Tuberculous Hypertrophic Pachymeningitis
Adrian Kelly* and Patrick Lekgwara
Department of Neurosurgery, Sefako Makgatho Health Sciences University, South Africa
Submission: August 14, 2018;Published: August 27, 2018
*Corresponding author: Adrian Kelly, Department of Neurosurgery, 3rd floor Sciences Building, Sefako Makgatho Health Sciences University, Pretoria , South Africa, Tel: +27846773771; Email: adriankelly1000@yahoo.co.uk
How to cite this article: Adrian Kelly, Patrick L. Tuberculous Hypertrophic Pachymeningitis. Theranostics Brain Spine Neuro Disord. 2018; 3(4): 555620. DOI: 10.19080/TBSND.2018.03.555620
Abstract
Tuberculosis infection of the central nervous system is diagnosed in a variety of forms which include tuberculous meningitis, tuberculomas and tubercular abscesses, tuberculous vasculitis, tuberculous encephalitis, tuberculous subdural empyema and spinal tuberculosis. In addition, each of these diagnoses may present with secondary complications which include hydrocephalus, cranial nerve palsies and stroke. Considering tubercular meningitis specifically it is noted to be the most common diagnosis of central nervous system tuberculosis occurring most frequently in young children and individuals with untreated HIV infection. The classical form of tuberculous meningitis is as a basal meningitis comprising thick exudates which as the disease progresses complicates by causing hydrocephalus and infarcts secondary to an obliterative arteritis of the small perforator vessels. A much rarer form of tuberculous meningitis is as a pachymeningitis affecting the meninges of the cranial vault. In certain cases, the inflammatory response may become exuberant leading to a hypertrophic form of pachymeningitis where considerable nodular or linear thickening of the dura mater occurs. This hypertrophic reaction may be localized or extensive and the extent of the disease as well as its location dictates the presenting symptoms. Histologically granulomatous change with caseous necrosis and Langerhans giant cells are seen in the tuberculous form of the disease. We present a mini review of hypertrophic pachymeningitis with emphasis on the tuberculous form of the disease.
Keywords: Tuberculous hypertrophic pachymeningitis; tuberculous meningitis; Mycobacterium; Blood-brain barrier; Granulomatous diseases
Introduction
Tuberculous meningitis can affect individuals of all age groups but occurs more commonly in young children and in individuals with untreated HIV infection [1]. While only representing 1% of all cases of tuberculosis the associated morbidity is alarmingly disproportionate in that over half of individuals whom present either die or are severely disabled despite treatment [2]. Several studies have identified certain strains of Mycobacterium tuberculosis that have an increased affinity to cause tuberculous meningitis such as the Indo-Oceanic and East Asian Beijing lineages which pose an increased risk as compared to the lower risk afforded by for example the Euro-American lineage [3]. The specific bacterial gene enabling the bacteria to cross the blood-brain barrier has been identified as the Rv0931c which codes for a specific serine/threonine protein kinase allowing endothelial invasion [4]. Besides the pathogen itself several host factors play an important role in the development and nature (Figure 1) in which tubercular meningitis presents. In healthy adult’s anorexia and malaise followed by a progressive headache and vomiting are the commonest presenting symptoms which in the early stages makes tuberculous meningitis difficult to differentiate from common illnesses such as influenza [5].
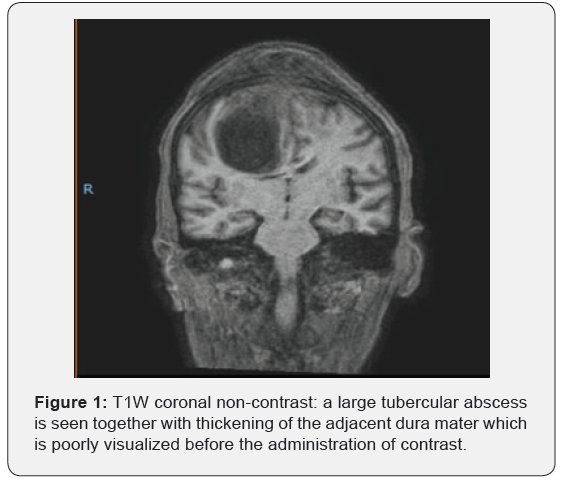
Neck stiffness is often not present during the early stages [6]. While classical described as a basal meningitis comprising thick exudates in certain cases tubercular meningitis may present as a pachymeningitis affecting the dura mater and leptomeninges of the cranial vault and tentorium [7]. In rare cases an exuberant inflammatory response may follow resulting in a hypertrophic form of the disease in which an inflammatory granulomatous mass is visualized, most commonly occurring in the skull base and posterior fossa in the cranial form of the disease [8]. As a disease hypertrophic pachymeningitis has several associations other than tuberculous which includes many of the well-known granulomatous diseases (Figure 2). These include syphilis, sarcoidosis, fungal and parasitic infections, Wegener’s granulomatosis, rheumatoid arthritis, mucopolysaccharidosis and multifocal fibrosclerosis [9]. There is also an idiopathic form of the disease which is histologically characterized by non-specific granulomatous change, proliferation of collagen fibers and an inflammatory cell infiltrate [10]. In questionable cases meningeal biopsy is hence essential to establish a diagnosis [11]. In this mini review we restrict our discussion to tuberculous hypertrophic pachymeningitis.
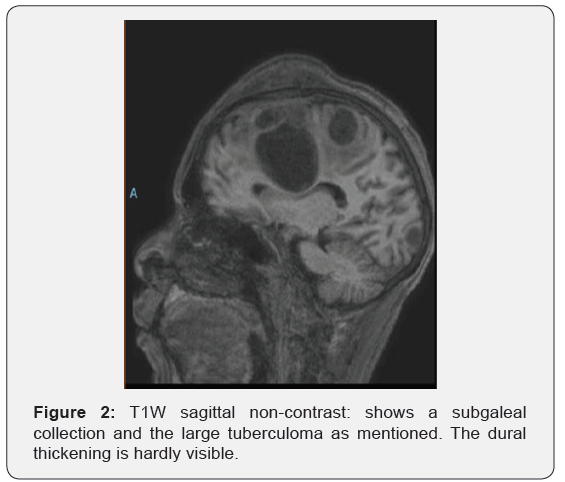
Results
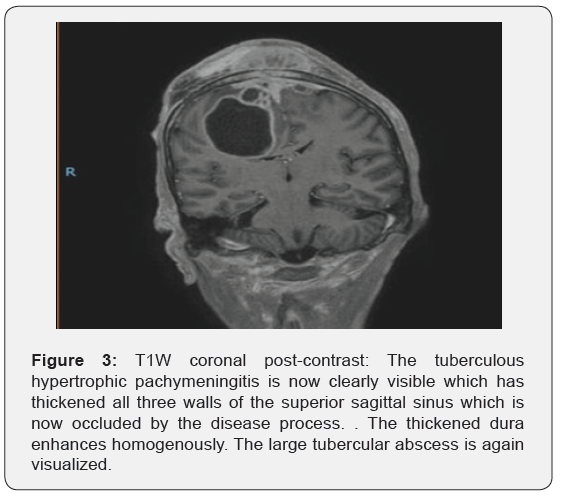
Patients with tuberculous hypertrophic pachymeningitis commonly present with generalized symptoms of raised intracranial pressure namely progressive headache, vomiting and blurring of vision. Other signs and symptoms are inconsistent and depend on the location of the disease but include cranial nerve palsies or a focal neurological deficit. Other complications include obstructive hydrocephalus and venous sinus thrombosis [12]. Neuroimaging reveals a dual based mass lesion which on MRI T1W imaging is isointense to hypo intense. MRI T2W (Figure 3).imaging shows the lesion to be similarly hypo intense. Post gadolinium administration the lesion shows a vivid homogenous enhancement pattern [12]. These signal characteristics are attributed to the fibrosis and necrosis of the dura mater and the proportion of each allows for some variation from these classical imaging characteristics [12,13].
Unfortunately, tuberculous hypertrophic pachymeningitis is progressive without treatment and once diagnosed by biopsy the aim of therapy is to arrest the inflammatory process with or without surgical debulking of the mass lesion itself. In terms of medical therapy systemic corticosteroids, cyclophosphamide and azathioprine in combination with anti-tuberculous treatment are established treatment options however come with significant side effects especially in patients co-infected with HIV [14-16]. The clinical outcome on purely medical therapy is non-consistent and ranges from a complete resolution of the mass lesion to progression of the disease requiring surgical debulking. In terms of response to therapy several studies note clinical improvement to correlate poorly with follow-up imaging studies and emphasize the former rather than the latter as the yardstick by which response to treatment should be evaluated [12,15,17].
Discussion
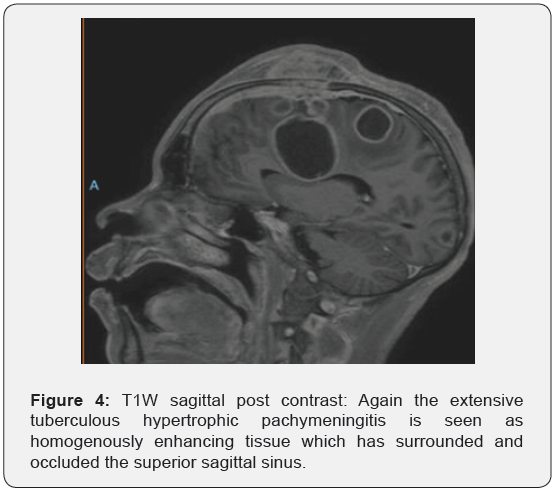
Tuberculous hypertrophic pachymeningitis is a rare inflammatory form of tuberculous meningitis that affects the dura mater and leptomeninges (Figure 4). It is this Dural involvement that accounts for the progressive headache which is the commonest presenting symptom in these patients [15]. The imaging characteristics described above are non-specific and the differential diagnosis includes meningioma a plaque, solitary fibroma, neurosarcoidosis, Dural carcinomatosis and lymphoma [18]. For this reason, biopsy is crucial to establish the diagnosis [19]. Fortunately, the histological characteristics of tuberculous hypertrophic pachymeningitis are relatively specific namely caseous necrosis and Langerhans giant cell infiltrates which differentiates it from the other causes of hypertrophic pachymeningitis [20]. Once confirmed to be tuberculous hypertrophic pachymeningitis patients are routinely started on systemic cortico-steroids and antituberculous treatment.
The benefits of administering corticosteroids together with antituberculous medication in the treatment of tuberculous meningitis have been clearly confirmed in seven randomized control trials where their use reduced death and disability by up to 30% [21]. In the context of tuberculous hypertrophic pachymeningitis the reduction in mass effect with the administration of corticosteroids is less predictable with some patients demonstrate a complete response while in others little change occurs requiring the use of second line agents or even surgical debulking [12,15,17]. What is noted is that the earlier the diagnosis is made and treatment instituted the greater the chance of clinical improvement [22]. Surgical debulking of the hypertrophic mass lesion has a role in treatment resistant cases and in patients deteriorating from mass effect and rapid clinical improvement is usually seen. Surgery alone is however rarely curative unless total excision is achieved [23].
Conclusion
Tuberculous hypertrophic pachymeningitis is a rare disorder with clinical outcome depending on expedient diagnosis as well as the extent and site of the disease. Patient factors such as coinfection with HIV are also important determinants of eventual outcome. Clinical response rather than radiological resolution is the cornerstone by which the effectiveness of treatment should be assessed.
References
- Zhang L, Tu D, Enarsen D (2000) The impact of directly-observed treatment on the epidemiology of tuberculosis in Beijing. Int J Tuberc Lung Dis 4(10): 904-910.
- Jarvis J, Meintjes G, Williams A, Brown Y, Crede T, et al. (2010) Adult meningitis in a setting of high HIV and TB prevalence: findings from 4961 suspected cases. BMC Infect Dis 10: 67.
- Caws M, Thwaites G, Dunstan S, Hawn TR, Lan NT, et al. (2008) The influence of host and bacterial genotype on the development of disseminated disease with Mycobacterium tuberculosis. PLos Pathog 4(3): e1000034.
- Be N, Bishal W, Jain S (2012) Role of Mycobacterium tuberculosis pknD in the pathogenesis of central nervous system tuberculosis. BMC Microbiol 12: 7.
- Smith H (1964) Tuberculous meningitis. Int J Neurol 4: 134-1357.
- Smith H, Vollum R (1954) The diagnosis of tuberculous meningitis. Br Med Bull 10: 140-144.
- van Well GT, Paes BF, Terwee CB, Springer P, Roord JJ, et al. (2009) Twenty years of pediatric tuberculous meningitis: a retrospective cohort study in the western cape of South Africa. Pediatrics 123(1): e1-8.
- Okimura Y, Tanno H, Kurasudani H, Suda S, Ono J, et al. (1989) A case of hypertrophic cranial pachymeningitis: assessment with CT and MR imaging. AJNR 10(3): 477-484.
- Berger J, Snodgrass S, Glaser J, Post M, Norenberg N, et al. (1989) Multifocal fibrosclerosis with hypertrophic intracranial pachymeningitis. Neurology 39(10): 1345-1349.
- Adler J, Sheridan W, Kosek J, Linder S (1991) Pachymeningitis associated with a pulmonary nodule. Neurosurgery 29(2): 283-287.
- Tariq R, Ahmed R (2012) Tuberculous hypertrophic Pachymeningitis presenting as visual blurring and headaches. J Pakistan Med Assoc 62(9): 966-968.
- Kazem I, Robinette N, Roosen N, Schaldenbrand M, Kin J (2005) Best cases from the AFIP: Idiopathic tumefactive hypertrophic pachymeningitis. Radiographics 25(4): 1075-1080.
- Panos G, Duarte G, Pooia F Panos G, Duarte G, Pooia F (2012) Idiopathic hypertrophic pachymeningitis mimicking neurosarcoidosis. Clinical Neurology and Neurosurgery 114(2): 176-178.
- Kupersmith M, Martin V, Heller G, Shah A, Mitnick H (2004) Idiopathic hypertrophic pachymeningitis. Neurology 62(5): 686-694.
- Wang Y, Fuh J, Ling J, Lu S, Wang S (2004) Headache profile in patients with hypertrophic cranial pachymeningitis. Headache 44(9): 916-923.
- Thwaites G, Nguyen D, Nguyen H, Hoang Thi Quy, Thi Tuong Oanh, Nguyen Thi Cam Thoa, et al. (2004) Dexa-methazone for the treatment of tuberculous meningitis in adolescents and adults. N Engl J Med 351: 1741-1751.
- Kleiter I, Hans V, Schuierer G, Marienhagen J, Han P, et al. (2004) Intraventricular cytarabine in a idiopathic hypertrophic pachymeningitis. J Neurol Neurosurg Psychiatry 75(9): 1346-1348.
- Martin N, Masson C, Henin D, Mompoint D, Marsault C, et al. (1989) Hypertrophic cranial pachymeningitis: Assessment with CT and MR imaging. AJNR 10(3): 477-484.
- Hatano N, Behari S, Nagatani T, Kimura M, Ooka K, et al. (1999) Idiopathic cranial pachymeningitis: clinic-radiological spectrum and therapeutic options. Neurosurgery 45(6): 1336-13343.
- Miranda M, Breiman A , Allain S, Deknuydt F, Altare F (2012) The Tuberculous Granuloma: An Unsuccessful Host Defence Mechanism Providing a Safety Shelter for the Bacteria? Clin Dev Immunol 2012: 139127.
- Prasad K, Singh M, (2008) Corticosteroids for managing tuberculous meningitis. Cochrane Database Syst Rev (1): CD002244.
- Lincoln E, Sordillo S, Davies P (1960) Tuberculous meningitis in children. J Pediatr 57: 807-823.
- Bucy P, Freeman L (1952) Hypertrophic spinal pachymeningitis. With special reference to appropriate surgical treatment. J Neurosurg 9(6): 564-578.






























