Transnasal Delivery of Fluoxetine HCL to Brain for Treating Depression
Sagar Atmaram Aher and Avinash Ramrao Tekade*
Department of Pharmaceutics and Pharmaceutical QA, Marathwada MitraMandal’s College of Pharmacy, India
Submission: September 17, 2019;Published: November 15, 2019
*Corresponding author: Avinash R Tekade, Department of Pharmaceutics and Pharmaceutical QA, Marathwada Mitra Mandal’s College of Pharmacy, Thergaon, Pune, Maharashtra 411033, India
How to cite this article: Sagar Atmaram Aher, Avinash Ramrao Tekade. Transnasal Delivery of Fluoxetine HCL to Brain for Treating 0030 Depression Theranostics Brain,Spine & Neural Disord. 2019; 4(2): 555633. DOI:10.19080/JOJS.2019.04.555633
Abstract
The objective of this study is to formulate a mucoadhesive microemulsion containing Fluoxetine HCl as antidepressant and Cedar oil as penetration enhancer. Water titration method was used to formulate the microemulsion using two different ratios of selected surfactant and cosurfactant (Tween 80 and Propylene Glycol). The prepared microemulsion showed mean globule size of 303.84nm. The prepared microemulsion is non-irritant for nasal mucosa which was confirmed by ciliotoxicity study using sheep nasal mucosa. It is clearly evident from ex vivo diffusion study that mucoadhesive microemulsion can deliver drug for longer time, the initial release was burst release in the first hour followed by sustained for next three hours, and increased diffusion will definitely help in reducing dose related side effects associated with the Fluoxetine HCl. Thus, present study concluded that the prepared microemulsion containing Fluoxetine HCl have potential to deliver the drug efficiently to the brain by nasal administration.
Keywords: Transnasal delivery Depression Fluoxetine Hcl Brain
Introduction
A common mental disorder termed as depression that limits psychosocial functioning and diminishes quality of life [1,2] It is different from emotional responses to daily life challenges and mood fluctuations also. It can lead to suicide if it is severe, according to WHO about 8 lakh peoples die due to suicide every year which is the second leading cause of death in age group of 15-29 years. Not all the peoples get effective treatments that are available for depression; barriers include lack of resources, lack of trained health-care providers, and social stigma associated with mental disorders. Another barrier to effective care is inaccurate assessment. Sometimes, people who are depressed are often not correctly diagnosed, and others who do not have the disorder are too often misdiagnosed and prescribed with antidepressants [1]. Major Risk factors includes Personal or family history of depression, Major life changes, trauma, or stress and Certain physical illnesses and medications.
Signs and Symptoms [2-4]
If anyone is suffering through the following signs and symptoms most of the day or for at least two weeks he/she might be depressed: Persistent sad, anxious, or “empty” mood, Feelings of hopelessness, or pessimism, Irritability, Feelings of guilt, worthlessness, or helplessness , Loss of interest or pleasure in hobbies and activities, Decreased energy or fatigue, Moving or talking more slowly, Feeling restless or having trouble sitting still, Difficulty concentrating,remembering, or making decisions, Difficulty sleeping, early-morning awakening, or oversleeping, Appetite and/or weight changes, Thoughts of death or suicide, or suicide attempts, Aches or pains, headaches, cramps, or digestive problems without a clear physical cause and/or that do not ease even with treatment.
Types [1,3]
It might be mild, moderate, or severe depending on number and severity of symptoms of depressive episodes.
a) Recurrent/ Persistent Depressive Disorder: This involves repeated depressive episodes.
b) Postpartum Depression: Women may suffer through postpartum depression i.e. experiences full-blown major depression during pregnancy or after delivery
c) Psychotic Depression: When a person has severe depression plus some form of psychosis that may be the psychotic depression, such as having disturbed false fixed beliefs (delusions) or hearing or seeing upsetting things that others cannot hear or see (hallucinations). The psychotic symptoms typically have a depressive “theme,” such as delusions of guilt, poverty, or illness.
d) Seasonal Affective Disorder: It is characterized by the onset of depression during the winter months, when there isless natural sunlight. This depression generally lifts during spring and summer.
e) Bipolar Affective Disorder: It consists of both manic and depressive episodes segregated by periods of normal mood.
Diagnosis and Treatment [1-3,5]
There are effective treatments available for moderate and severe type of depression. A health-care provider generally offers psychological treatments (like behavioural activation, cognitive behavioural therapy [CBT], and interpersonal psychotherapy [IPT]) or antidepressant like selective serotonin reuptake inhibitors [SSRIs] and tricyclic antidepressants [TCAs]. Healthcare providers should keep in mind the possible adverse effects associated with antidepressant medication, the ability to deliver either intervention (in terms of expertise, and/or treatment availability), and individual preferences. Different psychological treatment formats for consideration include individual and/or group face-to-face psychological treatments delivered by professionals and supervised lay therapists. Psychosocial treatments are also effective for mild depression. Antidepressants can be an effective form of treatment for moderate-severe depression but are not the first line of treatment for cases of mild depression. They should not be used for treating depression in children and are not the first line of treatment in adolescents, among whom they should be used with extra caution.
Antidepressants, including Fluoxetine HCl, can have a number of possible adverse effects. The FDA requires Prozac (Brand name of Fluoxetine HCl) to come with a black box warning stating that antidepressants may increase the risk of suicide in young generation. It can lead to suicidal thoughts, or a worsening of these, in children and young adults. Other possible side effects may include decreased libido and sexual dysfunction, anxiety and nervousness, abnormal dreams, sweating, diarrhoea, insomnia, drowsiness, and yawning, sinusitis, an inflammation of the mucous membrane. Nasal route will overcome some of these dose related side effects as it requires too less dose strength as compared to oral dose.
Nose to Brain Drug Delivery [5-8]
Traditionally, the nasal route has been exploited for delivery of drugs for the treatment of local diseases like nasal allergy, sinusitis, nasal infections and nasal congestion. In the last few decades, the nasal route has attracted wide attention as a reliable, safe, noninvasive and convenient route to accomplish faster and higher levels of drug absorption. The crucial reasons behind the interest in the nasal route are the highly vascularized epithelium of nasal mucosa, porous endothelial membrane, its ready accessibility, its large surface area for rapid drug absorption, rapid onset of action, lower enzyme levels compared with gastrointestinal tract and liver, high total blood flow per cm3 and direct drug transport to systemic circulation and to the brain, thereby avoiding first-pass hepatic metabolism and enhancing bioavailability.
The blood-brain barrier and the blood-cerebrospinal fluid barrier are major barriers for drug delivery towards central nervoussystem (CNS), as they resist entry of most molecules in the brain. In contrast, nose-to-brain delivery is a minimally invasive drug administration pathway, which bypasses the blood-brain barrier as the drug is directed from the nasal cavity to the brain.
Pathways and mechanisms [9,10]
While the precise mechanisms underlying intranasal drug delivery to the CNS are not entirely understood yet, an accumulating body of evidences demonstrates that pathways involving nerves connecting the nasal passages to the brain and spinal cord are important. Additionally, pathways involving the vasculature, CSF and lymphatic system have been employed in transport of molecules from nasal cavity to the CNS. It is possible that a combination of these pathways is responsible, although one pathway may predominate, depending on the properties of neurotherapeutics, the characteristics of formulations and the delivery device used.
Olfactory nerve pathway
In order for a drug to travel from the olfactory region in the nasal cavity to the CSF or brain parenchyma, it has to traverse the nasal olfactory epithelium and, depending on the pathway followed, also the arachnoid membrane surrounding the subarachnoid space.
Trigeminal nerve pathway
An important pathway connecting nasal passages to the CNS involves the trigeminal nerve (the largest nerve among all cranial nerves), which innervates the respiratory and olfactory epithelium of nasal passages and enters the CNS in the pons.
Microemulsions [11-13]
Microemulsions are clear, stable, isotropic mixtures of oil, water and surfactant, frequently in combination with a cosurfactant. Microemulsions act as potential drug carrier systems for oral, topical, and parenteral administration. They offer the advantage of spontaneous formation, ease of manufacturing and scale-up, thermodynamic stability, and improved drug solubilization and bioavailability. Preparing a pharmaceutically acceptable dosage form demands a clear understanding of the micro-emulsion structure, phase behaviour, factors leading to its thermodynamic stability and the potential uses and limitations of the microemulsion system. Knowledge of the various methods available to thoroughly characterize a microemulsion system is essential. Microemulsions have been shown to be able to protect labile drug, control drug release, increase drug solubility, increase bioavailability and reduce patient variability. Furthermore, it has proven possible to formulate preparations suitable for most routes of administration. There is still however a considerable amount of fundamental work characterizing the physico-chemical behavior of microemulsions that needs to be performed before they can live up to their potential as multipurpose drug delivery vehicles.
In this study attempt has been made to formulate mucoadhesive microemulsion by using Fluoxetine HCl as an antidepressant and cedar Wood oil as penetration enhancer and its antidepressant activity to get a synergistic action.
Materials and Methods
Drugs and excipients used
Fluoxetine Hydrochloride was received as a gift sample from Hetero Drugs, Hyderabad, India. Cedaroil was purchased from Manakarnika Aushadhalaya, Pune-India. Carbopol940 P was donated by Lubrizol life sciences, Mumbai-India. Tween 80 and Polyethylene Glycol was purchased from SK Enterprises Pune-India. All other chemicals and reagents used were of analytical reagent grade.
Methodology
Formulation development [9,14,15]: Solubility studies [16]. The solubility of Fluoxetine HCl in Water, pH 6.2, various oils (sandalwood oil, lavender oil and cedar oil), surfactants (Span 80 and Tween 80) and cosurfactants (PG, ethanol, Transcutol HP) was measured by dissolving an excess amount of Fluoxetine HCl in 2ml of each solution separately. The mixtures were shaken with the help of vertex mixer for 15min and kept overnight. Solution was filtered and supernatant was properly diluted and concentration of Fluoxetine HCl was quantified spectrophotometrically at 226nm wavelength.
Screening of oils for its penetration enhancing activity:
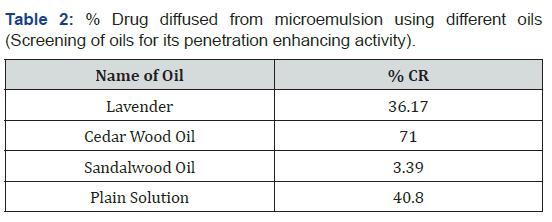
Various oils such as sandalwood oil, lavender oil and cedar oil having use in aroma therapy are evaluated for their penetration enhancing activity. The microemulsion formulations containing different oils are formulated and evaluated for the drug release by using 6 Station Franz Diffusion cell apparatus with buffer pH of 6.2
Construction of pseudo-ternary phase diagram [5,9]:
The pseudo ternary phase diagram of the oil, Smix and water were developed using the aqueous titration method. Based on the solubility studies, the selected oil (Cedar oil), surfactant (Tween 80), and cosurfactant (Propylene Glycol) were used to prepare the microemulsion. The surfactant and cosurfactant (Smix) were mixed at different mass ratios (1:1, and 1:2). Different combinations of oil and Smix were made so that the maximum ratios were covered in order to precisely delineate the boundaries of phases formed in the phasediagrams. Water was added drop-by-drop to this mixture under proper magnetic stirringat room temperature until the mixture became visually transparent and flowable.
Experimental design nasal gel
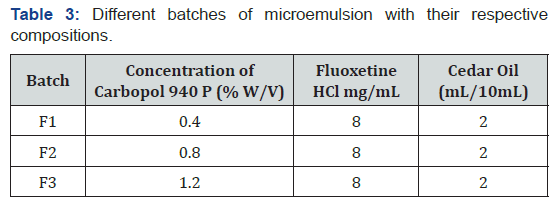
Selection of suitable polymer and excipient: Optimization technique is a vital aid to the formulator, as it helps in developing the best possible formulation design under a given set of conditions with minimum experimentation, saving considerable time, money and effort. The concentration range of the Corbopol 940 P for the preparation of mucoadhesive microemulsion by above mentioned method was optimized by trial and error basis.
Preparation of trial batches of microemulsion based in situ nasal gel
Oil phase: Oil phase of microemulsion was prepared by dissolving 40mg of Fluoxetine HCl in 20ml of Cedar oil, and then specified amount (70mL) of S mix i.e. Tween 80 (surfactant) & Propylene Glycol (cosurfactant) was added to above solution of API.
Water phase: Corbopol 940 P (4, 8, 12mg) was taken in a beaker to make 0.4%, 0.8% and 1.2% solutions and it was dispersed in ultrapure water and stirred till it dissolves completely. This aqueous phase was added dropwise to the oil phase under constantmixing with vortex mixer. After complete addition of water, microemulsion was vortexed for 15mins followed by homogenization with the help of ultra turrax. Effect of various concentrations of Carbopol 940 P on the physical characteristics of mucoadhesive microemulsion was studied. Fluoxetine HCl loaded nasal mucoadhesive microemulsion gel were prepared by selecting various concentrations of Carbopol940 P. The amount of Carbopol940 P was tried in the range of 0.2 - 1% W/V. Trial and error batches of varying concentrations were evaluated for particle size, drug content and drug release.
In this study, only one factor was evaluated for three levels, and experimental trials were performed at all three possible combinations. The amount of Carbopol940 P(X1) was selected as independent variable Mucoadhesive strength (Y1) and drug release (Y2) were selected as dependent variables. This factor might affect the designed characteristic of nasal mucoadhesive formulation were varied over three levels.
Evaluation Parameters
Droplet size & zeta potential [17]
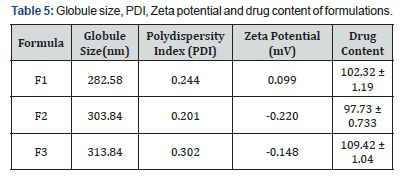
Droplet size and zeta potential were measured with the help of Nanophox particle size analyzer and Malvern zeta sizer 6.32(Malvern Instruments, Malvern, UK). The Fluoxetine HCL microemulsion was diluted in double-distilled water at 25°C for analysis. The mean Z-average and globule size was obtained by cumulate analysis using the NANOPHOX and MALVERN software.
Measurement of pH [9]
The pH of the microemulsion was measured with digital pH meter. The pH of the nasal cavity and pKa of a particular drug should be considered in order to optimize systemic absorption and to avoid nasal irritation. The delivery volume is limited due to the size of the nasal cavity. An upper limit of 20mg/dose and a volume of 25 to 200μl/ nostril have been suggested in order to: Avoid irritation of nasal mucosa, Allows the drug to be available in unionized form for absorption, Sustains normal physiology of ciliary movement.
Drug content [17,18]
To determine drug content of Microemulsion containing Fluoxetine HCL, 0.5ml of formulation was dissolved in 100ml of pH 6.2 phosphate buffer followed by stirring for 5 minutes and absorbance was recorded at 226nm with the proper dilutions in the same buffer solution. The drug content of all batches was determined by UV spectrometer.
Viscosity measurement
Higher the viscosity of the formulation greater is the contact time between the drug and the nasal mucosa thereby increasesin the time for drug permeation. At the same time, highly viscous formulations may interfere with the normal functions like ciliary beating, muco ciliary clearance and thus alter the permeability of drug. The viscosity values were measured using the Brookfield digital viscometer. The viscosities of all batches of nasal mucoadhesive microemulsion were measured with spindle number SV 18 at 20rpm.
Nasal ciliotoxicity studies [9,19,20]
The method described by Sheetal [9]. was used for this study; pieces of freshly excised sheep nasal mucosa with a thickness of 0.32mm were exposed to Mucoadhesive ME containing Fluoxetine HCl for 2h followed by thorough rinsing with PBS pH 6.2. In two other different sets of experiments isopropyl alcohol (a strong mucociliary toxin) and PBS pH 6.2 were used instead of Mucoadhesive ME containing for arriving at a comparative analysis of the extent of damage caused by the preparation. These pieces of mucosa were fixed in paraffin blocks and fine sections were taken that were stained properly by eosin and hematoxylin. The prepared slides were examined with an opticalmicroscope (B1 Advanced Series 223, Motic, Japan) and photomicrographs (magnification 400_) were taken.
Ex vivo drug release
To determine the release pattern of drug Fluoxetine HCl from microemulsion formulation, ex vivo drug diffusion study was carried by using 6 Stations Franz diffusion cell having two chambers a donor and a receptor compartment with 20mL capacity and 2cm diameter. A sheep nasal mucosa was fixed between donor and receptor chamber. Before starting the experiment, the piece of sheep nasal mucosa with desired dimension was soaked in the phosphate buffer pH 6.2 for 24h. The receptor compartment was filled with 20ml of phosphate buffer pH 6.2 and to maintain sink conditions for Fluoxetine HCl. The receptor solution was kept at constant temperature of 37 0C ± 0.50C and was maintained at constant magnetic stirring of 200rpm to mimic in vivo conditions. Simulated nasal fluid (0.45ml) was added to the dialysis membrane facing to the donor chamber and immediately 1ml of formulation was added to it. At specific time interval of 5min, 30min, 1h, 2h, 3h, 4h, and 5h, 1ml solution was withdrawn from the receptor compartment and1ml of blank receptor solution was added to the receptor chamber to maintain constant volume. The aliquots (1mL) removed was suitably diluted and analysed using UV spectrophotometer (Figure 1).
Stability studies [21-23]
Stability studies were performed as per ICH guidelines by storing formulations at 35±2 0C and 65 % RH for 30 days. The samples were analysed spectrometrically for the estimation of Drug Content, pH and Globule Size at days 0, 15 and 30.
Formulation Development and Evaluations
Solubility study of fluoxetine HCl
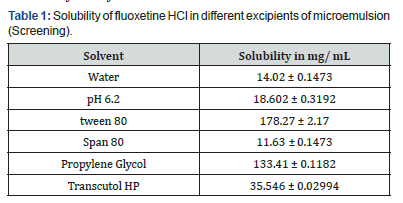
Table 1 shows solubility of Fluoxetine HCl in water (14.021mg/ mL), various surfactants and cosurfactants. The solubilisation of Fluoxetine HCl was highest in Tween 80 (178.27mg/mL) when compared with the other Surfactants. Additionally, Propylene Glycol was selected as a co-surfactant due to its efficient solubilizing effect (127.3mg/ml). All oils were evaluated for their penetration enhancing activity which is compared with plane aqueous solution of Fluoxetine HCl. It was found that the Cedar oil has highest penetration power amongst selected oils and sandalwood oil was having lowest penetration enhancing capability (Table 2).
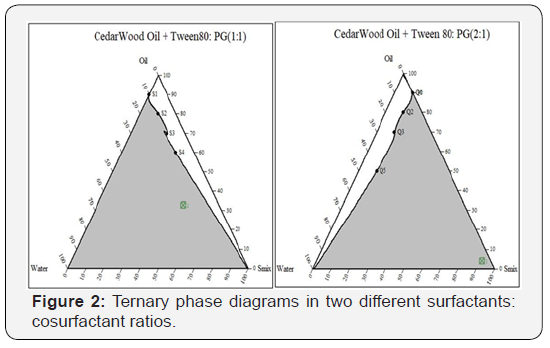
Pseudo ternary phase diagram
Based on solubility studies selected oil (Cedar oil, surfactant (Tween 80) and cosurfactant (Propylene Glycol) were used to prepare mucoadhesive microemulsion. The surfactant and cosurfactant were mixed at different mass ratios. Pseudo ternary phase diagram were constructed by using chemix software in which Smix was prepared at different mass ratios (1:1 and 1:2). In both the Smix ratios the microemulsion region was determined by visual observation highlighted in ternary phase Diagrams (Figure 2).
Viscosity measurement
Higher the viscosity of the formulation greater is the contact time between the drug and the nasal mucosa thereby increases in the time for drug permeation. At the same time, highly viscous formulations may interfere with the normal functions like ciliary beating, mucociliary clearance and thus alter the permeability of drug. The viscosity values were measured using the Brookfield digital viscometer. The viscosities of all batches of in situ nasal gel were measured before and after gelation by spindle number SV18 at 20rpm. (Table 3)
pH of the formulation
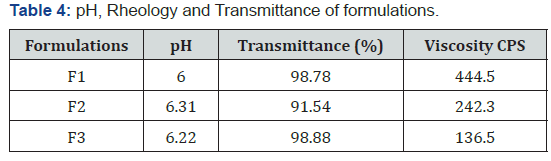
All formulations show pH range in between 4.5 and 6.4 which iswell suited to the nasal pH. Because, lysozyme is present in the nasal secretions, which is responsible for destroying certain microbes at acidic pH. Under alkaline pH lysozyme become inactive and thus nasal tissue becomes susceptible to microbial infection (Table 4).
Transmittance
Transmittance was found to be greater than 99% which confirmed that prepared Fluoxetine HCl microemulsion was transparent (Table 4).
Experimental Work
Drug content
Fluoxetine HCl content was found to be 97.73 % in optimized batch determined by UV spectroscopy
Globule size and zeta potential
The average Globule size of the optimized batch of Fluoxetine HCl microemulsion was found to be 303.84 nm. The homogenization speed significantly affects the particle size. Globule size was also found to be affected by concentration of Carbopol 940P. For controlled-delivery based on nanomedicine concept, the optimal formulation would be the one that would reveal the lowest particle size. The zeta potential of optimized batch was found to be -0.148mV (which indicates little stability of the formulation). The wide range of particle size may be due to swelling nature of polymer Carbopol 940P. The Particle size and zeta potential of the optimized batch is shown in (Table 5).
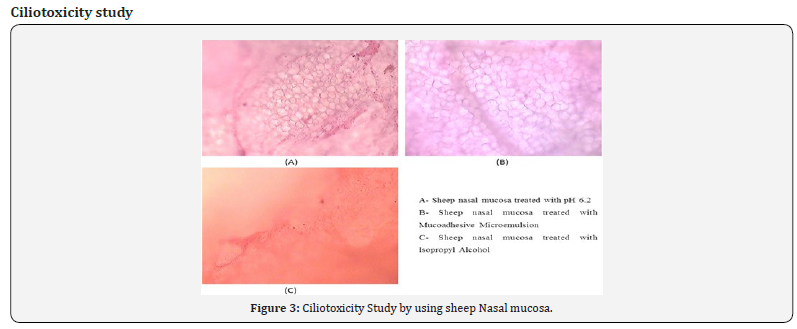
Photomicrographs of sheep nasal mucosa after treating with formulated microemulsion was observed for histopathological changes in comparison with the 6.2 pH Phosphate buffer and Isopropyl alcohol treated mucosa. Histological inference ofphotomicrographs shows that the section of mucosa treated with formulation had no degeneration of nasal epithelium along with no erosion. There was no sign of remarkable destructive effect of formulations on the treated nasal mucosa (Figure 3).
Ex vivo drug release studiesl
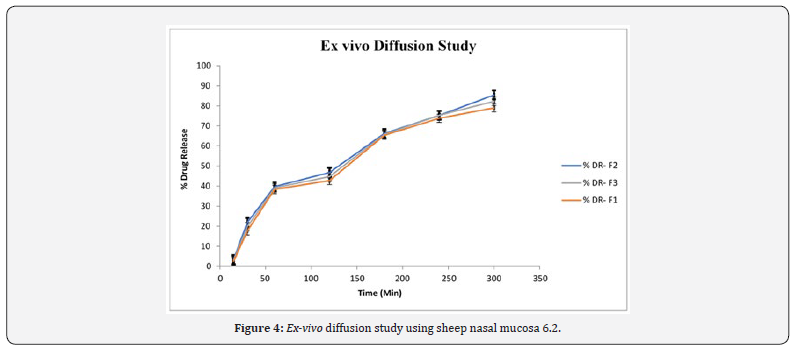
Formulations F1, F2 and F3 were subjected to Ex vivo release study with the help of 6 station Franz diffusion cell. This study was carried out using dissolution media, pH 6.2 phosphate buffer using sheep nasal mucosa. The release profile of Fluoxetine HCl from mucoadhesive microemulsion showed a burst release initially followed by a sustained release over the experimental period. The initial rates of drug release for Fluoxetine HCl were very rapid because the drug is hydrophilic in nature and was present in water phase (external phase) of microemulsion or due to incomplete gel formation but as the time progresses the release rate decreases. The Ex vivo release profiles of Fluoxetine HCl after 5hrs of F1 to F3 batches are depicted in (Figure 4).
Stability Study
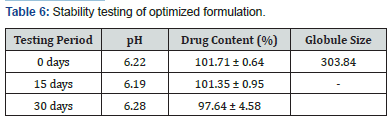
The formulation was assessed for stability according to ICH guidelines and the following parameters were taken into consideration pH, drug content and globule size and the formulation was found to be stable (Table 6).
Conclusion
The prepared microemulsion containing Fluoxetine HCl have potential to deliver the drug efficiently to the brain by nasal administration. The prepared microemulsion shown mean globulesize of 303.84nm. Also, it is non-irritant for nasal cavity which was confirmed by ciliotoxicity study by using sheep nasal mucosa. An evident from ex vivo diffusion study that it can deliver drug for longer time, thus the microemulsion containing Fluoxetine HCL might be a good option for future developments to deliver such drugs to the brain.
References
- https://www.who.int/en/news-room/fact-sheets/detail/depression
- Malhi GS, Mann JJ (2018) Depression. The Lancet, pp. 1-8.
- (2017) Depression and Other Common Mental Disorders: Global Health Estimates. Geneva: World Health Organization Licence: CC BY-NC-SA 3.0 IGO.
- Jackson SW (1986) Melancholia and depression: From Hippocratic Times to Modern Times. New Haven: Yale University Press, CT, USA.
- https://www.mentalhealthamerica.net/conditions/depression
- Sekerdag E (2017) Challenges of the Nose-to-Brain Route. Nanotechnology Methods for Neurological Diseases and Brain Tumors, pp. 103-113.
- Mandal S, Mandal SD, Chuttani K, Subudhi BB (2015) Mucoadhesive Microemulsion of Ibuprofen: Design and Evaluation for Brain Targeting Efficiency Through Intranasal Route. Brazilian Journal of Pharmaceutical Sciences 51(3): 721-731.
- Gänger S, Schindowski K (2018) Tailoring Formulations for Intranasal Nose-to-Brain Delivery: A Review on Architecture, Physico-Chemical Characteristics and Mucociliary Clearance of the Nasal Olfactory Mucosa. Pharmaceutics 10(3): 116.
- Pardeshi CV, Belgamwar VS (2013) Direct Nose to Brain Drug Delivery via integrated Nerve Pathways Bypassing the Blood–Brain Barrier: An Excellent Platform for Brain Targeting. Expert Opinion on Drug Delivery 10(7): 957-972.
- Agrawal M, Saraf S, Saraf S, Antimisiaris SG, Chougule MB, et al. (2018) Nose-To-Brain Drug Delivery: An Update on Clinical Challenges and Progress towards Approval of Anti-Alzheimer Drugs. Journal of Controlled Release 281: 139-177.
- Madhav S, Gupta D (2011) A Review on Microemulsion Based System. International Journal of Pharmaceutical Sciences and Research 2(8): 1888-1899.
- Espinoza LC, Vacacela M, Clares B, Garcia ML, Fabrega MJ, et al. (2018) Development of a Nasal Donepezil-loaded Microemulsion for the Treatment of Alzheimer ’s disease: in vitro and ex vivo CNS & Neurological Disorders - Drug Targets 17(1): 43-53.
- Yadav V, Jadhav P, Kanase K, Bodhe A, Dombe S (2018) Preparation and Evaluation of Microemulsion Containing Antihypertensive Drug. International Journal of Applied Pharmaceutics 10(5): 138-140.
- Patel V, Kukadiya H, Mashru R, Surti N, Mandal S (2010) Development of Microemulsion for Solubility Enhancement of Clopidogrel. Iran J Pharm Res 9(4): 327-334.
- Dixit GR, Mathur VB (2015) Microemulsions: Platform for Improvement of Solubility and Dissolution of Poorly Soluble Drugs. Asian Journal of Pharmaceutical and Clinical Research 8(5): 7-17.
- Patel RB, Patel MR, Bhatt KK, Patel BG (2013) Formulation Consideration and Characterization of Microemulsion Drug Delivery System for Transnasal Administration of Carbamazepine. Bulletin of Faculty of Pharmacy, Cairo University 51: 243-253.
- Kale SN, Deore SL (2016) Solubility Enhancement of Nebivolol by Micro Emulsion Technique. Journal of Young Pharmacist 8(4): 356-367.
- Solanki SS, Sarkar B, Dhanwani RK (2012) Microemulsion Drug Delivery System: For Bioavailability Enhancement of Ampelopsin. ISRN Pharmaceutics 1-4.
- Wang Y, Li M, Qian S, Zhang Q, Zhou L, et al. (2016) Zolpidem Mucoadhesive Formulations for Intranasal Delivery: Characterization, In Vitro Permeability, Pharmacokinetics, and Nasal Ciliotoxicity in Rats. Journal of Pharmaceutical Sciences 105(9): 2840-2847.
- Naik A, Nair H (2014) Formulation and Evaluation of Thermosensitive Biogels for Nose to Brain Delivery of Doxepin. Hindawi Publishing Corporation 20: 1-10.
- International Conference on Harmonisation (ICH) of Technical Requirements for Registration of Pharmaceuticals for Human use ICH Harmonised Tripartite Guideline Stability Testing of New Drug Substances and Products Q1A (R2).
- Vicentini FT, Vaz MM, Fonseca YM, Bentley MV, Fonseca MJ (2010) Characterization and stability study of a water-in-oil microemulsion incorporating quercetin. Drug Development and Industrial Pharmacy 37(1): 47–55.
- Thakkar H, Patel A, Chauhan N (2014) Formulation and optimization of mucoadhesive microemulsion containing mirtazapine for intranasal delivery. Chronicles of Young Scientists 5(1): 25.






























