensively overview the significant imaging findings in the dementia syndromes spectrum to improve the multidisciplinary approach to this complex condition.
Alzheimer’s Disease
Alzheimer’s disease (AD) is a neurodegenerative disease involving progressive deterioration and nerve cell loss [7]. It accounts for 50%–75% of dementia cases worldwide, and its prevalence doubles every five years after age 65 [8]. Patients with AD commonly experience episodic memory loss, which falls into the amnestic mild cognitive impairment category. Cognitive decline affects behavior, speech, visuospatial orientation, and the motor system [9]. AD is usually diagnosed by a cerebrospinal fluid (CSF) biomarker panel containing AB42, tau, and phosphotau, along with a calculation of the amyloid-tau index. Diagnostic imaging modalities include non-contrast brain magnetic resonance imaging (MRI), non-contrast computed tomography (CT), electroencephalogram (EEG), brain fluorodeoxyglucose (FDG)-positron emission tomography (PET), single-photon emission computerized tomography (SPECT), and brain amyloid- PET scan [10].
In structural MRI, gray matter atrophy is first observed in the entorhinal cortex and the hippocampus, which are parts of the medial temporal lobe responsible for conscious memory (Figure 1). As AD progresses, atrophy is preserved in the remainder of the medial temporal gyrus, para-hippocampus, and temporal pole, accompanied by white matter hyper intensities indicating demyelination. In diffusion-tensor imaging, there is increased mean diffusivity in the frontal, occipital, parietal, and temporal areas, as well as decreased fractional anisotropy in the cingulum, corpus callosum, and frontal white matter. Applying the Arterial spin labeling perfusion technique, hypoperfusion is noted in the posterior cingulate, precuneus, and occipital, temporal, and parietal cortical areas in AD. Functional MRI demonstrates increased brain activity in parietal and posterior cingulate regions indicating compensation due to medial temporal lobe dysfunction [11].
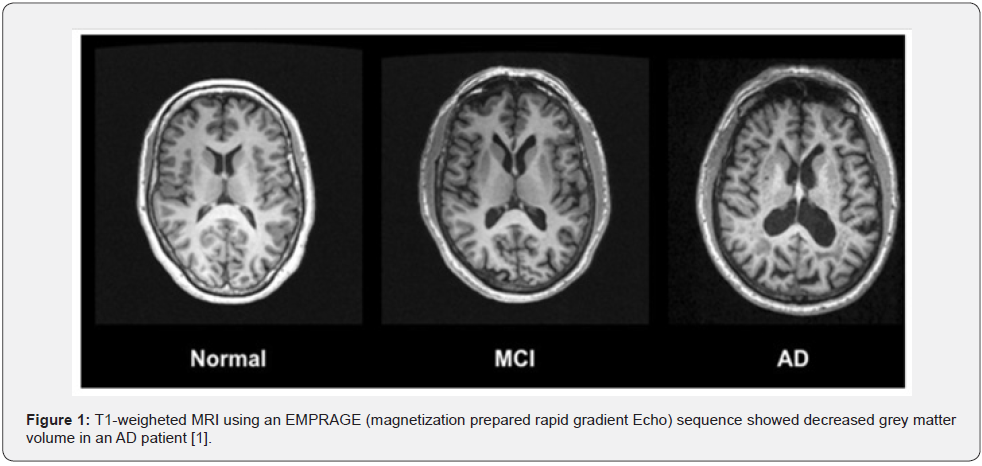
SPECT and PET can detect molecular changes that precede brain atrophy development. Using technetium-99m-hexamethyl propylene amine oxime (99mTc-HMPAO) or technetium-99methyl cysteine diethyl ester (99mTc-ECD), FDG-PET and SPECT might show patterns of hypometabolism and hypoperfusion in the posterior cingulate cortex in early disease stages. Following these changes, bilaterally and asymmetrically reduced posterior temporoparietal cortices are observed. PET scan usually demonstrates an increased uptake of radiotracers that bind to Aβ amyloid (i.e., PiB, florbetapir) in frontal, parietal, and temporal cortices. Finally, Tau neuroimaging has documented higher uptake of [18F] THK5117 in the cortex of AD patients, which correlates with the level of cognitive decline as the disease progresses [12].
Vascular Dementia
Vascular dementia (VD) results from multiple cerebral arterial infarcts leading to chronic ischemia. As a result, there is a gradual decline in the cognitive capacity associated with this condition, most notably in the elderly population. VD commonly affects one or more of the five cognitive domains (memory, executive function, visuospatial, language, and attention) [13]. The condition has been identified as the second most common cause of dementia in the elderly, accounting for 15-30% of cases worldwide [14]. Besides advanced age, several other risk factors contribute to vascular dementia, including diabetes mellitus, myocardial infarction, atrial fibrillation, epileptic seizures, sepsis, cardiac arrhythmias,congestive heart failure, global cerebral atrophy, medial temporal lobe atrophy, and changes in white matter [15]. There are numerous areas of the brain that may be affected by vascular dementia. Damage to the frontal and prefrontal cortex may lead to mood disorders such as anxiety, depression, and disorientation. Damage to the hippocampus, frontal lobe, and prefrontal cortex can lead to memory impairment, including short-term memory, long-term memory, and working memory. Subcortical and white matter damage can result in abnormal gait and mental processing difficulties. Finally, VD can also affect the frontal cortex executive function, resulting in impaired problem-solving, planning, and task-execution skills [16].
When vascular dementia is suspected, MRI is the preferred imaging modality for diagnosis. Typical findings of VD include multiple punctate or brain lesions in the white matter, known as leukoaraiosis. Moreover, MRI can display cortical microhemorrhages. The use of dynamic contrast-enhanced MRI (DCE-MRI), diffusion tensor imaging, and proton magnetic resonance spectroscopy (H-MRS) can be used to detect white matter ischemia. In H-MRS, N-acetyl aspartate and creatine levels are measured when injury to axons occurs, while in DCE-MRI, blood vessel permeability is measured, indicating the presence of neuroinflammation [13]. Sodium fluoride PET scan can be used to detect early signs of atherosclerosis (Figure 2). This is in contrast to conventional MRI, which can only detect late ischemic changes, which are irreversible [17,18].

Lewy Body Dementia
Lewy Body dementia (LBD) is a chronic degenerative neurological disease characterized by psychotic and Parkinsonlike symptoms [19]. The condition develops due to the intraneuronal deposition of cytoplasmic inclusion bodies with clumps of alpha-synuclein and ubiquitin (also known as Lewy bodies) [19,20]. LBD is the third most common form of dementia after vascular dementia and Alzheimer’s disease, accounting for 20% to 30% of all dementia cases. Men of advanced age are the most common population affected by this condition. Other risk factors include a family history of Parkinson’s disease or Lewy Body Dementia [21]. Clinical manifestations include a progressive decline in concentration and executive function, fluctuating cognitive function (with variation in concentration and drowsiness), visual hallucinations, and parkinsonian symptoms (i.e., muscular rigidity, tremor, and bradykinesia). Less common findings include sleep behavior disorder, autonomic dysfunction, depression, and sensitivity to antipsychotic medications. LBD is often an underdiagnosed condition due to a lack of understanding of its clinical features and common overlaps with symptoms of more prevalent dementia disorders [21-23]. The diagnosis of this type of dementia should therefore be based on a comprehensive approach. Diagnostic tests include a detailed history and physical examination, assessment of mental function, blood tests (i.e., vitamin B12 levels, chemistry panel, thyroid profile, syphilis, HIV), and imaging studies [22].
In LBD, brain MRI is the most commonly used imaging modality, in which it is possible to observe atrophy in numerous brain regions without a discernible pattern. MRI findings include generalized cerebral volume reduction (most pronounced in the frontal lobe and parietotemporal areas), expansion of the lateral ventricles, and relative focal atrophy of the midbrain, hypothalamus, and substantia innominata [19,23]. The sensitivity and specificity of susceptibility weighted imaging (SWI), an MRI technique, have been reported to be 93% and 87%, respectively, for detecting LBD. Nuclear medicine imaging techniques (SPECT/ PET) might reveal occipital hypoperfusion, which may assist in differentiating the condition from other types of dementia (particularly Alzheimer’s disease, which primarily exhibits frontal and temporal hypoperfusion patterns) [22,23].
Parkinson’s Disease
Parkinson’s disease (PD) is a neurodegenerative disorder characterized by generalized bradykinesia, motor symptoms (tremors or rigidity), and cognitive function deterioration [24]. The prevalence of Parkinson’s disease rises with age, affecting approximately 1% of the population over 60, primarily men. Clinical manifestations include motor symptoms, such as resting tremors, rigidity, bradykinesia, and gait disturbance. Cognitive decline, commonly called Parkinson’s disease dementia (PDD), usually presents later in the disease process [25].
PDD is diagnosed by a complete clinical examination and laboratory examination, especially to rule out possible pseudodementia syndromes [25,26]. In this condition, radiologic imaging plays an essential role in diagnosing and excluding other conditions, such as normal pressure hydrocephalus or subcortical stroke [27]. Brain MRI is usually the preferred imaging method for PDD. The most promising diagnostic evidence may be the absence of the swallow tail pattern of the susceptibility signal on axial imaging of the substantia nigra [28,29]. Aside from aiding in diagnosis, MRI can also be used to identify characteristics indicative of subsequent parkinsonism rather than the primary disease itself, such as ischemia of the small vessels [29]. Other imaging modalities include the dopamine transporter (DAT) scan, a method to detect decreased dopamine uptake in dopaminecontaining neurons. In PDD, the basal ganglia exhibit a low dopamine uptake [30].
Wernicke-Korsakoff Syndrome
Wernicke-Korsakoff syndrome (WKS) covers both Wernicke encephalopathy and Korsakoff syndrome. Wernicke encephalopathy is caused by thiamine deficiency [31]. The condition is commonly reversible if the deficit is corrected early. However, if left untreated, this condition can lead to Korsakoff syndrome, an irreversible neurodegenerative disorder. It is estimated that the prevalence of WKS ranges from 0% to 2%, with higher rates found in specific subpopulations (i.e., homeless or lowincome individuals, substance abusers, elderly individuals living alone, and patients in mental health facilities) [32,33]. Wernicke encephalopathy exhibits a triad of clinical manifestations: acute confusion, ataxia, and ophthalmoplegia (most commonly horizontal nystagmus and conjugate gaze palsies). This condition is known as Wernicke-Korskoff syndrome when symptoms such as memory loss (global amnesia) and confabulation occur as the disease progresses [33,34]
The diagnosis of WKS is typically based on a patient’s medical history and clinical manifestations, along with lab and imaging tests. Standard diagnostic imaging methods include contrastenhanced cross-sectional imaging (CT, MRI) and advanced MRI techniques [34]. A brain CT scan usually reveals a normal brain architecture, but the early-stage disease may manifest as hemorrhage, necrosis, and edema. On MRI T2-weighted image and FLAIR sequence, common findings include symmetrically increased signal intensity in the mammillary bodies, dorsomedial thalami, tectal plate, and periaqueductal gray matter around the third ventricle. MRI T1 weighted sequences typically display contrast enhancement in previously described brain regions, particularly the mammillary bodies [34,35]. In addition, diffusionweighted imaging (DWI) with Apparent Diffusion Coefficient (ADC) mapping may depict restricted diffusion in those same regions. Finally, MR spectroscopy may show decreased or normal NAA with the notable presence of lactate [35].
Normal Pressure Hydrocephalus
Normal pressure hydrocephalus (NPH) is one of the few causes of dementia that may be reversible. This condition can be divided into primary or idiopathic (unknown cause) and secondary to various brain diseases such as subarachnoid hemorrhage, trauma, meningitis, and stroke [36]. The prevalence of NPH ranges from 0.2% to 5.9%, with rates increasing in males over 80 [37]. The classic clinical triad, first described by Hakim and Adams in 1965, includes gait disturbance, urinary incontinence, and dementia, all of which can occur individually or in combination [36,38]. NPH dementia commonly exhibits frontal and subcortical deficits and can be one of the earliest signs of this condition [3].
The diagnosis of NPH is primarily based on the evaluation of medical history, neurologic examination, and brain imaging [38]. Brain MRI is usually the standard method for assessing iNPH. Typical findings include ventricular enlargement greater than 0.3cm (Evans index), callosal angle ranging between 40 to 90 degrees, and disproportionate widening of the ventricles compared to the cerebral sulci (Figure 3). Other imaging modalities include PET scan, which has proven useful in diagnosing NPH. In a retrospective study, Townley et al. found that patients with iNPH had significant regional hypometabolism in the dorsal striatum involving the caudate and putamen bilaterally compared with healthy controls and patients with other types of dementia [39].

Frontotemporal Dementia
Frontotemporal dementia (FTD), also known as Pick disease, is characterized by relatively progressive atrophy of the frontal and/or temporal lobe. Estimates of the prevalence of FTD before age 65 range from four to fifteen per 100,000 people, which is significantly lower than that of Alzheimer’s disease [40-43]. Nevertheless, this condition has a disproportionately large impact since it leads to early-onset dementia and all its adverse clinical and socioeconomic consequences. Clinical manifestations of FTD include behavioral disturbances (frequently lacking the necessary primal and interpersonal emotions, similar to individuals with an antisocial personality disorder) and cognitive decline, which is less pronounced than other dementia syndromes [44].
Patients should go through a neuropsychiatric evaluation, cognitive testing, and neuroimaging procedures to confirm the diagnosis of FTD [45]. FTD may be distinguished from other types of cognitive impairment using neuroimaging. Structural imaging (MRI, CT) often reveals anterior temporal and frontal atrophy, and functional imaging may exhibit decreased perfusion over the front temporal lobes, ventromedial frontal cortex, and orbitofrontal cortex [46,47]. SPECT scanning may identify hypoperfusion in the ventromedial frontal region, even before structural atrophy becomes evident [45]. (Figure 4) [48].

Conclusion
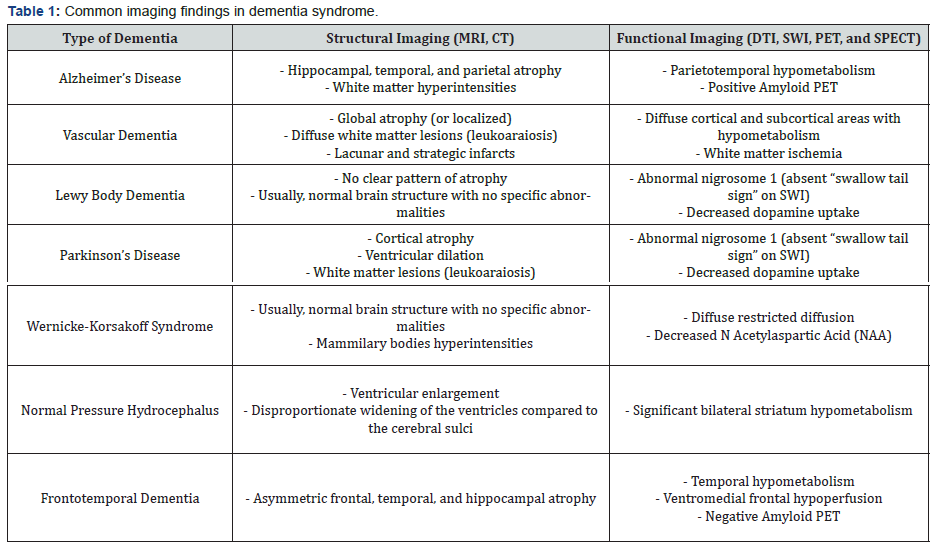
Identifying the dementia syndrome spectrum is primarily determined by the neurological clinical findings presented by the patients. However, most of these syndromes have similar characteristic features of cognitive decline, making an accurate assessment particularly challenging. Therefore, structural imaging methods (MRI, CT) and those that estimate functionality (DTI, MR spectroscopy, PET, and SPECT) are critical when evaluating this complex neurological disorder. This review article described the most common imaging findings associated with the most prevalent forms of dementia. A brief summary of the findings is presented in table 1. While current literature provides adequate evidence regarding the radiologic findings in dementia, actual image studies have significant limitations. For example, neurodegenerative disorders could reflect similar anatomical changes on brain imaging or display normal structures/functions despite the presence of neurological symptoms. It is expected that with the advancement of imaging technology in the future, a more comprehensive depiction and overview of these syndromes will be obtained, which in turn will contribute to a more precise evaluation of these patients.
References
- Mahalingam S, Chen MK (2019) Neuroimaging in Dementias. Semin Neurol 39(2): 188-199.
- Gale SA, Acar D, Daffner KR (2018) Dementia. Am J Med 131(10): 1161-1169.
- Kalaria RN, Maestre GE, Arizaga R, Friedland RP, Galasko D, et al. (2008) World Federation of Neurology Dementia Research Group. Alzheimer's disease and vascular dementia in developing countries: prevalence, management, and risk factors. Lancet Neurol 7(9): 812-826.
- Forette F, Boller F (1991) Hypertension and the risk of dementia in the elderly. Am J Med 90(3A): 14S-19S.
- Rizzi L, Rosset I, Roriz CM (2014) Global epidemiology of dementia: Alzheimer's and vascular types. Biomed Res Int.
- Dening T, Sandilyan MB (2015) Dementia: definitions and types. Nurs Stand 29(37): 37-42.
- Alzheimer A, Stelzmann RA, Schnitzlein HN, Murtagh FR (1995) An English translation of Alzheimer's 1907 paper, "Uber eine eigenartige Erkankung der Hirnrinde 8(6): 429-431.
- DeTure MA, Dickson DW (2019) The neuropathological diagnosis of Alzheimer's disease. Mol Neurodegener 14(1): 32.
- Atri A (2019) The Alzheimer's Disease Clinical Spectrum: Diagnosis and Management. Med Clin North Am 103(2): 263-293.
- Chandra A, Dervenoulas G, Politis M (2019) Alzheimer’s Disease Neuroimaging Initiative. Magnetic resonance imaging in Alzheimer's disease and mild cognitive impairment. J Neurol 266(6): 1293-1302.
- Valotassiou V, Malamitsi J, Papatriantafyllou J, Dardiotis E, Tsougos I, et al. (2018) SPECT and PET imaging in Alzheimer's disease. Ann Nucl Med 32(9): 583-593.
- Uwagbai O, Kalish VB (2022) Vascular Dementia. In: StatPearls. Treasure Island (FL): StatPearls Publishing, USA.
- Wolters FJ, Ikram MA (2019) Epidemiology of Vascular Dementia. Arterioscler Thromb Vasc Biol 39(8): 1542-1549.
- Pinkston JB, Alekseeva N, González Toledo E (2009) Stroke and dementia. Neurol Res 31(8): 824-31.
- Venkat P, Chopp M, Chen J (2015) Models and mechanisms of vascular dementia. Exp Neurol 272: 97-108.
- Heiss WD, Rosenberg GA, Thiel A, Berlot R, De Reuck J (2016) Neuroimaging in vascular cognitive impairment: a state-of-the-art review. BMC Med 14(1): 174.
- Haider A, Spurling BC, Sánchez MJC (2022) Lewy Body Dementia. In: StatPearls. Treasure Island (FL): StatPearls Publishing, USA.
- King E, O Brien JT, Donaghy P, Morris C, Barnett N, et al. (2019) Peripheral inflammation in mild cognitive impairment with possible and probable Lewy body disease and Alzheimer's disease. Int Psychogeriatr 31(4): 551-560.
- Durcan R, Donaghy P, Osborne C, Taylor JP, Thomas AJ (2019) Imaging in prodromal dementia with Lewy bodies: Where do we stand? Int J Geriatr Psychiatry 34(5): 635-646.
- Whitwell JL, Weigand SD, Shiung MM, Boeve BF, Ferman TJ, et al. (2007) Focal atrophy in dementia with Lewy bodies on MRI: a distinct pattern from Alzheimer's disease. Brain 130(3): 708-719.
- Durcan R, Donaghy P, Osborne C, Taylor JP, Thomas AJ (2019) Imaging in prodromal dementia with Lewy bodies: Where do we stand? Int J Geriatr Psychiatry 34(5): 635-646.
- Whitwell JL, Weigand SD, Shiung MM, Boeve BF, Ferman TJ, et al. (2007) Focal atrophy in dementia with Lewy bodies on MRI: a distinct pattern from Alzheimer's disease. Brain 130(Pt 3): 708-719.
- Harding AJ, Broe GA, Halliday GM (2002) Visual hallucinations in Lewy body disease relate to Lewy bodies in the temporal lobe. Brain 125(Pt 2): 391-403.
- Mirpour S, Turkbey EB, Marashdeh W, El Khouli R, Subramaniam RM (2018) Impact of DAT-SPECT on Management of Patients Suspected of Parkinsonism. Clin Nucl Med 43(10): 710-714.
- Alexoudi A, Alexoudi I, Gatzonis S (2018) Parkinson's disease pathogenesis, evolution and alternative pathways: A review. Rev Neurol (Paris) 174(10): 699-704.
- Kabra A, Sharma R, Kabra R, Baghel US (2018) Emerging and Alternative Therapies for Parkinson Disease: An Updated Review. Curr Pharm Des 24(22): 2573-2582.
- Ehgoetz MKA, Shine JM, Walton CC, Georgiades MJ, Gilat M, et al. (2018) Evidence for subtypes of freezing of gait in Parkinson's disease. Mov Disord 33(7): 1174-1178.
- Chung SJ, Yoo HS, Lee HS, Oh JS, Kim JS, et al. (2018) The Pattern of Striatal Dopamine Depletion as a Prognostic Marker in De Novo Parkinson Disease. Clin Nucl Med 43(11): 787-792.
- Gratton C, Koller JM, Shannon W, Greene DJ, Maiti B, et al. (2019) Emergent Functional Network Effects in Parkinson Disease. Cereb Cortex 29(6): 2509-2523.
- Berg D, Adler CH, Bloem BR, Chan P, Gasser T, et al. (2018) Movement disorder society criteria for clinically established early Parkinson's disease. Mov Disord 33(10): 1643-1646.
- Gerridzen IJ, Joling KJ, Depla MF, Veenhuizen RB, Verschuur EML, et al. (2019) Awareness and its relationships with neuropsychiatric symptoms in people with Korsakoff syndrome or other alcohol-related cognitive disorders living in specialized nursing homes. Int J Geriatr Psychiatry 34(6): 836-845.
- Zuccoli G, Gallucci M, Capellades J, Regnicolo L, Tumiati B, et al. (2007) Wernicke encephalopathy: MR findings at clinical presentation in twenty-six alcoholic and nonalcoholic patients. AJNR Am J Neuroradiol 28(7): 1328-1331.
- Heirene R, John B, Roderique DG (2018) Identification and Evaluation of Neuropsychological Tools Used in the Assessment of Alcohol-Related Cognitive Impairment: A Systematic Review. Front Psychol.
- Loh Y, Watson WD, Verma A, Krapiva P (2005) Restricted diffusion of the splenium in acute Wernicke's encephalopathy. J Neuroimaging 15(4): 373-375.
- Sabatini JS, Schutz PGL, Feltrin F, Teive HAG, Camargo CHF (2016) Wernicke's encephalopathy with chorea: Neuroimaging findings. Dement Neuropsychol 10(4): 370-372.
- Hakim S, Adams RD (1965) The special clinical problem of symptomatic hydrocephalus with normal cerebrospinal fluid pressure. Observations on cerebrospinal fluid hydrodynamics. J Neurol Sci 2(4): 307-327.
- Brean A, Eide PK (2008) Prevalence of probable idiopathic normal pressure hydrocephalus in a Norwegian population. Acta Neurol Scand 118(1): 48-53.
- Shprecher D, Schwalb J, Kurlan R (2008) Normal pressure hydrocephalus: diagnosis and treatment. Curr Neurol Neurosci Rep 8(5): 371-376.
- Yin R, Wen J, Wei J (2021) Progression in Neuroimaging of Normal Pressure Hydrocephalus. Front Neurol 12: 700269.
- Rabinovici GD, Miller BL (2010) Frontotemporal lobar degeneration: epidemiology, pathophysiology, diagnosis and management. CNS Drugs 24(5): 375-398.
- Mackenzie IR, Neumann M, Baborie A, Sampathu DM, Du Plessis D, et al. (2011) A harmonized classification system for FTLD-TDP pathology. Acta Neuropathol 122(1): 111-113.
- Rohrer JD, Guerreiro R, Vandrovcova J, Uphill J, Reiman D, et al. (2009) The heritability and genetics of frontotemporal lobar degeneration. Neurology 73(18): 1451-1456.
- Rohrer JD, Warren JD (2011) Phenotypic signatures of genetic frontotemporal dementia. Curr Opin Neurol 24(6): 542-549.
- Neary D, Snowden J, Mann D (2005) Frontotemporal dementia. Lancet Neurol 4(11): 771-780.
- Hodges JR (2001) Frontotemporal dementia (Pick's disease): clinical features and assessment. Neurology 56(11 Suppl 4): S6-10.
- Mendez MF, Shapira JS (2005) Loss of insight and functional neuroimaging in frontotemporal dementia. J Neuropsychiatry Clin Neurosci. Summer 17(3): 413-416.
- Talbot PR, Snowden JS, Lloyd JJ, Neary D, Testa HJ (1995) The contribution of single photon emission tomography to the clinical differentiation of degenerative cortical brain disorders. J Neurol 242(9): 579-586.
- Bang J, Spina S, Miller BL (2015) Frontotemporal dementia. Lancet 386(10004): 1672-1682.






























