Post-Surgery of Glioma, Delirium and Anti IGF-I Therapy
Jerzy Trojan1*, Basílio Vagner2, Beatriz H Aristizabal2, Ignacio Briceno3* and Annabelle Trojan4
1ICGT Bogota / Paris, Laboratories Legrand, Bogota and INSERM, Paris, France
2HPTU Hospital, and Antioquia University, Medellin, Colombia
3Institute of Human Genetics, PUJ - Javeriana University, Bogota, Colombia
4Faculty of Medicine, FUJNC - Corpas University, Bogota, Colombia
Submission: August 31, 2018;Published: September 07, 2018
*Corresponding author: Jerzy Trojan and Ignacio Briceno, Institute of Human Genetics, PUJ University, Bogota, Colombia, Tel: +57 320 993 18 85; Email: genetherapy@hotmail.fr
How to cite this article: Jerzy Trojan, Basílio Vagner, Beatriz H Aristizabal, Annabelle Trojan. Post-Surgery of Glioma: Delirium and Anti IGF-I Therapy. Theranostics Brain Spine Neuro Disord. 2018; 3(5): 555622. DOI: 10.19080/TBSND.2018.03.555622
Abstract
Postoperative delirium (POD) is one of common adverse outcome in patients after major surgery including brain tumor therapy. The presented review summarizes the theories of etiology and possible pathophysiological mechanism of delirium, as well as the laboratory diagnosis and the current treatment of delirium. In pathogenesis, the role of activation of inflammatory response is underlined in relation with alterations in neurohormonal signaling including growth factor IGF-I. This growth factor is postulated as a principal regulator of the connection between brain and pathophysiological mechanism as inflammation, cognitive impairment of delirium and brain neoplastic processes. The inflammatory response in delirium is accompanied by immunosuppression and increased synthesis of IGF-I factor. This “switch of the light on” factor of any pathological processes is also characteristic for glioblastoma multiforme, GBM, presenting an overexpression of IGF-I and immunosuppression. Logically, as long as a therapy of GBM and its POD is considered, the IGF-I and immunosuppression should be targeted in both processes. The treatment of glioblastoma, whose mortality remains close to 100%, still constitutes the challenge. Applying the post-operative immuno-gene therapy based on anti - gene anti IGF-I approach inducing in vivo immune response, this strategy has permitted to stop the neoplastic development. By the way, post-surgery by-sides effects including delirium were not observed. The treatment has increased the median survival of GBM patients up to two years (in current post-operative pharmacology therapy, the maximum survival is as 14 and rarely up to 18 months).
Keywords: Glioma; Post-surgery Delirium; Inflammatory response; IGF-I; Anti-gene immunotherapy
Introduction
Glioma and Delirium
Glioblastoma multiforme, GBM, constitutes the most aggressive malignant brain tumor [1,2]. GBM presents a dismal outcome and an obvious impact on quality of life, because of neurological, physical and cognitive problems, as well as personality and behavioral changes [3]. Using radiotherapy and chemotherapy, the mortality remains close to 100% and the median survival, using conventional therapy, is 9-14 months. Current pharmacology increases the survival to 15 and rarely to 18 months [4]. The etiology of glioma is still being investigated using molecular biology techniques [5]. New or proposed therapies are based either on immune treatment or on immuno-gene strategies [6-8]. The immunogen therapy is postoperative therapy based on genetically modified glioma cells (anti - tumor vaccines) provided from surgical biopsy. Nevertheless, we need to consider a factor of post trauma lesions of glioblastoma surgery, especially delirium. Moreover, the delirium constitutes a postoperative neurological morbidity in glioblastoma whose risk factors, incidence, and prognostic implications remain undefined [9]. Delirium is characterized by fluctuating disturbances in attention, memory, orientation, perception, psychomotor behavior, and sleep. Postoperative delirium (POD) is a common adverse outcome in patients after major surgery with incidence of up to 65%; POD has been linked to higher rates of inpatient mortality and morbidity [10,11].
Etiology of Delirium
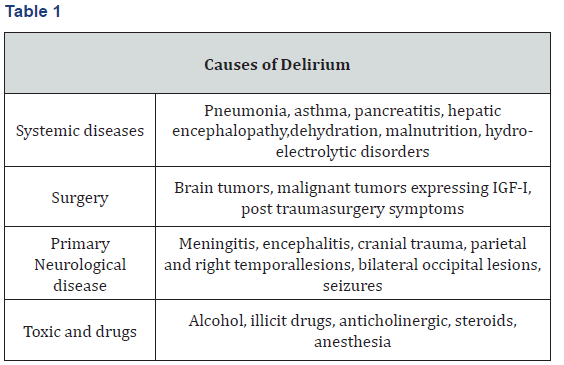
The etiology of delirium is complex. Known delirium risk factors include, among others, previous cognitive impairment, dementia, opioid pain treatment, exposure to benzodiazepines. There is a great diversity of factors for delirium [9,12-14] (Table 1). To help identify these mechanisms, the authors suggest that delirium etiologies may be classified into two major categories:
(a) direct brain insults, for example, hypotension, hypoxia, hypercapnia, brain infarcts, brain hemorrhage, brain tumor, trauma, and drugs and
(b) aberrant stress responses, induced by aberrations in the normally adaptive systemic and central nervous system (CNS) responses to stressors such as infection, surgical trauma, anxiety, and so forth. In summary there are four main causes that can trigger delirium: systemic medical conditions, primary neurological disease and medicine or toxic, either for use or removal of this (Table 1).
Discussion
Pathophysiology of delirium
Despite being a common disease, its pathophysiology remains poorly understood [12,13]. In pathogenesis, the following elements could play a role: activation of inflammatory response, oxidative stress, apoptosis, and alterations in neurohormonal signaling including IGF-I [11]. Especially IGF-I is postulated as a principal regulator of the connection between brain and pathophysiological mechanism as inflammation and neoplastic processes [8,15]. The relationships between IGF-I, cognitive impairment and inflammatory processes support a possible role in delirium pathogenesis [14]. As to the inflammatory response and delirium, the symptoms of delirium certainly occur in humans exposed to infection, surgery, or injury. The alterations of behavior occurring during immune stimulation appear to be coordinated by CNS synthesis of pro-inflammatory mediators such as cytokines and prostaglandins, nitric oxygen, accompanied by increased synthesis of cortisol and IGF-I [1].
Moreover, the activation of autonomous nervous system (catecholamines), immune response (PMNs cells, lymphocytes, macrophages) [16,17] and AFP [18]. Presence, are included in the processes. By the way, there are a number of routes by which a systemic inflammatory signal can be transduced into the brain s. This hyperinflammatory response is accompanied by increasing immunosuppression as well humoral as cellular, the last concerning monocytes, NK cells and T lymphocytes - with diminished expression of HLA-DR (MHC-I, MHC-II, TAP 1, TAP 2 [19-22]. Considering IGF-I, as well precancerous diagnostic of gliomas, as the most efficient post-surgery gene therapy of this tumor, are both targeting IGF-I [23]. In this context, as far as delirium is considered being a postsurgery lesion, it exists an association between serum insulin-like growth factor-I (IGF-I) levels and postoperative delirium, POD; in other pathologies, the recent clinical studies have assessed also a preoperative serum IGF-I levels as a predictor of incident delirium [11]. We need underline a strike difference: as IGF-I increased serum level is related to precancerous diagnostic [24,25]. a statistically significant association between lower preoperative plasma IGF-I levels and increased incidence and severity of POD was demonstrated [11].
According to some observations, hypothalamic–pituitary– adrenal axis hyperresponsiveness and a less frequent suppression of the GH/IGF-1 axis in response to acute stress are possibly involved in delirium pathophysiology [26]. To predict development of POD, other authors are studying the problem using 2 tumor-specific (bihemispheric tumors and tumor size) and 3 patient-specific (age, psychiatric history, and chronic pulmonary disease) factors [9]. Several neurotransmitter alterations and deficiencies have been held responsible for the occurrence of delirium as the synthesis and release of acetylcholine, noradrenaline, serotonin, dopamine, GABA, and glutamate. Changes in the other neurotransmitters very probably also play an important role, an excess of glutamate and dopamine being held responsible for cellular damage and the psychotic features of delirium [10,27]. Moreover, the intracellular pathways (involving G proteins, calcium, the phosphatidylinositol cascade, the cyclic nucleotides AMP and GMP, and protein phosphorylation) and signal transduction systems including receptor of IGF-I, and its role in glioma pathophysiology (TK, PI3k, AKT, GSK3, Bcl2), are of importance in the synthesis and release of neurotransmitters [28,29].
Diagnosis and Treatment of Delirium
As described above, delirium may be caused by many pathophysiological changes and its cause is not always obvious [30]. In 66% of cases it is unknown, and diagnosis is not done [31]. The diagnosis is important because some patients can progress to convulsions, stupor, coma and death [31,32]. The most important laboratory diagnostic exams to establish the delirium’s etiology, besides the clinical symptoms, are laboratory tests: hemogram with sedimentation, electrolytes, blood urea nitrogen (BUN), creatinine, glucose, arterial blood gases, inflammatory cytokines (IL-1b, IL-10, IL-12, IL-15, IL-17, IL-18, IL-23, IFNg, TNFa, TNFb), THS, T4 free, serum IGF-I, TGFbeta and AFP as well as molecules HLA, TAP-1 and- 2. Moreover, lumbar puncture and toxic presence in blood and urine are performed [9,11,14,22,33]. In the management of delirium, the most important aspect is to try to detect the etiology and treat it of according to the established protocols for each one [34].
Antipsychotic drugs are recommended for symptoms of delirium. Haloperidol is effective and is the most studied drug to treat delirium, having few anticholinergic effects, little cardiovascular effects, high potency and low sedative effect. Atypical antipsychotics such as risperidone, olanzapine, quetiapine have demonstrated utility in delirium. Benzodiazepines are effective alternative treatment when antipsychotics are not effective [35- 37]. As far as post-surgery delirium is related to glioma, the first point of treatment is to stop a neoplastic reappearance of the tumor. Fortunately, from some years, the application of immunotherapy and immunogen therapy targeting growth factors, TGFbeta or IGF-I, has stopped the symptoms related to neoplastic development, including delirium, due to anti-tumor immune response [8,38]. Especially the immunogen therapy, based on anti – gene anti IGFapproach, was efficient. This therapy is targeting IGF-I and an immunosuppression, both elements present as well in delirium as in glioma [23].
Targeting the IGF-I system has emerged as a useful method to reduce glial malignant development. In practice, when human glioma cells, derived from glioblastoma biopsy, are transfected in vitro with vectors expressing either IGF-I antisense RNA or inducing IGF RNA-DNA triple helix, the synthesis of IGF-I is stopped on translation or transcription levels, respectively (anti–gene strategy). Down regulation in the expression of IGF-I coincides with the reappearance of B7 and MHC class I antigens at the surface of transfected cells (immunogenicity). When injected subcutaneously, the transfected cancer cells, “vaccines”, initiate an immune reaction involving CD8+ and CD28+ lymphocytes, followed by tumor regression. The patients treated by classical surgery and radiotherapy, were “vaccinated” by three successive injections. The median survival of treated patients was 2 years, on some cases 3 and 4 years [23].
Conclusion
In presented review, it has only been possible to review some of the main research issues concerning the etiology, pathophysiology, diagnosis and treatment of delirium. Further investigation into the pathophysiological mechanisms of delirium requires the use of homogeneous populations of patients, combining different objective methods for characterization of delirium, together with the simultaneously measuring of biochemical and neurotransmitter parameters and functional brain imaging. However, these types of time consuming studies, demand a combined effort of different clinical research centers to continue basic studies on delirium, with the ultimate aim of finding effective intervention strategies for this very complicated syndrome. It seems, that to determine the true prevalence of biochemical and clinical changes accompanied the delirium, only shortly signaled in the reported review, prospective investigations are needed using proper definitions of personality and behavioral changes [3]. Especially the study of pathophysiology of delirium, in the context of postoperative trauma, including surgery of glioma treatment, should consider the relation between inflammatory and immune responses, both related to IGF-I presence [8,11,14,26].
References
- Trojan J, Cloix JF, Ardourel MY, Chatel M, Anthony DD (2007) Insulinlike growth factor type 1 biology and targeting in malignant glioma. Neurosci 145(3): 795-811.
- Wrensch M, Rice T, Miike R, McMillan A, Lamborn KR, et al. (2006) Diagnostic, treatment, and demographic factors influencing survival in a population-based study of adult glioma patients in the San Francisco Bay Area. Neuro-Oncol 8(1): 12-26.
- Zwinkels H, Dirven L, Vissers T, Habets EJJ, Vos MJ, et al. (2016) Prevalence of changes in personality and behavior in adult glioma patients: a systematic review. Neuro-Oncol Pract 3(4): 222-223.
- Stupp R, Hegi ME, van den Bent MJ, Mason WP, Weller M, et al. (2006) Changing paradigms--an update on the multidisciplinary management of malignant glioma. Oncologist 11(2): 165-180.
- Jiang R, Mircean C, Shmulevich I, Cogdell D, Jia Y, et al. (2006) Pathway alterations during glioma progression revealed by reverse phase protein lysate arrays. Proteomics 6(10): 2964-2971.
- Kjaergaard J, Wang L, Kuriyama H, Shu S, Plautz GE (2005) Active immunotherapy for advanced intracranial murine tumors by using dendritic cell-tumor cell fusion vaccines. J Neurosurg 103(1): 156-164.
- Dietrich PY, Dutoit V, Tran Thang NN Walker PR (2010) T cell immunotherapy for malignant glioma: toward a combined approach. Curr Opinion Onco 22(6): 604-610.
- Trojan J (2017) Cancer immunogene therapy. Anti - gene anti IGF-I approach. Case of glioblastoma. Ed. Lambert Academic Publishers (LAP), Saarbrucken, Germany, pp. 1-140.
- Flanigan PM, Jahangiri A, Weinstein D, Dayani F, Chandra A, et al. (2018) Postoperative delirium in glioblastoma patients: Risk factors and prognostic implications. Neurosurgery.
- Meagher D (2009) Motor subtypes of delirium: past, present and future. Int Rev Psychiatry 21(1): 59-73.
- Yen TE, Allen JC, Rivelli SK, Patterson SC, Metcalf MR, et al. (2016) Association between serum IGF-I levels and postoperative delirium in elderly subjects undergoing elective knee arthroplasty. Sci Rep 6.
- Flacker JM, Lipsitz LA (1999) Neural mechanisms of delirium: current hypotheses and evolving concepts. J Gerontol A Biol Sci Med Sci 54(6): B239-B246.
- Trzepacz PT, Baker RW, Greenhouse J (1988) A symptom rating-scale for delirium. Psychiatry Res 23(1): 89-97.
- Adamis D, Meagher D (2011) Insulin-Like Growth Factor I and the pathogenesis of delirium: A review of current evidence. J Aging Res.
- Pollak MN, Schernhammer ES, Hankinson SE (2004) Insulin-like growth factors and neoplasia. Nat Rev Cancer 4(7): 505-518.
- Westborough JP (2000) The stress response to trauma and surgery. Br J Anaesth 85(1): 109-117.
- Lenz A, Franklin GA, Cheadle WG (2007) Systemic inflammation after trauma. Injury 38(12): 1336-1345.
- Trojan J, Naval J, Jusforgues H, Uriel J (1989) Alphafetoprotein (AFP) in granulomatous inflammation of the mouse. Brit J Exp Path 70(4): 469-478.
- Dantzer R, Capuron L, Irwin MR, Millers AH, Ollat H, et al. (2008) Identification and treatment of symptoms associated with inflammation in medically ill patients. Psychoneuroendocrino 33(1): 18-29.
- Bandyopadhyay G, Laudanski K, Li F, Lentz C, Bankey P, et al. (2007) Negative signaling contributes to T-cell anergy in trauma patients. Crit Care Med 35(3): 794-801.
- Trabado S, Van Binh PN, Martin C, Lafarge Frayssinet C, Lone YC, et al. (2007) Stimulation of anti-melanoma immune effectors via modified tumour cells exhibiting inhibited IGF-I and low CD9 Biomed & Pharmacother 61(8): 494-498.
- Pan Y, Trojan J, Guo Y, Anthony DD (2013) Rescue of MHC-1 antigen processing machinery by down-regulation in expression of IGF-I in human glioblastoma cells. PLoS One 8(3): e58428.
- Trojan J (2018) Brain - from development to neoplasia and gene therapy solution. Ed. Lambert Academic Publishers (LAP), Saarbrucken, Germany, pp.s 1-1500.
- Zumkeller W (2002) IGFs and IGF-binding proteins as diagnostic markers and biological modulators in brain tumors. Expert Rev Mol Diagn 2(5): 473-477.
- Trojan A, Aristizabal B, Jay LM, Castillo T, Penagos P, et al. (2016) Testing of IGF-I biomarker in an ethical context. Adv Modern Onco Res 2(4).
- Cerejeira J, Batista P, Nogueira V (2013) The stress response to surgery and postoperative delirium. Evidence of hypothalamic-pituitaryadrenal axis hyperresponsiveness and decreased suppression of the GH/IGF-1 axis. J Geriatric Psychiatry Neuro 26(3): 185-194.
- Benke T, Delazer M, Bartha L, Auer A (2003) Basal ganglia lesions and the theory of fronto-subcortical loops: neuropsychological findings in two patients with left caudate lesions. Neurocase 9(1): 70-85.
- Lammie GA (2000) Pathology of small vessel stroke. Br Med Bull 56(2): 296-306.
- Trojan J, Anthony DD (2011) Antisense strategies in therapy of gliomas. Curr Signal Transd T 6(3): 411-423.
- Burns A, Gallagley A, Byrne J (2004) Delirium. J Neurol Neurosurg Psychiatry 75(3): 362-367.
- Leonard M, Raju B, Conroy M, Donnelly S, Trzepacz PT, et al. (2008) Reversibility of delirium in terminally ill patients and predictors of mortality. Palliat Med 22(7): 848-854.
- Samuels SC, Evers MM (2002) Delirium. Pragmatic guidance for managing a common, confounding, and sometimes lethal condition. Geriatrics 57(6): 33-38.
- Pae CU, Marks DM, Patkar AA, Masand P (2008) Delirium: underrecognized and undertreated. Curr Treat Options Neurol 10(5): 386-395.
- Miller MO (2008) Evaluation and management of delirium in hospitalized older patients. Am Fam Physician 78(11): 1265-1270.
- Skrobik YK, Bergeron N, Dumont M, Gottfried SB (2004) Olanzapine vs Haloperidol: treating delirium in a critical care setting. Intensive Care Med 30(3): 444-449.
- Lonergan E, Britton AM, Luxenberg J, Wyller T (2007) Antipsychotics for delirium. Cochrane Database SYst Rev 18(2): CD005594.
- Gagnon PR (2008) Treatment of delirium in supportive and palliative care. Curr Opin Support Palliat Care 2(1): 60-66.
- Hau P, Jachimczak P, Bogdahn U (2009) Treatment of malignant gliomas with TGF-beta2 antisense oligonucleotides. Expert Rev Anticancer Ther 9(11): 1663-1674.






























