A Review Study on A Polymer Optical Nano sensor Named «Pourya-Zar Sensor« for Detection of ((Cu)) Heavy Metal Ions in Aquatic Environments
Pourya Zarshenas1*, Mehran AmirBeygi2, Elaheh Haghbin2 and Mohammad Amin Abdoos3
1Department of Inorganic Chemistry, Faculty of Chemistry & Petroleum Sciences, Shahid Beheshti University (SBU), Tehran, Iran
2Department of Polymer Chemistry, Faculty of Chemistry & Petroleum Sciences, Shahid Beheshti University (SBU), Tehran, Iran
3Medicinal Plants and Drugs Research Institute, Shahid Beheshti University (SBU), Tehran, Iran
Submission: March 09, 2023; Published: May 09, 2023
*Corresponding author: Pourya Zarshenas, Department of Inorganic Chemistry, Faculty of Chemistry & Petroleum Sciences, Shahid Beheshti University (SBU), Tehran, Iran
Pourya Z, Mehran A, Elaheh H, Mohammad Amin A. A Review Study on A Polymer Optical Nano sensor Named «Pourya-Zar Sensor« for Detection of ((Cu)) Heavy Metal Ions in Aquatic Environments. Recent Adv Petrochem Sci. 2023; 7(5): 555724.DOI: 10.19080/RAPSCI.2023.07.555724
Abstract
Environment can be referred to as the surroundings within which humans exist. These are made up of: the land, the water and the atmosphere of the earth; microorganisms, plant and animal life; any part or combination of the first two items on this list and the interrelationships among and between them and the physical, chemical, aesthetic and cultural properties and conditions of the foregoing that influence human health and well-being. It is also characterized by a number of spheres that influence its behavior and intrinsic value. The most important sphere of the environment is the biosphere because it harbors living organisms. This is the sphere where you find living organisms (plants and animals) interacting with each and their nonliving environment (soil, air and water). In the late centuries, industrialization and globalization have impaired pristine environments and their ability to foster life. This has introduced components that compromise the holistic functioning of the environment and its intrinsic values [1]. An environment can be polluted or contaminated. Pollution differs from contamination; however, contaminants can be pollutants, and pose detrimental impact on the environment. From literature, pollution is defined as the introduction by man, directly or indirectly, of substances or energy into the environment resulting in such deleterious effects as harm to living resources, hazards to human health, hindrance to environmental activities and impairment of quality for use of the environment and reduction of amenities. Contamination on the other hand is the presence of elevated concentrations of substances in the environment above the natural background level for the area and for the organism. Environmental pollution can be referred to undesirable and unwanted changes in physical, chemical and biological characteristics of air, water and soil which is harmful for living organisms-both animal and plants. Pollution can take the form of chemical substances or energy, such as noise, heat or light [2]. Pollutants, the elements of pollution, can either be foreign substances/energies or naturally occurring contaminants. Environmental pollutants continue to be a world concern and one of the great challenges faced by the global society. Pollutants can be naturally occurring compounds or foreign matter which when in contact with the environment cause adverse changes. There are different types of pollutants, namely inorganic, organic and biological. Irrespective of pollutants falling under different categories, they all receive considerable attention due to the impacts they introduce to the environment. The relationship between environmental pollution and world population has become an inarguable directly proportional relationship as it can be seen that the amount of potentially toxic substances released into the environment is increasing with the alarming growth in global population. This issue has led to pollution being a significant problem facing the environment. Industrial, agricultural and domestic wastes contribute to environmental pollution, which causes adverse harm to human and animal health. From such sources, inorganic pollutants are released. Inorganic pollutants are usually substances of mineral origin, with metals, salts and minerals being examples [2]. Studies have reported inorganic pollutants as material found naturally but have been altered by human production to increase their number in the environment. Inorganic substances enter the environment through different anthropogenic activities such as mine drainage, smelting, metallurgical and chemical processes, as well as natural processes. These pollutants are toxic due to the accumulation in the food chains [3]. Organic pollution can be briefly defined as biodegradable contaminants in an environment. These sources of pollution are naturally found and caused by the environment, but anthropogenic activity has also been contributing to their intensive production to meet human needs. Some of the common organic pollutants which have been noted to be of special concern are human waste, food waste, polychlorinated biphenyls (PCBs), polybrominated diphenyl ethers (PBDEs), polycyclic aromatic hydrocarbons (PAHs), pesticides, petroleum and organochlorine pesticides (OCPs) [4]. The definition of a sensor does not precisely define what physical elements constitute the sensor. For example, what portion of a thermocouple is the sensor? Is it solely the bimetallic junction? Does it include the wires used for transmission purposes? Does it include any packaging or signal processing? On the basis of information in the current technical literature, the committee chose to adopt the following definitions:
Sensor element: The fundamental transduction mechanism (e.g., a material) that converts one form of energy into another. Some sensors may incorporate more than one sensor element (e.g., a compound sensor).
Sensor: A sensor element including its physical packaging and external connections (e.g., electrical or optical).
Sensor system: A sensor and its assorted signal processing hardware (analog or digital) with the processing either in or on the same package or discrete from the sensor itself. In order to describe and characterize the performance of a sensor, a large and specific vocabulary is required. Several excellent references, which provide a basic review of transducer characteristics, are cited in the bibliography. Lion (1969) introduced a classification of principles according to the form of energy in which sensor signals were received and generated, which yielded a matrix of effects
In this article, we intend to investigate water pollution by heavy metals. Finally, we will review the methods for characterizing heavy metals by chemical sensors. So, Ladies and gentlemen! Welcome to the age of pollution.
Keywords: Heavy Metals; Water Pollution; Nano Sensors; Polymer Sensors; Optical Sensors; Water Environments; Cu; Copper
Introduction
Heavy metals are defined as metallic elements that have a relatively high density compared to water [1]. With the assumption that heaviness and toxicity are inter-related, heavy metals also include metalloids, such as arsenic, that are able to induce toxicity at low level of exposure [2]. In recent years, there has been an increasing ecological and global public health concern associated with environmental contamination by these metals. Also, human exposure has risen dramatically as a result of an exponential increase of their use in several industrial, agricultural, domestic and technological applications [3].
Reported sources of heavy metals in the environment include geogenic, industrial, agricultural, pharmaceutical, domestic effluents, and atmospheric sources [4]. Environmental pollution is very prominent in point source areas such as mining, foundries and smelters, and other metal-based industrial operations. Although heavy metals are naturally occurring elements that are found throughout the earth’s crust, most environmental contamination and human exposure result from anthropogenic activities such as mining and smelting operations, industrial production and use, and domestic and agricultural use of metals and metal-containing compounds [4-7].
Environmental contamination can also occur through metal corrosion, atmospheric deposition, soil erosion of metal ions and leaching of heavy metals, sediment re-suspension and metal evaporation from water resources to soil and ground water [8]. Natural phenomena such as weathering and volcanic eruptions have also been reported to significantly contribute to heavy metal pollution. Industrial sources include metal processing in refineries, coal burning in power plants, petroleum combustion, nuclear power stations and high-tension lines, plastics, textiles, microelectronics, wood preservation and paper processing plants [9-11].
It has been reported that metals such as cobalt (Co), copper (Cu), chromium (Cr), iron (Fe), magnesium (Mg), manganese (Mn), molybdenum (Mo), nickel (Ni), selenium (Se) and zinc (Zn) are essential nutrients that are required for various biochemical and physiological functions [12]. Inadequate supply of these micro-nutrients results in a variety of deficiency diseases or syndromes [12]. Heavy metals are also considered as trace elements because of their presence in trace concentrations (ppb range to less than 10ppm) in various environmental matrices [13]. Their bioavailability is influenced by physical factors such as temperature, phase association, adsorption and sequestration.
It is also affected by chemical factors that influence speciation at thermodynamic equilibrium, complexation kinetics, lipid solubility and octanol/water partition coefficients [14]. Biological factors such as species characteristics, trophic interactions, and biochemical/physiological adaptation, also play an important role [15].
The essential heavy metals exert biochemical and physiological functions in plants and animals. They are important constituents of several key enzymes and play important roles in various oxidation-reduction reactions [12].
Copper for example serves as an essential co-factor for several oxidative stress-related enzymes including catalase, superoxide dismutase, peroxidase, cytochrome c oxidases, ferroxidases, monoamine oxidase, and dopamine β-monooxygenase [16- 18]. Hence, it is an essential nutrient that is incorporated into a number of metalloenzymes involved in hemoglobin formation, carbohydrate metabolism, catecholamine biosynthesis, and crosslinking of collagen, elastin, and hair keratin. The ability of copper to cycle between an oxidized state, Cu(II), and reduced state, Cu(I), is used by cuproenzymes involved in redox reactions [16-18].
However, it is this property of copper that also makes it potentially toxic because the transitions between Cu(II) and Cu(I) can result in the generation of superoxide and hydroxyl radicals [16-19]. Also, excessive exposure to copper has been linked to cellular damage leading to Wilson disease in humans [18,19]. Similar to copper, several other essential elements are required for biologic functioning, however, an excess amount of such metals produces cellular and tissue damage leading to a variety of adverse effects and human diseases. For some including chromium and copper, there is a very narrow range of concentrations between beneficial and toxic effects [19,20].
Other metals such as aluminum (Al), antinomy (Sb), arsenic (As), barium (Ba), beryllium (Be), bismuth (Bi), cadmium (Cd), gallium (Ga), germanium (Ge), gold (Au), indium (In), lead (Pb), lithium (Li), mercury (Hg), nickel (Ni), platinum (Pt), silver (Ag), strontium (Sr), tellurium (Te), thallium (Tl), tin (Sn), titanium (Ti), vanadium (V) and uranium (U) have no established biological functions and are considered as non-essential metals [20]. In biological systems, heavy metals have been reported to affect cellular organelles and components such as cell membrane, mitochondrial, lysosome, endoplasmic reticulum, nuclei, and some enzymes involved in metabolism, detoxification, and damage repair [21].
Metal ions have been found to interact with cell components such as DNA and nuclear proteins, causing DNA damage and conformational changes that may lead to cell cycle modulation, carcinogenesis or apoptosis [20-22]. Several studies from our laboratory have demonstrated that reactive oxygen species (ROS) production and oxidative stress play a key role in the toxicity and carcinogenicity of metals such as arsenic [23- 25], cadmium [26], chromium [27,28], lead [29, 30], and mercury [31,32]. Because of their high degree of toxicity, these five elements rank among the priority metals that are of great public health significance.
They are all systemic toxicants that are known to induce multiple organ damage, even at lower levels of exposure. According to the United States Environmental Protection Agency (U.S. EPA), and the International Agency for Research on Cancer (IARC), these metals are also classified as either “known” or “probable” human carcinogens based on epidemiological and experimental studies showing an association between exposure and cancer incidence in humans and animals. Heavy metal-induced toxicity and carcinogenicity involves many mechanistic aspects, some of which are not clearly elucidated or understood.
However, each metal is known to have unique features and physic-chemical properties that confer to its specific toxicological mechanisms of action. This review provides an analysis of the environmental occurrence, production and use, potential for human exposure, and molecular mechanisms of toxicity, genotoxicity, and carcinogenicity of arsenic, cadmium, chromium, lead, and mercury. Heavy metals are defined as metallic elements that have a relatively high density compared to water. Heavy metals like Chromium (Cr), Cadmium (Cd), Mercury (Hg), Lead (Pb), Nickel (Ni), and Thallium (Tl) are potentially hazardous in combined or elemental forms [33-40].
Heavy metals are highly soluble in the aquatic environments and therefore they can be absorbed easily by living organisms. Previous studies have detected heavy metals in the gills, liver, and muscles tissues of various species of fish in contaminated marine ecosystems. Once the heavy metals enter the food chain, they may end up accumulating in the human body. Since most heavy metals are widely applied in industries, exposure and contamination of the workers and residents near such facilities is likely to occur. Heavy metals above allowable limits will often lead to disadvantageous effects on humans, other organisms and the environment at large. Allowable safe limits of heavy metals in food samples are associated with low health risks in humans.
The level of toxicity of some selected metals for humans follows the sequence Co < Al < Cr < Pb < Ni < Zn < Cu < Cd < Hg. The harmful effects of heavy metals in humans depend on their dosage, rate of emission and period of exposure. Some of the heavy metals that have received more attention for the last decades are Hg, Cd, and Pb7. The adverse health effects that are associated with Hg and mercuric compounds in humans includes possible carcinogens; damage of the brain, lungs and kidneys; damage of developing fetuses; high blood pressure or heart rate; vomiting and diarrhea; skin rashes and eye irritation8. The US EPA’s regulatory limit of Hg in drinking water is 2 parts per billion (ppb). The WHO recommended safe limits of Hg in wastewater and soils for agriculture are 0.0019 and 0.05 ppm respectively.
Chronic toxicity of Cd in children includes damages of respiratory, renal, skeletal and cardiovascular systems as well as development of cancers of the lungs, kidneys, prostate and stomach. Exposure of people to Cd includes eating contaminated food, smoking cigarettes, and working in cadmium-contaminated workplaces and in primary metal industries. A study carried out in Iran reported that the level of Cd was higher than the maximum permissible limit (MPL) in canned fish samples, and this was due to discharge of heavy metal rich pollutants into aquatic ecosystems.
The US EPA’s regulatory limit of Cd in drinking water is 5 ppb or 0.005 parts per million (ppm). The WHO recommended safe limits of Cd in both wastewater and soils for agriculture is 0.003 ppm [40-50].
Exposure to Pb can occur through inhalation of contaminated dust particles and aerosols or by ingesting contaminated food and water. Lead poisoning in humans damages the kidneys, liver, heart, brain, skeleton and the nervous system. Initial symptoms of poisoning associated with exposure to Lead may include headache, dullness, memory loss and being irritable. Lead poisoning may cause disturbance of hemoglobin synthesis and anemia. In children, chronic exposure to low levels of Lead may decrease their intelligence capacity. According to the International Agency for Research on Cancer (IARC), Lead is a possible carcinogenic substance in humans. The regulatory limit of Pb in drinking water according to US EPA is 15 ppb. The WHO recommended safe limits of Pb in wastewater and soils used for agriculture are 0.01 and 0.1 ppm respectively.
Chromium is widely used in metallurgy, electroplating, and in the manufacturing of paints, pigments, preservatives, pulp and paper among others. The introduction of Chromium into the environment is often through sewage and fertilizers. Hexavalent Chromium compounds including chromates of Ca, Zn, Sr, and Pb are highly soluble in water, toxic and carcinogenic. Furthermore, compounds of Chromium have been associated with slow healing ulcers. It has also been reported that Chromate compounds can destroy DNA in cells. The WHO recommended safe limits for Cr (hexavalent) in wastewater and soils used for agriculture are 0.05 and 0.1 ppm respectively.
Thallium is a soft, tasteless, odorless white, blue metal in its pure form, and it oxidizes to thallium oxide when exposed to air. Sources of Tl include electronics, optical glasses, semi-conductors, mercury lamp among others. Humans become exposed to Tl through ingestion, inhalation, and dermal exposure. Thallium is highly toxic with a lethal dose of 6 to 40 mg/kg. Thallium poisoning is associated with anorexia, vomiting, gastrointestinal bleeding, abdominal pain, polyneuropathy, alopecia, renal failure, skin erythema, seizures, emotional changes, autonomic dysfunction, cardio toxicity, and coma among others. In China, the recommended safe limit of thallium in drinking water is 0.0001 ppm. The WHO recommended safe limits for Tl in both wastewater and agricultural soils were not given in the literature accessed.
Nickel is a silver- colored metal used in making stainless steel, electronics, and coins among other uses. Globally, the release of Ni to the environment is estimated to vary from 150,000 to 180,000 metric tons per year. Exposure of Ni to humans is through food, air and water. Previous study has shown that ingestion of dust contaminated with Nickel was the main exposure pathway of the heavy metal by local residents when compared to inhalation and dermal pathways. Upon exposure to Nickel, an individual may show increased levels of Ni in his or her tissues and urine. The disadvantageous effects of nickel on human health may include dermatitis, allergy, organ diseases, and cancer of the respiratory system. The recommended safe limits by WHO for Ni in wastewater and agricultural soils are 0.02 and 0.05 ppm respectively.
Wastewater from factories may contain heavy metals which with time accumulate in the soil deposits along wastewater channels as well as in organisms that inhabit such channels. Exposure of humans to contaminated wastewater is often possible especially in urban highly populated areas or where the wastewater is reused for agricultural activities. Previous studies however have shown that effective reuse of wastewater is a major challenge in many countries of the world. The current study was designed to establish the concentration of Hg, Pb, Cr, Tl, Cd and Ni in samples of wastewater and soil obtained from open wastewater channels in selected sites in Nairobi’s industrial area, Kenya.
There are many informal urban settlements/villages that neighbor Nairobi industrial area and some of the wastewater channels drain into a tributary of Nairobi River which flows across these villages. Clogged wastewater channels enhance overflow of the wastewater into the surrounding areas through surface runoff when it rains. Therefore, the current study aims to highlight the potential health risks that may ensue when humans, livestock and crops become directly or indirectly exposed to the heavy metal contaminated wastewater and soils from the open channels in the study area. It is envisaged that the results obtained from the study will inform and justify on the need to adopt good wastewater management including prioritizing on effective wastewater treatment and reuse in Kenyan major urban areas. Previous studies elsewhere have shown that the degree of wastewater treatment determines the applicability of the reclaimed water.
Collection of Water and Soil Samples
A standard 350 ml dipper was used to collect samples of wastewater in triplicate portions, from open channels and put into clean reagent plastic bottles. Two separate portions of wastewater destined for determination of heavy metals were acidified with concentrated hydrochloric acid (HCL) and concentrated nitric acid (HNO3) respectively to a pH ranging between 1.5 and 2.0 as described by Davies. The third sample of wastewater was left plain with no acidification. The samples were then labeled. Control samples of clean water were collected from tap water in randomly selected premises in the study area. Soil samples in triplicates were also collected from the open channels using a hand metallic soil scoop and then packaged into brown paper bags and labeled. The soil scoops were cleaned after every scooping was done. Both water and soil samples collected were immediately transferred to Kenyatta University Science Complex Laboratory for further processing.
Physico-Chemical Parameters of Water Samples
Both physical and chemical parameters of the water samples were measured and recorded at the collection site. These included temperature, pH, electrical conductivity, and turbidity. Electronic devices capable of recording the parameters at the same time (HANNA Instruments, H1991300, and Romania) were used.
Preparation of Soil Samples for Heavy Metal Analysis
In the laboratory, the wet soil samples from each sampling site were spread on brown papers to dry under room temperature. They were then ground, sieved, weighed, and packaged in small brown envelopes and labeled. The labels included site, date of collection, and weight in grams.
Standard Limits of Heavy Metals in Wastewater, Drinking Water, Soils, & Vegetables
The standard limits of Hg, Pb, Cd, Cr, Tl and Ni in drinking water, wastewater (effluents), agricultural soils, and vegetables, as recommended by WHO; China (both Chinese Ministry of Health (CMH) & The National Standard of China); Kenya (both National Environment Management Authority (NEMA) & Kenya Bureau of Standards (KEBS); USA EPA; India (CPCB) and World Bank (WB) were retrieved from the available literature. The determined level of heavy metals in the field samples were then compared to these standard limits in order to establish whether the level of pollution in the open channels was above the locally and internationally accepted standards in addition to making reliable conclusions.
Analysis of Heavy Metals in Samples of Water and Soil
The analyses were carried out at Mineral Laboratories, Bureau Veritas Commodities Ltd, Vancouver, Canada. The protocols included aqua regia digestion ultra-trace inductively coupled plasma mass spectroscopy (ICP-MS) for soil samples; and ICP-MS (solutions >0.1% Total Dissolved Solids (TDS) for water samples as described by the American Herbal Products Association (AHPA). Briefly, the digest solution was nebulized, and sample aerosols transferred to argon plasma. The high temperature plasma then produced ions, which were then introduced into the mass spectrometer, which then sorted out the ions according to their mass-to-charge ration. The ions were then quantified with an electron multiplier detector. Certificates of analysis and quality control reports for all the samples analyzed were awarded by the Bureau Veritas, Canada.
Data Analysis
Statistical Package for Social Sciences (SPSS) for Windows (Version 20) at 5% level of significance was used. Descriptive statistics involved computing the mean, standard error (SE), and standard deviation (SD) for the different variables measured in water and soil samples. One-way analysis of variance (ANOVA) was used to establish the significant differences within and between groups. Tukey’s and Games-Howell Post hoc tests were carried out to establish the pairs of variables that were significantly different. Correlation analysis was carried out to establish the nature of relationship, level of significance between concentrations of heavy metals in different samples. Pairwise correlations of coefficients for the levels of selected heavy metals in wastewater and soils were computed.
Physico-chemical parameters of wastewater samples
The conductivity of wastewater obtained from Chief’s Camp (B-2), Railways (sites C & D) and Sinai (G) was high and ranged between 770.00 ± 11.55 to 1134.33 ± 180.39 μS/cm. The conductivity levels of wastewater samples from the remaining sites ranged from 366.33 ± 33.79 to 556.00 ± 27.62 μS/cm. Increased conductivity corresponded to increased turbidity of wastewater and vice versa. Therefore, high turbidity of wastewater was similarly recorded at Railways upper section (D), Sinai (G) and Railways lower section (C) sites and it measured 535.33 ± 41.60, 544.67 ± 21.17 and 562.00 ± 84.33 ppm respectively. The chief’s camp (B-1) had the lowest turbidity at 160.33 ± 0.88 ppm. The pH of wastewater samples ranged from 7.28 to 8.78 while the temperature ranged from 16.75 to 26.05 °C.
Standard Limits of Heavy Metals in Drinking Water, Wastewater, Soils, and Vegetables
The recommended limits of selected heavy metals according to WHO, Kenya (NEMA & KEBS), China (CMH & the National Standard of China), US EPA, India (CPCB) and World Bank were retrieved and recorded from the literature accessed. The standard levels for Tl were not given for wastewater, soils, and vegetables in the literature accessed. However, allowable level of Tl in drinking water, recommended by Chinese Ministry of Health and US EPA was available. The Tl limit level in surface water as recommended by US EPA was also recorded. Standards of heavy metals in agricultural soils in Kenya were missing in the literature accessed.
Levels of Heavy Metals in Wastewater and Tap Water
The wastewater samples had high Pb levels followed by Cr and the lowest was Tl. The mean concentration of heavy metals analyzed in wastewater samples, in an ascending order was Tl < Cd < Hg < Ni < Cr < Pb. This trend applied for all samples of wastewater that were acidified immediately after collection. The mean concentration of chromium was the highest at 24.2 ppb followed by nickel at 2.90 ppb in the wastewater samples that were not digested by acids immediately after collection hence an ascending order of Tl < Cd < Hg < Pb < Ni < Cr.
The mean concentration of Hg in the wastewater samples was <0.0001 ppm and this was lower than the standards set by WHO, World Bank (WB), Kenya, India and China but greater than 0.00003 ppm which is the standard set by US EPA. The level of Hg in wastewater samples from all the sampling sites was below the method detection limit (MDL) which had been set at 0.1 ppb. Similarly, the average levels of Pb for acid digested wastewater samples in 5 out of 8 (62.5%) sites (2 sites at Chief’s camp; Davis & Shirtliff, Kartasi and Donholm) had high Pb levels that ranged from 0.011 to 0.032 ppm, and this was above the recommended limits of Pb in wastewater set by WHO, Kenya, and US-EPA.
The mean concentration of Cr in wastewater samples from all the sampling sites ranged between 0.00308 to 0.00812 ppm which was between 84% to 99% less than the recommended limits by WHO, China, Kenya, US EPA, WB and India. The wastewater samples collected at Sinai (G1) had the highest concentration of chromium at 0.0507 ppm, but which was within the maximum limit recommended level set by WHO and US EPA. The mean concentration of Ni in wastewater in all sampling sites was 0.004ppm and this was within the recommended limits set by WHO, China, Kenya, US EPA, WB and India. Nickel level was significantly high in wastewater samples obtained from Railways Lower (C2) with a mean concentration of 21.7 ppb and at Sinai (G1) with a concentration of 19.4 ppb.
The mean levels of Tl in the wastewater were about 100 000 times less than the US EPA (SWQS) recommended limits. The mean concentration of Cd in wastewater in all the sampling sites was 0.000087 ppm which was far less than the recommended limits by WHO, WB, US EPA, China, Kenya, and India. The level of Hg in samples of tap water was below the MDL which had been set at 0.1 ppb or 0.0001 ppm. Similarly, the levels of Pb, Cd, Cr, Tl and Ni in the samples of tap water ranged between 0.00001 and 0.0016 mg/ml (ppm) which were far below the standard limits set by WHO, Chinese Ministry of Health and Kenya (NEMA), US EPA.
The Levels of Heavy Metals in Soil Samples
The mean concentration ± SE (standard error) of heavy metals in soil samples was highest for Pb and lowest for Hg in an ascending sequence of Hg < Tl < Cd < Ni < Cr < Pb. The concentration of Pb in soil samples from Davis & Shirtliff site was 471.17 ± 117.5 ppm compared to samples collected from Chief’s Camp (B) and Railways Lower (C) sites that were at 255.50 ± 91.20 and 211.00 ± 8.26 ppm respectively. Soil samples from Sinai site had the lowest level of Pb at 59.92 ± 8.42 ppm. Relatively higher levels of Hg were recorded for soil samples collected at Chief’s Camp (B), Railways Lower (C), and Davis & Shirtliff (E), which were at 223.75, 121.00, and 106.67 ppb respectively.
The concentration of Cd and Tl in the soil samples ranged from 0.2 ± 0.05 to 1.90 ± 1.40 ppm and 0.23 ± 0.01 to 0.50 ± 0.06 ppm respectively. Soil samples from Chief’s Camp (B) site had the highest level of Cd and Tl while samples from Donholm (H) site had the lowest. The concentration of Cr and Ni ranged between 21.37 ± 9.87 to 81.17 ± 3.80 and 11.70 ± 0.44 to 29.87 ± 1.90 ppm respectively for the soil samples obtained from the study area.
When compared to the standard limits, the mean concentration of Hg and Ni in soil samples was 0.085 and 18.81 ppm respectively and this was below the recommended limits set by China and USEPA but above WHO limits for agricultural and gardening soils.
The mean concentration of Cr in the soil samples was 45.19 ppm and it was above the limits set by WHO and US EPA. The average levels of Pb and Cd in soil samples was also above the recommended limits set by WHO, China but within the US EPA limit for agricultural and gardening soils. The mean concentration of Tl in the soil samples was 0.38 ppm, however the soils standards for Tl in agricultural soils for WHO, US EPA, China and Kenya were not available. It was observed that the mean concentration of heavy metals analyzed in the soil samples was between 800 to 13,500 times more than the mean concentration of the same metals in wastewater samples at the same sampling site.
Correlation of heavy metals level in different samples
Pairwise comparison of the level of heavy metals in wastewater and soils samples yielded several strong significant positive correlations (P < 0.05). Such pairs included Cd & Hg in soils samples; and Tl (wastewater) & Cd (soil); where an increase in one element corresponded to an increase in the other. The strong positive correlations indicated a close association of the elements in samples of soil and wastewater. A negative correlation between Tl (soil) & Cr (wastewater), r = - 0.641, P = 0.087, was also observed. At Sinai sampling site (G), Inter-elemental correlation levels of metals for the samples of wastewater and soils samples were computed.
A significant positive correlation was obtained for pairs Cd & Pb; Ni & Pb in samples of wastewater. Similarly, a strong positive correlation coefficient (r = 0.995) for Cr & Pb that was near significance level (P = 0.061), was recorded for soil samples obtained from Sinai. Conductivity is a measure of the water’s ability to conduct electricity and it is directly related to the concentration of ions in the water. Significant changes in the conductivity of water directly compromise its quality. In the current study, conductivity of the wastewater was highest for samples obtained from Railways lower (C), Railways upper (D) and Sinai (G) sampling sites and it ranged from 1072 to 1134 μS/ cm.
This range was higher than WHO range for electrical conductivity of water which is between 400 to 600 μS/cm. The wastewater samples from the remaining sites had conductivity levels that were within the WHO standards. The source of the conductive ions in wastewater may have been the dissolved substances including pollutants. At railways sampling sites (C and D), combined organic sewage material overflow that had been directed into the open wastewater channel may have introduced dissolved ions into the channels hence increasing conductivity. This was in line with a previous study by Mbui and colleagues who reported that domestic effluents discharge into the river increases the electrical conductivity.
The road construction activities near Sinai sampling site (G) which involved heavy machinery plus nearby industries may have contributed to increased particles and ions in the wastewater channels hence accounting for the raised turbidity and conductivity levels of the wastewater samples. It was noted that increased turbidity corresponded to increased conductivity and vice versa. Turbidity indicates total suspended solids in water, and it is a principle parameter of wastewater effluent monitoring and therefore it can be used to evaluate wastewater treatment plant efficiency and compliance to discharge requirements. Both conductivity and turbidity are important parameters in measuring the quality of water post treatment.
The temperature of wastewater samples ranged from 16.75 to 26.05 °C, which was within the recommended WHO range of 20 to 32 °C. The pH of wastewater samples ranged from 7.28 to 8.78 and similarly was within WHO range of 6.5 to 8.5. Microbial degradative activities in wastewater are dependent on temperature, pH, presence of organic substances and types of microbes. Therefore, elevated temperature in wastewater tends to support increased bio-degradative reactions in presence of increased organic substances. The organic substances and types of microbes in the wastewater were not determined in this study.
During the third United Nations Environment Assembly hosted by Kenya at UNEP headquarters (Gigiri) in December 2017, Kenya promised to improve the lives of its people by cleaning up air, land and water. Industrial, sewage and domestic wastes have been finding their way into Nairobi River in Kenya, hence making the river unsuitable for use. According to UN Environment, over 80% of the world’s wastewater is released into the environment without treatment, polluting the fields where plants grow, lakes and rivers. Such pollutants can easily flow from the environment into humans directly or indirectly. Water and soil pollution with heavy metals in developing countries emanate from poor disposal of industrial and urban wastes.
Municipal and industrial wastewater should be treated as a strategy of minimizing the contaminants before reusing wastewater. However, health impact assessment of treated wastewater should be carried out to identify the hazards and risk factors that may be associated with wastewater reuse. The current study established that the levels of Cr, Cd, and Ni in wastewater were below the limits set by WHO, US EPA, WB and Kenya. Similarly, the Hg level in wastewater was below the limits set by WHO, China, WB, India and Kenya (NEMA) but slightly above the limit set by US EPA. The level of Pb in the wastewater was above limits set by WHO, US EPA, WB and Kenya but lower than the limits set by China and India.
The public health concern in terms of wastewater in Nairobi industrial area is therefore Hg and Pb levels which were above the limits set by US EPA and WHO respectively. Standards for thallium in wastewater were not available and therefore it was difficult to make a conclusion on whether the levels obtained were high or low. Previous reports however, have shown that water quality within Nairobi River catchment area in Kenya has degraded due to intensive land use hence increasing Hg and Pb levels and surpassing the critical guidelines of WHO. In the current study area, humans can become exposed to such pollutants when surface runoff finds its way into residential areas or utilizing the contaminated soils for agricultural activities.
Heavy metal accumulation in soils leads to increased phyto - accumulation of such metals in the crops grown. Clogging of open wastewater channels with solid wastes, mud and overgrown vegetation can enhance surface runoff of the wastewater to surrounding areas. One of the factors which may increase the chance of exposure to metal pollutants in the study area is the presence of dense population in the informal settlements near Nairobi industrial area. Some of the open wastewater channels pass through these settlements or drain into Nairobi River which then flow across these settlements. However, treatment of wastewater for reuse is a common practice in many countries since it can alleviate natural water shortage and minimize contaminants finding their way into natural aquatic ecosystems.
According to a study carried out in Greece, the annual percent contribution of treated wastewater in the total irrigation water volume in Thermos and Nafpaktos was 87.8% and >100% respectively. The same should be adopted in Nairobi to minimize the contaminants in wastewater and to provide adequate water for agricultural activities. Tap water samples (controls) that were randomly collected from the study area were analyzed to establish the heavy metals levels and compared to wastewater samples. The levels of all the heavy metals studied in tap water samples ranged from <0.00001 to 0.0016 ppm and these levels were far below limits set by WHO, US EPA, Chinese Ministry of Health (CMH) and Kenya (NEMA).
The tap water sampled from residential and hotels in the study area was therefore safe, high quality, and acceptable in terms of Hg, Pb, Cd, Cr, Tl and Ni levels. The domestic pigs observed at Kartasi industries sampling site, scavenging for edibles from the mud and vegetation clogged open wastewater channels, was evidence that there were residential areas nearby in the study area. The sampling sites in the current study were actually near the densely populated informal settlements (slums) that included Sinai, Mukuru kwa Njenga, and Land Mawe. The samples of wastewater and soil sediments from Kartasi sampling site had levels of heavy metals that were above the MDL except for Hg level in wastewater.
It is worth noting that even low levels of environmental metal pollutants can accumulate with time in exposed humans and animals. Previous studies show that livestock are prone to general problems of industrial pollution. A study in Namibia established that pasture grass that was obtained from around waste dumpsites had higher levels of heavy metals. It is possible for heavy metals to accumulate in the tissues and organs of domestic animals that become exposed to contaminated environments, materials, and fodder. The concentration of Hg and Cd was shown to be high in the liver, kidney, and muscle samples of organically and conventionally produced pigs in Czech Republic.
Therefore, the scavenging pigs in the open wastewater channels in industrial areas, that were observed in the current study, can serve to directly or indirectly spread the heavy metal pollutants from such channels into humans. When the heavy metals pollutants from the channels accumulate in the pigs’ muscles with time, then the quality of pork from such animals is compromised and it may become a health risk Heavy metals occur naturally in soils following the weathering processes of the underlying rocks. Availability of heavy metals in soils is influenced by environmental conditions that determine the pH and organic matter content in soils.
Heavy metal contamination of the soils may pose risks and hazards to humans and ecosystems through direct contact or ingestion, food chain, contaminated drinking water, reduced food quality among others. The concentration of the Lead (Pb) in the soil samples at Davis & Shirtliff sampling site was 471.17 ± 117.5 ppm and this was above the normal range of Pb (2 to 300 ppm) in the soils. The worldwide Pb concentration for surface soil averages 32 mg/kg (ppm) and it ranges from 10 to 67 mg/kg (ppm) implying that the levels at Davis & Shirtliff were significantly above this limit. The average Pb level in the soil samples collected from Chief’s camp (B) and Railways Lower (C) were 255.50 ± 91.20 and 211.00 ± 8.26 ppm respectively and they were skewing towards the upper limit of normal range as described by Gardea-Torresdey and colleagues but above the range reported by Pendias & Pendias.
The soil samples from Sinai site had the lowest level of Pb at 59.92 ± 8.42 ppm which was skewed towards the lower limit of normal range of Pb in soils. Soil sediments in the wastewater channels may enrich with pollutants present in wastewater with time. Increased Pb content in soils recovered from the open wastewater channels is a health hazard to workers who regularly clean up the channels especially when they are ignorant about the need to maximize safety measures. Lead has been associated with multiple organ problems and cancers. The soil samples collected from Chief’s camp (B), Railways Lower (C), and Davis & Shirtliff (E) sampling sites had relatively higher Hg levels, but which were within the limits set by China and US EPA for agricultural soils.
The average concentration of Cr and Ni in the soil samples from the study area which ranged between 21.37 ± 9.87 to 81.17 ± 3.80 and 11.70 ± 0.44 to 29.87 ± 1.90 ppm respectively were below the allowable limits recommended by China and US EPA but above the limits recommended by WHO for agricultural soils. It is important to note that even soils that are contaminated with low levels of heavy metals can contribute to bioaccumulation of such elements with time in organisms that are in higher trophic levels in a food chain. Pollution of the environment with traces of heavy metals from anthropogenic sources should not therefore be ignored.
The mean concentration of heavy metals was higher in soils than in wastewater samples. This was in line with a previous report by Khan and his colleagues which explained that contaminated wastewater can lead to a build-up of heavy metals in soils. Inter-elemental analysis of the metals showed several strong and positive correlations. This suggested that, these metals were from the same source, most likely the industries whose wastes were draining into the open channels in the study area.
This explanation was in line with previous studies carried out in Nigeria and Pakistan. The significant correlation coefficients between pairs of metals in samples of wastewater and soils may be a pointer of a common source of heavy metal pollution in the study area, most likely anthropogenic activities.
This study showed that wastewater and soils samples from open waste channels in Nairobi industrial area contained heavy metals. Of the metals studied, the mean concentration of Ni, Cr and Pb were relatively higher than those of Tl, Hg, and Cd in the samples analyzed. The levels of Hg, Cr, Cd and Ni in wastewater samples were within the allowable limits set by WHO, WB, Kenya, China and India. The mean level of Hg in wastewater was <0.0001ppm and this was a public health concern in the study area, based on the US EPA allowable limit of Hg in wastewater that is set at 0.00003 ppm. The level of Tl in wastewater samples was below the limit set by US EPA and this was commendable.
The mean concentration of Pb in wastewater was above the allowable limits set by WHO, WB, US EPA and Kenya in 5 out of 8 sampling sites, hence becoming a public health concern in the study area. The levels of Pb, Hg, Cr, Cd, and Ni in open drainage channels soil samples were above the limits set by WHO for agricultural and gardening soils. The mean concentration of heavy metals was relatively higher in soil than in wastewater samples at each sampling site. This was evidence of a build-up of toxic metals in the soils found in open waste channels.
There was adequate evidence of clogging of the wastewater channels with mud and overgrown vegetation hence facilitating overflow and spread of contaminated wastewater and soils from the channels to residential areas nearby during the rainy seasons. The presence of domestic pigs scavenging from the open channels suggested a likely pathway through which the metallic contaminants could eventually find their way into humans. Therefore, there is need to formulate and adopt policies, strict rules among others that would translate to excellent wastewater management and treatment infrastructure hence minimizing environmental pollution and its associated health hazards as well as avail adequate reclaimed water for urban agricultural activities.
Frequent inspections and unclogging of the open waste channels should be carried out to enhance faster flow and to minimize possible spread of heavy metal contaminated wastewater to the densely populated informal settlements/ villages that neighbor Nairobi industrial area. Residents living nearby should be made aware of the health hazards that could emanate from exposure to untreated wastewater through public education and awareness campaigns. We the authors recommend the determination of heavy metals in pork available in the study area in order to provide possible evidence of bioaccumulation of these metal contaminants in human food.
Copper(Cu)
Copper is a mineral and natural component of soils. It is an essential nutrient for humans and plants. Industrial pollution, domestic wastewater, mining wastewater, and weathering of copper-bearing rocks are major sources of copper in surface and ground waters. Discharges of copper into sewer systems from some residential areas have also been found. Leading areas of use for copper and its alloys are the plumbing, electrical, and electronic industries. Copper compounds are used as agricultural fungicides, algaecides in water reservoirs, in food supplements, and fertilizers. In Washington, most copper in drinking water comes from corrosion of household plumbing. Public water supplies are required to conduct treatment to reduce corrosion when more than 10 percent of the tap water samples exceed 1.3 parts per million (ppm) of copper. Copper from plumbing corrosion can accumulate overnight. Flushing the water from the tap for 30 to 45 seconds can reduce the copper that can accumulate when the household plumbing is not in use.
Health Issues
Copper is an essential mineral in the diet. Too much copper, however, can cause health problems. Major food sources of copper are shellfish, nuts, grains, leafy vegetables, and stone fruits. Typical sources of copper from food range from less than 2 milligrams (mg.) to 5 mg. per day. Health studies have found that copper in drinking water can add 4 to 45 percent more copper to a person’s diet than what is in food sources. Absorption studies have indicated that the body excretes about half of ingested copper, which offers some protection against copper poisoning. Copper is widely distributed within the tissues of the body but accumulates primarily in the liver and kidneys. The National Academy of Science recommends 2-3 mg of copper in the daily diet. Severe cases of copper poisoning have led to anemia and to the disruption of liver and kidney functions. Individuals with Wilson’s and Menke’s diseases (genetic disorders resulting in abnormal copper absorption and metabolism) are at higher risk from copper exposure than the general public and can have serious health problems.
Detecting Copper in Drinking Water
Blue-green stains on plumbing fixtures are usually an indication of the presence of copper in drinking water. Some factors that affect copper levels in household drinking water are:
• Acidic water (low pH)
• Soft water (low in calcium and magnesium)
• High chlorine residual levels
• Long standing time in pipes
• Elevated water temperature
Backflow Prevention for Carbonated Beverage Dispensers
There is a great risk of copper contamination from carbonated or acidic beverages that have been in contact with copper tubing, fixtures, or containers. The carbon dioxide used in soft drink dispensers is under pressure. Where it is mixed with the incoming water, there is a potential for carbon dioxide to backflow through the incoming water supply. The carbon dioxide then can corrode copper plumbing, which can add copper to soft drinks. The surest way to prevent the carbon dioxide from contacting copper plumbing pipes is to install a reduced pressure backflow prevention assembly (RPBA). This assembly should be installed on the incoming water supply pipe before the carbon dioxide injection point. Copper piping should not be used past the injection point. (Some utilities may allow the substitution of a dual-check valve with an intermediate vent in place of the RPBA).
The Federal-Provincial-Territorial Committee on Drinking Water (CDW) has assessed the available information on copper with the intent of updating the current drinking water guideline and guideline technical document on copper in drinking water. The purpose of this consultation is to solicit comments on the proposed guideline, on the approach used for its development and on the potential economic costs of implementing it, as well as to determine the availability of additional exposure data.
The existing guideline on copper, last updated in 1992, established an aesthetic objective of ≤1.0 mg/L, to ensure palatability and to minimize staining of laundry and plumbing fixtures. This new updated document proposes a health-based maximum acceptable concentration (MAC) of 2 mg/L for copper in drinking water. The proposed MAC is considered to be protective of both short-term effects (i.e., gastro-intestinal effects) as well as longer-term effects (including potential effects in the liver and kidneys). The document is based on currently available scientific studies and approaches, taking into consideration the mode of action. It provides exposure information as well as sampling considerations, analytical methods and treatment technologies for copper in drinking water. The document also proposes an aesthetic objective (AO) of 1 mg/L, based on taste and staining considerations.
The CDW has requested that the guideline technical document on copper in drinking water be made available to the public and open for comment. Comments are appreciated, with accompanying rationale, where required. Comments can be sent to the CDW Secretariat via email at water_eau@hc-sc.gc.ca. If this is not feasible, comments may be sent by mail to the CDW Secretariat, Water and Air Quality Bureau, Health Canada, 3rd Floor, 269 Laurier Avenue West, A.L. 4903D, Ottawa, Ontario K1A 0K9. All comments must be received before May 25, 2018.
Comments received as part of this consultation will be shared with the appropriate CDW member, along with the name and affiliation of their author. Authors who do not want their name and affiliation shared with their CDW member should provide a statement to this effect along with their comments. It should be noted that this guideline technical document on copper in drinking water will be revised following evaluation of comments received, and a drinking water guideline will be established, if required. This document should be considered as a draft for comment only.
Health effects
Copper is an essential element in humans. Copper deficiency may cause several health effects but is not expected to be a concern in Canada, based on copper intake from food. The U.S. National Academy of Medicine (formerly called the Institute of Medicine) has established recommended daily allowances of 900 µg/day for adults and 340-890 µg/day for children, as well as tolerable upper intake levels of 10,000 µg/day for adults, and between 1,000 and 8,000 µg/day for children. International agencies have determined that the data available on copper are not sufficient to classify it with respect to carcinogenicity. Short-term exposure to copper may result in effects in the gastrointestinal tract (nausea, pain and vomiting, diarrhea). Long-term effects are less well documented; current evidence indicates that, in the general population, chronic exposure to very high levels of copper may lead to effects in the liver and kidney. The proposed MAC was developed based on bottle-fed infants (aged 0 to 6 months) and is considered to be protective of all health effects, both short-term and longer-term.
Aesthetic Considerations
The presence of copper can affect the taste of the water and cause the staining of laundry and plumbing fixtures at levels below the proposed MAC. Although there are no adverse effects associated with such levels, they will affect the acceptability of the water by consumers. Although the proposed AO for copper in drinking water is 1 mg/L (1000 µg/L), some individuals can readily perceive copper at levels below the AO. Nevertheless, corrosion control practices should target the MAC and not the elimination of aesthetic concerns as this could jeopardize other water quality priorities such as lead control. Utilities may choose to use the AO to trigger a site-specific or localized water quality/ corrosion investigation.
Exposure
Copper occurs naturally in the Earth’s crust, either in mineral deposits or, less frequently, as a metal. Copper can enter water sources from natural processes, such as soil weathering, and human activities such as agriculture, mining, and manufacturing. The National Plumbing Code of Canada considers copper to be an acceptable material for service lines and plumbing systems. Consequently, copper in drinking water can also result from the corrosion of copper-containing pipes and fittings in distribution and plumbing systems, depending on the chemistry of the water. Canadians can be exposed to copper through food, drinking water, air, soil and consumer products. Although the most significant source of copper intake is food for the general population, the bioavailability of copper in drinking water may be greater than that in food. Based on the physical and chemical properties of copper, inhalation and dermal absorption of copper are not expected from exposure to drinking water.
Analysis and Treatment
The establishment of a drinking water guideline must take into consideration the ability to measure the contaminant. There are several methods available for the analysis of total copper in drinking water. Based on the capacity of commercial laboratories in Canada, analytical methods are available to reliably measure total copper in drinking water below the proposed MAC. These methods should include sample preparation to ensure that they are able to detect both dissolved and particulate copper. Copper levels in source water are typically very low. Although there are treatment technologies that can remove copper efficiently at the treatment plant, municipal treatment is not generally an effective strategy. This is because copper is an acceptable material for service lines and plumbing systems. However, in certain water qualities, copper may be released into drinking water from pipes, service lines and brass fittings. The treatment approach for copper is generally focused on corrosion control through approaches such as water quality adjustments and the use of corrosion inhibitors.
As the primary source of copper in drinking water is the leaching from plumbing and distribution system components, drinking water treatment devices offer an effective option at the residential level, although their use should not be considered to be a permanent solution. There are a number of certified residential treatment devices available that can remove copper from drinking water to below the proposed MAC.
Application of the Guideline
Note: Specific guidance related to the implementation of drinking water guidelines should be obtained from the appropriate drinking water authority in the affected jurisdiction. The proposed MAC for copper is established to be protective of both short-term gastrointestinal effects and longer-term effects in bottle fed infants as well as in the general population. Considering that copper levels at the consumer’s tap may be significantly higher than levels at the treatment plant or in the distribution system, strategies to reduce exposure to copper will need to focus on controlling corrosion within the distribution and plumbing systems. The water quality factors that have the greatest effect on copper corrosion are pH and alkalinity. Dissolved oxygen and/or chlorine residual/oxidants are also important considerations for copper. Copper monitoring should focus on areas known or likely to have new copper piping and where water quality conditions are potentially corrosive (e.g., low pH, dead ends, low or high alkalinity waters) and in consecutive systems.
An exceedance of the proposed MAC should be investigated and followed by the appropriate corrective actions to reduce the copper levels below the MAC. These actions include, but are not limited to, resampling, public education and corrosion control measures. However, these actions should be based on an assessment of the cause of the exceedance using appropriate protocols, such as those found in Guidance on Controlling Corrosion in Drinking Water Distribution Systems.
Monitoring
Sampling protocols will differ, depending on the desired objective (i.e., identifying sources of copper, controlling corrosion, assessing compliance, estimating exposure to copper). As monitoring of copper at the tap can be done using different sampling protocols, it is important that the selected protocol be appropriate to meet the desired objective. It is important to note that although the analysis of metals can be undertaken from the same sample bottle, sample sites for copper and lead differ. As such, copper results from lead sampling sites are unlikely to properly identify areas with copper issues.
The objective of sampling protocols in this document is to monitor typical community exposure to total copper to determine whether there are concerns about human health effects. Compliance monitoring should be conducted at the consumer’s tap and focus on areas known or likely to have new copper pipe installations and should include areas or zones (geographical areas within which the quality of drinking water is considered approximately uniform) supplied by potentially corrosive water (e.g., low pH, low or high alkalinity). Specifically, priority should be given to sites known to have new copper plumbing or when the water supply has a pH < 7, with or without disinfection or oxidation processes and alkalinity <25 mg CaCO3 /L, or pH>7 and alkalinity >200 mg CaCO3/L with disinfection or oxidation processes, regardless of the age of copper.
The addition of corrosion inhibitors can prevent the formation of stable malachite scales or displace sorbed oxyanions (e.g., copper) from distribution system piping. A water system using orthophosphate treatment for corrosion control having a PO4 residual less than 3.3 mg PO4/L, should also monitor for copper. Daycare facilities should also be prioritized for monitoring to ensure that the most sensitive population (i.e., infants) is captured. Sampling should be conducted at least once per year, with the number of sites to be monitored determined based on the size of the drinking water system and the type of building. The frequency may be reduced if no failures have occurred in a defined period as determined by the regulator, or if water quality conditions are not corrosive to copper.
Science and Technical Considerations Identity, use and sources in the environment Chemical and physical properties
Copper (Cu, CAS Registry No.7440-50-8) is a transition element that naturally occurs in four oxidation states: elemental copper metal (Cu(0) or Cu0), cuprous (monovalent)ion (Cu(I) or Cu+), cupric (divalent)ion (Cu(II) or Cu2+) and rarely trivalent copper ion (Cu(III) or Cu3+). Copper has two stable (63Cu and 65Cu) and 27 less stable isotopes. Copper metal has a reddish-brown color, an atomic mass of 63.55 Da, a density of 8.92 g/cm3 and a vaporization point of 2350°C (Georgopoulos et al., 2001:Turnlund et al., 1998; Riedel, 2008; EFSA, 2015 Kiaune and Singhasemanon, 2011; Ellingsen et al., 2015). In the sections below, “total copper” will be used when referring to all of the major oxidation states. However, “copper” will be used when referring to both Cu(I) and Cu(II), which cycle from one to the other (see section 9.1), are soluble, bioavailable, and considered the most relevant from a toxicological standpoint (see sections 4.3, 8 and 9.3); otherwise, the specific form will be used.
Sources and Uses
With an average concentration of 50 ppm in the Earth’s crust, copper occurs in several primary mineral deposits, mostly as sulphide ores (the most abundant being chalcopyrite, and chalcocite) and less frequently in its metallic form (CCME, 1999; Mauceri and Mishra, 2014). In general, weathering of these primary mineral deposits liberates secondary forms of copper including copper carbonate ores (e.g., malachite and azurite) and copper oxide ores (e.g., cuprite) (CCME, 1999; ATSDR, 2004; Ellingsen et al., 2007; Riedel, 2008; Mauceri and Mishra, 2014). In addition, anthropogenic activities like mining, municipal and industrial wastes management, agriculture (e.g., pesticides), and water treatment (e.g., copper sulphate pentahydrate in algaecides) can generate localized copper deposits and increase levels in the neighborhood (ATSDR, 2004; Mauceri and Mishra, 2014). Thus, copper can enter water sources from natural processes (e.g., soil weathering), human activities (e.g., agricultural runoffs), or from the corrosion of pipes/fittings in water distribution systems; the latter depends on water chemistry (e.g., low pH, high temperature, reduced hardness) (WHO, 1998; NRC, 2000; Ellingsen et al., 2007).
In Canada, the mining industry has produced 580,082; 652,595 and 695,513 tonnes of total copper in 2012, 2013 and 2014, respectively (British Geological Survey, 2016). Owing to its malleability, conductivity, alloying potential and resistance to corrosion and wear, copper metal is used in many products, such as pipes, paint pigments, antifouling agents, refinery oils, and in construction materials such as flashing electronic components, tubes, sheets, and rods for roofing (ATSDR, 2004; WHO, 2011; Stewart et al., 2012; Gaetke et al., 2014; Ellingsen et al., 2015). As a component of bronze and brass, it is used in marine equipment, cooking utensils, coins, to name a few. Copper nanoparticles are increasing in use for example in antimicrobial products such as biomedical and surgical devices (Ingle et al., 2014), or in copper nanowires (Yang et al., 2014). In addition, total copper is used by the health industry as a component of birth control pills, intrauterine devices (IUDs), dental amalgam, anti-microbial/ bacterial agents, vitamin and mineral supplements, and by the food industry as a colouring agent (Barceloux, 1999; Grass et al., 2011; Park et al., 2012; Stewart et al., 2012; Shaligram and Campbell, 2013). Finally, copper is used as an active ingredient in pesticides (fungicides, algaecides, and antimicrobials). There are currently nine copper compounds (e.g., cuprous oxide, copper sulphate, copper carbonate) registered as pesticides, for use in combination with other active ingredients, in Canada (Health Canada, 2009a).
Copper is present in tap water principally as a result of dissolution or leaching (i.e., corrosion) from components of distribution and plumbing systems that contain copper, particularly from the extensive use of copper pipes in household plumbing. In addition, fittings or service connections to homes and brass fittings may also represent a source of copper (Schock and Neff, 1988; Kimbrough, 2001, 2007, 2009). Copper leaching will vary with the types of alloys used and the manufacturing and fabrication processes (Lytle and Schock, 1996, 2000; Oliphant and Schock, 1996; Dudi et al., 2005). Several factors can influence copper corrosion, including the age of the piping and the water quality characteristics in the system. These factors are discussed in greater detail in section 7.2.
Copper release may result in blue water, which can stain bathroom fixtures and hair and may be caused by the presence of particulate copper. It is more difficult to determine the cause of particulate copper corrosion, but it is thought to primarily be the result of microbiological, chemical, or physical factors, or some combination of factors that also result in pitting corrosion and/or pipe failure (Edwards et al. 2000; Bremer et al. 2001; Arnold et al., 2012). The most significant home plumbing failures that occur as a result of internal corrosion are copper pitting and pinhole leaks. Pitting corrosion is not a well understood cause of copper release, but it can cause significant damage to copper plumbing, leading to leaks that can also cause substantial damage to homes and result in mould growth (Scardina et al., 2008). Pitting can occur relatively rapidly (a few months to years) after installation in cold water plumbing; whereas pitting generally takes many years (typically more than 8 years) to perforate pipe walls in hot water systems (Ferguson et al., 1996).
Environmental Fate
In the environment, copper metal and Cu (I) are unstable. Conversely, Cu (II) is more stable and the most abundant and frequently encountered species in the environment, including water and biological systems (Georgopoulos et al., 2001; Ellingsen et al., 2007; Riedel, 2008; Shike, 2009; Kiaune and Singhasemanon, 2011; Omur-Ozbek and Dietrich, 2011; Gaetke et al., 2014; Ellingsen et al., 2015). Hence, in general, surface oxidation leads to copper (I) oxide or hydroxide; but, due to its instability, Cu (I) is frequently oxidized to Cu(II) (WHO, 2004). Cu (II) is subject to redox reactions and tends to be hydrated. The hydrated Cu (II) ion rarely occurs free, but usually adsorbs to particulate matter or a variety of dissolved (inorganic and organic) compounds such as hydrated metal oxides (including those of iron, aluminum and manganese), polyvalent organic anions and humic acids (WHO, 1996, 2004; IPCS, 1998).
In the air, copper is mainly removed from the atmosphere by wet deposition (rain and snow), followed by gravitational settling and dry deposition (Kieber et al., 2004; WHO, 2004). In soil, copper strongly adsorbs to particles and has low mobility, with greatest retention at neutral pH (Health Canada, 2009a). Applied copper usually persists in topsoil, accumulating in upper layers (≈15 cm) (CCME, 1999; Kiaune and Singhasemanon, 2011). Its fate and availability in soils depend on many soil features: soil types (high mobility in sand vs. clays), pH (acidity and high reductive capacity favor release from oxide complexes), cation exchange capacity, water content, organic/inorganic matter content, redox potential, and the presence of oxides (e.g., of iron, manganese, aluminum) and clay (IPCS, 1998; Kiaune and Singhasemanon, 2011). Also, copper does not bioaccumulate, hydrolyze or breakdown in soil (Health Canada, 2009a).
In water, copper speciation is complex and influenced by many factors such as water source (e.g., Cu(II) is less bioavailable in hard water than in soft water), water chemistry (i.e., alkalinity, ionic strength, dissolved oxygen, pH and redox potential), sediments-water clays interaction (pH-dependent), presence of complexing (inorganic and organic) ligands and suspended particulate matter and carbon (WHO, 2004; Cuppett et al., 2006; Kiaune and Singhasemanon, 2011). The formation of Cu(I) and Cu(II)complexes can impact the solubility, color, and taste of water. Thus, when dissolved, Cu(I) is colorless while most Cu(II)- compounds impart a light blue-green colour and a metallic, bitter taste to drinking water (Georgopoulos et al., 2001; Cuppett et al., 2006; Omur-Ozbek and Dietrich, 2011). In raw water, copper primarily occurs as complexed Cu(II) and to a lesser extent as precipitated Cu(II) particles (Cu(II)-particulate matter), assuming typical pH values and oxidation-reduction potentials (Lide, 1993).
The primary copper-complexing ligands are dissolved molecules such as organic (e.g., dissolved organic carbon or matter) and inorganic species. On one hand, copper-organic complexes and copper-particulate matter compounds usually precipitate, rendering them non-bioavailable (Kiaune and Singhasemanon, 2011). On the other hand, inorganic ligands commonly form Cu(II) and Cu(I) complexes, alongside the ubiquitous copper hydroxo complexes; the latter, however, mostly precipitate (commonly at pH 6.5-12) as copper hydroxide (Cuppett et al., 2006). Thus, the further dissolution of the remaining copper complexes and the relative stability of Cu(II) as compared to Cu(I) makes free Cu(II) the most prevalent species in drinking water (Cuppett et al., 2006; Kiaune and Singhasemanon, 2011; Omur-Ozbek and Dietrich, 2011). Therefore, the main focus of this document will be on dissolved copper.
Aesthetic Characteristics
Copper in water can be detected through taste or visual inspection (i.e., colouration). Taste thresholds for copper have ranged from 0.0035 to > 5 mg/L (Zacarias et al., 2001; OmurOzbek and Dietrich, 2011) and 0.4-0.8 mg/L (Cuppett et al., 2006); reported taste threshold values vary significantly due to different sensory test methods used, the concentrations of the metal tested, the type of water used, and the type of data analysis performed. The aesthetic properties of copper are affected by solubility, with soluble copper being more readily tasted than the particulate form (Cuppett et al., 2006). However, both Cu(I) and Cu(II) ions are equally detectable producing a weak bitter and salty taste and an astringent mouth feel (Omur-Ozbek and Dietrich, 2011). OmurOzbek and Dietrich (2011) estimated that 79% of the population would be able to taste copper at a concentration of 2 mg/L, while 72% would be able to taste it at 1 mg/L. The visual detection of copper in water is a function of light path through the water, deposited particles, and the concentration of copper in water (Edwards et al., 2000). For example, 0.3 mg/L would be visible in a white bathtub at a distance of approximately 460 mm (18 inches) (typical observation distance), but 3 mg/L of copper would need to be present for the copper particulate to be noticeable in a glass of water.
Exposure
Canadians can be exposed to total copper via food, drinking water, air, soil and consumer products. For the general population, food is the major source of exposure (more than 90% of dietary intake), followed by drinking water (less than 10% of dietary intake) and copper-containing supplements (WHO, 2004; Sadhra et al., 2007; Cockell et al., 2008; de Romana et al., 2011; Shaligram and Campbell, 2013; Mauceri and Mishra, 2014; EFSA, 2015). However, drinking water can be a significant source of exposure if distributed through corroded pipes, especially in the case of infants fed formula (fortified, non-soy-based) reconstituted with water from such sources (Cockell et al., 2008; Chambers et al., 2010). Copper speciation in water differs from that in food (NRC, 2000) and this can influence its solubility and bioavailability. In fact, in food, copper is covalently bound to macromolecules (IOM, 2001; OEHHA, 2008; Ceko et al., 2014). In contrast, copper occurs free or complexed with ligands in drinking water. Thus, copper in drinking water can readily reach the bloodstream, bypassing the liver processing. Consequently, the bioavailability of copper in drinking water ingested in a fasted state is considered greater than that in food (OEHHA, 2008; Mauceri and Mishra, 2014; Ellingsen et al., 2015).
Food
The Canadian Total Diet Study (TDS) is a Health Canada initiative that measures the dietary concentrations of different chemicals in food and uses these data to estimate dietary intakes for different age-sex groups of the Canadian population (Health Canada, 2009b). Based on the TDS, the average intakes of dietary copper were estimated between 1993 and 2007 for Canadians of all age groups in 7 cities (Montreal, Ottawa, St John’s, Vancouver, Winnipeg, Toronto, and Halifax). These estimates (µg/ kg bodyweight [bw] per day) were 40-91 for 0-6 months old, 36-57 for 6 months-4 years old, 36-48 for 5-11 years old, 19-35 for 12-19 years old, and 13-32 for 20 years old and above. Based on copper intake for male and female 20+ years of age, an adult Canadian weighing 70 kg would have a dietary exposure to copper of 910-2,240 µg/day.
Based on the data collected as part of the TDS conducted between 1993 and 2007, copper concentrations in common food items vary in Canada, with the highest concentrations being food in organ meats (offal) such as liver and kidney. Lower copper levels are found in milk (25-70 µg/kg) , while infant milk formulas have concentrations of 650-850 µg/L, and human breast milk was reported to have 200-300 µg/L (Lonnerdal, 1998; Bauerly et al., 2005). The World Health Organization (WHO) and the Food and Agricultural Organization (FAO) recommended a minimum of 35 μg Cu/100 kcal formula (liquid or powder) intended to be marketed in order to comply with normal nutritional requirements of infants. They also acknowledged that levels adjustment may be needed in regions with high copper levels (FAO and WHO, 2011). Even if the copper content of human breast milk is lower than that of formula, this form of copper is highly bioavailable (Lonnerdal, 1996, 1998; Bauerly et al., 2005). In contrast, phytates (zinc and copper chelators found at high levels in soy-based infant formula) and soy proteins in infant formula reduce copper bioavailability (Gibson, 1994; NRC, 2000; IOM, 2001; WHO, 2004; EFSA, 2006, 2009; Stern et al., 2007; OEHHA, 2008; Collins and Klevay, 2011).
Water
Exposure to copper in drinking water can be properly assessed only by monitoring copper levels at the tap. This is because copper is present in tap water principally as a result of dissolution (corrosion) from components of distribution and household plumbing systems that contain copper. However, the concentration of copper is less variable both across a system and within an individual site (Kirmeyer et al., 1994; Schock et al., 1995), making the assessment of copper exposure from drinking water much less challenging than for lead. Monitoring of copper at the tap can be done using different sampling protocols; the selection of an appropriate protocol must take into consideration the desired objective, such as identifying sources of copper, effectively controlling corrosion or estimating exposure to copper.
Canadian Exposure to Copper from Drinking Water
Generally, across the provinces and territories, median copper levels at the tap were low (below 1 mg/L); however there are cases where high levels of copper release have been observed. In Prince Edward Island, dissolved copper (filtered through a 0.45 µm filter prior to sample preservation) was measured in ground water, municipal central water supply plants, private central water systems, and private wells and stand-alone system. Data from 2013-2015 were analyzed. For municipal central water supply plants, 48% of samples were below the detection limit (0.005 mg/L), and for the samples above the detection limit (n=117), the median dissolved copper level was 0.03 mg/L, with a maximum reported value of 0.9 mg/L. For municipal central water supplies, the 95th percentile was 0.12 mg/L, and the 97th percentile was 0.19 mg/L. In private central water systems, 67% of samples were below the detection limit, and for those samples above the detection limit (n=13), the median dissolved copper level was 0.013 mg/L, with a maximum reported value of 0.044 mg/L. For private wells, and stand-alone systems, 27% of samples were below the detection limit, and for those samples above the detection limit (n=2798), the median dissolved copper level was 0.017 mg/L, with a maximum reported value of 1.8 mg/L.
In Newfoundland, for the 62 source water samples taken in 2013-2014, all were found to be above the detection limit, and the median total copper level was 0.0020 mg/L, with a range of 0.001-0.073 mg/L. For source water samples, the 95th percentile was 0.031 mg/L, and the 97th percentile was 0.033 mg/L. For the 1968 tap water samples reported from 2013-2014, 97% were above the detection limit. For the tap water samples above the detection limit (n=1905), the median total copper level was 0.035 mg/L, with a range of 0.001-2.5 mg/L. For tap water samples, the 95th percentile was 0.4 mg/L and the 97th percentile was 0.49 mg/L. In Nova Scotia, for the 845 municipal treated water samples reported from 2005-2012, 73% of the total copper were above the detection limit of 0.002 mg/L. For the treated water samples above the detection limit, the median total copper level was 0.014 mg/L, with a range of 0.002-0.490 mg/L. The treated water samples 95th percentile was 0.12 mg/L, and the 97th percentile was 0.17 mg/L.
In New Brunswick, for copper samples from 2010-2015 the detection limit ranged from 0.000026 mg/L to 0.0004 mg/L. Samples are prepared by nitric acid preservation and analyzed for dissolved plus dilute acid labile trace copper, which is approximate to a total copper sample. For raw water, 71% of total copper samples were below the detection limit; for samples above the detection limit (n=367) the median copper level was 0.006 mg/L with a maximum of 8.3 mg/L. For water treatment plant samples, all the total copper samples were above the detection limit (n=17), with a median of 0.029 mg/L and a maximum of 0.11 mg/L. For water treatment plant samples, the 95th percentile was 0.083 mg/L, and the 97th percentile was 0.094 mg/L. For the distribution system samples, 20% were below the detection limit; for samples above the detection limit (n=1129), the median copper level was 0.021 mg/L with a maximum of 3.1 mg/L. For distribution system samples, the 95th percentile was 0. 48 mg/L, and the 97th percentile was 0.63 mg/L.
In Quebec, copper sampling is conducted at the tap following a five-minute flush. Of the 17,863 reported total copper samples from 2012-2015, 12.5% were below the detection limit (0.001- 0.01 mg/L), and 119 samples (0.7%) exceeded 1.0 mg/L. The median total copper concentration was 0.028 mg/L, and the maximum value detected was 15 mg/L. In Manitoba, total copper samples from 2009-2015 were analysed, and the detection limit was reported as 0.001 mg/L. For raw water, 6% of samples were below the detection limit, and for those samples above the detection limit (n=969), the median copper level was 0.003 mg/L, with a maximum of 0.94 mg/L. For raw water samples, the total copper 95th percentile was 0.03 mg/L, and the 97th percentile was 0.05 mg/L. For distributed water, all of the samples were above the detection limit, and the median copper level was 0.016 mg/L with a maximum reported level of 0.193 mg/L and minimum reported level of 0.002 mg/L (n=9). For distributed water samples, the 95th percentile was 0.133 mg/L, and the 97th percentile was 0.157 mg/L. For treated drinking water, 2% of the samples were below the detection limit, and for those samples above the detection limit (n=1026), the median copper level was 0.009 mg/L with a maximum of 0.8 mg/L. For treated water samples, the 95th percentile was 0.12 mg/L, and the 97th percentile was 0.19 mg/L. For untreated drinking water, 6% of the total copper samples were below the detection limit, and for those samples above the detection limit (n=165) the median copper level was 0.005 mg/L with a maximum of 0.62 mg/L. For disinfected drinking water samples, the 95th percentile was 0.08 mg/L, and the 97th percentile was 0.13 mg/L.
In Saskatchewan, of the 2423 total copper samples taken from distribution sites in 2005-2015, 90% were above the detection limit, with a median of 0.012 mg/L, and a range of 0.002-3.9 mg/L. The 95th percentile was 0.12 mg/L and the 97th percentile was 0.18 mg/L. In Alberta, for dissolved copper samples from 2010- 2015 the detection limit ranged from 0.002-0.003 mg/L. For raw water/entering the water treatment plant samples 43% were below the detection limit, and the median copper level was 0.005 mg/L with a maximum of 0.025 mg/L for those samples above the detection limit (n=131). For samples taken exiting the water treatment plant, 94% were below the detection limit, and the median copper level was 0.003 mg/L with a maximum of 0.17 mg/L for those samples above the detection limit (n=9). For water leaving the treatment plant, the 95th percentile was 0.105 mg/L, and the 97th percentile was 0.131 mg/L. For distribution system samples, 82% of samples were below the detection limit, and the median copper level was 0.004 mg/L, with a maximum of 0.147 mg/L for those samples above the detection limit (n=50). For distribution system samples, the 95th percentile was 0.047 mg/L, and the 97th percentile was 0.096 mg/L.
In British Columbia, for total copper samples from 2010- 2015, 11% were below the detection limit (0.0002-0.005 mg/L). For those samples above the detection limit (n=473), the median copper level was 0.0077 mg/L, with a maximum reported value of 0.549 mg/L. The 95th percentile was 0.0824 mg/L, and the 97th percentile was 0.13 mg/L. In the Yukon, samples from 2012-2015 taken from 20 communities were examined, and 19% of raw water and 10% of treated water total copper samples were below the detection limit (0.001-0.002 mg/L). For raw water samples above the detection limit (n=51), the median total copper level was 0.0029 mg/L, with a maximum reported value of 0.13 mg/L.
For raw water samples, the total copper 95th percentile was 0.054 mg/L, and the 97th percentile was 0.09 mg/L. For treated water samples above the detection limit (n=26), the median total copper level was 0.003 mg/L, with a maximum reported value of 0.0712 mg/L. For treated water samples, the total copper 95th percentile was 0.067 mg/L, and the 97th percentile was 0.07 mg/L.
High Canadian household tap water copper levels have also been documented in the literature. In a 1985 study found that 50% of rural Nova Scotia homes had copper levels exceeding 1 mg/L. The 55 homes surveyed were all served by private wells in three different areas of bedrock (granite, quartzite, and limestone). The authors did not find the Langelier, Ryznar, or Aggressiveness indexes, chloride to sulphate mass ratio (CSMR), alkalinity, or pH to be good predictors of corrosion. They also noted that from house to house, there was a large degree of variation of metal (copper, lead, zinc) mobilization (Maessen et al., 1985).
Sampling to Assess Copper Exposure from Drinking Water
Sampling protocols will differ, depending on the desired objective (i.e., identifying sources of copper, controlling corrosion, assessing compliance, estimating exposure to copper). As monitoring of copper at the tap can be done using different sampling protocols, it is important that the selected protocol be appropriate to meet the desired objective. Monitoring for typical exposure to total copper should be conducted at the consumer’s tap, with priority given to identifying homes with new copper plumbing or supplied with water having low or unstable pH or situated near dead ends, as these are likely to have the highest copper concentrations. Daycare facilities should also be prioritized for monitoring, to ensure that the most sensitive population (i.e., bottle-fed infants) is captured. Sampling should be conducted at least once per year, with the number of sites to be monitored determined based on the size of the drinking water system and the water quality characteristics.
Air
The Canadian National Air Pollution Surveillance Program reported copper concentrations in fine (PM2.5) particulates in 2014 (Environment Canada, 2014). 75.2% of samples exceeded the detection limit of 0.28-0.85 ng/m3, 14 samples were above 50 ng/m3 and 7 were above 100 ng/m3. The mean was 3.4 ng/m3 , the median was 1.2 ng/m3 , the 75th percentile was 2.4 ng/m3 , and the maximum was 430.3 ng/m3 . Similar results were observed in a Health Canada study of indoor and outdoor air (n=3,000 sites in Edmonton, Halifax, Ottawa, Sault-Saint-Marie, and Windsor), where average concentrations of 2.9 ng/m3 and medians < 2 ng/ m3 were reported (Health Canada, 2015). Indoor airborne copper concentrations of 8.2 ng/m3 (range of 3.8-17.8) and 11.4 ng/ m3 (range of 3.9-56.9) were reported in 10 Canadian rural and urban homes (PM2.5 filter), respectively (Rasmussen et al., 2006). Copper was shown to be mainly associated with the organic phase of house dust, increasing its bioavailability (Rasmussen et al., 2008). Based on these low concentrations of copper in ambient air, intake from air would be negligible compared with that from other media.
Consumer Products
Canadian quantitative estimates of total copper exposure from consumer products are rather scarce. However, copper compounds, e.g., copper(II) acetate, carbonate, chloride, sulfate, are part of various medicinal products available in Canada, and can be consumed in oral natural therapy formulae at maximum doses of 0.7 mg/day for children 1-3 years of age, up to 8 mg/ day for adults ≥ 19 years of age (Health Canada, 2007). The use of copper IUDs may also result in daily exposure of about 80 µg copper and less, with decreasing levels after the first few weeks from insertion (IPCS, 1998; Gaetke et al., 2014; Ellingsen et al., 2015). Although amalgams have been pointed out as a source of copper, exposure from this source is considered minimal (IPCS, 1998).
Soil
Copper distribution in rocks and soil varies as a reflection of the mineralogy of parent material and proximity to anthropogenic sources. Total copper mean concentration in Canada was estimated at 20 mg/kg soil (range: 2-100 mg/kg), with lower levels in the Canadian Shield (11 mg/kg) and higher levels in the Cordilleran region (46 mg/kg) (CCME, 1999). Levels of 41-65 mg/kg were reported in Ontario parklands, 25 mg/kg in western Manitoba, and 17-19 mg/kg in rural Alberta. Street dusts in Halifax had levels of 87 mg/kg. Copper was measured in garden soil (geometric mean [GM]: 12.3 mg/kg; 95th percentile: 19.4 mg/ kg), house dust (GM: 170.7 mg/kg; 95th percentile: 489.0 mg/kg), and street dust (GM: 38.1 mg/kg; 95th percentile: 236.2 mg/kg) from 50, 48 and 45 residences in Ottawa, respectively (Rasmussen et al., 2001).
Copper is used as a dietary supplement for farmed animals (Nicholson et al., 1999; Xiong et al., 2010) with some of the copper absorbed by the animal and the remainder excreted. The concentration of copper in manure has been reported in a study in China as 699.6 mg Cu/kg (pigs); 31.8 mg Cu/kg (cattle); 81.8 mg Cu/kg (chickens) and; 66.85 mg Cu/kg (sheep). In a study in England and Wales, 360 mg Cu/kg was found in pig manure, 80 mg Cu/kg in poultry manure and 50 mg Cu/kg in cattle manure. In a Canadian study examining run-off from manure compost windrows (containing a mixture of cattle manure and barley straw), the copper concentration in the compost ranged from 49 to 55 mg Cu/kg (Larney et al., 2014). Run-off from the manure was measured periodically over a 224-day period. On day 18 (first measurement), the copper concentration ranged from 0.3 to 0.62 mg/L. The copper concentration in run-off peaked at 0.78 mg/L on day 26. As the compost matured, the copper concentration significantly declined, and the copper concentration decreased by 67% between days 26 and 40.
Blood and Urine Levels in the Canadian Population
Copper levels in the blood and urine of Canadians are available from the Canadian Health Measures Survey (cycle 2) conducted by Statistics Canada, Health Canada and the Public Health Agency of Canada. As part of this cross-sectional study, health data and biological specimens were collected at 257 sites from 6,070 Canadians aged 3-79 years, distributed among six age groups (3-5, 6-11, 12-19, 20-39, 40-59 and 60-79 years). The geometric mean for whole blood copper concentration was 900 μg/L (95% confidence interval [CI] = 900-910 μg/L; n = 6,070), and the geometric mean for urinary copper concentration was 10 μg/L (95% CI = 10-11 μg/L; n = 6,311), for the total Canadian population aged 3-79 years (Health Canada, 2013). A study conducted by the Government of the Northwest Territories and Health Canada between 1994 and 1999 found that the geometric mean for maternal and umbilical cord plasma copper concentrations for 523 women from arctic Canada was 2.1 mg/L (Butler Walker et al., 2006). Levels did not differ between ethnic groups.
Multi-Route Exposure
Based on the physical and chemical properties of copper, drinking water exposure through the dermal and inhalation routes is likely negligible. Exposure to copper in vapors while showering is not expected since it is non-volatile, and no reports on copper absorption by inhalation were found. Although the generation of mists during showering could allow for inhalation of copper in aerosol form, the typical multi-route assessment, which measures inhalation exposure to volatile chemicals from showering and bathing, would not accurately represent copper aerosols inhaled during a showering event. There is little information on the dermal absorption of copper; however, bathing and showering are not expected to be significant sources of exposure, as indicated in a risk assessment of dissolved trace metals in drinking water. As a result, dermal and inhalation exposure during showering and bathing are unlikely to contribute significantly to the total exposure to copper through drinking water.
Analytical Methods
The United States Environmental Protection Agency (U.S. EPA) has several approved methods for measuring copper in drinking water:
• EPA 200.5 Rev 4.2 (U.S. EPA, 2003a)
• EPA 200.7 Rev 4.4 (U.S. EPA, 1994a)
• EPA 200.8 Rev 5.4 (U.S. EPA, 1994b)
• EPA 200.9 Rev 2.2 (U.S. EPA, 1994c)
• ASTM D1688-95A, ASTM D1688-02A, ASTM D1688- 07A, D1688-12A (ASTM, 1995, 2002, 2007, 2012).
• ASTM D1688-95C, ASTM D1688-02C, ASTM D1688- 07C, D1688-12C (ASTM, 1995, 2002, 2007, 2012).
• Hach 8026, Hach 10272 (Hach, 2014, 2016)
• SM 3111B (1992, 1995, 2005, 2012) and the online version of SM3111B (1999) (APHA et al., 1992, 1995, 1999, 2005, 2012)
• SM 3113B (1992, 1995, 2005, 2012) (APHA et al., 1992, 1995, 1999, 2004, 2005, 2010, 2012)
• SM 3120 B (1992, 1995, 1998, 2005, 2012) and the online version of SM 3120B (1999) (APHA et al., 1992, 1995, 1999, 2005, 2012).
The method detection limits (MDLs) for the methods listed above range between 0.02 and 500 µg/L. It should be noted, however, that MDLs are dependent on the sample matrix, instrumentation and selected operating conditions and will also vary between individual laboratories. In selecting an analytical method, consideration should be given to the water matrix to minimize the effect of interfering agents by using the prescribed sample pre-treatment or mitigation steps can help eliminate or decrease the effects of interfering agents.
ASTM D1688A, ASTMD1688C, EPA Rev 2.2, SM 3111B, SM 3113B are all atomic absorption-based methods. The light source is specific to each element measured, as each metal has a characteristic absorption wavelength (APHA et al., 2012). ASTM D1688 uses an atomic absorption direct method, with a method range of 0.5-5 mg/L (ASTM, 1995, 2002, 2007, 2012); whereas ASTM D1688C uses an atomic absorption graphite furnace method, with a method range of 5-100 µg/L (ASTM, 1995, 2002, 2007, 2012). EPA 200.9 Rev 2.2 uses a graphite furnace atomic absorption method, with a method detection limit of 0.7 µg/L (U.S. EPA, 1994c). SM 3111B (1992, 1995, 2005, 2012) and the online version of SM 3111B (1999) use a flame atomic absorption spectrometry method, with an instrument detection limit of 0.01 mg/L (APHA et al., 1992, 1995, 1999, 2005, 2012). SM 3113B (1992, 1995, 2005, 2012) and the online version of SM 3113B (1999, 2004, 2010) use an electrothermal atomic absorption spectrometry method with an estimated detection level of 0.7 µg/L with an optimum concentration range of 5-100 µg/L (APHA et al., 1992, 1995, 1999, 2004, 2005, 2010, 2012). Flame atomic absorption is subject to several interferences primarily chemical interferences, where there is a lack of absorption by atoms because they are bound in molecules, or they ionize in the flame. The electrothermal/graphite furnace method generally has detection levels 20 to 1000 times lower than flame-based methods as a result of increased atom density within the furnace (APHA et al., 2012). However, the electrothermal/graphite furnace method is also subject to more interferences, including spectral, matrix and memory interferences (U.S. EPA, 1994c) and may require increased analysis time compared to flame based methods (APHA et al., 2012).
EPA 200.7 Rev 4.4 uses an inductively coupled plasma atomic emission spectroscopy (ICP-AES) method, with a method detection limit of 0.003 mg/L (U.S. EPA, 1994a). EPA 200.5 Rev 4.2 uses an axially viewed ICP-AES method, with a method detection limit of 0.3 µg/L and a calculated reporting limit of 0.7 µg/L (U.S. EPA, 2003a). EPA 200.8 Rev 5.4 uses an inductively coupled plasma mass spectroscopy (ICP-MS) method, with a method detection limit of 0.5 µg/L in scanning mode and 0.02 µg/L in selective ion monitoring mode (U.S. EPA, 1994b). SM 3120 B (1992, 1995, 1998, 2005, 2012) and the online version of SM 3120B (1999) use an inductively coupled plasma method, with an estimated detection level of 6 µg/L (APHA et al., 1992, 1995, 1999, 2005, 2012). Sources of interference can include matrix effects and isobaric elemental and polyatomic ion interferences. Generally, for both ICP-AES and ICP-MS methods, the presence of high dissolved solids in a sample may interfere with ion transmission, and interferences can occur when dissolved solids exceed 0.2% (w/v) (U.S. EPA, 1994b; APHA et al., 2012).
Determination of copper by the colorimetric methods Hach 8026 and 10272 relies on the complexation of Cu(I) with bicinchoninic acid. Hach 8026 has a method range of 0.04-5 mg/L (Hach, 2014), and Hach 10272 has a method detection limit of 0.06 mg/L (Hach, 2016). In the colorimetric method, the Cu(II) is chemically reduced to Cu(I), and the concentration is determined through spectrophotometry at 560 nm. There are a number of interfering agents for Hach 8026 (Al, C3+ N-, Fe3+, Ag+ , hardness, pH ≤ 2) and Hach 10272 (CN- , Ag+ , pH ≤ 2) that can be mitigated through the use of a chelating agent or a prescribed pH adjustment. The current practical quantitation limit (PQL) is 0.05 mg/L (U.S. EPA,2009). In the second-six-year review of existing national primary drinking water regulations, the U.S. EPA indicated that although the laboratory passing rate was high, there were insufficient or inconclusive performance evaluation and proficiency testing data for copper to justify lowering the PQL (U.S. EPA, 2009).
Sample Preparation
The concentration of copper is much less variable than the concentration of lead, both across a system and within an individual site (Kirmeyer et al., 1994; Schock et al., 1995), making the analysis of total copper generally easier. Sample preparation to ensure that both the particulate and dissolved fractions of copper are capable of being detected is best practice, especially for diagnostic purposes. Generally, all methods listed above follow the same preservation steps, including the use of 0.15% nitric acid, a 16-hour holding time and the addition of hydrochloric acid for hot digestion when the sample turbidity is above 1 nephelometric turbidity unit (NTU). The standard acid preservation (pH < 2) has been shown to quantify total copper in water samples (U.S. EPA, 1994b; APHA et al., 2012). However, similarly to lead and other metals, when particles of copper are present in a sample, they may not be well dispersed and may settle to the bottom of the sampling bottle, resulting in turbidity below 1 NTU. As such, the current protocol may underestimate total copper in drinking water when particulate copper is present and best practice for the preservation step should be considered. Increasing the nitric acid strength to 2% for the preservation step may result in better recovery for most forms of particulate copper as has been observed with lead particulates (Haas et al., 2013; Triantafyllidou et al., 2013; Clark et al., 2014).
Best practice leading to a better estimation of total metals include ensuring that no aliquot or volume transfers occur prior to preservation or analysis, in situ sample preservation where feasible, maintaining a minimum holding time of 16 hours after preservation, thoroughly mixing the sample prior to analysis and taking the aliquot directly from the original sample bottle (Cartier et al., 2013; Haas et al., 2013; Triantafyllidou et al., 2013). It is important to note that the addition of 2% nitric acid should be undertaken by qualified personnel and using appropriate precautions. To this end, if sampling is conducted by homeowners, the sample should only be acidified and held upon arrival at the laboratory.
Treatment Technology
Municipal Scale
Historically, copper release has been considered an aesthetic nuisance in the case of blue water or a financial nuisance in the case of pipe failure attributed to pitting corrosion/pinhole leaks. Copper levels in source water are typically very low, and copper is generally introduced into drinking water after it leaves the treatment plant. The treatment approach for copper is generally focused on corrosion control. The approaches used for corrosion control include water quality adjustments (e.g., pH, alkalinity) and the use of corrosion inhibitors. The selection of an appropriate strategy for minimizing copper at the tap and, thus copper exposure, will depend on many factors including the characteristics of the raw water supply and the type of corrosion (Health Canada, 2009c). Utilities should undertake pilot-scale or pipe rig studies to understand how water quality changes to control copper will affect various chemical constituents in the treated water (e.g., iron and manganese) as well as materials in the distribution system (e.g., scale stability, lead-bearing components).
Site-specific investigations can be a useful tool when copper exceedances are limited to an individual building or neighborhood. An investigation of a widespread and rapid copper plumbing failure (2 months after installation) in a German hospital, Fischer et al. (1992) found evidence of both uniform and pitting corrosion in copper pipes. Biofilm was also found layered between corrosion products and implicated as a contributing factor to copper release. The authors also found that the areas that experienced greater corrosion were in intensively branched, horizontal sections of the pipe network that experienced long stagnation times. Arnold et al. (2012) conducted a site-specific investigation at a newly built U.S. school where copper release exceeded 6 mg/L and blue water was observed. Through targeted investigations, a practical long-term treatment strategy (removal of natural organic matter [NOM]) was identified for the school.
Treatment Considerations
Although copper levels in source water tend to be low, conventional water treatment technologies have been demonstrated both at the bench/pilot-scale as well as full scale to remove copper. Stover and Kincannon (1983) conducted bench/ pilot scales treatability studies using raw groundwater (pH 6). pH adjustments using lime decreased copper from 0.1 mg/L to < 0.001 mg/L (at pH 9.1 and 9.9) and 0.008 (at pH 11.3). Activated carbon adsorption was shown to also decrease copper from 0.1 mg/L to < 0 .001 mg/L (Stover and Kincannon, 1983). A survey of the performance of twelve water treatment plants in Colorado and California also provided some evidence of copper removal from source water (average influent concentration of 0.03 mg/L) through microstrainer (14%) , clarifier (26%) and filtration (37%) steps, for an overall removal of 49% (Zemansky, 1974). A comparison of three granular activated carbon (GAC) filter media was conducted in a river water supplied water treatment plant in Southern Poland. The water treatment plant consisted of two parallel treatment trains prior to the GAC filters consisting of either pre-ozonation, coagulation, flocculation, sedimentation and rapid sand filtration; or pre-ozonation, coagulation, clarification/ flocculation, rapid sand filtration and ozonation. The GAC filters were followed by storage where final disinfection occurred. The water entering the GAC filters was pH 6.8, 0.2 NTU.
The authors found that the copper concentration increased by 130-253% on the Chemviron F-300 filter media and by 1965% on the Norit ROW 08 filter media over the course of the first year of operation (influent concentration not reported). On the third filter media (WG-12), adsorption of copper was irregular and some desorption (release) of copper was observed. (Dabioch et al., 2013). For public water systems, the U.S. EPA has identified point-of-use (POU) ion exchange (using cationic resins) and reverse osmosis (RO) as small systems (i.e., serving fewer than 10 000 people) compliance technologies for copper removal (U.S. EPA, 1998). These technologies are also relevant for residentialscale treatment (see section 7.). The alkalinity of finished water is affected by the use of the reverse osmosis process. This process removes sodium, sulphate, chloride, calcium and bicarbonate ions and results in finished water that is corrosive (Taylor and Wiesner, 1999). This underlines the importance of process adjustments such as addition of a base and aeration of the permeate stream to recover alkalinity prior to distribution.
Distribution System Considerations
The National Plumbing Code of Canada (NPC) allows copper as an acceptable material for service lines and plumbing systems (NRCC, 2010), without respect to water quality. All provinces and territories use the NPC as the basis for their plumbing regulations.
However, in certain water qualities, both soluble and particulate copper may be released into drinking water from copper plumbing pipes, copper service lines and copper in brass fittings used in both home plumbing systems and in distribution mains, to levels exceeding the proposed MAC.
Factors Affecting Uniform Copper Corrosion
Schock and Lytle (2011) report that for general uniform copper corrosion and copper release, the most important variables are pH, alkalinity, redox potential (ORP) and plumbing age. Several other factors can also influence uniform copper corrosion including the stagnation time of the water and flow in pipes, temperature and other water quality conditions as well as NOM and biofilms.
39.5. pH
The effect of pH on the solubility of the corrosion products formed during the corrosion process is often the key to understanding the concentration of metals at the tap. Typically, distributed water with higher pH decreases the solubility of the corrosion products formed in the distribution system. Copper corrosion increases rapidly as the pH drops below 6; in addition, uniform corrosion rates can be high at low pH values (below about pH 7), causing metal thinning. At higher pH values (above about pH 8), copper corrosion problems are almost always associated with non-uniform or pitting corrosion processes (Edwards et al., 1994a; Ferguson et al., 1996). Edwards et al. (1994b) found that for new copper surfaces exposed to simple solutions that contained bicarbonate, chloride, nitrate, perchlorate or sulphate, increasing the pH from 5.5 to 7.0, roughly halved corrosion rates, but that further increases in pH yielded only subtle changes. The prediction of copper levels in drinking water relies on the solubility and physical properties of the Cu(II) oxide, hydroxide and basic carbonate solids that comprise most scales in copper water systems (Schock et al., 1995).
In the Cu(II) hydroxide model of Schock et al. (1995), a decrease in copper solubility with higher pH is evident. Above a pH of approximately 9.5, an upturn in solubility is predicted, caused by carbonate and hydroxide complexes increasing the solubility of Cu(II) hydroxide. Copper solids also age much more rapidly, transforming to much less soluble Cu(II) hydroxide phases, at higher pH and higher temperatures (Hidmi et Edwards, 1999). Copper solubility was found to be high in cold, high-alkalinity and high-sulphate groundwater up to about pH 7.9 (Edwards et al., 1994a).
Alkalinity
Alkalinity is a measure of the capacity of the water to neutralize acids and serves to control the buffer intensity of most water systems. Therefore, a minimum amount of alkalinity is necessary to provide a stable pH throughout the distribution system for corrosion control. Alkalinity is influenced by the presence of the conjugate bases of inorganic carbon (HCO3- and CO32-), organic acids and OH-ion and is expressed in mg CaCO3/L (Edzwald and Tobiason, 2011). Alkalinity is generally used interchangeably with dissolved inorganic carbon (DIC). The relationship between alkalinity, pH and cuprosolvency is complex, with the form of the controlling copper solid playing a critical role. Hard, high alkalinity groundwaters are particularly aggressive toward copper materials (Schock and Lytle, 2011). For example, copper releases of 1.4-2.4 mg/L were observed in high alkalinity (273 CaCO3 mg/L) and pH 7.4 water from tap water samples from relatively new copper plumbing (< 6 years old) in large buildings. The addition of orthophosphate at 3mg/L was sufficient to decrease copper release to below 1.3 mg/L (Grace et al., 2012).
Similarly, Schock and Fox (2001) found that in a high alkalinity (280 mg CaCO3 /L), pH 7.3 water, the 90th percentile copper levels exceeded 1.63 mg/L. Edwards et al., (1999) found that a combination of low pH (< 7.8) and high alkalinity (> 74 mg CaCO3/L) produced the worst-case 90th-percentile copper levels (Edwards et al., 1999). Laboratory and utility experience have demonstrated that copper corrosion releases are worse at higher alkalinity (Edwards et al., 1994b, 1996; Schock et al., 1995; Ferguson et al., 1996; Broo et al., 1998). These effects are likely due to the formation of soluble Cu(II) bicarbonate and carbonate complexes (Schock et al., 1995; Edwards et al., 1996) and in certain waters such as those that do not form insoluble malachite scale (Arnold et al., 2012). Both carbonate complexation and pH have a strong effect on copper solubility in relatively new plumbing systems. Low alkalinity (< 25 mg CaCO3/L) also proved to be problematic under utility experience (Schock et al., 1995). For high-alkalinity waters, the only practical solutions to reduced cuprosolvency are lime softening, removal of bicarbonate or addition of rather large amounts of orthophosphate (U.S. EPA, 2003b). Lower copper concentrations can be associated with higher alkalinity when the formation of the less soluble malachite and tenorite has been favoured (Schock et al., 1995; Edwards et al., 2002).
Redox Potential
The importance of redox potential (ORP) lies mostly in defining the conditions under which Cu(II) ions are formed (i.e., high oxidizing potential). At lower redox potentials, copper will not be subject to corrosion, or the formation of highly insoluble copper(I) oxide or hydroxide solids will occur, resulting in extremely low copper levels in the water (Schock et al., 1995; Ferguson et al., 1996). Many untreated groundwaters of neutral pH and high alkalinity fall into this category. The rate of release of copper into the water is influenced by the level of oxidizing agents in the system, especially free chlorine. Even low levels (i.e., 0.2 mg/L) affect the oxidation and corrosion rate (Schock and Lytle, 2011). Reiber (1989) has shown that the chlorine effect is much more important than dissolved oxygen in normal drinking water situations.
Hypochlorous acid is the predominant form of free chlorine below pH 7.5 in drinking water and the dominant oxidant on the copper surface (Atlas et al., 1982; Reiber, 1987, 1989; Hong and Macauley, 1998). Free chlorine residual was shown to increase the copper corrosion rate at lower pH (Atlas et al., 1982; Reiber, 1989). Conversely, free chlorine residual was shown to decrease the copper corrosion rate at pH 9.3 (Edwards and Ferguson, 1993; Edwards et al., 1999). However, Schock et al. (1995) concluded that free chlorine species would affect the equilibrium solubility of copper by stabilizing copper (II) solid phases, which results in a substantially higher level of copper release. The authors did not observe any direct effects of free chlorine on copper (II) solubility other than the change in valence state and, hence, the indirect change in potential of cuprosolvency.
Sometimes, copper levels in water that has been stagnant in the plumbing system can increase for as many as 48 to 72 hours, especially in relatively rare cases of blue water (Edwards et al., 2000). For this reason, an overnight standing sample is not necessarily representative of a worst-case scenario for exposure to copper (Schock et al., 1995). Conversely, the depletion of oxidants may decrease copper levels (Edwards et al., 1996; Schock and Lytle, 2011). The introduction of oxidative processes such as those for iron or manganese removal, or the initiation of disinfection after low-ORP conditions, could increase, decrease, or have no effect on copper concentration depending on the circumstances.
Limited information has been reported in the literature about the effect of other disinfectants on copper. A bench-scale study by Rahman et al. (2007) examined the effect of three disinfectants (chlorine, chlorine dioxide, and chloramines) under two pH (7.2 and 8.5) and alkalinity (10 or 100 mg CaCO3/L) conditions. The authors found that there was no significant difference in dissolved copper release for the disinfectant-free control compared to any of the disinfectants used in the study. Generally, the study found that the application of disinfectant lowered the copper release, with the lowest total copper release found under the pH 8.5, 10 mg CaCO3/L and 1.0 mg Cl2/L conditions. The authors suggested that the lower biomass (not assessed in the study) might explain part of the benefit to disinfectant, and that the long stagnation period (72 h) would result in low chlorine residuals. The authors also suggested that in the case of chlorine dioxide the presence of chlorite (a by-product) may have acted as a corrosion inhibitor.
Boyd et al. (2010) studied the effects of changing disinfectants from free chlorine to chloramines and vice versa on leaching rates of metals and concentrations of metals from lead, brass, and copper components in the distribution system. Local tap water was used, and water quality parameters (disinfectant residual, pH, alkalinity, and orthophosphate concentration) were monitored and maintained. The authors conducted pipe loop testing using new copper tubing and bronze piping (as a surrogate for standard brass) with different galvanic couplings (copper-bronze, leadbronze, and lead-copper couplings). Copper concentrations in unpassivated copper and bronze pipe loops were sensitive to free chlorine and chloramines but the effects were transient and not related to a specific disinfectant. A study assessed the leaching potential for several components tested under NSF/ANSI Standard 61 protocols with both chlorinated and chlorinated water. The study also tested these components using 28 waters (chlorinated and chloraminated) collected from utilities throughout the U.S. These utilities were also surveyed on copper and lead release issues. The authors found that neither chlorine nor chloramine was a dominant factor towards the leaching of copper under the test conditions for both the synthetic and utility waters. This mirrored the results from the utility survey that indicated few problems with copper (and lead) release when chloramines were used for secondary disinfection (Sandvig et al., 2012).
Pipe Age and Copper Solubility
Copper release into the drinking water largely depends on the type of scale formed within the plumbing system and whether they are relatively thin and adherent films of corrosion products such as copper(I) oxide (cuprite, Cu2O), copper(II) oxide (tenorite, CuO), or copper carbonate (malachite, Cu2(OH)2CO3) (Schock and Lytle, 2011). It can be assumed that at a given age, a corrosion product governs the release of copper into the drinking water. In uniform corrosion, solubility is highest for Cu(I) hydroxide [Cu(OH)2]and lowest for tenorite [CuO] and malachite [Cu2(OH)2CO3] (Schock et al., 1995). Similarly, Lagos et al. (2001) observed a linear relationship between pipe age and mean dissolved copper concentration in water. The more stable scales (malachite and tenorite) were also found to be the oldest and the least stable scale (langite), the youngest. Copper concentrations continue to decrease with the increasing age of plumbing materials, even after 10 or 20 years of service, when tenorite or malachite scales tend to predominate (Sharrett et al., 1982; Neuman, 1995; Edwards and McNeill, 2002). Sulphate and phosphate can decrease copper concentrations by forming bronchantite and copper(II) phosphate in the short term but, in the long term, they may prevent the formation of the stable tenorite and malachite scales (Edwards et al., 2002; Schock and Sandvig, 2009; Arnold et al., 2012).
Stagnation Time, Water Age and Flow
Long copper pipe of small diameter produces the greatest concentrations of copper upon stagnation (Kuch and Wagner, 1983; Ferguson et al., 1996). Copper behaviour is complex with respect to the stagnation of the water. Copper levels will initially increase upon stagnation of the water, but can subsequently decrease or increase, depending on the oxidant levels. Lytle and Schock (2000) showed that copper levels increased rapidly with the stagnation time of the water, but only until dissolved oxygen (DO) fell below 1 mg/L, after which they dropped significantly. Sorg et al. (1999) also observed that in softened water, copper concentrations increased to maximum levels of 4.4 and 6.8 mg/L after about 20-25 h of standing time, then dropped to 0.5 mg/L after 72-92 h.
Peak concentrations corresponded to the time when the DO was reduced to 1 mg/L or less. In non-softened water, the maximum was reached in less than 8 h, because the DO decreased more rapidly in the pipe loop exposed to non-softened water. Typically, water in a conventional home has a water age of 1 day compared to green buildings, where water age has been found to range from 2.7 days (net-zero energy house), 8 days (in a LEEDGold healthcare facility) and 1-6 months (net-zero office) (Rhoads et al., 2016). Increased water age has been associated with deterioration of water quality (loss of disinfectant residual, pH decrease) (Masters et al., 2015) as well as increases in microbial population (Masters et al., 2015; Rhoads et al., 2016) and could increase the propensity of water towards copper corrosion. As green building technology becomes more prevalent, consideration should be made for the implications of increased water age on water quality and corrosion. High flow velocities can sometimes be associated with erosion corrosion or the mechanical removal of the protective scale (impingement attack) in copper pipes. Water flowing at high velocity, combined with corrosive water quality, can rapidly deteriorate pipe materials. For example, the combination of high Cl2, continuous flow velocity with low-pH and high-alkalinity is extremely aggressive toward copper (Sarver et al., 2011; Schock and Lytle, 2011).
Temperature
No simple relationship exists between temperature and corrosion processes, because temperature influences several water quality parameters, such as dissolved oxygen solubility, solution viscosity, diffusion rates, activity coefficients, enthalpies of reactions, compound solubility, oxidation rates and biological activities (McNeill and Edwards, 2002). These parameters, in turn, influence the corrosion rate, the properties of the scales formed and the leaching of materials into the distribution system. In hot water systems of building plumbing, a high-water temperature (>60°C), in the presence of high flow velocity, is a factor in erosion corrosion (Ferguson et al., 1996). In a survey of the release of copper corrosion by-products into the drinking water of highrise buildings and single-family homes in the Greater Vancouver Regional District, Singh and Mavinic (1991) noted that copper concentrations in water run through cold water taps were typically one-third of copper concentrations in water run through hot water taps. A laboratory experiment that compared copper release at 4, 20, 24 and 60°C in a soft, low alkalinity water showed higher copper release at 60°C, but little difference in copper release between 4°C and 24°C (Boulay and Edwards, 2001). However, copper hydroxide solubility was shown to decrease with increasing temperature (Edwards et al., 1996; Hidmi and Edwards, 1999). In a survey of 365 utilities under the U.S. EPA Lead and Copper Rule, no significant trend between temperature and copper levels was found (Dodrill and Edwards, 1995).
Chloride and Sulpha
Chloride has traditionally been reported to be aggressive towards copper but has been found to be less detrimental for cuprosolvency than originally expected (Edwards et al., 1994b). High concentrations of chloride (71 mg/L) were shown to reduce the rate of copper corrosion at pH 7-8 (Edwards et al., 1994a, 1994b, 1996; Broo et al., 1997, 1999). Edwards and McNeill (2002) suggested that this dichotomy might be reconciled when long-term effects are considered instead of short-term effects: chloride increases copper corrosion rates over the short term. However, with aging, the copper surface becomes well protected by the corrosion products formed. Sulphate is a strong corrosion catalyst implicated in the pitting corrosion of copper (Schock, 1990; Edwards et al., 1994b; Ferguson et al., 1996; Berghult et al., 1999). Although sulphate was shown to decrease concentrations of copper in new copper materials, it was found that high sulphate concentrations resulted in higher copper levels in the experimental water once the copper material had aged (i.e., transitioned from copper(II) hydroxide to CuO) (Edwards et al., 2002). The authors concluded that this was due to the ability of sulphate to prevent the formation of the more stable and less soluble malachite and tenorite scales, supporting the hypothesis of Schock et al. (1995) that aqueous sulphate complexes are not likely to significantly influence cuprosolvency in potable water [51-60].
Natural Organic Matter
Research in copper plumbing pitting has indicated that some NOM may alleviate the propensity of a water to cause pitting attacks by promoting the formation of more protective scales (Campbell, 1954a, 1954b, 1971; Campbell and Turner, 1983; Edwards et al., 1994a; Korshin et al., 1996; Edwards and Sprague, 2001). However, NOM contains strong complexing groups and has been shown to increase the solubility of copper corrosion products (Korshin et al., 1996; Rehring and Edwards, 1996; Broo et al., 1998, 1999; Berghult et al., 1999, 2001; Edwards et al., 1999; Boulay and Edwards, 2001; Edwards and Sprague, 2001). Nevertheless, the significance of NOM to cuprosolvency relative to competing ligands has not been conclusively determined (Schock et al., 1995; Ferguson et al., 1996). A study found that NOM initially promoted pitting under certain narrow water quality and NOM concentration ranges. However, over long periods of time, a reduction in pitting at the higher NOM concentrations was observed (Korshin et al., 1996). Generally, the study concluded that NOM interacts with the copper through sorption, increasing the rate of copper leaching and the dispersion of the corrosion scales.
Mechanistic work by Edwards and Sprague (2001) elucidated some of the complex and apparently contradictory effects of NOM on copper. NOM was shown to contribute to copper by-product release through complexation and/or colloid mobilisation. NOM can interfere with pipe ageing through promoting the formation of highly soluble copper(II) hydroxide, copper carbonate and copper-NOM complex formation and thus, prevent the formation of the more stable solid, malachite. Conversely, NOM can serve as a food source for bacteria, which in turn consumes dissolved oxygen, triggering re-deposition of copper onto the pipe in the presence of a suitable catalyst (e.g., chloride). In practice, Arnold et al. (2012) demonstrated, at the bench-scale, that NOM removal was a practical and effective method to decrease copper release in a school experiencing blue-water issues. NOM removal increased dissolved oxygen (and the corrosion rate) and subsequently increased scale formation. The authors suggested that in the absence of NOM, the corrosion rate increased, accelerating the natural ageing process. Visual inspection of the copper pipes revealed a blue-green solid, which is associated with the stable crystalline solid, malachite.
Microbiologically Influenced Corrosion
Microbiologically influenced corrosion (MIC) results from a reaction between the pipe material and organisms, their metabolic by-products, or both (Schock and Lytle, 2011). Microbial activity can affect pH, metal solubility, and the oxidation-reduction potential of the surrounding microenvironment. Biofilms are surface associated communities of bacteria and their interaction with copper surfaces is complex. Bacteria can interact with the surface in physically, metabolically, or as storage reservoir for released metals. As biofilms form on the surface, corrosive microenvironments can form, for example, the area under the biofilm becomes oxygen deprived (anaerobic) and can act as an anode to the neighbouring metallic surface (cathode), thereby driving a localised corrosion event (Javaherdashti, 2008). Acidic metabolic products can also create local pH decreases (Little et al., 1991) and create bulk water pH decreases (Davidson et al., 1996). Bacteria in a biofilm produce sticky exopolymeric substances (EPS) that tend to have a negative charge, providing a binding site for positively charged metals ions (Qin-qin et al., 2012). Specifically, biofilm has been shown to act as a reservoir for released copper (Davidson et al., 1996; Lehtola et al., 2006; Qinqin et al., 2012; Payne et al., 2016), representing another potential source of copper exposure in a biofilm detachment event.
There have been several bench-scale studies, as well as autopsies of corroded copper plumbing, that have indicated a significant role for bacteria and biofilm in cuprosolvency. Dutkiewicz and Fallowfield (1998) found that copper exposed to Agrobacterium sp., isolated from a copper pipe biofilm found in Adelaide, Australia, increased copper release compared to a sterile control. In an Australian study, Sphingomonas spp. and Acidovorax sp. have been identified as species associated with corroding copper surfaces, and a species of Acidovorax has been observed to accumulate copper when grown in a laboratory setting on a copper-containing food source (Critchley et al., 2004). Conversely, other bacteria, Stenotrophomonas maltophilia, were observed to have protective effects on copper, which the authors hypothesized could be a result of the binding of the chloride ions to the EPS (Critchley et al., 2003).
Copper has antimicrobial properties (Grass et al., 2011), and in drinking water delayed biofilm formation on copper pipes has been observed when compared to polyethylene pipes. Although after 200 days there was a significant decrease in virus numbers on copper pipe biofilm, there was no significant difference in microbial numbers (Lehtola et al., 2004). Biofilms have also been implied as causative or contributing agents to both blue water events (Arens et al., 1996; Critchley et al., 2004) and pitting corrosion (Fischer et al., 1992; Reyes et al., 2008; Burleigh et al., 2014).
Blue Water Events
Copper dissolution can cause blue or green water events and is frequently characterized by a fine dispersion of copper corrosion products. Particulate copper appears light blue as a copper hydroxide and black as copper sulphide. Visual perception of a blue water issue is challenging as the coloured water can be flushed away through the first flush of a toilet or tap. The distance of the observer from the coloured water also affects the perception, with 0.3 mg/L visible at the typical observation distance from a bathtub, but 3 mg/L would need to present for the copper particulate to be noticeable in a glass of water (Edwards et al., 2000). No single cause has been identified for blue water events; they are thought to result from a combination of microbial activity and certain water quality conditions. Critchley et al. (2004) successfully induced blue water in a laboratory setting by inoculating copper pipe rigs with non-sterile copper corrosion extracts, although the water used was not representative of drinking water. The authors noted a nine-week lag time before blue water was apparent suggesting that there is an initial establishment phase to initiate this type of corrosion. Previously, Arens et al. (1996) investigated a blue water event in recently commissioned copper pipes. MIC was suspected as the causative agent and Arens et al. (1996) found that various food sources (e.g., sealing putty) increased microbial activity and copper release. Arnold et al. (2012) also reported blue water and elevated copper (>6 mg/L) in new copper plumbing, in a high alkalinity (200 mg CaCO3/L), pH 7.15-7.4 water. NOM removal provided long-term copper stability through accelerated pipe ageing.
Brass Alloys
Brasses are, basically, alloys of copper and zinc, with other minor constituents, such as lead. Brass dezincification is thought to be an either a selective leaching of zinc or a multi-stage process through which zinc and copper are leached through uniform corrosion and copper is redeposited, creating a net effect of zinc leaching and both mechanisms leave behind a porous weakened copper surface (Lytle and Schock, 1996; Sarver et al., 2011). Zinc oxide corrosion products can build-up forming a “meringue” structure that restricts flow, which can subsequently cause leaks and bursts. Brasses are particularly vulnerable to dezincification in low alkalinity, high chloride water (Sarver et al., 2011). Ideally, brass used in premise plumbing should be low zinc to help make the brass dezincification resistant. However, it should also be noted that in cross-linked polyethylene (PEX) plumbing systems, higher zinc content brass (yellow brass) fittings are frequently used. There have been widespread plumbing failures associated with PEX and dezincification of brass fittings documented in North America (U.S. District Court, 2011; Ontario Superior Court of Justice, 2011; Superior Court Province of Quebec, 2011).
In addition to the financial ramifications of brass corrosion, other metals are also released, including copper. Lytle and Schock (1996) demonstrated that the composition of the brass alloy was a significant factor in the amount of copper released and that small changes in pH were a strong influence on copper release. A pH 8.5 and 7, with red brasses (low zinc) released more copper than yellow brasses (high zinc) and some red brasses released similar copper levels as pure copper. Brasses at pH 7 released more copper than at pH 8.5. It should also be noted that at pH 7, the copper releases were more sporadic. In practice brass can represent a significant source of copper release. A cluster of Southern California homes with all plastic plumbing, brass fittings were the major source of copper release with medians ranging from 85-185 µg/L over the course of three sampling events.
Pitting Corrosion
Pitting corrosion is a form of non-uniform corrosion in all types of copper pipe and tubing and can ultimately lead to pinhole leaks, a costly plumbing failure (Scardina et al., 2008). Pitting corrosion can occur where a localized corrosion cell forms as a result of several mechanisms such as a differential in pH or oxygen on the pipe surface. In the localized corrosion cell, the cathodic area is protected, and the anodic area is oxidized, electrons are released, and copper is depleted, creating a pit. The pit can ultimately breach the wall of the copper, creating a small pinhole. If unfavorable water quality conditions occur prior to the formation of the protective scale, then a serious pitting corrosion may occur (Schock and Lytle, 2011). The causal pit initiation mechanism could be a result of one or a combination of several factors including surface imperfections, carbon residues from manufacturing, soldering flux, unfavorable water quality or microbiological activity.
There are certain water quality, microbiological and flow conditions that appear to induce or propagate pits and include: waters with high pH, low alkalinity, the presence of chloride (Lytle and Schock, 2008; Lytle and Nadagouda, 2010) and sulphides (Jacobs et al., 1998; Jacobs and Edwards, 2000); chlorine, high pH, low alkalinity (Sarver et al., 2011); the presence of some biofilms (Fischer et al., 1992; Reyes et al., 2008; Burleigh et al., 2014); pipe configuration (Fisher et al., 1992) and high water velocity (Custalow, 2009). Lytle and Schock (2011) describe the three types of copper pitting (based on chemistry and physical features): cold-water (type I), hot-water (type II) and soft-water (type III) as well as conditions typically associated with each type. It is important to note that sulphate and chloride are typically associated in one way or another to all forms of pitting although the relative importance of chloride versus sulfate in copper pitting corrosion is still a subject of debate (Schock and Lytle, 2011).
In bench-scale experiments, the addition of 3 mg PO4 /L or increasing DIC to 50 mg/L (from 5-25 mg/L) was effective in preventing pit initiation at pH 9 (Lytle and Schock, 2008). It has been theorised that phosphate may displace other anions from the anode and compete for binding sites with Cu2+ to form a relatively stable scale and that alkalinity decreases pit initiation by either, limiting pH differentials on the surface or by forming copper carbonates and decreasing precipitation on the surface. Ha and Scully (2013) suggested that phosphate (as Na2HPO4) addition inhibits pit initiation through increasing passivation and suppressing the dissolution of copper. Lytle and Nadagouda (2010) analysed the elemental composition of copper pits taken from in-home plumbing and found that chloride was concentrated in the floor of the pits as well as in cracks or imperfections near the surface, suggesting that chloride plays an important role in the propagation of pits. Although the presence of chloride has been demonstrated to play a fundamental role in pit initiation, Edwards et al. (1994a) hypothesized that given longer exposures (with greater scale formation) that chloride could potentially have a protective effect. Sulphides have been shown to be significant factors in inducing copper corrosion and have been associated with pitting at pH 9.2 (Jacobs and Edwards, 2000).
Burleigh et al. (2014) evaluated copper pitting in copper tubing from multiple cold-water lines in Santa Fe, New Mexico and found evidence of Actinobacteria deep inside the pits. The authors suggested that Actinobacteria role in copper pitting could be through either the excretion of an acidic metabolic product or using copper as an energy source (Burleigh et al., 2014). Reyes et al (2008) compared two communities in Chile, one rural (pH 6.2,CaCO3 = 63 mg/L, no chlorine) and one urban (pH = 6.9, CaCO3 = 98 mg/L, chlorinated), and although the two communities had similar water quality, they had different stagnant copper levels. The rural homes had stagnant copper levels of 5 mg/L compared to the urban homes where stagnant copper levels were 0.4 mg/L. When investigating the copper release in rural homes, the authors found that only one quarter of the copper release could be attributed to water quality factors and the microbiological component was suspected as another causal agent of copper release. In the rural homes pipes with biofilm, surface pitting was observed; whereas in the absence of bacterial biofilms no pitting was observed. High water velocity has also been demonstrated to increase pitting in high pH chlorinated waters (Custalow, 2009) and are likely a result of the continuous resupply of oxidants to the cathodic regions of a localised corrosion cell (Sarver et al., 2011). Leaking pipes present an additional health risk as they could lead to mould growth (Scardina et al., 2008), affecting indoor air quality (Health Canada, 2016).
Impact of Treatment and/or Water Quality Changes on Copper Release
Distribution system deposits and corrosion scales lie in delicate balance with the aqueous environment and utilities should be aware that changes in water quality can affect the stability of distribution system scale. Some treatment technologies can increase copper in drinking water by changing water quality parameters that impact copper release. Changing water treatment to address one issue could inadvertently negatively impact another water treatment objective. For example, switching from a sulphate to chloride containing coagulant has been shown to increase CSMR and exacerbate lead release in premise plumbing material (Edwards and Triantafyllidou, 2010).
In the anion exchange process, when removing contaminants such as uranium, freshly regenerated ion exchange resin removes bicarbonate ions. This causes reductions in pH and total alkalinity during the initial 100 bed volumes (BVs) of a run and may require raising the pH of the treated water at the beginning of a run to avoid corrosion (Clifford, 1999; Wang et al., 2010; Clifford et al., 2011). Similarly, frequent regeneration of an ion exchange resin can have an impact on corrosion. In a case study in Maine, frequent regeneration of the ion exchange resin was required to reduce the levels of uranium in the waste stream (residuals). This resulted in a significant and continual decrease of pH and subsequent leaching of copper and lead into the drinking water (Lowry, 2009, 2010).
Since reverse osmosis (RO) continually and completely removes alkalinity in water, it will continually lower the pH of treated water and increase its corrosivity. Therefore, the product water pH must be adjusted to avoid corrosion issues in the distribution system such as the leaching of lead and copper (Schock and Lytle, 2011; U.S. EPA, 2012). Additionally, changing from lime softening, which balances hardness and alkalinity with a high pH, to ion exchange softening can leave a high alkalinity, which is corrosive to copper (Schock and Lytle, 2011). Similarly changes to finished water to address corrosion concerns can cause other unintended changes in the distribution system. For example, raising pH in finished water to greater than 9 in order to decrease copper release from premise plumbing, would precipitate dissolved manganese in the treated water, which could lead to unwanted aesthetic issues.
Additionally, it is important to consider that distribution system deposits are susceptible to changes in water quality. Distribution system deposits are comprised of the underlying corroded material (e.g. cast iron) as well as the accumulated trace inorganics that have accumulated over the service life of the water main. Friedman et al. (2010) found that trace inorganic contaminants (including barium lead nickel, vanadium, arsenic, radium, chromium and uranium) had accumulated in manganese deposits in distribution systems. Gerke et al. (2016) reported copper accumulation in both chlorinated and chloraminated distribution system manganese deposits. The stability of deposited inorganics is dependent upon maintaining stable water quality conditions (e.g., pH, ORP, corrosion control), as well as avoiding the uncontrolled blending of source water (ground and surface), and the uncontrolled blending of chlorinated and chloraminated water (Friedman et al., 2010). Before changing water quality to achieve a new treatment goal, such as corrosion control, utilities should undertake pilot-scale studies to understand how a proposed water quality amendment will affect various components of their distribution system.
Mitigation of Copper Release
The selection of a mitigation strategy for copper should take into consideration other metals present since different strategies, including water quality adjustments, may impact various metals differently. The use of test pipe-rigs under site specific water quality conditions is recommended, as the testing process can be helpful in selecting an effective corrosion control strategy and aid in anticipating unintended impacts. Generally, the level of trace metals increases upon stagnation of the water but may vary according to water quality. For example, in standing water in plumbing systems where oxidants are present, copper levels can increase for up to 48 to 72 hours. As such, flushing the water present in the plumbing system can significantly reduce the levels of copper and is, therefore, considered a mitigation strategy. However, flushing the cold-water tap may not be sufficient to reduce the levels of copper (Singh and Mavinic, 1991; Murphy, 1993; Barn et al., 2014). Periodic flushing throughout the day and extensive flushing following long stagnation periods (vacation periods, weekends) may therefore be advisable to provide suitable water quality throughout the day. Reducing exposure to copper can also be achieved, as an interim measure, by the use of drinking water treatment devices. It must be noted that in situations where high levels of copper occur, drinking water treatment devices may have reduced capacity and require more frequent replacement of the filter.
Alkalinity and pH
Copper solubility is strongly influenced by both alkalinity (through carbonate complexation) and pH, for relatively new plumbing systems (Edwards et al., 1994b; Dodrill and Edwards, 1995). However, it was also noted that high alkalinity groundwaters are aggressive toward copper materials (Schock et al., 1995; Vik et al., 1996; Schock and Lytle, 2011). The adjustment of pH at the water treatment plant is the most common method used for reducing corrosion in drinking water distribution systems and leaching of contaminants, such as copper, in the distributed water. Raising the pH is an effective method for reducing copper corrosion and thus, minimizing copper levels in drinking water. Generally, uniform corrosion of copper is best controlled at pH > 7, depending on alkalinity (Schock and Lytle, 2011).
Treatment chemicals used for adjusting pH include caustic soda, potash, lime and sulfuric acid. Minimal pH adjustment can also be achieved using CO2, with either sodium carbonate (Na2CO3) or sodium bicarbonate (NaHCO3); however, this combination primarily adjusts alkalinity. Aeration is a non-chemical treatment method that introduces air in the water, removing CO2 and resulting in a pH increase. Another method of adjusting pH is the use of limestone contactors: water flows through a bed of crushed limestone, dissolving the limestone and causing the pH (as well as alkalinity and calcium level) to increase. These last two methods offer less precise control of the pH adjustment. A combination of lime or caustic soda and carbon dioxide offers more precise control, as it allows the pH and alkalinity of the water to be independently adjusted (Schock, 1999).
Alkalinity, in combination with pH, serves to control the buffer intensity of most water systems. Therefore, sufficient alkalinity is necessary to provide a stable pH throughout the distribution system for corrosion control of copper (and other metals such as lead). Alkalinity can improve the buffering capacity at a given pH. Raising or lowering the pH can substantially increase the buffering capacity of the water as seen in buffer intensity plots developed by Schock and Lytle (2011). This was demonstrated in practice by Clement et al. (1998) in a field study in Concord, New Hampshire, where minor adjustments in pH and alkalinity resulted in greater pH stability in the distribution system and improved corrosion control. In very low alkalinity waters, adjustment of the alkalinity can be achieved by adding carbonate ion with either Na2CO3 or NaHCO3. As stated previously, these chemicals are also used to adjust pH because they also contribute carbonate (CO32-) or bicarbonate (HCO3-) ions. It is important to note that the number of carbonate ions available is a complex function of pH, temperature, and other water quality parameters (Schock, 1999).
Addition of Corrosion Inhibitors
Phosphate- and silicate-based corrosion inhibitors are typically used for corrosion control in drinking water. The most commonly used inhibitors are phosphate corrosion inhibitors with or without zinc, which include orthophosphate or polyphosphate (typically, blended polyphosphates). Silicate corrosion inhibitors (i.e. sodium silicates) have also been used for corrosion control. The success of corrosion inhibitors is often based on pilot- or bench-scale testing and depends on both the water quality and the conditions prevailing in the distribution system. The effectiveness of corrosion inhibitors is largely dependent on maintaining a residual of the inhibitor throughout the distribution system and on the pH and alkalinity of the water. Measuring the concentration of inhibitors within the distribution system is part of good corrosion control practice. Generally, direct correlations between the residual concentration of inhibitors in the distribution system and the levels of metals, including copper at the tap, are not possible.
Health Canada recommends that water utilities choose drinking water additives, such as corrosion inhibitors, that have been certified as conforming to the applicable NSF/ANSI health-based standard. Phosphate- and silicate-based corrosion inhibitors are included in NSF/ANSI Standard 60, Drinking Water Treatment Chemicals-Health Effects (NSF International, 2015a). These standards have been designed to safeguard drinking water by ensuring that additives meet minimum health effects requirements and thus are safe for use in drinking water.
Phosphate-Based Inhibitors
Orthophosphate and zinc orthophosphate are the inhibitors most often reported in the literature as being successful in reducing copper levels in drinking water (Bancroft, 1988; Reiber, 1989; Boffardi, 1993; Johnson et al., 1993; Dodrill and Edwards, 1995; Rezania and Anderl, 1995, 1997; Schock et al., 1995; Boireau et al., 1997; MacQuarrie et al., 1997; Churchill et al., 2000; Schock and Fox, 2001; Becker, 2002; Dudi, 2004; Kirmeyer et al., 2004). Orthophosphate is generally used for lead control when pH control or pH and alkalinity adjustment are not sufficient to control corrosion (Vik et al., 1996), and the effects on copper are secondary considerations.
Some authors have reported that the use of orthophosphate may reduce copper levels in the short term, but that in the long term the formation of more stable scales such as malachite and tenorite may be prevented (Schock and Clement, 1998; Edwards et al., 2001; Cantor et al., 2003; Schock and Sandvig, 2009). There is evidence that ineffective treatment for copper with phosphate was successful when higher dosages were applied or when pH and orthophosphate dosages were optimized (Schock et al., 1996; Schock and Fox, 2001). Schock and Fox (2001) demonstrated successful copper control in high-alkalinity water with orthophosphate when pH and alkalinity adjustments were not successful. Similarly, Grace et al. (2012) found that orthophosphate dosed at 3 mg/L effectively decreased copper levels below 1.3 mg/L from 1.4-2.4 mg/L in a high alkalinity (273 mg/L) neutral pH (7.4) water. Bench-scale results also indicated that a pH increase to 8.4 may also have been successful; however due to the high alkalinity of the water this option was not selected over concerns of calcium carbonate deposition on distribution system components (Grace et al., 2012).
A survey of 365 water utilities under the U.S. EPA Lead and Copper Rule also revealed that utilities using orthophosphate had significantly lower copper levels only when pH was below 7.8 and alkalinity was below 74 mg CaCO3/L as (Dodrill and Edwards, 1995). Limited data are available on the impact of polyphosphate on copper solubility. Generally, polyphosphates increase the corrosion rate of copper (Vik et al., 1996). In a case study, Cantor et al. (2000) reported that the use of a polyphosphate blend, at one of the three utilities studied, increased copper levels at the tap. In a copper pipe rig study, Edwards et al. (2002) reported that although polyphosphate generally reduced soluble copper concentrations, they also significantly increased them at pH 7.2 and alkalinity of 300 mg CaCO3/L since polyphosphates hinder the formation of the more stable malachite scales.
Silicate-Based Inhibitors
Only limited data are available on the impact of sodium silicate on copper solubility. As sodium silicate is a basic compound, it is always associated with an increase in pH, making it difficult to attribute reductions in copper concentrations to sodium silicate alone when an increase in pH may also result in a decrease in copper concentrations. A study conducted by Schock (2005) in a medium-sized utility was able to solve problems from iron in source water as well as lead and copper leaching in the plumbing system. The problems were solved simultaneously through the addition of sodium silicate with chlorination. Sodium silicate was added to the three wells that contained elevated levels of iron and manganese and that serviced homes containing lead service lines. A fourth well required only chlorination and pH adjustment with sodium hydroxide. At the three wells, an initial silicate dose of 25- 30 mg/L increased the pH from 6.3 to 7.5 and immediately resulted in 55% and 87% reductions in lead and copper levels, respectively. An increase in the silicate dose to 45-55 mg/L increased the pH to 7.5 and resulted in an even greater reduction in the lead and copper levels (0.002 mg/L and 0.27 mg/L, respectively).
Lintereur et al. (2011) compared three different sodium silicate dosages (3 mg/L, 6 mg/L, and 12 mg/L) and found that sodium silicate decreased copper release compared to the control (i.e., no treatment) and pH increase control. The decrease appeared to be dose dependant, with the lowest copper releases observed at the highest doses of sodium silicate. Additionally, scale analysis revealed a silicate-copper scale on coupons retrieved from the silicate treated systems, providing evidence that a silicate scale is at least partly responsible for the inhibitory action of sodium silicates. Woszczynski et al. (2015) found that sodium silicates (18 mg-Si/L, pH 7.3 and pH 6.3) did not control copper when compared to treatment by phosphate (0.8 mg-PO4 /L, pH 7.3).The authors noted that silicate performance was affected by pH and suggested that water quality conditions could affect sodium silicate performance.
Mitigation of Blue Water Events
Blue water remains a poorly understood phenomenon, and there is limited guidance on its management. Short term management strategies include identified heat-shock (> 60ºC) (Arens et al., 1996) or a one-time super chlorination Edwards et al. (2000), although the blue water could reoccur. Arnold et al. (2012) found that NOM removal through GAC provided a practical long-term solution for a blue water issue. Sequestering agents, such as polyphosphate, will mask the copper release, improving the aesthetic quality without decreasing copper levels and may, in some cases, increase copper levels. Cantor et al. (2000) reported on an incident in Wisconsin, where polyphosphates were used to sequester iron in one of three communities with a hard, high alkalinity groundwater. Subsequent to this treatment, copper related illnesses were reported to and confirmed by the Wisconsin Department of Health and Family Services (Knobeloch et al., 1994). The highest copper levels (up to 5 mg/L) were found in new and newly remodeled homes and the levels decreased over time once polyphosphate was no longer dosed (Cantor et al., 2000). Generally, the use of sequestering agents is not recommended for mitigating copper release.
Mitigation of Copper Pitting and Dezincification
Given the variety of water quality, microbiological and flow condition factors that can affect the propensity towards copper pitting, utilities should consider using tools such as those found in Sarver et al. (2011). These tools help utilities avoid or mitigate copper pitting and address key water quality changes including the removal of NOM, phosphate, silicate as well as waters with chlorine, high pH or low alkalinity. In addition, utilities should conduct pilot-testing to evaluate their particular water quality and any proposed or potential changes to water quality to both diagnose a problem and to identify workable solutions (Lytle and Schock, 2008; Lytle et al., 2012). A low-cost pipe-loop system is described in Lytle et al. (2012) and could serve as an evaluative tool for utilities.
The water quality conditions associated with a tendency towards dezincification are not as clearly characterized when compared to the copper pitting phenomenon. However, high alkalinity low chloride water is associated with decreased dezincification (Sarver et al., 2011). Lytle and Schock (1996) found that orthophosphate did not provide a clear benefit at pH 7 and 8.5 although they suggested that orthophosphate might be more effective for copper leaching from brass at pH 6.5 (Lytle and Schock, 1996). Sarver et al. (2011) suggested that utilities determine the aggressiveness of their particular water quality towards brass alloys and communicate this information to stakeholders (consumers and developers) and encourage the use of dezincification resistant brass alloys.
Impact of Copper Ions on Lead Release
Copper has also been observed to play a role in lead release in downstream plumbing. Clark et al. (2015) observed copper deposition onto lead surfaces in certain water conditions significantly increased lead release. The copper deposits were hypothesized to act as small galvanic connections on the lead surface. The solubility of copper directly correlated to copper deposition and release of lead. Based on bench-scale experiments, Hu et al. (2012) deduced that between 37- 92% of the Cu2in chloraminated water and 62-91% of Cu2+ in chlorinated water accumulated on new lead pipe under stagnation conditions. Copper deposition was also observed to be affected by flow and stagnant conditions, as lead release was greatest from the chlorinated system under flow-through conditions; whereas lead release was greatest from the chloraminated system under stagnant conditions. Although, the specific water quality and flow conditions that exacerbate the effects of copper deposition still require further investigation, the role of copper deposition on lead release could be a significant consideration when upstream copper tubing is followed by lead bearing materials, such as in the case of partial lead service line replacements.
Residential Scale
It is not generally recommended that drinking water treatment devices be used to provide additional treatment to municipally treated water. However, as the primary source of copper in drinking water is the leaching from plumbing and distribution system components, a private residential drinking water treatment device is the best option for reducing copper concentrations in drinking water at the tap. However, the use of such devices should not be considered to be a permanent solution.
Before a treatment device is installed, the water should be tested to determine general water chemistry and to verify the concentration of copper. Periodic testing by an accredited laboratory should be conducted on both the water entering the treatment device and the finished water to verify that the treatment device is effective. Products that use adsorption technology can lose removal capacity through usage and time and need to be maintained and/or replaced. Consumers should verify the expected longevity of the adsorption media in their treatment device as per the manufacturer’s recommendations and service the device when required.
Health Canada does not recommend specific brands of drinking water treatment devices, but it strongly recommends that consumers use devices that have been certified by an accredited certification body as meeting the appropriate NSF/ANSI drinking water treatment unit standard(s). These standards have been designed to safeguard drinking water by helping to ensure the material safety and performance of products that come into contact with drinking water. Certification organizations provide assurance that a product conforms to applicable standards and must be accredited by the Standards Council of Canada (SCC).
Drinking water treatment devices can be installed at the faucet (POU) or at the location where water enters the home (pointof-entry or POE) in residential settings to reduce contaminant concentrations. POU systems are preferred for the removal of copper, as copper levels may increase in the plumbing system and because exposure to the contaminants from drinking water is a concern only if the contaminants are ingested (i.e., inhalation and dermal absorption are not significant routes of exposure). As such, POU treatment devices installed at individual drinking water taps are considered to be the best approach to reduce concentrations to safe levels immediately before consumption or for preparation of food or beverages.
A number of certified residential treatment devices are available that can remove copper from drinking water to below the proposed MAC of 2 mg/L. Adsorption (i.e., carbon block/ resin), RO and distillation technologies are effective treatment technologies at the residential scale for the removal of copper at the tap. Certified residential treatment devices using adsorption, RO and distillation are currently available for the reduction of copper in drinking water. For a drinking water treatment device to be certified to NSF/ANSI Standard 53 (Drinking Water Treatment Units-Health Effects) for the removal of copper, the device must be capable of reducing an influent copper concentration of 3 mg/L to a maximum final (effluent) copper concentration of less than 1.3 mg/L (NSF/ANSI, 2015b). Treatment devices that are certified to remove copper under NSF/ANSI Standard 53 are generally based on activated carbon adsorption technology.
RO systems certified to NSF/ANSI Standard 58 (Reverse Osmosis Drinking Water Treatment Systems) may also be certified for the reduction of copper to achieve a final concentration below 1.3 mg/L (NSF/ANSI, 2015c). RO systems certified to this standard are intended for POU installation only. This is because water treated by an RO system may be corrosive to internal plumbing components. RO requires larger quantities of influent water to obtain the required volume of drinking water, because these systems reject part of the influent water. A consumer may need to pretreat the influent water to reduce fouling and extend the service life of the membrane.
Distillation systems certified to NSF/ANSI Standard 62 (Drinking Water Distillation Systems) can also be certified for the reduction of copper to achieve a final concentration below 1.3 mg/L (NSF/ANSI, 2015d). Distillation systems that would be certified to this standard are also intended for POU installation, for the same reasons described above. The distillation process is effective for the reduction of copper but requires an electrical energy input. Health Canada strongly recommends that any components used in these treatment systems be certified to NSF/ ANSI Standard 61- Drinking Water System Components Health Effects (NSF/ANSI, 2016a) and NSF/ANSI Standard 372 - Drinking Water Systems Components Lead Content (NSF/ANSI, 2016b).
Toxicokinetic
The toxicokinetics, storage and handling of copper are regulated by homeostatic mechanisms (Petris et al., 1996; Peña et al., 1999).
Absorption
In humans, dietary Cu(II) is mainly absorbed from the upper gastrointestinal (GI) tract after oral exposure, with maximum absorption occurring within1-3 hours of exposure (Linder, 1991; Lonnerdal, 1996; Tapiero et al., 2003; Stern et al., 2007; Shike, 2009; Corkins, 2011). Studies on copper absorption in humans are limited; mean absorption rates of 50-55% (range, 12-60%) have been reported for normal copper intakes (0.6-1.6 mg/day) from mixed diets, as was a theoretical maximum absorptive capacity of 63-75% (Turnlund et al., 1989; Wapnir, 1998; NRC, 2000; Tapiero et al., 2003; Turnlund et al., 2005; Klevay, 2010; Collins and Klevay, 2011; EFSA, 2015; Ellingsen et al., 2015). However, with typical diets in developed countries, copper absorption is considered to average 30-40% (Wapnir, 1998). Dietary Cu(II) absorption is modulated by several factors, including an individual’s copper status and intake, age, the amount of metal in the GI tract, the valence state, and the presence of other dietary factors or (essential and nonessential) competing metals (Turnlund et al., 1989; Wapnir, 1998; NRC, 2000; EFSA, 2006; Stern et al., 2007; van den Berghe and Klomp, 2009; Stern, 2010; Collins and Klevay, 2011; de Romana et al., 2011; Gaetke et al., 2014).
Although limited, human studies using different methodologies (e.g., fecal copper excretion, 65Cu retention) have demonstrated that absorption rates vary inversely with dietary copper intake. More specifically, absorption rates higher than 50% have been reported with copper intakes below 1 mg/day whereas absorption rates less than 20% have been reported with copper intakes above 5 mg/day (Turnlund et al., 1989, 1998; Stern et al., 2007). In contrast, copper retention (or balance) increases with increasing dietary intakes. ). For example, an increase in copper retention from 0.06 mg/day when dietary intake was 1.6 mg/day to 0.67 mg/day with dietary intake of 7.8 mg/day was reported. However, the amount retained decreases with time, suggesting restoration of normal balance over time (Turnlund et al., 2005).
The solubility of Cu(II) complexes influences their bioavailabilities (OEHHA, 2008; Mauceri and Mishra, 2014; EFSA, 2015). This was observed in a study by Lind and Glynn (1999) in which high levels of Cu(II) were measured in the blood of rats given copper in deionized water as compared to rats given a fulvic acid mixture in infant formula. Furthermore, developmental stage has been reported to influence Cu(II) absorption, with higher intestinal absorption anticipated in neonates (Lonnerdal, 1998; Bauerly et al., 2005; Collins and Klevay, 2011). This may be explained by immature digestive and homeostatic copper mechanisms (WHO, 2004; Araya et al., 2005a; Collins and Klevay, 2011). Finally, it has been suggested that Cu(II) absorption increases during pregnancy and cancer (Tapiero et al., 2003).
Distribution
Upon release from the GI tract, Cu(II) is immediately bound to either albumin, transcuprein, or low-molecular weight molecules (e.g., peptides and amino acids, especially histidine) before it reaches the hepatic portal circulation (Linder, 1991; IPCS, 1998; Linder, 2002; Talcott, 2013; Ellingsen et al., 2015). During this first pass, around 40% of copper is taken up by the liver (Gaetke et al., 2014; Pal, 2014; EFSA, 2015). Cu(II) then reaches the posthepatic circulation bound to ceruloplasmin (Cp) for distribution throughout the body. Hence, under human physiological conditions, plasma copper is usually viewed in two pools, one covalently bound to Cp (about 75-98% of plasma copper) and the remainder (about 7% or less), also known as ‘free copper’, which is loosely bound to albumin and low-molecular weight molecules (IPCS, 1998; Bradberry, 2007; Montes et al., 2014; Ellingsen et al., 2015). Cp is a ferroxidase synthesized and bound to copper within the liver, for the delivery of the nutrient to peripheral tissues (OEHHA, 2008; Shike, 2009; Montes et al., 2014). Consequently, increases in copper and Cp parallel one another (Turnlund et al., 2004). Moreover, Cp is an acute phase protein that levels, and activity can be increased by hormone replacement therapy (e.g., oral contraceptives, steroid hormones), response to several physiological alterations (e.g., inflammation, some nonspecific diseases, some cancers, pregnancy and other hormonal perturbations) and smoking (Kehoe et al., 2000; Turnlund et al., 2004; Shike, 2009; Collins and Klevay, 2011). There is a welldocumented sex difference in Cp levels and activity with women exhibiting higher levels (Kehoe et al., 2000).
“Free copper” mostly includes free Cu(I) and Cu(II) and, to a lesser extent, complexes of these ions with labile ligands, in body fluids (Donnelly et al., 2007). In addition, ‘free copper’ constitutes the bulk of the exchangeable copper pool in blood plasma and (presumably) interstitial fluid which levels have been estimated around 2×10-16 M or 43±30 mg (Linder, 2001; Harvey et al., 2005). Its levels are tightly controlled by homeostatic mechanisms (IOM, 2001; Bertinato and L’Abbe, 2004; Cockell et al., 2008; Collins and Klevay, 2011) and very low within cells (Rae et al., 1999; Rosenzweig, 2001; Prohaska, 2008; Boal and Rosenzweig, 2009; Kaplan and Lutsenko, 2009; Brewer, 2010). Some of these mechanisms are materialized by transporters such as ATP7A, ATP7B (Kim et al., 2008; Qian et al., 2012; Montes et al., 2014), storage proteins such as metallothionein (Tapia et al., 2004) and chaperones such as copper chaperone for superoxide dismutase (SOD), cytochrome c oxidase copper chaperone (COX17) and ATOX1 (Shike, 2009; Bertinato et al., 2010; Boveris et al., 2012; Talcott, 2013).
Thus, copper is distributed to all tissues (including the liver) as Cu(II)-Cp bound and a volume of distribution of 1.95L/kg has been estimated in healthy individuals (OEHHA, 2008; Shike, 2009; Montes et al., 2014). The average total body content of copper is about 110 mg (range, 50-150 mg) (Turnlund et al., 2005), with approximately 50% being found in muscle and bone; however, its highest concentrations are found in the liver (about 40µg/g dry weight or 8-10% of the body’s total content) and the brain (about 5 µg/g) (Prohaska, 1990; Turnlund, 1998; Gaggelli et al., 2006; Gurusamy and Davidson, 2007; Madsen and Gitlin, 2007; Collins and Klevay, 2011; Kumar et al., 2015). It is noteworthy that in humans of all ages, there are documented sex differences in plasma copper levels. Women, particularly those taking estrogens, have higher plasma levels (Ellingsen et al., 2015). In humans, fetal copper levels increase during gestation, reaching their maximum around the third trimester (13-1,218 µg/g wet weight), then decline with age (Zlotkin et al., 1995; Lonnerdal, 1996; Bauerly et al., 2005; Michaluk and Kochman, 2007). Quantitative estimates of copper in infant livers are quite variable. Hepatic levels above 90% of the total body copper (4-5 mg/kg bw) have been reported for neonates (WHO, 1996). In contrast, in one study, neonatal levels similar to adults were reported, while in another, neonatal to adult ratios as high as 16:1were suggested (EFSA, 2015 and references therein).
Metabolism
Copper metabolism involves all the processes implicated during its absorption, distribution, biotransformation and utilization by cells within the body. The intestines and liver are the key metabolic organs and steps within each of them could be considered as pre-systemic and post-systemic metabolism, respectively.
Pre-Systemic Metabolism
In mammals, ingested Cu(II)is first reduced to Cu(I) by enterocyte metalloreductases for either storage by metallothionein (MT), use by intestinal enzymes or blood absorption (by the transporter affected by Menkes disease). Upon leaving the gut, Cu(I) is reoxidized to Cu(II), bound to albumin, and then secreted into the portal circulation (Peña et al., 1999; Shike, 2009; Ellingsen et al., 2015).
Post-Systemic Metabolism
The liver plays a key role in storage, uptake regulation and biliary excretion (Talcott, 2013; Ellingsen et al., 2015). When Cu(II)-albumin enters hepatocytes, it is first reduced to Cu(I). The latter is further complexed by glutathione (GSH) before being incorporated as Cu(I)-GSH complex into either MT (for storage) or various copper chaperones. Later, these chaperones transfer copper to Cp or the numerous copper-requiring nascent apo-proteins within the hepatocytic cytoplasm and subcellular compartments. However, the most significant of these hepatic copper metabolism pathways is the transfer of copper from a chaperone to ATPase7B (the transporter affected in Wilson’s disease) with further coupling to Cp. The Cu(II)-Cp complex is then either excreted into the bile or secreted into systemic circulation (IPCS, 1998; Roelofsen et al., 2000; Linder, 2002; Bradberry, 2007; de Romana et al., 2011; Boveris et al., 2012; Gitlin, 2014).
Elimination
In humans, absorbed copper is primarily excreted from the body by feces (up to 98% or ~2,500 µg/day), and, to a lesser extent, through urine, sweat, menses, and hair (Turnlund et al., 1990; Wijmenga and Klomp, 2004; Stern et al., 2007; Mauceri and Mishra, 2014). Biliary copper excretion accounts for almost all of the fecal copper and is proportional to dietary copper to some degree (Linder, 1991; Turnlund et al., 1998; Harvey et al., 2005; Stern et al., 2007). It represents the main mechanism of regulating total body copper (Scott and Turnlund, 1994; Turnlund, 1998; Miller et al., 2006; van den Berghe and Klomp, 2009). 12-days averaged daily fecal excretion of 100% (1.6 mg/ day) and 91% (7.1 mg/day) have been reported in 9 men after 18- day exposure to low (1.6 mg/day) and high (7.8 mg/day) dietary intakes, respectively (Turnlund et al., 2005). It is noteworthy that, in contrast, intravenous infusion of 460 µg 63Cu resulted in significantly higher (p < 0.05) excretion (46%) during high intakes than during low intakes (27%; standard error = 2.3) (Turnlund et al., 2005).
Quantitative estimates of urinary excretion are scarce. However, excretion increases with increasing intakes (Turnlund et al., 2004, 2005) and average levels lower than 3% are generally reported (WHO, 1996; Turnlund et al., 2004, 2005). Turnlund et al. (2004, 2005) reported average urinary excretion for 9 men of 26 µg/day and 20 µg/day (range, 18.5-30.9; standard error on the means = 3.3) during high (7.8 mg/day) and low (1.6 mg/day) intake period, respectively. Even if the authors didn’t estimate daily losses in hair, they found significantly increased hair concentrations (mean ± standard deviation [SD]) during periods of high copper dietary intakes as compared to low intakes: from 9.2 ± 3.1 to 21.1 ± 5.9 µg/g hair (range, 3-40 µg/g hair), as measured on the 15th day of an 18-day exposure period (Turnlund et al., 2004, 2005). These levels were not significantly different between subjects (Turnlund et al., 2004). Linder (1991) has estimated a total daily excretion from hair, nails, skin, and sweat of 200 µg in adults. A half-life of 2-5 weeks was estimated for copper in healthy adults following oral administration (Ellingsen et al., 2007). Biliary excretion is limited in early life due to a poorly developed biliary transport system at the time of birth. Excretory losses of 50 µg/kg bw per day have been estimated for infants (Aggett, 1999).
Health effects Effects in humans Essentiality
Copper is an essential element. Its ability to cycle between the oxidized (Cu(II)) and reduced (Cu(I)) forms provide the catalytic properties of various metalloenzymes that are necessary for metabolism (e.g., of glucose) and cellular respiration (e.g., SOD, cytochrome c oxidase) (IOM, 2001; Bertinato and L’Abbe, 2004; Gitlin, 2014). Moreover, copper plays a role in many physiological processes including neurological, immunological, connective tissues formation, thermal regulation, and erythropoiesis (Tapiero et al., 2003; Stern et al., 2007; Gambling et al., 2011).
Copper deficiency can lead to abnormalities of these systems; cases of copper deficiency have been reported in premature infants fed unfortified milk formulas, in severely malnourished infants with chronic diarrhea, and in patients receiving chronic parenteral nutrition without copper supplementation (IOM, 2001). Copper deficiency is characterized by anemia, leukopenia, neutropenia, neurological and bone abnormalities, among other symptoms. Deficiency is not expected in the Canadian population, as food is the main source of intake, and the Canadian TDS (Health Canada, 2011) shows that the population is meeting the recommended daily allowances (RDAs) established by the Institute of Medicine (IOM, 2001) and Health Canada (2010). The RDA is “the average daily intake level sufficient to meet the nutrient requirements of 97.5% of the apparently healthy population of a given sex and age” (de Romana et al., 2011). RDAs for copper have been established at 900 µg/day for adults (19-70+ year-old) of both genders and 340-890 µg/day for children (1 to ≤ 18 years old) (IOM, 2001; Health Canada, 2010). No RDA was established for infants, but adequate intakes of 200 µg/day for 0–6-month-olds and 220 µg per/day for 7-12 month-olds were established based on copper levels in human milk (200-250 µg/L).
Due to the potential for adverse health effects from ingestion of high amounts of copper, tolerable upper intake levels (ULs) have been estimated at 1,000 µg/day for 1–3-year-olds, 3,000 µg/ day for 4–8-year-olds, 5,000 µg/day for 9–13-year-olds, 8,000 µg/ day for 14-18 year-olds, 10,000 µg/day for those ≥19 year-olds, and 8,000-10,000 µg/day for pregnant and lactating women (IOM, 2001). The ULs were based on absence of liver or GI effects in a small and poorly detailed 12-week double-blind study of backpain management in seven human adults (Pratt et al., 1985), indicating no adverse effects of diets contributing 10 mg/day of copper, as the gluconate salt.
Copper Toxicity Syndromes
Copper dyshomeostasis can cause adverse health effects including conditions resulting from inherited metabolic disorders such as Menkes’ disease (MKD) and Wilson’s disease (WD) (Cockell et al., 2008; Govindaraju et al., 2013; Gaetke et al., 2014; Jazvinscak Jembrek et al., 2014). MKD, a rare X-linked recessive disorder, results from a mutation on the gene encoding ATP7A translocase. Symptoms in patients with MKD are consistent with those of a severe copper deficiency. Thus, MKD (and its milder variant occipital horn syndrome [OHS]) is characterized by reduced systemic Cu(II) absorption from the gut and blood-brain barrier, excessive copper accumulation in gut mucosa, low serum copper and Cp levels and low copper levels in liver and brain (NRC, 2000; Shike, 2009; Montes et al., 2014; Ellingsen et al., 2015). Most MKD patients die during early childhood (within their first 10 years, usually before three years of age) while OHS patients may live longer (Kaler, 1998; Camakaris et al., 1999; Schaefer and Gitlin, 1999).
WD (an hepatolenticular degeneration) is a monogenic autosomal recessive disorder that results from one mutation, among over 500 identified, in the gene encoding ATP7B translocase (Harris and Gitlin, 1996; Schilsky, 1996; Shike, 2009; Boveris et al., 2012; Bandmann et al., 2015; Ellingsen et al., 2015; Kumar et al., 2015). WD is characterized by impaired biliary excretion of excess copper, deficient efflux of copper from cells, impaired ceruloplasmin synthesis, which results in copper accumulation in certain tissues (particularly the liver, brain and kidneys). Consequently, WD patients frequently experience liver failure (e.g., disease, cirrhosis) and a variety of neuropsychiatric/ behavioral symptoms (Das and Ray, 2006; Machado et al., 2008; Shike, 2009; Montes et al., 2014). Usually, liver dysfunction occurs in the first decade of life and neurological dysfunction in the second (Ala et al., 2007). WD patients are normally under medical supervision. Heterozygous carriers of the defective WD gene have been speculated to be sensitive populations, but evidence for this, other than increased hepatic copper levels, is lacking (IPCS, 1998; NRC, 2000; WHO, 2004).
Other extremely rare but well documented childhood copper toxicosis conditions are: Indian childhood cirrhosis (ICC, a condition primarily affecting children under 5 years old), endemic Tyrolean infantile cirrhosis (TIC), and idiopathic copper toxicosis (ICT) (Madsen and Gitlin, 2007; Pal, 2014). These diseases are characterized by extremely high copper concentrations in the liver during early childhood, leading to liver enlargement, pericellular fibrosis, necrosis, and even death (Muller et al., 1996; Tanner, 1998; Johncilla and Mitchell, 2011). However, the etiology of these syndromes seems more complex, potentially involving, in addition to genetic components, environmental factors (e.g., very high copper intakes) (Olivares and Uauy, 1996; Scheinberg and Sternlieb, 1996; ATSDR, 2004; OEHHA, 2008; Shike, 2009; Ellingsen et al., 2007, 2015). Finally, individuals with glucose6-phosphate dehydrogenase deficiencies may be sensitive to haemolytic anemia in response to copper overexposure (OEHHA, 2008).
Case Reports
There are several reports of ingested copper poisoning with the main targets including the GI tract, liver, kidneys and haematopoietic/central nervous systems (IPCS, 1998; NRC, 2000). During 1992 and 1993, the Wisconsin Division of Health investigated cases of gastrointestinal upsets thought to be related to copper in drinking water. They concluded that the symptoms occurred most frequently in infants and children and among residents of newly constructed or renovated homes (Knobeloch et al., 1994). In adults, anecdotal reports suggest that the immediate symptoms following the consumption of contaminated beverages or drinking water include a metallic taste and GI disruption (e.g., nausea and epigastric pain and vomiting combined, diarrhea), usually occurring like symptoms of food poisoning, within 15-60 minutes, and at concentrations as low as 3-4 mg Cu(II)/L drinking water. Nausea and vomiting (generally not persistent) are usually more common than diarrhea. Anorexia, headache, dizziness, hypotension and shock have also been reported. Delayed symptoms, at higher doses, include anemia, hemoglobinuria, jaundice, liver and renal failure, and even death. Histological findings upon autopsy include kidney lesions (e.g., glomeruli congestion, necrosis) and gastric mucosa erosions (Wyllie, 1957; Spitalny et al., 1984; Mehta et al., 1985; Gulliver, 1991; Araya et al., 2001). Rhabdomyolysis (muscle injury) has also been reported (Jantsch et al., 1984; Takeda et al., 2000).
In children, limited data indicates similar effects. Indeed, case reports have attributed adverse effects (e.g., diarrhea and weight loss) to levels ranging from 0.22 to 1.0 mg Cu(II)/L drinking water (Stenhammar, 1999) or 1.0 to 6.5 mg Cu(II)/L drinking water (Berg et al., 1981). Also, neonates and infants appear to be more sensitive due to their enhanced intestinal absorption and immature hepatobiliary system (Collins and Klevay, 2011). Finally, there are few case reports of chronic exposure. According to the available data, chronic ingestion of excessive copper may cause GI effects in infants, and liver and renal failure in adults (O’Donohue et al., 1993, 1999; Stenhammar, 1999; Cech et al., 2006; Stern et al., 2007).
Short-Term Exposure
Gastrointestinal symptoms, usually defined in most studies as the occurrence of one or more outcome symptoms of nausea, epigastric pain, vomiting, or diarrhea, have been identified as primary effects from short-term exposure to ingested Cu(II). Nausea is the earliest (occurring within 15 minutes to one hour) and most frequent symptom and is transient (Pizarro et al., 1999a; Pizarro et al., 1999b; Araya et al., 2001; Olivares et al., 1998; 2001).
In a double-blind randomized prospective study consisting of sixty healthy Chilean women (15/group), acute GI effects from ingested copper were identified by (Pizarro et al., 1999b) following a two-week-exposure to drinking water with at least 3 mg Cu(II)/L (as copper sulphate). In this study, GI effects (nausea, abdominal pain, or vomiting) were significantly increased (overall χ2 = 11.3, p<0.01) but not diarrhea. Although thresholds for specific symptoms could not be derived, nausea was identified as a good indicator for the effects evaluation. Also, there was a range of sensitivity among subjects and most symptoms disappeared when the exposure ceased. Therefore, a lowest observed adverse effect level (LOAEL) of 3 mg Cu(II)/L based on nausea, abdominal pain, and vomiting combined can be identified from this study.
With nausea identified as the earliest symptom of GI effects, several efforts were conducted to estimate a dose threshold. Olivares et al. (2001) used a randomized, Latin square design to administer 0, 2, 4, 6, 8, 10, or 12 mg Cu(II)/L purified water or orange-flavored drink, as copper sulphate, to apparently healthy Chilean adults. In this study, the subjects (47-61/group) received, after an overnight fast, a fixed volume of 200 mL of the solution, during a 1-hour session, once per week, for a theoretical maximum of 12 weeks. This resulted in exposure dose of 0, 0.4, 0.8, 1.2, 1.6, 2 and 2.4 mg Cu(II), respectively. There were no age- or genderrelated differences between the groups and no observed response up to 0.4 mg (= no observed adverse effect level [NOAEL] based on nausea). However, mild nausea shortly after ingestion was most frequently reported, starting at 0.8 mg Cu(II), then plateauing at 1.6-2.4 mg. The vomiting incidence, which first appeared at 1.2 mg, was low (11.5%), with a two-fold increase between 2 and 2.4 mg. The concomitant occurrence of nausea and vomiting was less frequent, with the highest incidence at 2.4 mg (12% of subjects).
Diarrhea and abdominal cramps were rare within the range of concentrations studied.
Because a range of sensitivity had been reported earlier (Pizarro et al., 1999b), the authors used a benchmark dose (BMD) approach to derive the lower bound on the 95% confidence interval (CI) on the drinking water concentration that would produce a response in 5% of the population (BMDL05). The resulting BMDL05 were 0.4 mg Cu(II) (2 mg/L) and 0.84 mg Cu(II) (4.2 mg/L) for apparent and confirmed cases of nausea, respectively. The BMDL05 for apparent cases of nausea is very close to the NOAEL of 0.4 mg Cu(II) based on nausea; the LOAEL based on nausea was 0.8 mg Cu(II), which was also the NOAEL for vomiting.
The same group of investigators reported on a prospective, double-blind controlled multi-site (Chile, Northern Ireland and United States) phase I study in apparently healthy adult volunteers, also designed to determine the nausea threshold (Araya et al., 2001). After an overnight fast, subjects (179/ group) were randomly given 0, 0.4, 0.8, 1.2, or 1.6 mg Cu(II) (as copper sulphate salt) in a bolus of 200 mL distilled-deionized water (equivalent to 0, 2, 4, 6, or 8 mg Cu(II)/L) once per week over five consecutive weeks. The results also suggested that women may be more sensitive. As copper dose increased, female subjects reported significantly more occurrences of nausea and GI symptoms than male subjects (odds relative to males, 2.66, p < 0.004; and 1.68, p< 0.05, respectively). Nausea, which was transient, was the most prevalent symptom (average prevalence = 27.3% among all subjects) and was frequently reported within the first fifteen minutes. Also, there was a clear dose-response to (1) combined positive GI symptoms (nausea, vomiting, diarrhea or abdominal pain) and (2) nausea alone. Statistically significant greater reporting of effects occurred at 1.2 and 1.6 mg Cu(II).
Unlike abdominal pain and diarrhea, nausea and GI symptoms as a whole were strongly predicted by Cu(II) concentrations, with incidences significantly increasing (P < 0.0001) with increasing concentrations (odds of 0.66; 3.53, 7.67, and 17.15 for nausea and 0.87, 1.83, 3.54, and 7.29 for GI symptoms, for 2, 4, 6, and 8 mg/L, respectively; p < 0.0001). Both nausea and other GI symptoms were significantly increased (p < 0.002) at 1.2 mg Cu(II) but not at 0.8 mg Cu(II) (LOAEL and NOAEL, respectively). The performed dose-response modeling determined that the BMDL05 for nausea alone was 0.7-0.8 mg Cu(II) (3.5-4 mg/L). The upper bound of this latter range is similar to the NOAEL of 0.8 mg based on nausea and other GI symptoms. Overall, the findings (nausea occurrence, no correlation with diarrhea and a LOAEL ≥ 3 mg Cu(II)/L) were consistent with that from Olivares et al. (2001) and Pizarro et al. (1999b).
The above phase I results from Araya et al. (2001), in which copper was delivered in distilled-deionized water, were confirmed in the phase II of this study (Araya et al., 2003a) in another multisites double-blind study in 269 adult women receiving Cu(II) sulphate in 200 mL bottled natural spring water, once per week, for 11consecutive weeks. In this study, a 3 × 3 two-way factorial design was used to administered to the subjects (58 to 73/site) 0.4, 0.8, 1.2 mg Cu(II) as the sulphate salt in 100, 150 and 200 mL of water. The 0 and 1.6 mg Cu(II) doses were added at the 200 mL level. It is worth noting here that, all the other short-term studies used a constant dosing volume of 200 mL The results from (Araya et al., 2003a) showed that both the copper dose (p = 0.0001) and the volume of water (p = 0.032) significantly contribute to the nausea incidence. The latter increased with an increase in copper dose and decrease in water volume. In addition, a benchmark dose (BMD) analysis was performed for nausea incidence for the 200 mL bolus dose. The calculated BMD for a 10% response was 0.94 mg Cu(II) (4.8 mg/L; p = 0.94) and the corresponding BMDL10 was 0.84 mg Cu(II) (4.2 mg/L). The BMDL10 is very close to the NOAEL of 0.8 mg Cu (II) (4 mg/L); this NOAEL identified for women is also the same as the NOAEL based on nausea and other GI symptoms identified for both men and women in the phase I of the study by Araya et al. (2001). Overall, from the above shortterm studies, the lowest effect level identified is a BMDL05 of 0.7mg (or 3.5 mg/L) based on nausea alone and non-continuous exposure (Araya et al., 2001).
Long-term exposure
Chronic copper toxicity is usually observed in patients with Wilson’s disease, hemochromatosis and infantile cirrhosis (Olivares et al., 1998; Mauceri and Mishra, 2014). While WD involves more than simple accumulation of copper, it is likely that some of the typical WD symptoms are related to long-term exposure to excessive copper (see section 9.1.2). However, the current evidence, mostly from mechanistic data, as well as limited case and clinical studies, indicates that in the general population, chronic exposure to very high levels, above those associated with GI illnesses, may lead to liver and renal damage (O’Donohue et al., 1993; IPCS, 1998; O’Donohue et al., 1999; Pizarro et al., 1999b; Stenhammar, 1999; Stern et al., 2007).
Gastrointestinal Effects
Araya et al. (2004) conducted a randomized, doubleblind, two-month-community-based study on 1365 apparently healthy adults (327 to 355/test group) receiving up to 6 mg Cu(II)/L. Copper-containing water was used for drinking and the preparation of other beverages (e.g., tea, coffee, infusions, and soups) and mean fluid consumption was 1.5 L/day. Unlike the previous findings from short-term exposure (Pizarro et al., 1999b; Araya et al., 2001, 2003a; Olivares et al., 2001), abdominal pain and nausea were most prevalent and symptoms reporting significantly decreased over time for both sexes, suggesting an adaptive response. However, more importantly, a LOAEL of 6 mg Cu(II) (4 mg/L) was identified in women, the most sensitive population. This LOAEL which was no longer valid by week 4, is similar to the BMDL05 of 4 mg/L, based on nausea, identified previously for short-term exposure (Araya et al., 2001, 2003a; Olivares et al., 2001). Also, as with the short-term studies, the symptom risks were positively associated with copper concentration and the female sex, and negatively related to time. In fact, the risk of GI effects was significantly higher in female during the first week, but this was no longer the case by week 4. Overall, the authors found that Cu(II) concentration and dosing volume were the main determinants of the response, the applied dose and vehicle being less important.
A prospective study by Olivares et al. (1998) showed that infants who continuously received up to 31.48 μmol Cu(II)/L drinking water as copper sulphate (i.e., 2 mg/L) during the last nine months of their first year of life remained asymptomatic, grew normally, and did not present with significant increased risk of diarrhea when compared to those receiving less than 1.57 μmol Cu(II)/L (i.e., less than 0.1 mg/L). The main goal of this study was to verify the tolerance and safety of chronic exposure of infants to the previous provisional guideline value (WHO, 1993) for the general population. This placebo-controlled study randomly assigned 128 apparently healthy Chilean infants (3 months of age), from the same socioeconomic level and weighing more than 2 kg at birth, to receive, drinking water with either 1.57 μmol Cu(II)/L or 31.48 μmol Cu(II)/L, daily for 9 months. Breast-fed infants were exclusively breast-fed until at least 6 months of age and those given formula were partially or totally weaned from the breast by 3 months of age. More specifically, the 3-month olds infants were divided into four groups at the beginning of the study as follows: 1) group 1: formula-fed infants receiving 31.48 μmol Cu(II)/L (n = 56); 2) group 2: formula-fed infants receiving 1.57 μmol Cu(II)/L (n = 27); 3) group 3: breast-fed infants receiving 31.48 μmol Cu(II)/L (n = 24); and 4) group 4: breastfed infants receiving 1.57 μmol Cu(II)/L water (n = 21). Mothers of breast-fed infants (groups 3 and 4) also received water with the corresponding Cu(II) levels. Thus, two litres of drinking water were prepared for consumption daily, by the mothers, either from a fresh copper sulphate-containing standard solution (0.8 g Cu (II)/L) for the exposed groups (1 and 3), or from an equivalent volume of placebo for the non-exposed groups (2 and 4).
This placebo was not clearly defined except that it was organoleptically identical to the copper-based solution. The water for daily consumption was used for drinking, in preparation of meals, and in preparations (by dilution) of the powdered cow’s milk and the powdered cow’s milk-based formula. The latter was fortified with 7.87 µmol copper/L. Any additional water needed was prepared as per the same approach. Formula-fed infants started receiving solid food at 3 months of age whilst breastfed infants began weaning and receiving unfortified cow’s milk formula and solid food at 6 months of age. Although breast milk Cu(II) was not considered when calculating total intake, dietary copper from foods, milk formula and water used for meals and formula preparation was. During the study, a field worker visited each home weekly, recording water intake, GI, respiratory, and other illnesses, as recalled by mothers. Each infant also underwent monthly clinical and anthropometric evaluations.
Overall, chronic ingestion of up to 31.48 µmol Cu/L by infants (in their formula [F], foods and drinking water), from three months to one year old, did not result in any acute or chronic effects. Specifically, there were no differences in morbidity episodes (i.e., diarrhea and respiratory infections) among the four studied groups, as indicated by the performed analyses. At 9 months, there was a significant difference in Cp activity between the Cu(II) exposed subjects (groups 1 and 3) as compared to the unexposed (groups 2 and 4) (350 ± 85 mg/L vs. 322 ± 75 mg/L; F = 5.42; p = 0.02), which was more pronounced in the breast-fed groups (groups 3 and 4).
However, the overall copper status (based on serum copper levels, Cp protein levels or activity, erythrocyte Cu/Zn-SOD, erythrocyte metallothionein) of the infants did not significantly change at 6, 9, and 12 months of age. Also, no significant differences were found in the liver function (based on bilirubin, SGOT, serum glutamic pyruvic transaminase, and gamma glutamyl transferase [GGT]) among the infants at 6, 9, and 12 months of age. The authors reported the results for 3 time points (4 to 6 months, 6 to 9 months and 9 to 12 months). But as discussed earlier (section 9.1.3), infants are at increased risk of copper exposure during their first 6 months of life, when their hepatobiliary system is still immature. Since no toxic effect was reported for the entire studied period, results from the highest exposure window, months 4 to 6 (equivalent to a 4-month exposure), will be considered. According to the authors, for this exposure window, the corresponding figures for total daily intakes (mean ± SD) were: 318.7 ± 107.3 µg/ kg bw per day (group 1); 122.7 ± 107.4 µg/kg bw per day (group 2); 52.2 ± 48.5 µg/kg bw per day (group 3); 37.8 ± 36.7 µg/kg bw per day (group 4).
Liver and Renal Effects
Liver toxicity from copper overexposure has mostly been studied and reported in sensitive populations (e.g., WD, childhood cirrhosis syndromes); however, there have been a few reports studying individuals from the general population. Indeed, a significant transient increase in liver enzyme activities (glutamic-oxaloacetic aminotransferase (GOT), glutamic-pyruvic transaminase (GPT) and gamma glutamyl transferase (GGT); ANOVA p-value < 0.01) was observed in a prospective controlled trial in apparently healthy Chilean adults (n = 87, with serum Cp levels below the 0.05 and above the 0.95 quantiles of the distribution curve) receiving 10 mg Cu(II)/day(0.17 mg/kg bw per day assuming an average 60-kg adult) (as sulphate salt) in capsules for 60 days (Araya et al., 2005b). However, not only were the results within normal clinical ranges, but the participants remained asymptomatic and the increased liver enzyme activities ceased one year after the termination of the study.
In a double-blind study by Pratt et al. (1985), there was no evidence of liver or GI effects in seven adult humans supplemented with 10 mg Cu/day (0.08 mg/kg bw twice a day) as gluconate salt in capsules for 12 weeks. Furthermore, no significant changes were reported in:(1) hair, serum and urine copper levels; (2) hematocrit, serum triglycerides and cholesterol; and (3) serum enzymes such as alkaline phosphatase (AP), GGT, GOT and lactate dehydrogenase (LDH). A European multi-site, double-blind, placebo-controlled, food-trial also found no association between liver function and copper supplementation (in sequence: 0.05 mg/kg bw per day as copper sulfate salt, and 0.12 mg/kg bw per day as copper amino acid chelates) for 6 weeks (O’Connor et al., 2003).
No significant changes in indicators of liver function (aspartate aminotransferase (AST), alanine aminotransferase (ALT), GGT) were observed in adults who ingested up to 6 mg Cu(II)/L (0.10 mg/kg bw per day for an average 60-kg adult) as the sulphate salt for two months (Araya et al., 2003b). Long-term exposure of infants and young children to drinking water containing copper was not associated with a significant increase in the prevalence of liver disease (Fewtrell et al., 1996; Olivares et al., 1998; Zietz et al., 2003a).
Biochemical Effects
In general, the results of studies that lasted less than 3 months showed that copper supplementation did not alter the indexes of its status. Thus, no significant changes in markers of copper status (serum, erythrocyte, and mononuclear cell copper concentrations; serum Cp; non-Cp-bound copper pool; SOD activity) were detected in healthy adults given either up to 6 mg Cu(II)/L as the sulphate (~ 0.20 mg/kg bw per day for 2 L water/day) (Araya et al., 2003b, 2004; Mendez et al., 2004), tablets of 10 mg/day (0.17 mg/kg bw per day) copper sulphate for two months (Mendez et al., 2004), or diet supplemented with up to 6 mg Cu(II)/day (0.09 mg/kg bw per day) as the sulphate salt or amino acid chelates (Kehoe et al., 2000; Turnlund et al., 2004). However, some changes observed after supplementation for a longer period of time may suggest an adverse effect of high copper intake. Thus, a study was conducted in 9 men to determine the long-term effects of high copper intake (Turnlund et al., 2004). After a 5-month supplementation with up to 7.8 mg Cu(II)/day (0.11 mg/kg bw per day for an average 70-kg adult), changes were observed in several function markers. Indeed, for the immune function, there were significant decreases (p < 0.05) in the percentage of circulating neutrophils, serum interleukin 2R, and the antibody titer against the Beijing strain of influenza. For the antioxidant defense, the urinary thiobarbituric acid reactive substances (TBARS), an indirect measure of oxidative damage, was significantly higher (p < 0.05). Additionally, small but significant changes were observed in some indexes of copper status (e.g., Cp activity and SOD activity), although concentrations of plasma copper and Cp did not change (Turnlund et al., 2004).
In conclusion, for exposure less than 3 months, the results were negative. Indeed, adults supplementation with up to 0.17 mg/kg bw per day did not significantly alter the indices of liver damage (O’Connor et al., 2003; Araya et al., 2005b) or copper status (Kehoe et al., 2000; Mendez et al., 2004; Turnlund et al., 2004); the same picture was observed after long-term exposure to drinking water with up to 0.2 mg/kg bw per day (Araya et al., 2003b; Araya et al., 2004; Mendez et al., 2004). However, for exposure above 5 months, the results suggest adverse effects of high copper intake over longer period of time (Turnlund et al., 2004). Therefore, given the paucity of chronic exposure studies, perhaps such data (Turnlund et al., 2004) and the symptoms experienced by WD patients could shed light on the types of adverse effects that might be expected from chronic copper overexposure [61-70].
Neurotoxicity
Copper-enzymes play an essential role in neurological functions. Multiple reviews have reported conflicting results on the association between copper exposure and neurodegenerative conditions such as Alzheimer’s disease (AD) and Parkinson’s disease (Bush et al., 2003; Squitti et al., 2006; Rivera-Mancia et al., 2010; Tiffany-Castiglioni et al., 2011; Montes et al., 2014; Pal et al., 2014). For example, two case-control studies in Italy have found that serum copper levels were significantly higher in AD patients compared to age or sex-matched controls (P-value < 0.001) and correlated with lower neuropsychological scores; however, the copper serum levels reported were within normal range (Squitti et al., 2002; Brenner, 2003). Also, senile plaques and neuropils of the amygdala of 9 AD patients were found to contain significantly higher levels of copper compared with neuropils of the amygdala of 5 control subjects (P-value < 0.05) (Lovell et al., 1998) and compared with non-plaque regions of 2 AD patients, However, copper levels also correlated with those of zinc (Miller et al., 2006).
Other studies have not supported the above results. For example, copper levels in cerebrospinal fluid and serum were not significantly different between Alzheimer’s disease patients (n = 26) and matched controls (n = 28) (Molina et al., 1998). In another study, patients with lower plasma copper had a significantly poorer performance on the AD’s Assessment Scalecognitive subscale compared with patients with higher plasma copper (ANOVA p-value < 0.001; all patients (n = 32) had normal plasma copper levels) (Pajonk et al., 2005). Finally, higher levels of AD biomarkers were correlated with lower plasma copper levels among AD patients (Mann-Whitney p-value = 0.03, total n = 29) (Kessler et al., 2006). In conclusion, from the above results, it appears that the specific role of copper in neurodegenerative diseases, which are multifactorial, remains unclear.
Carcinogenicity
Numerous epidemiological studies have investigated the association between copper status or intake, serum copper and various types of cancers. In general, the evidence of the carcinogenicity of copper is inconclusive and there is no clear or substantiated evidence of an aetiological role of the nutrient in the cancer development in humans. A case-control study in France (Senesse et al., 2004) found an increase in the odds of colorectal cancer in the highest quartile of copper intake compared to the lowest quartile (odds ration [OR] = 2.4, 95% CI 1.3-4.6, p-value <0.01). Another case-control study nested within a large Dutch chronic diseases cohort (n > 10,000) found an increased risk of death from cancer (OR = 3.7, 95% CI 1.5-9.1) in participants with serum copper > 1.4 mg/L compared to 1.1-1.4 mg/L after adjusting for BMI, cholesterol, education and blood pressure; however, important cofactors were not adjusted for (Kok et al., 1988).
Furthermore, a nested case-control study (n=133 cases, 241 controls) of a worker cohort (n=5,000) in Washington also found an increased risk of cancer in subjects with serum copper at 126- 276 µg/dL compared with those having 43-92 µg/dL (OR=2.4, 95% CI 1.1-5.1). However, according to the authors, the findings rather suggested that increased serum copper was a consequence of the disease state, i.e., cancer (Coates et al., 1989). Finally, a nested case-cohort study from a random sample of cases (n=46) from a cohort of English women found an increased odds of breast cancer (OR 3.2, 95% CI 1.1-9.4) for women with plasma copper above 1.3 mg/L compared to 1.0-1.2 mg/L after adjusting for age, weight, and family history; however, there was no dose-response and other factors could explain the association (Overvad et al., 1993). Moreover, many case-control studies have not supported associations between copper intake or serum levels and the risk of breast or oesophageal cancers (Cavallo et al., 1991; Dabek et al., 1992; Prasad et al., 1992).
Furthermore, no association between copper intake and lung cancer was observed in a retrospective cohort study (Mahabir et al., 2010). In a cohort study of associations between multiple antioxidants and cancers in women, the risks of non-Hodgkin’s lymphoma, diffuse large B-cell lymphoma or follicular lymphoma were not increased in participants with higher copper in take (Thompson et al., 2010). In conclusion, the results from the above studies are equivocal since (1) individual exposure was poorly characterized and (2) increased serum copper concentrations might be related to alterations in copper handling as a result of disease state (cancer in this case). In addition, as has been mentioned earlier, more than 90% of serum copper is normally bound to Cp. The latter is an acute phase protein whose levels increase during stress (see section 8.2).
Genotoxicity
In general, copper is not expected to be genotoxic with normal intakes. However, increases in genotoxicity and oxidative damage have been shown in patients suffering from copper dyshomeostasis conditions (NRC, 2000). For example, DNA damage was observed in hepatocytes from patients with Indian Childhood Cirrhosis, and this was postulated to result from excessive accumulation of copper in the nucleus, leading to the production of free radicals (Prasad et al., 1996). Also, bulky DNA adducts (vs. 8-hydroxydeoxyguanosine adducts) were observed in the livers of Wilson’s disease patients (Carmichael et al., 1995). Moreover, a 6-week-double-blind crossover trial (n = 15-17) found no significant differences in leucocyte DNA damage (Comet assay) in participants supplemented with 3 mg/day copper sulphate or 3 or 6 mg/day copper glycine chelates compared with the placebos (t-test p-value > 0.05) (O’Connor et al., 2003).
Developmental and Reproductive Toxicity
There is no convincing evidence of reproductive or developmental toxicity in humans related to oral copper intake (IPCS, 1998; NRC, 2000; EFSA, 2006; Heinemann et al., 2015). However, it should be noted that women with untreated WD frequently experience spontaneous abortions (NRC, 2000).
Effects on Experimental Animals
Copper toxicity varies between animal and chemical species, and target organs are primarily those involved in the absorption and excretion of the metal (i.e., liver, kidneys, forestomach). Liver and kidney toxicities were the main endpoints reported in young rodents exposed to high copper doses for short/sub-chronic durations, and recoveries suggestive of physiological adaptations to high copper intake have been observed in rats (Haywood, 1980, 1985; Haywood and Loughran, 1985; Haywood et al., 1985a, 1985b; Fuentealba and Haywood, 1988; Fuentealba et al., 1989a, 1989b). Effects on the central nervous system have also been observed; however, the body of evidence for these effects is weak and inconclusive. Exposure to high levels of copper via drinking water appears to be more toxic than via the diet, and rats were generally more sensitive than mice. Copper salts, and particularly Cu(II) sulphate, were commonly used and the toxicity is attributed to the ionic form. No quality study of chronic duration was found.
Acute Toxicity
Oral lethal doses (LD50) of 15-1600 mg Cu/kg bw have been reported for different forms of copper across different species; for example, in rats, the LD50 is 120-300 mg/kg bw for Cu(II) sulphate and 15-90 mg/kg bw for Cu(II) chloride (Stern et al., 2007; OEHHA, 2008; Ellingsen et al., 2015). Death is usually preceded by bleeding of the stomach, cardiovascular dysfunctions, and paralysis (IPCS, 1998). The stomach being the primary site for the emetic response, vomiting is induced within a few minutes in Beagle and Mongrel dogs and ferrets exposed to aqueous solution of Cu(II) sulphate (WHO, 2004). Most studies using vomiting as an indicator may not have observed a response in mouse and rat at levels that might be comparable to those eliciting a response in other species; it is important to note, however, that rats and mice do not vomit (Horn et al., 2013). Soluble salts (e.g., Cu(II) sulphate) generally elicit greater responses than insoluble forms, and rodents were reported to be more sensitive than ferrets, sheep, dogs and cats (IPCS, 1998; WHO, 2004).
Short-Term and Sub-Chronic Toxicity
Liver Effects
Liver inflammation (i.e., multifocal hepatitis, accumulation of macrophages, increased liver enzyme activity in the serum) and necrosis of hepatocytes have been observed in young (~6- 10 weeks of age) and adults F344 rats of both sexes ingesting 1,500-16,000 ppm Cu(II) sulphate in feed for 2-18 weeks (Hebert et al., 1993; NTP, 1993; Fuentealba et al., 2000; Aburto et al., 2001). Effects were more pronounced in both male and young rats. Moreover, alterations in the integrity and size of lysosomes, endoplasmic reticulum, mitochondria, and nucleus have been observed in rats fed 1,500 mg Cu/kg feed for 16 weeks (Fuentealba and Haywood, 1988).
No evidence of copper toxicity or liver damage was observed in infant rhesus monkeys administered 6.6 mg Cu(II) sulphate/L (900 µg/kg bw per day, n=5) until 5 months of age; however, increased number of Küpffer cells were observed in the liver, suggestive of inflammation (Araya et al., 2005a). Moreover, no clinical, biochemical, or histological evidence of liver damage were observed in newborn or adults capuchin monkeys (2 females, 2 males each) exposed for 36 months to, 5.5 mg and 7.5 mg Cu(II) gluconate/kg per day, respectively; however, an increase in inflammatory genes (e.g., HGF, TGFβ and NFκB) was found (Araya et al., 2012). Finally, no adverse effects were noted in Beagle dogs fed with Cu(II) gluconate at levels up to 8.4 mg/kg bw per day for 6 to 12 months, and the slight liver function changes observed at the highest dose were reversed after stopping the exposure (NRC, 2000).
Kidney Effects
Increases in cytoplasmic protein droplets were observed in the renal convoluted tubules of male rats given water containing 300 ppm Cu(II) sulphate for 2 weeks (Hebert et al., 1993; NTP, 1993). Similar effects were observed in rats receiving feed with 2,000 ppm Cu(II) sulphate and more for 2-15 weeks (Haywood, 1980; Hebert et al., 1993; NTP, 1993). The effects were more pronounced in males.
Other Effects
Hyperplasia and hyperkeratosis of the forestomach’s limiting ridge mucosa were observed in both sexes of rats and mice at doses of 2,000 ppm Cu(II) sulphate and more in food for 2-13 weeks, with rats being more sensitive (Hebert et al., 1993; NTP, 1993). Symptoms varied from oedema and inflammation in the stroma to papilliferous ridge with expansions of the stromal core and the squamous mucosa in severely affected rats. Bone marrow cell depletion decreased spleen and bone marrow hematopoiesis, transient changes in haematological parameters, organ damages, and death have been observed in rats consuming high doses of copper (over 2,000 ppm) (NTP, 1993; Bertinato et al., 2010).
Carcinogenicity
Copper has not been shown to be carcinogenic in animals. Indeed, the limited available data does not suggest that copper or its salts are carcinogenic in animals with normal copper homeostasis. Specifically, the available data have several weaknesses that hinder any adequate assessment such as: ‘dated studies, small group sizes, single oral dose tested; limited extent of histopathological examination, inadequate reports’ (NRC, 2000; EFSA, 2006; Abe et al., 2008; OEHHA, 2008). No chronic/ cancer inhalation studies in animals were identified. However, the data for the Long-Evans cinnamon (LEC) rat, a model for Wilson disease, supports the hypothesis that copper-induced cirrhosis can be an aetiological factor in hepatic cancer (NRC, 2000).
Genotoxicity
Although some positive responses have been observed in the Mutatox and SOS Chromotest assays (Codina et al., 1995), copper is generally not genotoxic. Moreover, microbial mutation assays with different copper salts were negative. Indeed, no mutations were induced by Cu(II) sulphate in the TA98, TA 100, TA102, TA1535 or TA1537 strains of S. Typhimurium (Moriya et al., 1983). Also, no mutations were induced by Cu(II) gluconate in the Ames test or by Cu(II) chloride in E. coli WP2 with or without microsomal activation (Wong, 1988; Hujita et al., 1998).
Neurotoxicity
There is some evidence for a relationship between copper exposure and neurotoxicity. For example, lower anti-oxidative activity and alterations in dopamine kinetics were observed in the brains of rats exposed to high levels of copper (1 g Cu/L, or 46 mg Cu/kg bw) via drinking water for 1-11 months (De Vries et al., 1986; Ozcelik and Uzun, 2009) and both alterations in amyloidbeta protein processing and learning impairment were observed in mouse and rabbit models of AD chronically exposed to copper (Sparks and Schreurs, 2003; Kitazawa et al., 2009). However, no increased neurological sensitivities were identified in LEC rats and in Bedlington terriers, genetic animal models for copper toxicosis (NRC, 2000; Pal et al., 2013), and no neurobehavioral manifestations were observed in rats exposed to 1 ppm Cu(II) sulphate in their drinking water for 6 weeks (Fujiwara et al., 2006)(Fujiwara et al., 2006). Hence, the results for neurotoxicity are equivocal and adverse effects were only observed following exposure to very high doses and/or with the use of animals with genetic anomalies.
Reproductive and Developmental Toxicity
Some studies have reported changes in testes, seminal vesicles, uteri and ovaries in rats, mice, and bank voles after exposure to high levels of copper (27-200 mg/kg bw per day) (EFSA, 2006; Roychoudhury et al., 2015). Despite that there were some reports of teratogenicity and feto-lethality following ingestion of copper at high doses (above 80-390 mg/kg bw), these outcomes were attributed to dam toxicity (IPCS, 1998; NRC, 2000; EFSA, 2006). Moreover, no adverse effects were reported for any of the reproductive parameters studied in rats and mice of either sex exposed to Cu(II) sulphate (up to 8,000 mg/kg of feed or 140 mg/kg bw) for 13 weeks (NTP, 1993).
Mode of Action
Copper exerts its toxicity via two main pathways: The neuronal pathway in the case of GI effects, and the cellular toxicity pathway (especially oxidative stress) for the other adverse effects. In order to manifest toxic effects, copper must be in its free ionic form or free copper. The following sections briefly describe the proposed mode of action for copper-induced toxicity.
Gastrointestinal Effects
Copper exerts its GI tract toxicity by direct irritation of the stomach, activating the release of serotonin by enterochromaffin cells (NRC, 2000; Bradberry, 2007; OEHHA, 2008). The mode of action of this neuromuscular response is a reflex mechanism mediated by the central nervous system. As part of a study involving several emetic agents, Bhandari and Andrews (1991) concluded that copper sulfate causes emesis through GI chemoreceptors in the vagal nerve afferents and that this process most likely involves activation of 5-hydroxytryptamine-4 receptors in some manner. These receptors are receptor subtypes that respond to serotonin. Serotonin can sensitize gastric mucosa parasympathetic nerve endings, activating the vomit centre in the brain. The latter mediates efferent signaling messages to the GI tract, initiating a response (OEHHA, 2008). Stomach infusion with copper sulphate solution resulted in vomiting in 7/9 ferrets as compared to 1/9 animals with duodenum infusion with similar solution (Makale and King, 1992). Dogs with severed vagus and sympathetic gastric nerves showed impaired emetic response (Fukui et al., 1994). The latter is modulated by many factors such as: individual sensitivity, copper form, and the nature of gastric content. Adaptation in the response has also been demonstrated (NRC, 2000). Since the GI responses of copper are related to portal of entry effects, the appropriate metric for risk assessment is the concentration.
Other Adverse Effects
Cellular toxicity is the main and most studied mode of action by which copper exerts its other adverse effects (Gaetke and Chow, 2003; Gaetke et al., 2014). Several mechanisms have been proposed to explain copper-induced cellular toxicity. However, oxidative damage to membranes and biological macromolecules is the most studied and consistent of them (Bremner, 1998; Gaetke and Chow, 2003; Jomova and Valko, 2011; Mauceri and Mishra, 2014). Free copper has been identified as the toxic moiety leading to oxidative stress (EFSA, 2006; Bertinato et al., 2010; Stern, 2010; Montes et al., 2014). Both Cu(I) and Cu(II) ions possess redox potential and can catalyze the production of reactive oxygen species (ROS), especially the hydroxyl radical (OH•). However, in order to exert its cytotoxicity, the Cu(II) ion must be reduced (e.g., by GSH or ascorbate) to the Cu(I) ion (EFSA, 2006; Bertinato et al., 2010; Stern, 2010; Formigari et al., 2013; Montes et al., 2014) which can catalyze the formation of OH• from hydrogen peroxide (H2O2) via a Haber-Weiss driven Fenton reaction (Bremner, 1998; Bradberry, 2007; Bertinato et al., 2010; Stern, 2010; Formigari et al., 2013; Gaetke et al., 2014; Jazvinscak Jembrek et al., 2014; Montes et al., 2014). Indeed, copper-induced oxidative stress involves the formation of the extremely reactive hydroxyl radical (OH•) which is capable of initiating one of the three main cellular events most frequently reported (Gaetke et al., 2014; Jazvinscak Jembrek et al., 2014). The first event, the peroxidation of membrane lipids, is the consequence of a hydrogen abstraction from an unsaturated fatty acid by the hydroxyl radical, leading to a lipid radical. One consequence of this process is the breakdown of hepatic lysosome membranes (Burkitt, 2001; Letelier et al., 2010; Gaetke et al., 2014).
The second key event, the oxidation of proteins, results from a hydrogen abstraction from an amino-bearing carbon by OH•, to form a carbon-centered protein radical. This leads to, for example, the deactivation of enzymes. The third key event, DNA damage, is the consequence of the DNA strands breaks and oxidation of bases by OH•, leading for example to mutations (Gaetke and Chow, 2003; Jomova and Valko, 2011; Gaetke et al., 2014). These key oxidation events can lead to inflammation and cell death (Yoshida et al., 1993; NRC, 2000; Jomova and Valko, 2011; Formigari et al., 2013; Gaetke et al., 2014) which potentially explain copper’s toxic effects as well as the molecular and morphological alterations observed in the liver, kidney, brain, and red blood cells at high doses in animal studies (Eskici and Axelsen, 2012; Shaligram and Campbell, 2013; Jazvinscak Jembrek et al., 2014). Also, since copper-induced ROS is time and dose-dependent, this mode of action has long been proposed to explain the liver toxicity induced by the metal (Formigari et al., 2013). In addition, the current evidence suggests that the hepatic mitochondrion is an important target in copper-induced liver oxidative damage (Jiménez Del Rı́o and Vélez-Pardo, 2004; EFSA, 2006; Kawakami et al., 2008; Paris et al., 2009; Tardito et al., 2011; Formigari et al., 2013; Gaetke et al., 2014).
Thus, data from experimental studies support the ROSinduced oxidative damage as the mode of action of copper toxicity. For example, lipid peroxidation, protein oxidation, decline in the mitochondrial membrane potential, and mitochondrial expulsion of cytochrome c, all preceding cell death, were reported following exposure to excess copper (Kumar et al., 2015). Also, rats fed high levels of dietary copper exhibited increased peroxidation of the mitochondrial membrane lipids as measured by increased levels of conjugated dienes and TBARS (Sokol et al., 1990). In addition, the formation of hydroxyl radical has been measured in bile of rats dosed with copper and ascorbic acid (Kadiiska et al., 1992).
Furthermore, neurotoxic endpoints observed at high doses could be explained by the production of ROS, such as release of a hydroxyl radical, a by-product of the monoamine oxidase metabolism (Eskici and Axelsen, 2012; Formigari et al., 2013; Govindaraju et al., 2013; Shaligram and Campbell, 2013; Zhao et al., 2013; Gaetke et al., 2014; Jazvinscak Jembrek et al., 2014; Montes et al., 2014). Also, the depletion of GSH reductase (which also depends on NADPH) and generation of hydroxyl radicals could also lead to haemolytic effects (IPCS, 1998; Grass et al., 2011). Furthermore, the methaemoglobinemia often observed in cases of acute copper poisoning may be a consequence of the direct oxidization of the haem iron from the ferrous (Fe2+) to the ferric (Fe3+) form by copper (IPCS, 1998; Bradberry, 2007).
Other modes of action such as the direct interaction of copper with essential macromolecules and the displacement of cofactors from metalloenzymes have also been proposed. Thus, being a component of many metalloenzymes, it is also likely that high concentrations of copper can affect proteins containing sulfhydryl groups by directly interacting with macromolecules (e.g., DNA, biological membranes, proteins, etc.). The irreversible and nonspecific binding of copper to sulfhydryl groups can alter the structure of macromolecules (e.g., GSH, monooxygenases, etc.) and inactivate them. This mode of action has been proposed to explain the haemolytic anemia and methaemoglobinemia observed following acute copper sulphate poisoning. Copper can enter red blood cells, where it can bind to sulfhydryl groups and inactivate glucose-6-phosphate dehydrogenase, which is essential for generating NADPH, an intermediate in the electron transfer chain (Pamp et al., 2005).
Classification and Assessment
The current data are insufficient to classify copper with respect to carcinogenicity. The U.S. EPA (1988) has classified copper in Group D, not classifiable with respect to its carcinogenic risk to humans. The International Agency for Research on Cancer has not classified copper and its soluble salt (e.g., sulphate, chloride) with respect to carcinogenicity.
Non-Cancer Risk Assessment
Copper is an essential element involved in many biological processes (e.g., as a component of metalloenzymes), however, both copper deficiency and overexposure can result in adverse effects. As discussed in section 9.1.1, copper deficiency and overexposure are rare in healthy Canadians consuming a balanced diet since homeostatic mechanisms regulate nutrient levels (Health Canada, 2011). The health effects from copper exposure may be influenced by the medium of exposure. Copper in food is subject to liver’s first-pass effect which can potentially reduce the systemic dose; free copper, the toxic moiety of concern in drinking water, may not be subject to such an effect which may significantly increase the internal dose (Brewer, 2009, 2014).
The database of human studies on health effects associated with ingested copper is large and contains adequate data for determining an acceptable concentration in drinking water. In general, short-term exposures results in GI tract effects, usually observed at copper levels greater than 3 mg/L water (see sections 9.1.3 and 9.1.4). Also, when considering the current evidence (mainly from animal and mechanistic studies) it appears that the primary effect from chronic overexposure is metal accumulation in the liver and kidneys, potentially leading to structural and biochemical damage (Linder and Hazegh-Azam, 1996; IPCS, 1998; Tapiero et al., 2003; Cockell et al., 2008).
Several randomized controlled studies have investigated the adverse health effects of copper exposure, although mostly in adults; the results from these studies indicate that shortterm oral exposure to copper in drinking water while in a fasted state leads to GI effects (usually nausea, epigastric pain and vomiting combined, and diarrhea), which although transient, are the most sensitive clinical endpoints (Pizarro et al., 1999b; Araya et al., 2001, 2003a; Olivares et al., 2001). Nausea was the most frequently reported symptom generally occurring within 15 minutes following exposure; diarrhea was less frequently reported within the range of studied doses and women appeared to be more sensitive. In addition, the observed GI effects were found to disappear when exposure ceased suggesting an adaptive response with time (Araya et al., 2004). Furthermore, the results indicate that the concentration of copper (i.e., the bolus dose in water on an empty stomach divided by the dosing volume) is the primary determinant of the incidence of nausea (Araya et al., 2001, 2003a) and thus, with support from the data on the mode of action, is considered as the most appropriate dose metric for risk assessment. It is noteworthy, however, that although the above studies (except Araya et al., 2004) were conducted for up to 12 weeks, the exposure was not continuous and copper was consistently administered via single bolus dosing (using tap or distilled-deionized water) for short time periods (i.e., once/week) which may not accurately reflect the typical chronic low-dose drinking water exposure.
Although the long-term exposure to copper has not been extensively studied, limited results suggest an effect of time. Indeed, available data from drinking water exposure of adults for less than 3 months confirmed the results from short-term exposure studies (Araya et al., 2004). However, the results from adult supplementation studies suggest adverse health effects following exposure to high levels intakes over longer periods of time (over 5 months; Turnlund et al., 2004). Subgroups identified as being vulnerable to chronic copper overexposure are individuals with copper-metabolism-related genetic disorders (such as WD patients), individuals with glucose-6-phosphate dehydrogenase deficiencies, children with childhood cirrhosis syndromes (ICC, ICT and TIC), and infants (less than one year-old). Of these subgroups, infants are considered the most vulnerable since they are likely to have the highest exposure due to the immaturity of their hepatobiliary system and their higher rates of copper absorption.
A randomized prospective study by Olivares et al. (1998) investigated the effects of copper exposure in breast- and formulafed infants (3 to 12 months old) who ingested drinking water with high (31.48 µmol Cu(II)/L or 2 mg/L) or low (less than 1.57 µmol Cu(II)/L or 0.1 mg/L) copper levels (as Cu (II) sulphate) for nine months via different means of consumption (e.g., food, formula, drinking water). Clinical and biochemical findings were recorded at six, nine and twelve months of age. Both over the course of the study and upon study termination, the subjects presented no acute (i.e., cases of diarrhea or respiratory symptom) or chronic (i.e., differences in markers of liver function) clinical effects following exposure to drinking water containing 2 mg/L of copper (corresponding to a mean ± SD daily intake of 318.7 ± 107.3µg/kg bw per day) when compared to controls.
The Olivares et al. (1998) study has been chosen as the critical study for deriving a proposed MAC since it was conducted in infants, involved continuous exposure to Cu(II) via drinking water for a longer period than studies in adults (usually up to three months), and took into account several temporal windows, including infants 4-6 months when the hepatobiliary system is still immature and gut absorption is greatest. Since formula-fed infants are expected to have the highest exposure to copper, the upper bound of the NOAEL, based on GI effects and liver function, for the formula-fed exposure sub-group (mean ± SD = 318.7 ± 107.3 µg/kg bw per day, experienced by 56 infants aged 4-6 months) was chosen as the point of departure; this upper bound dose (i.e., 426 µg/kg bw per day) represents the highest dose administered where no adverse effects were observed.
Although there are some uncertainties in the database for copper toxicity (including limited data on copper exposure in infants aged less than 6 months, limited data on the degree of variability in copper absorption among early age groups, as well as a lack of chronic exposure studies), the point of departure identified from Olivares et al. (1998) is considered conservative since no symptoms of copper toxicity were observed at this dose over the entire duration of the study (9 months); furthermore, due to the homeostatic regulation of copper absorption and excretion, the absorption of copper has been shown to decline as exposure to dietary copper increases (see section 8.1). As a result, an uncertainty factor accounting for database deficiencies is not warranted and thus 426 µg/kg bw per day is identified as the tolerable daily intake (TDI) for free copper.
The WHO (1993) set a provisional health-based guideline value of 2 mg/L for copper in drinking water from a provisional maximum tolerable daily intake (PMTDI) of 0.5 mg/kg proposed by JECFA in 1982 and an allocation factor of 10% of the PMTDI to drinking water. The PMTDI was based on liver toxicity, observed in an industry-funded study in dog (Shanaman et al., 1972) and the guideline was deemed provisional due to the uncertainties related to copper toxicity in humans (WHO, 1996). However, from an assessment conducted in 2003, the WHO maintained the value of 2 mg/L stated that is was based ‘to be protective against acute GI effects of copper and provide an adequate margin of safety in population with normal copper homeostasis’ (WHO, 2011). In 2004, the guideline value of 2 mg/L was confirmed, but was no longer provisional based on (Olivares et al., 1998, 2001; Pizarro et al., 1999b, 2001; Araya et al., 20012003a; Zietz et al., 2003b).
The U.S. Environmental Protection Agency (U.S. EPA, 1991) has set the maximum contaminant level goal (MCLG) and maximum contaminant level (MCL) for copper in drinking water at 1.3 mg/L. The U.S. EPA’s MCLG for copper was based on adverse gastrointestinal tract effects following acute exposure and was derived from a case study of nurses who consumed an alcoholic beverage that was stored in copper containers (Wyllie, 1957). A secondary Maximum Contaminant Level (SMCL) for copper has been established at 1 mg/L based on aesthetic considerations (taste and blue/green staining). However, the U.S. EPA regulates levels of copper through the Lead and Copper Rule (U.S. EPA, 1991, 2000), a treatment-based rule, which established an action level of 1.3 mg/L for copper in drinking water. Large water systems (with more than 50,000 connections), unless determined to be non-corrosive, are required to install “optimal corrosion control treatment” and meet specified water quality operating limits requirements. If the 90th percentile of copper concentrations in samples taken at customer taps at sites distributed with a specified prioritization (first-draw samples that have stagnated for at least 6 hours) exceeds the action level of 1.3 mg/L, the system must undertake a number of additional actions to control corrosion and provide public education. The number of sites, frequency of monitoring and scope of required actions vary with system size. Currently, a major revision of the Lead and Copper Rule is under way but is not expected to be finalized until after 2017 [71-80].
The California EPA (OEHHA, 2008) has developed a nonregulatory public health goal of 0.3 mg/L for copper in drinking water, based on infants as a sensitive population and is “believed adequate to protect against any adverse acute or chronic effects” from copper exposure. This goal was based on the absence of an adverse effect in Olivares et al. (1998) clinical study of infants exposed to copper in drinking water, and took into consideration “data from other studies on GI effects as the key endpoint”. The Australian Drinking Water Guidelines has set two levels for copper: 1 mg/L for aesthetics to prevent taste and staining problems; and 2 mg/L to prevent health issues (NHMRC, 2011).
Rationale
Copper is an essential nutrient that occurs naturally in the Earth’s crust, either in mineral deposits or, less frequently, in its metallic form. The U.S National Academy of Medicine (formerly called the Institute of Medicine) has established both recommended daily intakes and tolerable upper intake levels for copper. Copper is used in a wide range of products, including as a result of it being a component of bronze and brass, and is considered by the National Plumbing Code of Canada to be an acceptable material for service lines and plumbing systems. The main anthropogenic sources of copper in the environment include mining, agricultural run-off, waste management and water treatment. Canadians are mainly exposed to copper through the ingestion of food, drinking water and nutritional supplements. International agencies have determined that the data available on copper is not sufficient to classify it with respect to carcinogenicity. Health effects can be associated with both copper deficiency, and exposure to copper at levels much higher than the recommended daily intake. Copper deficiency is not considered to be a concern in Canada.
A maximum acceptable concentration (MAC) of 2 mg/L (2000 µg/L) is proposed for total copper in drinking water. The proposed MAC is considered to be protective of both short-term effects (i.e., GI effects) as well as longer-term effects (including potential effects in the liver and kidneys). The proposed MAC is achievable by available treatment technology and measurable by available analytical methods. However, because copper is generally found in drinking water as a result of leaching in the distribution and plumbing systems, strategies for reducing exposure to copper from drinking water should focus on corrosion control through approaches such as water quality adjustments and the use of corrosion inhibitors. As part of its ongoing guideline review process, Health Canada will continue to monitor new research in this area and recommend any change to the guideline that it deems necessary.
British Columbia
The proposed change to the copper guidelines to maintain the aesthetic objective (AO) at 1.0 mg/L as well as the addition of a maximum acceptable concentration (MAC) of 2.0 mg/L based on studies citing an association between long term exposure to copper and liver and kidney effects is anticipated to have some notable shifts in how copper in drinking water is measured. Similar to lead, a notable impact of the new MAC will be the shift in focus from centralized sampling to point of consumptionbased programs. Sampling of buildings, particularly high-risk institutions such as schools and daycares, will remain a cost pressure. Questions of whether this is the responsibility of water suppliers or building owners will be raised. Given that the data on water concentrations reported in section 5.2 shows very few exceedances of the MAC, it is anticipated the proposed change in the guideline will have little impact on the water delivered by water supply systems. However, as leaching from fixtures within individual buildings is likely the most significant contributor to copper concentration in water, it is therefore difficult to anticipate copper concentrations delivered at individual taps, as this will vary from building to building, depending on plumbing, and system to system, depending on how corrosive water is.
The proposed addition of a maximum acceptable concentration will also mean significantly different messaging from what health officials have historically given regarding copper. It will also require the need for clear and meaningful advice to those that have been consuming water sources that exceed the proposed MAC, who have previously been told that elevated copper is merely an aesthetic concern. Yukon, It is not possible to quantify the impact of the proposed technical document and MAC for copper at this point in time for Yukon. The Yukon drinking water regulatory framework is primarily focused on water treatment and provision of safe drinking water as it leaves the water treatment plant to the distribution system. Provisions of the Drinking Water Regulation specify specific monitoring requirements for raw and treated water (at the point of water leaving the plant). While there is a requirement for monitoring of free chlorine, total coliforms and E.coli within the distribution system, there are currently no specific requirements for routine monitoring of other parameters.
Environmental Health Services (EHS) regulatory mandate extends to curb stop for large public drinking water systems. Routine compliance monitoring and regulatory requirements for large public drinking water system owners beyond this point is not within the current mandate. Further policy development would be needed to incorporate testing at the tap. Currently all Yukon large public drinking water systems meet the proposed MAC for copper entering the distribution system. EHS will be doing policy development in terms of monitoring of the distribution system for copper and other metals over the next few months.
Environmental Impacts of Heavy Metals
The presence of heavy metals in the environment leads to a number of adverse impacts. Such impacts affect all spheres of the environment, that is, hydrosphere, lithosphere, biosphere and atmosphere. Until the impacts are dealt with, health and mortality problems break out, as well as the disturbance of food chains.
Effect of Heavy Metals Contamination
Heavy metals contamination is becoming a serious issue of concern around the world as it has gained momentum due to the increase in the use and processing of heavy metals during various activities to meet the needs of the rapidly growing population. Soil, water and air are the major environmental compartments which are affected by heavy metals pollution.
Effect on Soil
Emissions from activities and sources such as industrial activities, mine tailings, disposal of high metal wastes, leaded gasoline and paints, land application of fertilisers, animal manures, sewage sludge, pesticides, wastewater irrigation, coal combustion residues and spillage of petrochemicals lead to soil contamination by heavy metals. Soils have been noted to be the major sinks for heavy metals released into the environment by aforementioned anthropogenic activities. Most heavy metals do not undergo microbial or chemical degradation because they are nondegradable, and consequently their total concentrations last for a long time after being released to the environment.
The presence of heavy metals in soils is a serious issue due to their residence in food chains, thus destroying the entire ecosystem. As much as organic pollutants can be biodegradable, their biodegradation rate, however, is decreased by the presence of heavy metals in the environment, and this in turn doubles the environmental pollution, that is, organic pollutants and heavy metals thus present. There are various ways through which heavy metals present risks to humans, animals, plants, and ecosystems as a whole. Such ways include direct ingestion, absorption by plants, food chains, consumption of contaminated water and alteration of soil pH, porosity, color and its natural chemistry which in turn impact on the soil quality [81-96].
Effects on Water
Although there are many sources of water contamination, industrialization and urbanization are two of the culprits for the increased level of heavy metal water contamination. Heavy metals are transported by runoff from industries, municipalities and urban areas. Most of these metals end up accumulating in the soil and sediments of water bodies. Heavy metals can be found in traces in water sources and still be very toxic and impose serious health problems to humans and other ecosystems. This is because the toxicity level of a metal depends on factors such as the organisms which are exposed to it, its nature, its biological role and the period at which the organisms are exposed to the metal. Food chains and food webs symbolize the relationships amongst organisms. Therefore, the contamination of water by heavy metals actually affects all organisms. Humans, an example of organisms feeding at the highest level, are more prone to serious health problems because the concentrations of heavy metals increase in the food chain.
Effects on Air
Industrialisation and urbanisation, due to rapid world population growth, have recently made air pollution as a major environmental problem around the world. The air pollution was reported to have been accelerated by dust and particulate matters (PMs) particularly fine particles such as PM2.5 and PM10 which are released through natural and anthropogenic processes. Natural processes which release particulate matters into air include dust storms, soil erosion, volcanic eruptions and rock weathering, while anthropogenic activities are more industrial and transportation related. Particulate matters are important and require special attention as they can lead to serious health problems such as skin and eyes irritation, respiratory infections, premature mortality and cardiovascular diseases. These pollutants also cause deterioration of infrastructure, corrosion, formation of acid rain, eutrophication and haze. Amongst others, heavy metals such as group 1 metals (Cu, Cd, Pb), group 2 metals (Cr, Mn, Ni, V and Zn) and group 3 metals (Na, K, Ca, Ti, Al, Mg, Fe) originate from industrial areas, traffic and natural sources, respectively.
Treatment processes for acid mine water typically generate high-density sludge that is heterogeneous due to variety of metals, metalloids and anionic components, and this makes it difficult to dispose the sludge [19]. Recent research have therefore focused on the recovery of chemical species from acid mine drainage (AMD) and secondary sludge. This is aimed at recovering valuable resources and also enabling easier and safer disposal of the treated sludge, hence reducing their environmental footprints. Disposal of metal ladened waste to landfills and waste retention ponds/ heaps lead to secondary pollution of surface and subsurface water resources. It may also lead to soil contamination, hence affecting their productivity. In order to protect human health, plants, animals, soil and all the compartments of the environment, proper and careful attention should be given to remediation technologies of heavy metals. Most physical and chemical heavy metal remediation technologies require handling of large amounts of sludge, destroy surrounding ecosystems and are very expensive.
Precipitation
A variety of alkaline chemical reagents have been used over the years for neutralisation of acid mine drainage (AMD) in order to increase the pH and consequently precipitate and recover the metals. The most common alkaline reagents used for sequential recovery of minerals resources from AMD are limestone (CaCO3), caustic soda (NaOH), soda ash (Na2CO3<), quicklime (CaO), slaked lime (Ca(OH)2) and magnesium hydroxide (Mg(OH)2). Some processes have recovered metals at varying pH regimes and synthesized commercially valuable materials such as pigments and magnetite. Some minerals are recovered and sold to metallurgical industries, hence off-setting the treatment costs Table 1.
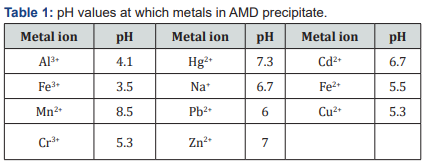
Adsorption
Adsorption occurs when an adsorbate adheres to the surface of an adsorbent. Due to reversibility and desorption capabilities, adsorption is regarded as the most effective and economically viable option for the removal of metals from aqueous solution. Although efficient, adsorption is not effective with very concentrated solution as the adsorbent easily gets saturated with the adsorbate. It is only feasible for very dilute solutions, is labor intensive because it requires frequent regeneration, and it is not selective in terms of metal attenuation. Adsorption is therefore not applied in a large scale of metal remediation.
Ion Exchange
Ion exchange is the exchange of ions between two or more electrolyte solutions. It can also refer to the exchange of ions on a solid substrate to soil solution. High cation exchange capacity clay and resins are commonly used for the uptake of metals from aqueous solutions. However, this method requires high labor and is limited to certain concentration of metals in the solution. This system also operates under specific temperature and pH. Natural and synthetic clays, zeolites and synthetic resins have been used for removal and attenuation of metals from wastewater.
Biosorption
Biosorption refers to the removal of pollutants from water systems using biological materials, and it entails the absorption, adsorption, ion exchange, surface complexation and precipitation. Biosorbents have an advantage of accessibility, efficiency and capacity. This process is readily and easily available. Regeneration is easy, hence making it very favorable. However, when the concentration of the feed solution is very high, the process easily reaches a breakthrough, thus limiting further pollutant removal.
Membrane Technologies
The use of membrane technologies for the recovery of acid mine drainage is very effective for water that has high concentration of pollutants. It uses the concentration gradients phenomenon or the opposite which is reverse osmosis. There are different types of membranes that are used for mine water treatment including: ultrafiltration, nano-filtration, reverse osmosis, microfiltration, and particle filtration.
Case study of South Africa Acid Mine Drainage
South Africa is well endowed by mineral reserves, and this has triggered its immense dependence on mineral resources for gross domestic product and economy. However, the legacy of coal and gold mining has left in its wake serious environmental problems. The major problem is acid mine drainage. Acid mine drainage (AMD) is formed from the hydro-geochemical weathering of sulphide-bearing rocks (pyrite, arsenopyrite and marcasite) in contact with water and oxygen. This reaction is also catalyzed by iron (Fe) and sulphur-oxidising microorganisms. In a nutshell, the formation of AMD can be summarized as follows:
The oxidation of sulphide to sulphate solubilises the ferrous iron (Fe(II)), which is subsequently oxidized to ferric iron (Fe(III)):
Either these reactions can occur spontaneously or can be catalyzed by microorganisms (sulphur- and iron-oxidizing bacteria) that derive energy from the oxidation reaction. The ferric cations produced can also oxidize additional pyrite into ferrous ions:
The net effect of these reactions is to produce H+ and maintain the solubility of the ferric iron. Because of the high acidity and elevated concentration of toxic and hazardous metals, AMD has been a prime issue of environmental concern that has globally raised public concern. The discharge of metalliferous drainage from mining activities has rendered the environment unfit to foster life. Pragmatic approaches need to be developed to counter this mining legacy that is perpetually degrading the environment and its precious resources. Research and piloted studies have indicated that active and passive approaches can be successfully adopted to treat acid mine drainage and remove potentially toxic chemical species. The presence of Al, Fe, Mn and sulphates is a prime concern in addition to the trace of Cu, Ni, Pb and Zn. Metalloids of as and earth alkali metal (Ca and Mg) are also present in significant levels. Several studies have shown the feasibility of treating acid mine drainage to acceptable levels as prescribed by different water quality guidelines, but the resultant sludge has been an issue of public concern due to its heterogeneous and complex nature loaded with metal species.
Based on that evidence, research studies have been firmly embedded on the recovery of valuable minerals from AMD. There are several mechanisms used for the recovery of chemical components from AMD including: precipitation, adsorption, biosorption, and ion exchange, desalination and membrane filtration. Out of those techniques, precipitation has been the promising technology due to the ability to handle large volumes of water with very little dosage. Adsorption and ion exchange have a challenge of poor efficiency at elevated concentrations and quick rate of saturation. Membrane technologies have the problem of generating brine that creates another environmental liability. Desalination has a problem of producing salts that has impurities, hence making them unsuitable for utilization. Freeze desalination has been the promising technology, but it has never been tried in a large scale.
Impacts of Heavy Metals in South Africa
South Africa’s geology is rich in coal and mineral reserves which contain key metals such as gold, platinum and copper. The significant volume of mineral and coal reserves has made mining serve as a backbone in the development and growth of the country’s economy. This is evident from the massive number of mines found around the country. However, mining has been noted to cause inimical impacts to the human health, organisms and environment as a whole, with water resources being the most common victim of the pollution. However, the above -mentioned concentrations depend on the pH of the AMD-concentrations decrease when pH increases. When exposed to such conditions, mortality and diseases are most likely to occur in organisms, as well as other health. In addition, AMD destroys ecosystems of organisms and also negatively impacts on the economy of the country. Heavy metals in active and abandoned mines in South Africa have impacted both surface and underground water.
Legal requirements of Water Quality
The National Environmental Management Act (NEMA) 108 of 1998, stipulates that everyone has the right to live in an environment which is safe and unlikely to pose any deleterious effects to their health. The legislative requirements for industrial effluents are primarily governed by the Department of Water Affairs DWS Water Quality Guidelines. This purpose requires that any person who uses water for industrial purposes shall purify or otherwise treat such water in accordance with requirements of DWA.
Deleterious effects of Acid Mine Drainage on Terrestrial and Aquatic Ecosystems
The introduction of effluents from mining activities into receiving streams can severely impact aquatic ecosystems through habitat destruction and impairment of water quality. This will eventually lead to a reduction in biodiversity of a given aquatic ecosystem and its ability to sustain life. The severity and extent of damage depends on a variety of factors including the frequency of influx, volume and chemistry of the drainage and the buffering capacity of the receiving stream.
Acidity
When metals in AMD are hydrolysed, they lower the pH of the water making it unsuitable for aquatic organisms to thrive. AMD is highly acidic (pH 2-4), and this promotes the dissolution of toxic metals. Those toxic species exert hazardous effects on terrestrial and aquatic organisms. Also, if the water is highly acidic, only acidophile microorganisms will thrive on such water with the rest of aquatic organisms migrating to other regions which are conducive to their survival. Many streams contaminated with AMD are largely devoid of life for a long way downstream. To some aquatic organisms, if the pH range falls below the tolerance range, probability of death is very high due to respiratory and osmoregulation failure. Acidic conditions are dominated by H+ which is adsorbed and pumps out Na from the body which is important in regulating body fluids.
Toxic Chemical Species
Exposure of aquatic and terrestrial organisms to potentially toxic metals and metalloids can have devastating impacts to living organisms. Toxic chemical species present in AMD have been reported to be toxic to aquatic and terrestrial organisms. They are associated with numerous diseases including cancer. Some of these chemical species may accumulate and be biomagnified in living organisms, hence threatening the life of higher trophic organisms such as birds. Lead causes blood disorders, kidney damage, miscarriages and reproductive disorders and is linked to various cancers. The exposure of living organisms to toxic chemical species in AMD can also lead to nausea, diarrhoea, liver and kidney damage, dermatitis, internal haemorrhage and respiratory problems. Epidemiological studies have shown a significant increase in the risk of lung, bladder, skin, liver and other cancers on exposure to these chemical species. The effects of Al, Fe, Mn, Cu, Mg and Zn on the health of living organisms are summarised in Table 2.
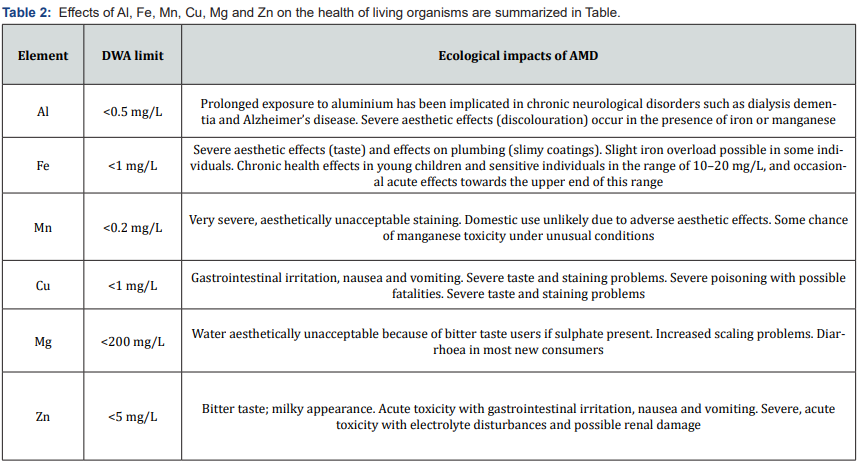
Why are traces of heavy metals in drinking water at all?
Heavy metals enter the natural water cycle via emissions during combustion processes with rainwater, it also happens in over fertilization in agriculture via leachate and the rainwater becomes contaminated when it falls.
In each country a governing body will determine which contaminants are permitted and to what limit. Waterworks control water to the point where it is fed into the public grid. There, drinking water converts into tap water and evades the supervision of the waterworks in the stretch of pipe to your tap where contamination can happen. A well-known example of this is high lead concentrations in tap water, which are due to outdated supply lines or house pipes made from lead. These pipes have been banned as components, yet cases of households with leaded water are still known.
How does lead from a pipe actually get into drinking water? Lead is known to be non-water soluble as a metal. The chemical process called substance migration is the cause of metals entering water. The surfaces of two substances on a molecular or atomic level interact. Not every metal behaves the same when migrating. The less a metal releases traces of its substance to liquids, the more food-grade it is and therefore useable. For example Copper and stainless steel are a higher food grade as they do not release traces when in contact with water.
The limit values protect us from acute symptoms of poisoning. However, long-term health consequences have not been taken into account with the permanent consumption of the smallest quantities of heavy metals. They can accumulate in the body over years and have a harmful effect on metabolism. They can disrupt natural cell functions or cause hormone failures. There are many illnesses associated with heavy metal stress, Cancer, Multiple Sclerosis, Parkinson’s and Alzheimer’s and even depression. In principle, heavy metals, like all toxins, effect the kidneys, liver and pancreas - organs that primarily process foreign bodies and harmful substances.
Today, water works and utilities use technologically advanced filter systems in drinking water treatment. The prescribed limit values can be largely met with the cleaning processes used. However, zero tolerance solutions are not to be expected from this field with the usual methods in the medium and long term.
In industrial applications, wherever pure water is of great importance reverse osmosis is used for drinking water extraction and purification. Reverse osmosis plants discharge particles and substances from the water via semipermeable membranes and artificially generated osmotic pressure. The final result is pure H2O. So why do water suppliers not use this procedure to remove all pollutants from the water? The answer is obvious: reverse osmosis works so well that it also removes substances that are valuable to the body. In the case of drinking water, essential minerals. However, waterworks - as well as mineral water bottlers - are prohibited by law from re-enriching their products retrospectively with what they previously withdrew from them. If natural mineral water is enriched with ingredients that are not already present in the natural spring water, it may only be marketed as table water.
Nanosensor
What they are…What they do?!
The term nanosensor is not clearly defined. Most definitions refer to a sensing device with at least one of its dimensions being smaller than 100 nm and for the purpose of collecting information on the nanoscale and transferring it into data for analysis. Nanotechnology deals with physical or chemical properties of matter at the nanoscale, which can be different from their bulk properties. Nano sensors can take advantage of these phenomena. Important characteristics and quality parameters of nano sensors can therefore be improved over the case of classically modeled sensors with merely reduced sensing parts and/or the transducer.
Therefore, nano sensors are not necessarily reduced in size to the nanoscale but could be larger devices that make use of the unique properties of nanomaterials to detect and measure events at the nanoscale. For instance, in noble metals such as silver or gold, nanostructures of smaller size than the de Broglie wavelength for electrons lead to an intense absorption in the visible/near-UV region that is absent in the spectrum of the bulk material. Nano sensors have been developed for the detection of gases, chemical and biochemical variables, as well as physical variables and the detection electromagnetic radiation.
Nano sensor fabrication
Nano sensors can be prepared by using different methods. Three common methods are top-down lithography, bottom-up fabrication (such as for instance controlled lateral epitaxial growth and atomic layer deposition), and self-assembled nanostructures (usually done with biomolecules, e.g., liposomes, that combine in such a way that the biochemical detection of an analyte is converted into an electrical signal).
Nanosensors Based on Nanoparticles and Nanoclusters
Nanoparticles, primarily noble metal ones, have outstanding size-dependent optical properties that have been used to build optical nano sensors. The spectrum of a phenomenon called the localized surface Plasmon resonance (LSPR) depends on the size, shape and material of the nanoparticle itself as well as the particle’s environment. The high sensitivity of LSPR sensors can approach the single-molecule limit of detection for large biomolecules. Apart from metal nanoparticles, optical nano sensors based on fluorescence measurements have been built with semiconductor quantum dots and other optical sensors have been developed with nanoscale probes that contain dyes whose fluorescence is quenched in the presence of the analyte to be determined; nanoparticle films have been used for gas sensors; magnetic nanoparticles bound to biorecognitive molecules (i.e. DNA, enzymes, etc.) have been used to enrich the analyte to be detected.
For example, researchers have developed an enzyme biomarker test based on gold nanoparticles that can detect enzyme markers of disease known as proteases in humans, animals and food products. This nanosensor indicates when proteases are present through a visible color-change reaction.
Nanosensors based on nanowires, nanofibers and carbon nanotubes
Most sensors based on carbon nanotubes (CNTs) are field effect transistors (FET) because, although CNTs are robust and inert structures, their electrical properties are extremely sensitive to the effects of charge transfer and chemical doping by various molecules. The functionalization of CNTs is important for making them selective to the target analyte, different types of sensors are based on molecular recognition interactions between functionalism CNT and target analytes. For instance, researchers have developed flexible hydrogen sensors using single-walled carbon nanotubes decorated with palladium nanoparticles.
Nanowires and nanofibers have also been used to build chemiresistive sensors for the diagnosis of diseases. They have been used to maximize gas sensor responses in exhaled breath analysis for the detection of volatile organic compounds (which are biomarkers for various diseases; for example, acetone, hydrogen sulfide, ammonia, and toluene can be used as biomarkers for evaluating diabetes, halitosis, kidney malfunction, and lung cancer, respectively). In one example, porous tin oxide (SnO2) nanofibers have been demonstrated to detect acetone at levels of around 0.1 ppm, which is eight times lower than the required gassensing level for diagnosing diabetes.
Nano sensors Based on Graphene
Another carbon nanomaterial, functionalized graphene, holds exceptional promise for biological and chemical sensors. Already, researchers have shown that the distinctive 2D structure of graphene oxide (GO), combined with its superpermeability to water molecules, leads to sensing devices with an unprecedented speed (“Ultrafast graphene sensor monitors your breath while you speak”). Scientists have found that chemical vapors change the noise spectra of graphene transistors, allowing them to perform selective gas sensing for many vapors with a single device made of pristine grapheme, no functionalization of the graphene surface required (“Selective gas sensing with pristine graphene”). Researchers also have begun to work with graphene foams, threedimensional structures of interconnected graphene sheets with extremely high conductivity. These structures are very promising as gas sensors (“Graphene foam detects explosives, emissions better than today’s gas sensors”) and as biosensors to detect diseases (see for instance: “Nanotechnology biosensor to detect biomarkers for Parkinson’s disease”).
Nano sensors Based on Bulk Nanostructured Materials
Whereas several properties of nanoparticles are useful for applications in nanosensors, their catalytic behavior is one of the most important with regard to electrochemical sensing devices. For instance, platinum nanoparticles supported on materials such as porous carbon or noble metals such as gold are reported to be relevant in the design of gas diffusion electrodes. Another property, their high surface area, makes nanoparticles suitable for immobilizing molecules, polymers or biomaterial coatings that allow the generation of composite materials with tunable surface properties. For example, modifying metal nanoparticles with pre designed receptor units and assembling them on surfaces could lead to new electrochemical sensors with tailored specificities. Simple and highly-selective electro analytical procedures can also be achieved by proper functionalization of nanoparticles. Finally, stable nanoparticles can substitute amplifying labels of limited stability, such as enzymes or liposomes, with equivalent or improved sensitivities.
Nano Sensors Based on Metal-Organic Frameworks (MOFs)
Metal-organic frameworks (MOFs) are organic-inorganic hybrid crystalline porous materials that consist of a regular array of positively charged metal ions surrounded by organic ‘linker’ molecules. The metal ions form nodes that bind the arms of the linkers together to form a repeating, cage-like structure. Due to this hollow structure, MOFs have an extraordinarily large internal surface area, which makes them ideal materials for gas sensing. By making the MOF from different metal atoms and organic linkers, researchers can create materials that selectively absorb specific gases into tailor-made pockets within the structure. One example is a thin-film a tailor-made MOF, coated onto an electrode that forms an electronic sensor that could detect traces of sulfur dioxide gas.
Other Examples of Nano sensor Applications
A simple single nanoscale sensor and light spectroscopy (fluorescence) can be used for sensing the difference in potential across a biological membrane such as a cell wall. This non-contact, optical voltage nanosensor is based on a Forster resonance energy transfer (FRET) sensor on a DNA origami. A graphene nanosensor enables real-time monitoring of insulin at physiologically relevant concentrations - down to a concentration of approximately 35 pM. This device employs an aptamer-based graphene field-effect transistor (GFET) nanosensor that can rapidly respond to external stimuli such as the binding between surface-immobilized aptamer molecules with insulin. This results in significant changes of the electrostatic charge characteristics in the close proximity of the graphene surface. A gel-based sensor technology can help medical physicists and oncologists effectively plan radiotherapy in the clinic, reduce accidental overexposures, and reduce radiationinduced toxicity. In contrast to traditional inflexible sensors, these plasmonic nano sensor gels indicate exposure levels as radiation delivered to the hydrogel increases the intensity of color developed in the gel.
By interfacing passive, wireless graphene nano sensors onto biomaterials via silk bio resorption, researchers have developed graphene nano sensor tattoos on teeth in order to monitor harmful oral bacteria. A great example of nano sensor technology is the ability of chips with molecular probes to detect intracellular biological parameters, i.e., from within a living cell.
Sensors For Intelligent Processing Of Structural Polymeric Composites
Structural polymeric composites are critically important to sustaining U.S. aerospace and defense superiority. The potential market for these materials, by the end of the century, is projected to be 90,000 metric tons, a sixfold increase in tonnage since 1986, with a tenfold increase in worldwide employment to 200,000 people (AIA, 1991). Improvements in the manufacture of products that use these materials will help in the realization of such projections. A polymeric composite typically has two primary microstructural components: a polymer material and reinforcing fibers. Two different generic types of polymers are used: thermosetting (the traditional choice) and thermoplastic (the more recent choice). Thermosetting polymers are initially low-viscosity liquids that can flow into a mold or around fibers. During the course of processing, thermosets react to increase molecular weight and viscosity, eventually becoming highly cross-linked, insoluble, infusible materials. On the other hand, thermoplastics are fully polymerized materials that melt and flow upon application of heat. Thermoplastics are processed well above their glass transition temperatures or melting points (if the material is semicrystalline) to reduce the melt viscosity and allow flow and to promote adhesion to the fibers.
The fibers in the composite are the component that provides the desired high stiffness and strength properties that make the material useful for structural applications. The polymer matrix protects the fiber, serves as a medium to transfer load between the fibers, and stabilizes the fiber when subjected to compression loading.
A composite structure is typically made by stacking layers of pre-impregnated (“prepreg”) material in prescribed directions on a tool form, compressing the stack, and then curing or consolidating the composite. In the case of high-performance thermosetting resins, the curing process requires the application of heat and pressure. For small-lot production, this curing step is usually accomplished in a large pressure vessel called an autoclave. Alternative processes, such as resin transfer molding, are preferred for higher-volume mass production. Consolidation of a thermoplastic resin does not require autoclave but does require the application of energy, such as heat. In addition, new composites are being developed that consist of blends of thermosets and thermoplastics; in these materials, curing and consolidation occur simultaneously and interactively to produce complex microstructures possessing functionally gradient material properties that can be tailored to optimize specific strength, processability, etc. Intelligent processing can improve the resultant properties of composite components. The discussion that follows examines the technologies required to fully implement intelligent processing with emphasis on the most critical sensor requirements.
At the present time, most of the structural polymeric composites used in the aerospace industry consist of a high strength/high stiffness fiber (e.g., graphite, boron, or aramid) embedded in a thermosetting organic-resin matrix binder (e.g., epoxy, bismaleimide, polyimide, polyester, or cyanate ester). These resins are complex chemical formulations with batch-to-batch chemical variations. Until recently, the conventional processing strategy for thermoset polymer composites was based on a priori models. These models cannot account for variations in material chemistry, part geometry, autoclave sizes and heating patterns, and tolling materials. As a consequence, they assume “one size fits all” and are developed for the worst-case processing conditions. Hence these models specify a safe (i.e., protracted) cure cycle.
In contrast, intelligent processing senses the state of the material during cure. The development of these models requires the definition of events that denote the changes in material state, e.g., flow, deformation, growth, etc. A processing system is also required that is empowered (through a model or set of conditionaction relations) to adjust the process parameters in response to changes in the materials state to achieve desired end-use properties. The first steps in developing an intelligent processing system are the mapping of these desired end-use properties to the actual material parameters that can be sensed and the processing variables that can be controlled. Some properties (such as residual stresses; void volume; and fiber location, volume, and orientation) can be inferred from engineered relationships with parameters that can be sensed in situ, while other properties (such as part strength and modulus) depend upon other engineering relationships to determine properties that cannot now be measured in situ.
Identifying “need-to-be-sensed” parameters can be accomplished through a review of available models of the process or by developing a mapping of properties to parameters and to variables. In the next step, a process engineer must assess available sensor technologies that can provide the necessary information either directly or indirectly. To accomplish this assessment, the framework developed can be used as a guide to list and detail the necessary parameters for evaluating and comparing different sensor technologies. With these descriptors, the process engineer can describe the range, tolerance, and limits of the sensing requirements and identify one or more candidate sensor technologies, and their respective capability and constraints.
Performance estimates and benefits of intelligent manufacturing can only be validated in a true manufacturing environment. One example is provided by the U.S. Air Force for an application that produces polymer composite replacement parts for the A-10 aircraft. In this case, sheet metal aluminum parts for the leading edge of the aircraft wing are being replaced with a hybrid carbon and aramid fiber/epoxy-resin composite to improve performance and extend the useful life of the aircraft. Initially the composite replacements were cured using a conventional (i.e., a priori model) cure cycle. But in 1990, the Air Force’s Sacramento Air Logistics Center implemented an intelligent processing system that achieved full cure in 1.5 hours versus 7 hours required for the conventional approach; that is, a 70 percent reduction in cure time. The intelligent manufacturing system was originally implemented using the polymeric material temperature (as measured by a thermocouple) as the basis for estimating the degree of cure. Autoclave temperature profiles during cure are presented for both approaches along with the resultant test values of selected material properties. This data confirmed that intelligent processing could produce parts with strength values statistically equivalent to those conventionally processed (Warnock and LeClair, 1992).
Even though this initial implementation was quite successful, it was not without difficulty. For instance, the thermocouple signal was “noisy” and hence many data points were needed to clearly establish the trendline. To identify various levels of candidate improvements, the framework presented in Chapter 2 was applied to relate sensing needs (e.g., resin temperature) to the currently available sensor technology (e.g., thermocouple); the results of this comparison are summarized in Appendix D. Some of the thermocouple shortfalls can be partially ameliorated, but nonetheless, a new sensor technology was clearly needed. For example, autoclave temperature does not directly relate to the degree of resin cure (i.e., extent of the formation of cross-linked polymer bonds).
One such multiuse technology which addresses both the limitation of thermocouples and the need for more microscopic property monitoring is photon scattering fiberoptic sensing, as described in Appendix D. Photon-scattering sensor technology uses the energy and momentum distribution in the scattered photon flux to extract a wealth of useful and interpretable information regarding the physical and chemical nature of the material being processed. Suitable electromagnetic radiation scattering measurements (i.e., Rayleigh, Brillouin, and Raman scattering) can theoretically provide a direct measurement of material properties that include bulk modulus, thermal diffusivity, sound attenuation factor, sonic speed, heat capacity ratio, chemical composition, bulk viscosity, shear viscosity, and the free energy of mixing (Maguire and Talley, 1995). The scattered flux can be detected by the receiving optical fiber at any given angle and transported at the speed of light to a remote spectrometer.
Near-infrared Raman spectroscopy, using fiber optics to provide a relatively simple optical contact with the material, can allow direct determination of the degree of cure. As an example, an epoxy-amine material has a Raman spectral peak at 1,250 cm-1 resulting from the vibration of the polymer’s epoxide ring structure and another one at 2,870 cm-1 arising from the stretching of C-H bonds. During cure, the number of the C-H bonds does not change significantly, while the number of epoxide groups notably decreases. Therefore, the 2,870 cm-1 peak can be used as an internal standard to determine the degree of change in the 1,250 cm-1 peak over time (Maguire and Talley, 1995). Sensor array useful in many intelligent processing applications. Continued research and development of multiuse sensor technology will provide crucial enabling science and understanding for addressing these intelligent processing sensor opportunities. The last section of this chapter discusses research needs and opportunities for photon-scattering sensor technology.
Thermoplastic Polymer Matrix Composites
Future systems, ranging from aircraft parts and printed circuit boards to applications that must survive in the rigors of space, will require further innovation in intelligent processing, particularly in the development of low-cost out-of-autoclave8 composite fabrication techniques and new material systems such as components made of thermoplastic matrix composites. A newly developed out-of-autoclave technique involves the use of a unique energy transduction mode known as direct electric heating that is, passing a current through the carbon reinforcing fibers (Miller and Van den Nieuwenhuizen, 1993). Electric heating of the fibers provides for a versatile temperature source capable of accommodating complex parts possessing varying curvature and laydown angles. It can produce a high strength, low void content composite. In the discussion that follows, processing needs, together with the unique hardware for part lay-up, are discussed to identify requirements for new sensor configurations. Thermoplastic material part quality is defined by the resultant physical properties, such as ultimate strength, void content, and elastic modulus. However, in-process measurements of some of these quantities (e.g., ultimate strength) cannot be directly obtained, since their measurement requires part destruction (University of Delaware, 1989). Void content, a commonly used measure of laminate quality, can be measured by x-rays, ultrasonic pulses, or determination of local electrical or by thermal conductivity using infrared sensors. Controlling void content during the process requires measuring both temperature and pressure in real-time during processing and making appropriate adjustments. Thus, temperature must be measured; if it begins to exceed the prescribed range, the tape head pressure can be adjusted accordingly across the face of the tape head to bring the temperature back in line.
Polymer Nanocomposites
Polymer Nanocomposite-Based Electrochemical Sensors and Biosensors
Polymer nanocomposites (PNCs) have electrochemical properties as transducers which can be used for the manufacturing of electrochemical sensors and biosensors. They possess significant variations in responsiveness, synthesis, and morphology, which help in a significant level of variations in conductivity. Afar from the economic aspect of the PNC-based sensors, the improved performance on the electronic side stands apart among its peers through the basal plane ratio of the nanofillers, method of doping, kinetic properties of the electrode, biological response and environmental impact. The impact of nanofillers in PNCs plays a significant role in sensing, processing, and actuating capabilities of the electrodes of electrochemical and biosensing applications.
The “active states of PNCs” rests on three pillars: high electrical conductivity rate, large surface area and fast electron rate which leads to best electricidal sensor outcomes. PNCs helps in the material technological advancement of electrochemical sensors which have high sensitivity and selectivity, lower detection limits, reproducibility and stability. All these increased used the PNCs in electrochemical sensor research which were manufactured through chemical synthesis or polymerization methods and could be easily scaled up for various applications. The electrochemical sensors along with the immunosensors and biosensors are becoming the norm of the day. Detections limits and sensing technologies are improved consistently due to developments happening in the unique properties of PNCs especially conductivity and electrochemical activity. The interactive fillers facilitate ion diffusion that impacts the sensing applications through intercalation into the PNC matrices leading to better stability of active electron transfer sites and detection limits. These active fillers help in reducing the layer thickness in PNC leading to ultrathin electrochemical detector technology. PNCs stand as an outstanding leader with significant advantages in large specific interaction surface area, reduced dimension of fillers and efficient electron transfer rate.
Electrochemical Sensors
PNCs are widely used in the development of electrochemical sensors. The electrochemical sensors are based on three categories of PNCs. PNCs of conductive polymers and inorganic nanomaterials, PNCs of conductive polymers and Grp, and PNCs of conductive polymers and CNTs. Once interaction has occurred between the PNC-based electrochemical sensors and the target analyte, an electronic signal is detected by the transduction system.
Electrochemical Sensors Based on Polymer Nanocomposites
Polymer Nanocomposites Based on Conductive Polymers and Inorganic Nanomaterials
Metal and metal oxide nanoparticles have been extensively studied as electrochemical sensing materials due to such beneficial features as their small size; unique chemical, physical, and electronic properties; flexibility in fabricating novel and improved sensing devices; and good sensitivity to the ambient condition. The assimilation of nanoparticles of metals into PNC matrices set the stage for enhanced electrocatalytic electrode detection leading to multiple modern-day applications. For example, a Zinc oxide nanoparticle intercalated into polypyrrole (ZnO-PPy) PNC showed excellent Xanthine detection by through xanthine oxidase enzyme immobilization. A glassy carbon electrode (GCE) modified with ultrathin polypyrrole nanosheets decorated with Ag nanoparticles was fabricated for the detection of hydrogen peroxide (H2O2).
The modified device showed high sensitivity toward the reduction of H2O2. A similar electrochemical sensor based on polypyrrole-platinum (PPy-Pt) PNC was fabricated for the detection of H2O2. Another voltammetric sensor based on a polyaniline-gold nanoparticle (PANI-AuNP) PNC deposited on GCE was used for the detection of epinephrine (EP) and uric acid (UA). Exploiting the advantages of PNCs, two GCEs modified with PANITiO2 and PANI-NiCo2O4PNC-based electrochemical sensors were developed for the detection of glucose. TiO2 nanotubes (TNTs) were intercalated into a PANI-TNT PNC composite for through hydrothermal method for the detection (a reported sensitivity of 11.4 μA mM-1) of glucose (a reported sensitivity of 11.4 μA mM-1) by the immobilization of glucose oxidase.
Polymer Nanocomposites Based on Conductive Polymers and Graphene
Graphene (Grp), an allotrope of carbon, has become the new material of interest and widely integrated into sensor research from the beginning of this millennium due to its unique properties of electrical conduction and 2-dimensional existence. Grp-PNCbased electrochemical sensors are used for electroanalytical detection of target molecules with high precision of selectivity and sensitivity, which showed spectacular detection limits over a wide range. An electrochemical sensor fabricated for the detection of 4-aminophenol (4-AP) using a PANI-Grp-GCEmodified device showed a detection limit of 6.5×10-8 M and sensitivity of 604.2 μAmM-1. A sensor was fabricated with a PANIGrp-based PNC onto an ITO plate with immobilized horseradish peroxidase enzyme with a sensitivity limit of 0.15 mA ng.mL-1. A PANI-Grp-GCE-based PNC sensor for the elimination of calcium antagonist lercanidipine in pharmaceutical formulations for medical purposes showed a detection limit in the range from 5 to 125 ng.mL-1. The same PANI-Grp-GCE-based PNC sensor showed the detection of nitazoxanide compound which was an added advantage. Electrochemical sensors based on PPy-based PNC are becoming popular these days due to their specific applications through their overoxidized form polypyrrole (PPyox). Fabrication of polypyrrole-graphene (PPyox/Grp) helped in the simultaneous detection of adenine and guanine through an electrodeposition method. PPy-Grp composite was electro-polymerized with pyrrole and graphene oxide (GO), followed by electrochemical reduction of GO composite. The electrochemical sensor’s significant improvement in the sensing of adenine and guanine is due to the specific structure of the nanocomposite. The adenine and guanine showed strong π-π interactions, and cationic selectivity.
The detection of Dopamine (DA) using PNCs was the Holy Grail in neurochemical studies ad it is a prominent neurotransmitter, which plays a role in neurological disorders such as Parkinson’s disease and schizophrenia. A poly(ionic liquid)-functionalized polypyrrole-graphene oxide (PIL-PPyGO)-based PNC electrochemical sensor was fabricated by the polymerization of 1-vinyl-3-ethylimidazolium bromide on N-vinyl imidazolium-modified PPy-GO films. The PILs helped in changing the surface charge which dispersibility of the PIL-PPyGO composite and helped in the detection of DA. Another sensor used for the detection of DA was a PNC-based poly (3,4-ethylene dioxythiophene)-graphene oxide (PEDOT-GO) fabricated by electrodeposition showed significant sensing capabilities. A onestep electrochemical redox synthesis process of PEDOT-Grp PNC film was done using simultaneous electrodeposition of PEDOT and electrochemical reduction of GO on a GCE with high detection sensing of the ascorbic acid molecules. In this sensor PEDOTGrp thin film PNC mediated the electron transfer between AO and electrode interface resulting in significant improvement in electrocatalytic activity and sensitivity of ascorbic acid molecules.
Jain et al. introduced the combination of Grp and a conducting PANI-Bi2O3 PNC, the synergic effect of which enhanced the performance of sensors used for the electrocatalytic oxidation of etodolac in pharmaceutical formulations.
In recent years, molecularly imprinted polymers (MIPs) with high selectivity, affinity, chemical stability, and easy preparation for the template molecule are a promising candidate for developing a new generation of recognition elements for sensors. A double-layered membrane-sensing interface was fabricated based on rGO-PANI nanocomposites and MIPs embedded with AuNPs for sensitive and selective detection of serotonin (5-hydroxytryptamine, 5-HT). The obtained sensor showed remarkable selectivity to serotonin against the interferences caused by ascorbic acid and other interferents with a good detection limit of 11.7 nmol.L-1.
Polymer Nanocomposites Based on Conductive Polymers and Carbon Nanotubes
PNCs based on conductive polymers helped in improving the sensing properties of the electrochemical sensors with enhanced selectivity and stability. A PPy-multiwalled carbon nanotube (MWCNT)-toluene sulfonic acid-based PNC was fabricated fr the detection of cholesterol with ITO-coated glass was the substrate for the sensor. The sensor showed high sensitivity and a fast response time of 9 s. Sodium dodecyl sulfate-doped PPyox) with carboxylic acid functionalized MWCNT-modified GCE were reported for the detection of the anticancer drug pemetrexed (PMX). The results showed that overoxidation of the PPy film conferred a negative charge density on the porous layer, which in turn enhances the adsorption of PMX. Xu et al. fabricated a carbon paste electrode (CPE) modified with a PEDOT-CNT nanocomposite. They used this electrode for the analysis of hydroquinone, DA, and nitrobenzene.
DNA Biosensors
PNCs are widely used these days in DNA biosensors. The medico biological field is growing leaping and bounds. In this era of 23 and me everything possible with DNA is bouncing through the boundaries of technology like DNA CRISPR editing, gene mapping. Biological agents for nefarious purposes and forensics. A basic DNA sensor work on a simple principle. You plant a DNA probe on a surface and this planted DNA chain hybridizes with its complementary pair. This hybridization technically called transduction can be detected optically and electrochemically. The electrochemical detection of transducers through electrochemical sensors leads us to DNA biosensors and are now extremely popular.
The schematic illustration of the most popular DNA biosensor based on polyaniline-gold nanoparticle-chitosan-graphene sheet (PANI-AuNP-Cts-GS) composite with a functional capture probe for the detection of BCR/ABL fusion gene in chronic myelogenous leukemia (CML). The capture probe used a hairpin structure and was dually labeled with a 5′-SH and a 3′-biotin. The biotin electrode probe was used for the detection of streptavidin-alkaline phosphatase (AP) enzyme which in turn cause an electrochemical signal caused by the catalytic reduction of 1-naphthyl phosphate to 1-naphthol picked up by Diffuse Pulse Voltammetry (DPV) with a detection range of 10-1000 pM. A DNA biosensor fabricated with PANI-AuNP PNC was used for the detection of Ag+. It works on the following principle: the electrochemical biosensor regenerates cysteine leading to the release of Ag+ from the cytosine to Ag+- Cytosine complex and reused again. The fabricated biosensor showed excellent selectivity with a good detection limit for silver ions. Another DNA electrochemical biosensor was developed using polyaniline nanofibers (PANI-nf) enrapturing AuNPs making (PANI-nf-AuNP), a PNC. Gold was used as the electrode for the detection of Staphylococcus aureus DNA from the PANI-nf-AuNPs sensor, where the detection concentration varied from 150×10- 12 to 1×10-6 mol.L-1. A DNA biosensor based on the PANI-Fe3O4- CNT PNC was manufactured for sensing Neisseria gonorrhoeae through a DNA probe. The fabricated biosensor showed sensing in the range from 1×10-19 to 1×10-6 M through DPV measurements. The most recent DNA biosensor based on PANI-AuNPs PNC detected the microRNA-16 using a streptavidin-AP conjugate to biotinylated target sequences through transduction with a detection limit of 0.1 nM. DNA biosensor made with polypyrrolepolyaniline-gold (PPy-PANI-Au) PNC responded to the target DNA through transduction, noncomplementary and single- and double-base-mismatched target DNA-chains with a detection limit between 1×10-6 and 1×10-13 M. Nanotube DNA biosensor based on polypyrrole and poly(3,4-ethylenedioxythiophene) (PPy-PEDOT) PNC, which was functionalized with Ag nanoparticles sensed DNA transduction through EIS detection. The DNA chains used for detection were thiol-capped on the modified sensor.
Electrochemical Immunosensors
PNCs are superior candidates for the fabrication of electrochemical Immunosensors, where the antibodies are the probes which form ionic complexes with the corresponding antigen pair with a specific target. Electrochemical Immunosensors are becoming widely used in clinical diagnosis applications, doping or impurities or detecting biological components in the food industry and detecting the biomolecules of environmental origin and impact. The most widely reported are discussed in this section and the immunosensors based on CNT-PPy-goat IgGs showed the interaction between the goat IgGs and its antigoat IgGs, which changes the charges at the sensor surface with changes in conductance level. The response time for the anti-goat IgG was 1 min.A label-free impedance immunosensor for human chorionic gonadotropin (hCG) detection using a PPy-PPa-hCGmodified carbon ink electrode was fabricated by the deposition of a PPy-pyrole-2-carboxylic acid copolymer. The hCG antibody was immobilized via the COOH groups of pyrrole-2-carboxylic acid, as a linker for covalent biomolecular immobilization. This immunosensor has a detection limit of the hCG antigen in the range of 100 pg mL-1 to 40 ng mL-1.
The next progress was PANI-AuNP hybrid electrochemical immunosensor with the gold electrode for the detection of prostate antigen (PSA). The immunosensor showed effective immobilization of anti-PSA with excellent sensing performance (1.4 μA M-1) and detection limit (0.6 pg mL-1) through effective electron transport. For the detection of aflatoxin B1, an electrochemical immunosensor based on a Grp-CP-AuNP-IL composite film was used. The fabrication was in a five-part series mode as Grp-CP-AuNP-IL pattern. Poly (DPB), 2,5-di-(2-thienyl)- 1-pyrrole-1-(p-benzoic acid) helped in the electrochemical stability as a CP. The covalent bonding through the antibody immobilization via carbonyl groups of the polymer helped in preventing the antibody loss, resulting in a detection limit of 1.0 fM. For ofloxacin detection, an immunosensor was fabricated based on a dual-amplification mechanism resulting from Au nanoclusters embedded in the pre-synthesized PPy film as the sensor platform and multienzyme antibody-functionalized gold nanorods as the label. The electrochemical response was in the range of 0.08 and 410 ng mL-1 with a low detection limit of 0.03 ng mL-1.
An electrochemical immunosensor was fabricated a PNC-based Grp-AuNP-poly-DPB-AuNP-IL for the detection of microcystin-LR through electrodeposition method on GCE. In this electrochemical sensor, the Grp-gold helped in the electron transfer of [Fe(CN)6]3-, and the poly 2,5-di-(2-thienyl)-1-pyrrole1-(p-benzoic acid)- gold nanoparticle (poly-DPB-AuNP) enhanced the electrical conduction and subsequent immobilization of the microcystin-LR antibody. A Grp-PANI-based PNC electrochemical sensor for the estradiol using horseradish peroxidase-graphene oxide-antibody (HRP-GO-Ab) was designed where carboxylated GO serves the antibody carrier property while the horseradish peroxidase helped in catalytic hydrogen reduction on the electrode. This estradiol immunosensor detected the estradiol in tap water and milk samples, with average recoveries of 97.25% and 96.6%, respectively.
The electrochemical immunosensors with electrochemiluminescence (ECL) sensing property was achieved through Quantum dots (QDs). This was based on graphene oxide nanosheet-polyaniline nanowire-CdSe quantum dot (GO-PANICdSe) which detected human interleukin-6 (IL-6). A ZnSe QDAzure I-AuNP-PEDOT-modified Pt electrode electrochemical immunosensor helped in the detection of alpha-fetoprotein (AFP) through electrochemiluminescence (ECL) sensing (detection limit ~1.1 fg mL-1). The sensing mechanism was as follows: ZnSe QDs immobilize the antibody, the nanoAu-PEDOT facilitated the electron transfer, and Azure I did the catalytic reduction of redox dye with two active amino groups.
PANI-AuNP-modified paper working electrodes (PANI-AuNP PWEs) were fabricated for the simultaneous determination of two tumor markers, carcinoembryonic antigen (CEA) and AFP, in real human serum samples. An electrochemical immunosensor for prolactin hormone was also constructed by immobilizing the antigen onto poly (pyrrolepropionic acid) CP and carbon nanotube (pPPA/CNT) hybrids deposited onto a GCE and labeled with AP enzyme with a reported detection limit of 104 ng mL-1. Polypyrrole (PPy)-based PNC was used to manufacture a bioelectrode for the detection of human C-reactive protein antigen (Ag-αCRP). This was made possible with the inorganic nanoparticles (3-mercaptopropionic acid (MPA)-capped Pt nanoparticles. First, the Ab-αCRP was immobilized covalently through specific carboxyl groups linkages through Pt(MPA)- NPs within the polypyrrole (PPy)-based PNC film by carbodiimide coupling. The resulted electrochemical immunosensor showed excellent fine probe orientation with a detection capacity of 10 ng mL-1-10 μg mL-1.
Another label-free impedimetric immunosensor based on multifunctional PNCs was based on (polypyrrole-pyrrolepropylic acid-reduced graphene oxide (PPy-PPa-rGO)) for the detection of mycotoxin aflatoxin B1. An enzyme-free electrochemical immunosensor modeled on the sandwich pattern was used for the detection of Carbohydrate Antigen72-4 (CA72-4). The sensing electrode was nanoporous gold (NPG) film and asymmetric multicomponent (AMNPs) nanoparticles based on PANI-Au was used as labels. The NPG helped in the creased immobilization of Ab1 on the electrode, while the PANI-Au AMNPs impacted on the reduction capability of the electrochemical immunosensor. The doping of AuNPs and PANI films with potassium ferricyanide over a gold electrode was used to detect a carcinoembryonic antigen (CEA). This PNC-based biocompatible electrochemical immunosensor showed excellent conductivity and redox electroactivity. The detection of CEA was analyzed through electrode response of [Fe (CN)6]3- as the redox mediator.
Results and Discussion
A schematic view of the detection of heavy metals in a hypothetical water environment (seas and oceans) by means of an optical polymeric nanosensor named »Pourya-Zar Sensor« Figure 1. Image of SEM analysis of Pourya-Zar{TiO2/Poly(ammba-dtz)} synthetic nanocomposite to detect heavy metals in water environments Figure 2. Image of TEM analysis of PouryaZar{TiO2/Poly(am-mba-dtz)} synthetic nanocomposite to detect heavy metals in water environments Figure 3. A view of the identification of different concentrations of heavy metal by optical polymer nanosensor Pourya-Zar{TiO2/Poly(am-mba-dtz)} in water environment Figure 4.
The Future of Sensor Technology
As technology develops, the use of sensors will continue to expand into every aspect of our lives. Engineers and scientists around the world will use sensors to enhance transportation systems, medical procedures, nanotechnology, mobile devices, virtual and augmented reality, and even Artificial Intelligence (AI).
References
- Fergusson JE (1990) The Heavy Elements: Chemistry, Environmental Impact and Health Effects. Oxford: Pergamon Press, United Kingdom.
- Duffus JH (2002) Heavy metals-a meaningless term? Pure Appl Chem 74(5): 793-807.
- Bradl H (2002) Heavy Metals in the Environment: Origin, Interaction and Remediation Volume 6. Academic Press, London.
- He ZL, Yang XE, Stoffella PJ (2005) Trace elements in agroecosystems and impacts on the environment. J Trace Elem Med Biol 19(2-3): 125-140.
- Goyer RA (2001) Toxic effects of metals. In: Klaassen CD, Cassarett and Doull’s Toxicology: The Basic Science of Poisons. McGraw-Hill Publisher, New York, USA, pp. 811-867.
- Herawati N, Suzuki S, Hayashi K, Rivai IF, Koyoma H (2000) Cadmium, copper and zinc levels in rice and soil of Japan, Indonesia and China by soil type. Bull Env Contam Toxicol 64(1): 33-39.
- Shallari S, Schwartz C, Hasko A, Morel JL (1998) Heavy metals in soils and plants of serpentine and industrial sites of Albania. Sci Total Environ 209(2-3): 133-142.
- Nriagu JO (1989) A global assessment of natural sources of atmospheric trace metals. Nature 338: 47-49.
- Arruti A, Fernández-Olmo I, Irabien A (2010) Evaluation of the contribution of local sources to trace metals levels in urban PM2.5 and PM10 in the Cantabria region (Northern Spain) J Environ Monit 12(7): 1451-1458.
- Sträter E, Westbeld A, Klemm O (2010) Pollution in coastal fog at Alto Patache, Northern Chile. Environ Sci Pollut Res Int 17(9): 1563-1573.
- Pacyna JM (1996) Monitoring and assessment of metal contaminants in the air. In: Chang LW, Magos L, Suzuli T, Toxicology of Metals. Boca Raton, CRC Press, Florida, USA, pp. 9-28.
- (1996) WHO/FAO/IAEA. World Health Organization. Trace Elements in Human Nutrition and Health, Geneva, Switzerland.
- Kabata-Pendia A (2001) Trace Elements in Soils and Plants. Boca Raton, CRC Press, Florida, U
- Hamelink JL, Landrum PF, Harold BL, William BH (1994) Bioavailability: Physical, Chemical, and Biological Interactions. Boca Raton, CRC Press Inc, Florida, USA.
- Verkleji JAS (1993) In: The effects of heavy metals stress on higher plants and their use as biomonitors In Plant as Bioindicators: Indicators of Heavy Metals in the Terrestrial Environment. Markert B, VCH, New York, USA, pp. 415-424.
- Stern BR (2010) Essentiality and toxicity in copper health risk assessment: overview, update and regulatory considerations. Toxicol Environ Health A 73(2): 114-127.
- Harvey LJ, McArdle HJ (2008) Biomarkers of copper status: a brief update. Br J Nutr 99(S3): S10-S13.
- (2002) Agency for Toxic Substances and Disease Registry (ATSDR) Toxicological Profile for Copper. GA: Centers for Disease Control, Atlanta, Georgia.
- Tchounwou P, Newsome C, Williams J, Glass K (2008) Copper-induced cytotoxicity and transcriptional activation of stress genes in human liver carcinoma cells. Metal Ions Biol Med 10: 285-290.
- Chang LW, Magos L, Suzuki T (1996) Toxicology of Metals. Boca Raton, CRC Press, Florida, USA.
- Wang S, Shi X (2001) Molecular mechanisms of metal toxicity and carcinogenesis. Mol Cell Biochem 222(1-2): 3-9.
- Beyersmann D, Hartwig A (2008) Carcinogenic metal compounds: recent insight into molecular and cellular mechanisms. Arch Toxicol 82(8): 493-512.
- Yedjou CG, Tchounwou PB (2006) Oxidative stress in human leukemia cells (HL-60), human liver carcinoma cells (HepG2) and human Jerkat-T cells exposed to arsenic trioxide. Metal Ions Biol Med 9: 298-303.
- Yedjou GC, Tchounwou PB (2007) In vitro cytotoxic and genotoxic effects of arsenic trioxide on human leukemia cells using the MTT and alkaline single cell gel electrophoresis (comet) assays. Mol Cell Biochem 301: 123-130.
- Tchounwou PB, Centeno JA, Patlolla AK (2004) Arsenic toxicity, mutagenesis and carcinogenesis - a health risk assessment and management approach. Mol Cell Biochem 255(1-2): 47-55.
- Tchounwou PB, Ishaque A, Schneider J (2001) Cytotoxicity and transcriptional activation of stress genes in human liver carcinoma cells (HepG2) exposed to cadmium chloride. Mol Cell Biochem 222(1-2): 21-28.
- Patlolla A, Barnes C, Field J, Hackett D, Tchounwou PB (2009) Potassium dichromate-induced cytotoxicity, genotoxicity and oxidative stress in human liver carcinoma (HepG2) cells. Int J Environ Res Public Health 6(2): 643-653.
- Patlolla A, Barnes C, Yedjou C, Velma V, Tchounwou PB (2009) Oxidative stress, DNA damage and antioxidant enzyme activity induced by hexavalent chromium in Sprague Dawley rats. Environ Toxicol 24(1): 66-73.
- Yedjou GC, Tchounwou PB (2008) N-acetyl-cysteine affords protection against lead-induced cytotoxicity and oxidative stress in human liver carcinoma (HepG2) cells. Intl J Environ Res Public Health 4(2): 132-137.
- Tchounwou PB, Yedjou CG, Foxx D, Ishaque A, Shen E (2004) Lead-induced cytotoxicity and transcriptional activation of stress genes in human liver carcinoma cells (HepG2) Mol Cell Biochem 255: 161-170.
- Sutton DJ, Tchounwou PB (2007) Mercury induces the externalization of phosphatidylserine in human proximal tubule (HK-2) cells. Intl J Environ Res Public Health 4(2): 138-144.
- Sutton D, Tchounwou PB, Ninashvili N, Shen E (2002) Mercury induces cytotoxicity, and transcriptionally activates stress genes in human liver carcinoma cells. Intl J Mol Sci 3(9): 965-984.
- (2000) Agency for Toxic Substances and Disease Registry (ATSDR) Toxicological Profile for Arsenic TP-92/09. Georgia: Center for Disease Control, Atlanta, Georgia.
- Tchounwou PB, Wilson B, Ishaque A (1999) Important considerations in the development of public health advisories for arsenic and arsenic-containing compounds in drinking water. Rev Environ Health 14(4): 211-229.
- Centeno JA, Tchounwou PB, Patlolla AK, Mullick FG, Murakat L, et al. (2005) Environmental pathology and health effects of arsenic poisoning: a critical review. In: Naidu R, Smith E, Smith J, Bhattacharya P, editors. Managing Arsenic In the Environment: From Soil to Human Health. Adelaide, CSIRO Publishing Corp, Australia.
- Rousselot P, Laboume S, Marolleau JP, Larghero T, Noguera ML, et al. (1999) Arsenic trioxide and melarsoprol induce apoptosis in plasma cell lines and in plasma cells from myeloma patients. Cancer Res 59(5): 1041-1048.
- (1978) National Research Council Canada (NRCC) Effects of Arsenic in the Environment. National Research Council of Canada, Canada, pp. 1-349.
- Morton WE, Dunnette DA (2001) Health effects of environmental arsenic. In: Nriagu JO, editor. Arsenic in the Environment Part II: Human Health and Ecosystem Effects. John Wiley & Sons, Inc, New York, pp. 17-34.
- (2001) National Research Council. Arsenic in Drinking Water.
- Tchounwou PB, Centeno JA (2008) Toxicologic pathology. In: Gad SC, editor. Handbook of Pre-Clinical Development. NY: John Wiley & Sons, New York, USA, pp. 551-580.
- Tchounwou PB, Patlolla AK, Centeno JA (2003) Carcinogenic and systemic health effects associated with arsenic exposure-a critical review. Toxicol Pathol 31(6): 575-588.
- Tchounwou PB, Wilson BA, Abdelgnani AA, Ishaque AB, Patlolla AK (2002) Differential cytotoxicity and gene expression in human liver carcinoma (HepG2) cells exposed to arsenic trioxide and monosodium acid methanearsonate (MSMA) Intl J Mol Sci 3(11): 1117-1132.
- Yedjou GC, Moore P, Tchounwou PB (2006) Dose and time dependent response of human leukemia (HL-60) cells to arsenic trioxide. Intl J Environ Res Public Health 3(2): 136-140.
- Chappell W, Beck B, Brown K, North D, Thornton I (1997) Inorganic arsenic: A need and an opportunity to improve risk assessment. Environ Health Perspect 105(10): 1060-1067.
- Centeno JA, Gray MA, Mullick FG, Tchounwou PB, Tseng C (2005) Arsenic in drinking water and health issues. In: Moore TA, Black A, Centeno JA, Harding JS, Trumm DA, editors. Metal Contaminants in New Zealand. Resolutionz Press, New Zealand, pp. 195-219.
- Abernathy CO, Liu YP, Longfellow D, Aposhian HV, Beck B, et al. (1999) Arsenic: health effects, mechanisms of actions and research issues. Environ Health Perspect 107(7): 593-597.
- Hughes MF (2002) Arsenic toxicity and potential mechanisms of action. Toxicol Lett 133(1): 1-16.
- Wang Z, Rossman TG (1996) In: The Toxicology of Metals. Cheng LW, editor. Vol. 1. Boca Raton, CRC Press, Florida, USA, pp. 221–243.
- Belton JC, Benson NC, Hanna ML, Taylor RT (1985) Growth inhibition and cytotoxic effects of three arsenic compounds on cultured Chinese hamster ovary cells. J Environ Sci Health 20A: 37-72.
- Li JH, Rossman TC (1989) Inhibition of DNA ligase activity by arsenite: A possible mechanism of its comutagenesis. Mol Toxicol 2(1): 1-9.
- Jha AN, Noditi M, Nilsson R, Natarajan AT (1992) Genotoxic effects of sodium arsenite on human cells. Mutat Res 284(2): 215-221.
- Hartmann A, Speit G (1994) Comparative investigations of the genotoxic effects of metals in the single cell gel assay and the sister-chromatid exchange test. Environ Mol Mutagen 23(4): 299-305.
- Patlolla A, Tchounwou PB (2005) Cytogenetic evaluation of arsenic trioxide toxicity in Sprague-Dawley rats. Mut Res – Gen Tox Environ Mutagen 587(1–2): 126-133.
- Basu A, Mahata J, Gupta S, Giri AK (2001) Genetic toxicology of a paradoxical human carcinogen, arsenic: a review. Mutat Res 488(2): 171-194.
- Landolph JR (1989) Molecular and cellular mechanisms of transformation of C3H/10T1/2C18 and diploid human fibroblasts by unique carcinogenic, non- mutagenic metal compounds. A review. Biol Trace Elem Res 21: 459-467.
- Takahashi M, Barrett JC, Tsutsui T (2002) Transformation by inorganic arsenic compounds of normal Syrian hamster embryo cells into a neoplastic state in which they become anchorage-independent and cause tumors in newborn hamsters. Int J Cancer 99(5): 629-634.
- Anderson D, Yu TW, Phillips BJ, Schemezer P (1994) The effect of various antioxidants and other modifying agents on oxygen-radical-generated DNA damage in human lymphocytes in the Comet assay. Mutation Res 307(1): 261-271.
- Saleha Banu B, Danadevi K, Kaiser Jamil, Ahuja YR, Visweswara Rao K, et al. (2001) In vivo genotoxic effect of arsenic trioxide in mice using comet assay. Toxicol 162(3): 171-177.
- Hartmann A, Peit G (1994) Comparative investigations of the genotoxic effects of metals in the single cell gel assay and the sister chromatid exchange test. Environ Mol Mutagen 23(4): 299-305.
- Barrett JC, Lamb PW, Wang TC, Lee TC (1989) Mechanisms of arsenic-induced cell transformation. Biol. Trace Ele Res 21: 421-429.
- Tchounwou PB, Yedjou CG, Dorsey WC (2003) Arsenic trioxide - induced transcriptional activation and expression of stress genes in human liver carcinoma cells (HepG2) Cell Mol Biol 49(7): 1071-1079.
- Zhao CQ, Young MR, Diwan BA, Coogan TP, Waalkes MP (1997) Association of arsenic-induced malignant transformation with DNA hypomethylation and aberrant gene expression. Proc Natl Acad Sci USA 94(20): 10907-10912.
- Liu Y, Guyton KZ, Gorospe M, Xu Q, Lee JC, et al. (1996) Differential activation of ERK, JNK/SAPK and P38/CSBP/RK map kinase family members during the cellular response to arsenite. Free Rad Biol Med 21: 771-781.
- Ludwig S, Hoffmeyer A, Goebeler M, Kilian K, Hafner H, et al. (1998) The stress inducer arsenite activates mitogen-activated protein kinases extracellular signal-regulated kinases 1 and 2 via a MAPK kinase 6/p38- dependent pathway. J Biol Chem 273: 1917-1922.
- Trouba KJ, Wauson EM, Vorce RL (2000) Sodium arsenite-induced dysregulation of proteins involved in proliferative signaling. Toxicol Appl Pharmacol 164(2): 161-170.
- Vogt BL, Rossman TG (2001) Effects of arsenite on p53, p21 and cyclin D expression in normal human fibroblasts- a possible mechanism for arsenite’s comutagenicity. Mutat Res 478(1–2): 159-168.
- Chen NY, Ma WY, Huang C, Ding M, Dong Z (2000) Activation of PKC is required for arsenite-induced signal transduction. J Environ Pathol Toxicol Oncol 19(3): 297-306.
- Porter AC, Fanger GR, Vaillancourt RR (1999) Signal tansduction pathways regulated by arsenate and arsenite. Oncogene 18(54): 7794-7802.
- Soignet SL, Frankel SR, Douer D, Tallman MS, Kantarjian H, et al. (2001) United States multicenter study of arsenic trioxide in relapsed acute promyelocytic leukemia. J Clin Oncol 19(18): 3852-3860.
- Murgo AJ (2001) Clinical trials of arsenic trioxide in hematologic and solid tumors: overview of the National Cancer Institute Cooperative Research and Development Studies. Oncologist 6(2): 22-28.
- Puccetti ES, Guller S, Orleth A, Bruggenolte N, Hoelzer D, et al. (2000) BCR-ABL mediates arsenic trioxide-induced apoptosis independently of its aberrant kinase activity. Cancer Res 60(13): 3409-3413.
- Seol JG, Park WH, Kim ES, Jung CW, Hyun JM, et al. (1999) Effect of arsenic trioxide on cell cycle arrest in head and neck cancer cell-line PCI-1. Biochem Biophys Res Commun 265(2): 400-404.
- Alemany M, Levin J (2000) The effects of arsenic trioxide on human Megakaryocytic leukemia cell lines with a comparison of its effects on other cell lineages. Leukemia Lymphoma 38(1–2): 153-163.
- Deaglio S, Canella D, Baj G, Arnulfo A, Waxman S, et al. (2001) Evidence of an immunologic mechanism behind the therapeutic effects of arsenic trioxide on myeloma cells. Leuk Res 25(3): 237-239.
- Tully DB, Collins BJ, Overstreet JD, Smith CS, Dinse GE, et al. (2000) Effects of arsenic, cadmium, chromium and lead on gene expression regulated by a battery of 13 different promoters in recombinant HepG2 cells. Toxicol Appl Pharmacol 168(2): 79-90.
- Lu T, Liu J, LeCluyse EL, Zhou YS, Cheng ML, et al. (2001) Application of cDNA microarray to the study of arsenic-induced liver diseases in the population of Guizhou, China. Toxicol Sci 59(1): 185-192.
- Harris CC (1991) Chemical and physical carcinogenesis: advances and perspectives. Cancer Res 51: 5023s-5044s.
- Graham-Evans B, Colhy HHP, Yu H, Tchounwou PB (2004) Arsenic-induced genotoxic and cytotoxic effects in human keratinocytes, melanocytes, and dendritic cells. Intl J Environ Res Public Health 1(2): 83-89.
- Stevens JJ, Graham B, Walker AM, Tchounwou PB, Rogers C (2010) The effects of arsenic trioxide on DNA synthesis and genotoxicity in human colon cancer cells. Intl J Environ Res Public Health 7(5): 2018-2032.
- Walker AM, Stevens JJ, Ndebele K, Tchounwou PB (2010) Arsenic trioxide modulates DNA synthesis and apoptosis in lung carcinoma cells. Intl J Environ Res Public Health 7(5): 1996-2007.
- Yedjou CG, Tchounwou PB (2009) Modulation of p53, c-fos, RARE, cyclin A and cyclin D1 expression in human leukemia (HL-60) cells exposed to arsenic trioxide. Mol Cell Biochem 331(1-2): 207-214.
- Yedjou C, Sutton LM, Tchounwou PB (2008) Genotoxic mechanisms of arsenic trioxide effect in human Jurkat T-lymphoma cells. Metal Ions Biol Med 10: 495-499.
- Brown E, Yedjou C, Tchounwou PB (2008) Cytotoxicty and oxidative stress in human liver carcinoma cells exposed to arsenic trioxide. Metal Ions Biol Med 10: 583-587.
- Yedjou CG, Thuisseu L, Tchounwou C, Gomes M, Howard C, et al. (2009) Ascorbic acid potentiation of arsenic trioxide anticancer activity against acute promyelocytic leukemia. Arch Drug Inf 2(4): 59-65.
- Yedjou C, Rogers C, Brown E, Tchounwou P (2008) Differential effect of ascorbic acid and n-acetyl-cysteine on arsenic trioxide - mediated oxidative stress in human leukemia (HL-60) cells. J Biochem Mol Tox 22(2): 85-92.
- Yedjou GC, Moore P, Tchounwou PB (2006) Dose- and time-dependent response of human leukemia (HL-60) cells to arsenic tic trioxide treatment. Intl J Environ Res Public Health 3(2): 136-140.
- Miller WH, Schipper HM, Lee JS, Singer J, Waxman S (2002) Mechanisms of action of arsenic trioxide - review. Cancer Res 62(14): 3893-3903.
- Gesamp (1987) IMO/FAO/UNESCO/WMO/WHO/IAEA/UN/UNEP Joint Group of Experts on the Scientific Aspects of Marine Pollution: Report of the seventeenth session. World Health Organization, Geneva, Switzerland.
- (1988) Wilson DN Association Cadmium. Cadmium - market trends and influences; Cadmium 87 Proceedings of the 6th International Cadmium Conference, London, pp. 9-16.
- (2009) U.S Environmental Protection Agency (EPA), Cadmium Compounds, USA.
- (1993) International Agency for Research on Cancer (IARC) Monographs - Cadmium. Lyon, France.
- Paschal DC, Burt V, Caudill SP, Gunter EW, Pirkle JL, et al. (2000) Exposure of the U.S. population aged 6 years and older to cadmium: 1988–1994. Arch Environ Contam Toxicol 38(3): 377-383.
- (2008) Agency for Toxic Substances and Disease Registry (ATSDR) Draft Toxicological Profile for Cadmium. Atlanta, Georgia.
- Satarug S, Baker JR, Urbenjapol S, Haswell-Elkins M, Reilly PE, et al. (2003) A global perspective on cadmium pollution and toxicity in non-occupationally exposed population. Toxicol Lett 137(1-2): 65-83.
- Davison AG, Fayers PM, Taylor AJ, Venables KM, Darbyshire J, et al. (1988) Cadmium fume inhalation and emphysema. Lancet 1(8587): 663-667.
- Mascagni P, Consonni D, Bregante G, Chiappino G, Toffoletto F (2003) Olfactory function in workers exposed to moderate airborne cadmium levels. Neurotoxicol 24(4-5): 717-724.






























