A Review Study on a Polymer Optical Nano Sensor Named »Pourya-Zar Sensor« for Detection of ((As)) Heavy Metal Ions in Aquatic Environments
Pourya Zarshenas1, Mehran Amir Beygi2, Elaheh Haghbin2 and Mohammad Amin Abdoos3
1Department of Inorganic Chemistry, Faculty of Chemistry & Petroleum Sciences, Shahid Beheshti University (SBU), Tehran, Iran
2Department of Polymer Chemistry, Faculty of Chemistry & Petroleum Sciences, Shahid Beheshti University (SBU), Tehran, Iran
3Medicinal Plants and Drugs Research Institute, Shahid Beheshti University (SBU), Tehran, Iran
4Medicinal Plants and Drugs Research Institute, Shahid Beheshti University (SBU), Tehran, Iran
Submission: March 09, 2023; Published: May 01, 2023
*Corresponding author: Pourya Zarshenas, Department of Inorganic Chemistry, Faculty of Chemistry & Petroleum Sciences, Shahid Beheshti University (SBU), Tehran, Iran
Pourya Z, Mehran Amir B, Elaheh H, Mohammad Amin A.A Review Study on a Polymer Optical Nano Sensor Named »Pourya-Zar Sensor« for Detection of ((As)) Heavy Metal Ions in Aquatic Environments. Recent Adv Petrochem Sci. 2023; 7(4): 555719.DOI: 10.19080/RAPSCI.2023.07.555719
Abstract
Environment can be referred to as the surroundings within which humans exist. These are made up of: the land, the water, and the atmosphere of the earth; microorganisms, plant, and animal life; any part or combination of the first two items on this list and the interrelationships among and between them and the physical, chemical, aesthetic and cultural properties and conditions of the foregoing that influence human health and well-being. It is also characterized by a number of spheres that influence its behavior and intrinsic value. The most important sphere of the environment is the biosphere because it harbors living organisms. This is the sphere where you find living organisms (plants and animals) interacting with each and their nonliving environment (soil, air and water). In the late centuries, industrialization and globalization have impaired pristine environments and their ability to foster life. This has introduced components that compromise the holistic functioning of the environment and its intrinsic values [1]. An environment can be polluted or contaminated. Pollution differs from contamination; however, contaminants can be pollutants, and pose detrimental impact on the environment. From literature, pollution is defined as the introduction by man, directly or indirectly, of substances or energy into the environment resulting in such deleterious effects as harm to living resources, hazards to human health, hindrance to environmental activities and impairment of quality for use of the environment and reduction of amenities. Contamination on the other hand is the presence of elevated concentrations of substances in the environment above the natural background level for the area and for the organism. Environmental pollution can be referred to undesirable and unwanted changes in physical, chemical, and biological characteristics of air, water and soil which are harmful for living organisms-both animal and plants. Pollution can take the form of chemical substances or energy, such as noise, heat, or light [2]. Pollutants, the elements of pollution, can either be foreign substances/energies or naturally occurring contaminants. Environmental pollutants continue to be a world concern and one of the great challenges faced by the global society. Pollutants can be naturally occurring compounds or foreign matter which when in contact with the environment cause adverse changes. There are different types of pollutants, namely inorganic, organic, and biological. Irrespective of pollutants falling under different categories, they all receive considerable attention due to the impacts they introduce to the environment. The relationship between environmental pollution and world population has become an inarguable directly proportional relationship as it can be seen that the amount of potentially toxic substances released into the environment is increasing with the alarming growth in global population. This issue has led to pollution being a significant problem facing the environment. Industrial, agricultural, and domestic wastes contribute to environmental pollution, which causes adverse harm to human and animal health. From such sources, inorganic pollutants are released. Inorganic pollutants are usually substances of mineral origin, with metals, salts and minerals being examples [2]. Studies have reported inorganic pollutants as material found naturally but have been altered by human production to increase their number in the environment. Inorganic substances enter the environment through different anthropogenic activities such as mine drainage, smelting, metallurgical and chemical processes, as well as natural processes. These pollutants are toxic due to the accumulation in the food chains [3].Organic pollution can be briefly defined as biodegradable contaminants in an environment. These sources of pollution are naturally found and caused by the environment, but anthropogenic activity has also been contributing to their intensive production to meet human needs. Some of the common organic pollutants which have been noted to be of special concern are human waste, food waste, polychlorinated biphenyls (PCBs), polybrominated diphenyl ethers (PBDEs), polycyclic aromatic hydrocarbons (PAHs), pesticides, petroleum, and organochlorine pesticides (OCPs) [4]. The definition of a sensor does not precisely define what physical elements constitute the sensor. For example, what portion of a thermocouple is the sensor? Is it solely the bimetallic junction? Does it include the wires used for transmission purposes? Does it include any packaging or signal processing? On the basis of information in the current technical literature, the committee chose to adopt the following definitions:
Sensor element: The fundamental transduction mechanism (e.g., a material) that converts one form of energy into another. Some sensors may incorporate more than one sensor element (e.g., a compound sensor).
Sensor: A sensor element including its physical packaging and external connections (e.g., electrical, or optical). Sensor system: A sensor and its assorted signal processing hardware (analog or digital) with the processing either in or on the same package or discrete from the sensor itself. In order to describe and characterize the performance of a sensor, a large and specific vocabulary is required. Several excellent references, which provide a basic review of transducer characteristics, are cited in the bibliography. Lion (1969) introduced a classification of principles according to the form of energy in which sensor signals were received and generated, which yielded a matrix of effects.
In this article, we intend to investigate water pollution by heavy metals. Finally, we will review the methods for characterizing heavy metals by chemical sensors. So, Ladies and gentlemen! Welcome to the age of pollution
Keywords: Heavy Metals; Water Pollution; Nano Sensors; Polymer Sensors; Optical Sensors; Water Environments; As; Arsenic
Abbreviations: PCBs: Polychlorinated Biphenyls (PCBs); PBDEs: Polybrominated Diphenyl Ethers; PAHs: Polycyclic Aromatic Hydrocarbons; OCPs: Organochlorine Pesticides
Introduction
Heavy metals are defined as metallic elements that have a relatively high density compared to water [1]. With the assumption that heaviness and toxicity are inter-related, heavy metals also include metalloids, such as arsenic, that are able to induce toxicity at low level of exposure [2]. In recent years, there has been an increasing ecological and global public health concern associated with environmental contamination by these metals. Also, human exposure has risen dramatically as a result of an exponential increase of their use in several industrial, agricultural, domestic, and technological applications [3]. Reported sources of heavy metals in the environment include geogenic, industrial, agricultural, pharmaceutical, domestic effluents, and atmospheric sources [4].
Environmental pollution is very prominent in point source areas such as mining, foundries and smelters, and other metalbased industrial operations. Although heavy metals are naturally occurring elements that are found throughout the earth’s crust, most environmental contamination and human exposure result from anthropogenic activities such as mining and smelting operations, industrial production and use, and domestic and agricultural use of metals and metal-containing compounds [4- 7]. Environmental contamination can also occur through metal corrosion, atmospheric deposition, soil erosion of metal ions and leaching of heavy metals, sediment re-suspension and metal evaporation from water resources to soil and ground water [8]. Natural phenomena such as weathering and volcanic eruptions have also been reported to significantly contribute to heavy metal pollution. Industrial sources include metal processing in refineries, coal burning in power plants, petroleum combustion, nuclear power stations and high-tension lines, plastics, textiles, microelectronics, wood preservation and paper processing plants [9-11].
It has been reported that metals such as cobalt (Co), copper (Cu), chromium (Cr), iron (Fe), magnesium (Mg), manganese (Mn), molybdenum (Mo), nickel (Ni), selenium (Se) and zinc (Zn) are essential nutrients that are required for various biochemical and physiological functions [12]. Inadequate supply of these micro-nutrients results in a variety of deficiency diseases or syndromes [12]. Heavy metals are also considered as trace elements because of their presence in trace concentrations (ppb range to less than 10ppm) in various environmental matrices [13]. Their bioavailability is influenced by physical factors such as temperature, phase association, adsorption and sequestration. It is also affected by chemical factors that influence speciation at thermodynamic equilibrium, complexation kinetics, lipid solubility and octanol/water partition coefficients [14]. Biological factors such as species characteristics, trophic interactions, and biochemical/physiological adaptation, also play an important role [15].
The essential heavy metals exert biochemical and physiological functions in plants and animals. They are important constituents of several key enzymes and play important roles in various oxidation-reduction reactions [12]. Copper for example serves as an essential co-factor for several oxidative stress-related enzymes including catalase, superoxide dismutase, peroxidase, cytochrome c oxidases, ferroxidases, monoamine oxidase, and dopamine β-monooxygenase [16-18]. Hence, it is an essential nutrient that is incorporated into a number of metalloenzymes involved in hemoglobin formation, carbohydrate metabolism, catecholamine biosynthesis, and cross-linking of collagen, elastin, and hair keratin.
The ability of copper to cycle between an oxidized state, Cu(II), and reduced state, Cu(I), is used by cuproenzymes involved in redox reactions [16-18]. However, it is this property of copper that also makes it potentially toxic because the transitions between Cu(II) and Cu(I) can result in the generation of superoxide and hydroxyl radicals [16-19]. Also, excessive exposure to copper has been linked to cellular damage leading to Wilson disease in humans [18,19]. Similar to copper, several other essential elements are required for biologic functioning, however, an excess amount of such metals produces cellular and tissue damage leading to a variety of adverse effects and human diseases. For some including chromium and copper, there is a very narrow range of concentrations between beneficial and toxic effects [19,20]. Other metals such as aluminum (Al), antinomy (Sb), arsenic (As), barium (Ba), beryllium (Be), bismuth (Bi), cadmium (Cd), gallium (Ga), germanium (Ge), gold (Au), indium (In), lead (Pb), lithium (Li), mercury (Hg), nickel (Ni), platinum (Pt), silver (Ag), strontium (Sr), tellurium (Te), thallium (Tl), tin (Sn), titanium (Ti), vanadium (V) and uranium (U) have no established biological functions and are considered as non-essential metals [20].
In biological systems, heavy metals have been reported to affect cellular organelles and components such as cell membrane, mitochondrial, lysosome, endoplasmic reticulum, nuclei, and some enzymes involved in metabolism, detoxification, and damage repair [21]. Metal ions have been found to interact with cell components such as DNA and nuclear proteins, causing DNA damage and conformational changes that may lead to cell cycle modulation, carcinogenesis, or apoptosis [20-22]. Several studies from our laboratory have demonstrated that reactive oxygen species (ROS) production and oxidative stress play a key role in the toxicity and carcinogenicity of metals such as arsenic [23- 25], cadmium [26], chromium [27,28], lead [29,30], and mercury [31,32].
Because of their high degree of toxicity, these five elements rank among the priority metals that are of great public health significance. They are all systemic toxicants that are known to induce multiple organ damage, even at lower levels of exposure. According to the United States Environmental Protection Agency (U.S. EPA), and the International Agency for Research on Cancer (IARC), these metals are also classified as either “known” or “probable” human carcinogens based on epidemiological and experimental studies showing an association between exposure and cancer incidence in humans and animals [33].
Heavy metal-induced toxicity and carcinogenicity involves many mechanistic aspects, some of which are not clearly elucidated or understood. However, each metal is known to have unique features and physic-chemical properties that confer to its specific toxicological mechanisms of action. This review provides an analysis of the environmental occurrence, production and use, potential for human exposure, and molecular mechanisms of toxicity, genotoxicity, and carcinogenicity of arsenic, cadmium, chromium, lead, and mercury [34].
Heavy metals are defined as metallic elements that have a relatively high density compared to water. Heavy metals like Chromium (Cr), Cadmium (Cd), Mercury (Hg), Lead (Pb), Nickel (Ni), and Thallium (Tl) are potentially hazardous in combined or elemental forms. Heavy metals are highly soluble in the aquatic environments and therefore they can be absorbed easily by living organisms. Previous studies have detected heavy metals in the gills, liver, and muscles tissues of various species of fish in contaminated marine ecosystems. Once the heavy metals enter the food chain, they may end up accumulating in the human body. Since most heavy metals are widely applied in industries, exposure and contamination of the workers and residents near such facilities is likely to occur. Heavy metals above allowable limits will often lead to disadvantageous effects in humans, other organisms and the environment at large. Allowable safe limits of heavy metals in food samples are associated with low health risks in humans [35-40].
The level of toxicity of some selected metals for humans follows the sequence Co < Al < Cr < Pb < Ni < Zn < Cu < Cd < Hg. The harmful effects of heavy metals in humans depend on their dosage, rate of emission and period of exposure. Some of the heavy metals that have received more attention for the last decades are Hg, Cd, and Pb7. The adverse health effects that are associated with Hg and mercuric compounds in humans includes possible carcinogens; damage of the brain, lungs, and kidneys; damage of developing fetuses; high blood pressure or heart rate; vomiting and diarrhea; skin rashes and eye irritation8. The US EPA’s regulatory limit of Hg in drinking water is 2 parts per billion (ppb). The WHO recommended safe limits of Hg in wastewater and soils for agriculture are 0.0019 and 0.05 ppm respectively [41-45].
Chronic toxicity of Cd in children includes damages of respiratory, renal, skeletal, and cardiovascular systems as well as development of cancers of the lungs, kidneys, prostate, and stomach. Exposure of people to Cd includes eating contaminated food, smoking cigarettes, and working in cadmium-contaminated workplaces and in primary metal industries. A study carried out in Iran reported that the level of Cd was higher than the maximum permissible limit (MPL) in canned fish samples, and this was due to discharge of heavy metal rich pollutants into aquatic ecosystems. The US EPA’s regulatory limit of Cd in drinking water is 5 ppb or 0.005 parts per million (ppm). The WHO recommended safe limits of Cd in both wastewater and soils for agriculture is 0.003 ppm [46-48].
Exposure to Pb can occur through inhalation of contaminated dust particles and aerosols or by ingesting contaminated food and water. Lead poisoning in humans damages the kidneys, liver, heart, brain, skeleton, and the nervous system. Initial symptoms of poisoning associated with exposure to Lead may include headache, dullness, memory loss and being irritable. Lead poisoning may cause disturbance of hemoglobin synthesis and anemia. In children, chronic exposure to low levels of Lead may decrease their intelligence capacity. According to the International Agency for Research on Cancer (IARC), Lead is a possible carcinogenic substance in humans. The regulatory limit of Pb in drinking water according to US EPA is 15 ppb. The WHO recommended safe limits of Pb in wastewater and soils used for agriculture are 0.01 and 0.1 ppm respectively [49,50].
Chromium is widely used in metallurgy, electroplating, and in the manufacturing of paints, pigments, preservatives, pulp, and paper among others. The introduction of Chromium into the environment is often through sewage and fertilizers. Hexavalent Chromium compounds including chromates of Ca, Zn, Sr, and Pb are highly soluble in water, toxic and carcinogenic. Furthermore, compounds of Chromium have been associated with slow healing ulcers. It has also been reported that Chromate compounds can destroy DNA in cells. The WHO recommended safe limits for Cr (hexavalent) in wastewater and soils used for agriculture are 0.05 and 0.1 ppm respectively [51].
Thallium is a soft, tasteless, odorless white, blue metal in its pure form, and it oxidizes to thallium oxide when exposed to air. Sources of Tl include electronics, optical glasses, semi-conductors, mercury lamp among others. Humans become exposed to Tl through ingestion, inhalation, and dermal exposure. Thallium is highly toxic with a lethal dose of 6 to 40 mg/kg. Thallium poisoning is associated with anorexia, vomiting, gastrointestinal bleeding, abdominal pain, polyneuropathy, alopecia, renal failure, skin erythema, seizures, emotional changes, autonomic dysfunction, cardio toxicity, and coma among others. In China, the recommended safe limit of thallium in drinking water is 0.0001 ppm. The WHO recommended safe limits for Tl in both wastewater and agricultural soils were not given in the literature accessed [52-55].
Nickel is a silver- colored metal used in making stainless steel, electronics, and coins among other uses. Globally, the release of Ni to the environment is estimated to vary from 150,000 to 180,000 metric tons per year. Exposure of Ni to humans is through food, air, and water. Previous study has shown that ingestion of dust contaminated with Nickel was the main exposure pathway of the heavy metal by local residents when compared to inhalation and dermal pathways. Upon exposure to Nickel, an individual may show increased levels of Ni in his or her tissues and urine. The disadvantageous effects of nickel on human health may include dermatitis, allergy, organ diseases, and cancer of the respiratory system. The recommended safe limits by WHO for Ni in wastewater and agricultural soils are 0.02 and 0.05 ppm respectively [56-59].
Wastewater from factories may contain heavy metals which with time accumulate in the soil deposits along wastewater channels as well as in organisms that inhabit such channels. Exposure of humans to contaminated wastewater is often possible especially in urban highly populated areas or where the wastewater is reused for agricultural activities. Previous studies however have shown that effective reuse of wastewater is a major challenge in many countries of the world.
The current study was designed to establish the concentration of Hg, Pb, Cr, Tl, Cd and Ni in samples of wastewater and soil obtained from open wastewater channels in selected sites in Nairobi’s industrial area, Kenya. There are many informal urban settlements/villages that neighbor Nairobi industrial area and some of the wastewater channels drain into a tributary of Nairobi River which flows across these villages. Clogged wastewater channels enhance overflow of the wastewater into the surrounding areas through surface runoff when it rains. Therefore, the current study aims to highlight the potential health risks that may ensue when humans, livestock and crops become directly or indirectly exposed to the heavy metal contaminated wastewater and soils from the open channels in the study area. It is envisaged that the results obtained from the study will inform and justify on the need to adopt good wastewater management including prioritizing on effective wastewater treatment and reuse in Kenyan major urban areas. Previous studies elsewhere have shown that the degree of wastewater treatment determines the applicability of the reclaimed water [60-65].
Physico-Chemical Parameters of Water Samples
Both physical and chemical parameters of the water samples were measured and recorded at the collection site. These included temperature, pH, electrical conductivity and turbidity. Electronic devices capable of recording the parameters at the same time (HANNA Instruments, H1991300, and Romania) were used.
Preparation of Soil Samples for Heavy Metal Analysis
In the laboratory, the wet soil samples from each sampling site were spread on brown papers to dry under room temperature. They were then ground, sieved, weighed, and packaged in small brown envelopes and labeled. The labels included site, date of collection, and weight in grams.
Standard Limits of Heavy Metals in Wastewater, Drinking Water, Soils & Vegetables
The standard limits of Hg, Pb, Cd, Cr, Tl and Ni in drinking water, wastewater (effluents), agricultural soils, and vegetables, as recommended by WHO; China (both Chinese Ministry of Health (CMH) & The National Standard of China); Kenya (both National Environment Management Authority (NEMA) & Kenya Bureau of Standards (KEBS); USA EPA; India (CPCB) and World Bank (WB) were retrieved from the available literature. The determined level of heavy metals in the field samples were then compared to these standard limits in order to establish whether the level of pollution in the open channels was above the locally and internationally accepted standards in addition to making reliable conclusions.
Analysis of Heavy Metals in Samples of Water and Soil
The analyses were carried out at Mineral Laboratories, Bureau Veritas Commodities Ltd, Vancouver, Canada. The protocols included aqua regia digestion ultra-trace inductively coupled plasma mass spectroscopy (ICP-MS) for soil samples; and ICP-MS (solutions >0.1% Total Dissolved Solids (TDS) for water samples as described by the American Herbal Products Association (AHPA). Briefly, the digest solution was nebulized, and sample aerosols transferred to argon plasma. The high temperature plasma then produced ions, which were then introduced into the mass spectrometer, which then sorted out the ions according to their mass-to-charge ration. The ions were then quantified with an electron multiplier detector. Certificates of analysis and quality control reports for all the samples analyzed were awarded by the Bureau Veritas, Canada.
Data analysis
Statistical Package for Social Sciences (SPSS) for Windows (Version 20) at 5% level of significance was used. Descriptive statistics involved computing the mean, standard error (SE), and standard deviation (SD) for the different variables measured in water and soil samples. One-way analysis of variance (ANOVA) was used to establish the significant differences within and between groups. Tukey’s and Games-Howell Post hoc tests were carried out to establish the pairs of variables that were significantly different. Correlation analysis was carried out to establish the nature of relationship, level of significance between concentrations of heavy metals in different samples. Pairwise correlations of coefficients for the levels of selected heavy metals in wastewater and soils were computed.
Physico-Chemical Parameters of Wastewater Samples
The conductivity of wastewater obtained from Chief’s Camp (B-2), Railways (sites C & D) and Sinai (G) was high and ranged between 770.00 ± 11.55 to 1134.33 ± 180.39 μS/cm. The conductivity levels of wastewater samples from the remaining sites ranged from 366.33 ± 33.79 to 556.00 ± 27.62 μS/cm. Increased conductivity corresponded to increased turbidity of wastewater and vice versa. Therefore, high turbidity of wastewater was similarly recorded at Railways upper section (D), Sinai (G) and Railways lower section (C) sites and it measured 535.33 ± 41.60, 544.67 ± 21.17 and 562.00 ± 84.33 ppm respectively. The chief’s camp (B-1) had the lowest turbidity at 160.33 ± 0.88 ppm. The pH of wastewater samples ranged from 7.28 to 8.78 while the temperature ranged from 16.75 to 26.05 °C.
Standard Limits of Heavy Metals in Drinking Water, Wastewater, Soils, and Vegetables
The recommended limits of selected heavy metals according to WHO, Kenya (NEMA & KEBS), China (CMH & The National Standard of China), US EPA, India (CPCB) and World Bank were retrieved and recorded from the literature accessed. The standard levels for Tl were not given for wastewater, soils, and vegetables in the literature accessed. However, allowable level of Tl in drinking water, recommended by Chinese Ministry of Health and US EPA was available. The Tl limit level in surface water as recommended by US EPA was also recorded. Standards of heavy metals in agricultural soils in Kenya were missing in the literature accessed.
Levels of Heavy Metals in Wastewater and Tap Water
The wastewater samples had high Pb levels followed by Cr and the lowest was Tl. The mean concentration of heavy metals analyzed in wastewater samples, in an ascending order was Tl < Cd < Hg < Ni < Cr < Pb. This trend applied to all samples of wastewater that were acidified immediately after collection. The mean concentration of chromium was the highest at 24.2 ppb followed by nickel at 2.90 ppb in the wastewater samples that were not digested by acids immediately after collection hence an ascending order of Tl < Cd < Hg < Pb < Ni < Cr.
The mean concentration of Hg in the wastewater samples was <0.0001 ppm and this was lower than the standards set by WHO, World Bank (WB), Kenya, India and China but greater than 0.00003 ppm which is the standard set by US EPA. The level of Hg in wastewater samples from all the sampling sites was below the method detection limit (MDL) which had been set at 0.1 ppb. Similarly, the average levels of Pb for acid digested wastewater samples in 5 out of 8 (62.5%) sites (2 sites at Chief’s camp; Davis & Shirtliff, Kartasi and Donholm) had high Pb levels that ranged from 0.011 to 0.032 ppm, and this was above the recommended limits of Pb in wastewater set by WHO, Kenya, and US-EPA.
The mean concentration of Cr in wastewater samples from all the sampling sites ranged between 0.00308 to 0.00812 ppm which was between 84% to 99% less than the recommended limits by WHO, China, Kenya, US EPA, WB, and India. The wastewater samples collected at Sinai (G1) had the highest concentration of chromium at 0.0507 ppm, but which was within the maximum limit recommended level set by WHO and US EPA. The mean concentration of Ni in wastewater in all sampling sites was 0.004ppm and this was within the recommended limits set by WHO, China, Kenya, US EPA, WB and India. Nickel level was significantly high in wastewater samples obtained from Railways Lower (C2) with a mean concentration of 21.7 ppb and at Sinai (G1) with a concentration of 19.4 ppb. The mean levels of Tl in the wastewater were about 100 000 times less than the US EPA (SWQS) recommended limits. The mean concentration of Cd in wastewater in all the sampling sites was 0.000087 ppm which was far less than the recommended limits by WHO, WB, US EPA, China, Kenya, and India. The level of Hg in samples of tap water was below the MDL which had been set at 0.1 ppb or 0.0001 ppm. Similarly, the levels of Pb, Cd, Cr, Tl and Ni in the samples of tap water ranged between 0.00001 and 0.0016 mg/ml (ppm) which were far below the standard limits set by WHO, Chinese Ministry of Health and Kenya (NEMA), US EPA [67-70].
The Levels of Heavy Metals in Soil Samples
The mean concentration ± SE (standard error) of heavy metals in soil samples was highest for Pb and lowest for Hg in an ascending sequence of Hg < Tl < Cd < Ni < Cr < Pb. The concentration of Pb in soil samples from Davis & Shirtliff site was 471.17 ± 117.5 ppm compared to samples collected from Chief’s Camp (B) and Railways Lower (C) sites that were at 255.50 ± 91.20 and 211.00 ± 8.26 ppm respectively. Soil samples from Sinai site had the lowest level of Pb at 59.92 ± 8.42 ppm. Relatively higher levels of Hg were recorded for soil samples collected at Chief’s Camp (B), Railways Lower (C), and Davis & Shirtliff (E), which were at 223.75, 121.00, and 106.67 ppb respectively. The concentration of Cd and Tl in the soil samples ranged from 0.2 ± 0.05 to 1.90 ± 1.40 ppm and 0.23 ± 0.01 to 0.50 ± 0.06 ppm respectively. Soil samples from Chief’s Camp (B) site had the highest level of Cd and Tl while samples from Donholm (H) site had the lowest. The concentration of Cr and Ni ranged between 21.37 ± 9.87 to 81.17 ± 3.80 and 11.70 ± 0.44 to 29.87 ± 1.90 ppm respectively for the soil samples obtained from the study area.
When compared to the standard limits, the mean concentration of Hg and Ni in soil samples was 0.085 and 18.81 ppm respectively and this was below the recommended limits set by China and USEPA but above WHO limits for agricultural and gardening soils. The mean concentration of Cr in the soil samples was 45.19 ppm and it was above the limits set by WHO and US EPA. The average levels of Pb and Cd in soil samples was also above the recommended limits set by WHO, China but within the US EPA limit for agricultural and gardening soils. The mean concentration of Tl in the soil samples was 0.38 ppm, however the soils standards for Tl in agricultural soils for WHO, US EPA, China and Kenya were not available. It was observed that the mean concentration of heavy metals analyzed in the soil samples was between 800 to 13,500 times more than the mean concentration of the same metals in wastewater samples at the same sampling site.
Correlation of Heavy Metals Level in Different Samples
Pairwise comparison of the level of heavy metals in wastewater and soils samples yielded several strong significant positive correlations (P < 0.05). Such pairs included Cd & Hg in soils samples; and Tl (wastewater) & Cd (soil); where an increase in one element corresponded to an increase in the other. The strong positive correlations indicated a close association of the elements in samples of soil and wastewater. A negative correlation between Tl (soil) & Cr (wastewater), r = - 0.641, P = 0.087, was also observed. At Sinai sampling site (G), Inter-elemental correlation levels of metals for the samples of wastewater and soils samples were computed. A significant positive correlation was obtained for pairs Cd & Pb; Ni & Pb in samples of wastewater. Similarly, a strong positive correlation coefficient (r = 0.995) for Cr & Pb that was near significance level (P = 0.061), was recorded for soil samples obtained from Sinai.
Conductivity is a measure of the water’s ability to conduct electricity and it is directly related to the concentration of ions in the water. Significant changes in the conductivity of water directly compromise its quality. In the current study, conductivity of the wastewater was highest for samples obtained from Railways lower (C), Railways upper (D) and Sinai (G) sampling sites and it ranged from 1072 to 1134 μS/cm. This range was higher than WHO range for electrical conductivity of water which is between 400 to 600 μS/cm. The wastewater samples from the remaining sites had conductivity levels that were within the WHO standards. The source of the conductive ions in wastewater may have been the dissolved substances including pollutants. At railways sampling sites (C and D), combined organic sewage material overflow that had been directed into the open wastewater channel may have introduced dissolved ions into the channels hence increasing conductivity
This was in line with a previous study by Mbui and colleagues who reported that domestic effluents discharge into the river increases the electrical conductivity. The road construction activities near Sinai sampling site (G) which involved heavy machinery plus nearby industries may have contributed to increased particles and ions in the wastewater channels hence accounting for the raised turbidity and conductivity levels of the wastewater samples. It was noted that increased turbidity corresponded to increased conductivity and vice versa. Turbidity indicates total suspended solids in water, and it is a principle parameter of wastewater effluent monitoring and therefore it can be used to evaluate wastewater treatment plant efficiency and compliance to discharge requirements. Both conductivity and turbidity are important parameters in measuring the quality of water post treatment.
The temperature of wastewater samples ranged from 16.75 to 26.05 °C, which was within the recommended WHO range of 20 to 32 °C. The pH of wastewater samples ranged from 7.28 to 8.78 and similarly was within WHO range of 6.5 to 8.5. Microbial degradative activities in wastewater are dependent on temperature, pH, presence of organic substances and types of microbes. Therefore, elevated temperature in wastewater tends to support increased bio-degradative reactions in presence of increased organic substances. The organic substances and types of microbes in the wastewater were not determined in this study.
During the third United Nations Environment Assembly hosted by Kenya at UNEP headquarters (Gigiri) in December 2017, Kenya promised to improve the lives of its people by cleaning up air, land and water. Industrial, sewage and domestic wastes have been finding their way into Nairobi River in Kenya, hence making the river unsuitable for use. According to the UN Environment, over 80% of the world’s wastewater is released into the environment without treatment, polluting the fields where plants grow, lakes and rivers. Such pollutants can easily flow from the environment into humans directly or indirectly. Water and soil pollution with heavy metals in developing countries emanate from poor disposal of industrial and urban wastes. Municipal and industrial wastewater should be treated as a strategy of minimizing the contaminants before reusing wastewater. However, health impact assessment of treated wastewater should be carried out to identify the hazards and risk factors that may be associated with wastewater reuse.
The current study established that the levels of Cr, Cd, and Ni in wastewater were below the limits set by WHO, US EPA, WB and Kenya. Similarly, the Hg level in wastewater was below the limits set by WHO, China, WB, India and Kenya (NEMA) but slightly above the limit set by US EPA. The level of Pb in the wastewater was above limits set by WHO, US EPA, WB and Kenya but lower than the limits set by China and India. The public health concern in terms of wastewater in Nairobi industrial area is therefore Hg and Pb levels which were above the limits set by US EPA and WHO respectively. Standards for thallium in wastewater were not available and therefore it was difficult to make a conclusion on whether the levels obtained were high or low. Previous reports however, have shown that water quality within Nairobi River catchment area in Kenya has degraded due to intensive land use hence increasing Hg and Pb levels and surpassing the critical guidelines of WHO.
In the current study area, humans can become exposed to such pollutants when surface runoff finds its way into residential areas or utilizing the contaminated soils for agricultural activities. Heavy metal accumulation in soils leads to increased phyto - accumulation of such metals in the crops grown. Clogging of open wastewater channels with solid wastes, mud and overgrown vegetation can enhance surface runoff of the wastewater to surrounding areas. One of the factors which may increase the chance of exposure to metal pollutants in the study area is the presence of dense population in the informal settlements near Nairobi industrial area. Some of the open wastewater channels pass through these settlements or drain into Nairobi River which then flow across these settlements. However, treatment of wastewater for reuse is a common practice in many countries since it can alleviate natural water shortage and minimize contaminants finding their way into natural aquatic ecosystems. According to a study carried out in Greece, the annual percent contribution of treated wastewater in the total irrigation water volume in Thermos and Nafpaktos was 87.8% and >100% respectively. The same should be adopted in Nairobi to minimize the contaminants in wastewater and to provide adequate water for agricultural activities [71-73].
Tap water samples (controls) that were randomly collected from the study area were analyzed to establish the heavy metals levels and compared to wastewater samples. The levels of all the heavy metals studied in tap water samples ranged from <0.00001 to 0.0016 ppm and these levels were far below limits set by WHO, US EPA, Chinese Ministry of Health (CMH) and Kenya (NEMA). The tap water sampled from residential and hotels in the study area was therefore safe, high quality, and acceptable in terms of Hg, Pb, Cd, Cr, Tl and Ni levels.
The domestic pigs observed at Kartasi industries sampling site, scavenging for edibles from the mud and vegetation clogged open wastewater channels, was evidence that there were residential areas nearby in the study area. The sampling sites in the current study were actually near the densely populated informal settlements (slums) that included Sinai, Mukuru kwa Njenga, and Land Mawe. The samples of wastewater and soil sediments from Kartasi sampling site had levels of heavy metals that were above the MDL except for Hg level in wastewater. It is worth noting that even low levels of environmental metal pollutants can accumulate with time in exposed humans and animals.
Previous studies show that livestock are prone to general problems of industrial pollution. A study in Namibia established that pasture grass that was obtained from around waste dumpsites had higher levels of heavy metals. It is possible for heavy metals to accumulate in the tissues and organs of domestic animals that become exposed to contaminated environments, materials, and fodder. The concentration of Hg and Cd was shown to be high in the liver, kidney, and muscle samples of organically and conventionally produced pigs in Czech Republic. Therefore, the scavenging pigs in the open wastewater channels in industrial areas, that were observed in the current study, can serve to directly or indirectly spread the heavy metal pollutants from such channels into humans. When the heavy metals pollutants from the channels accumulate in the pigs’ muscles with time, then the quality of pork from such animals is compromised and it may become a health risk.
Heavy metals occur naturally in soils following the weathering processes of the underlying rocks. Availability of heavy metals in soils is influenced by environmental conditions that determine the pH and organic matter content in soils. Heavy metal contamination of the soils may pose risks and hazards to humans and ecosystems through direct contact or ingestion, food chain, contaminated drinking water, reduced food quality among others. The concentration of the Lead (Pb) in the soil samples at Davis & Shirtliff sampling site was 471.17 ± 117.5 ppm and this was above the normal range of Pb (2 to 300 ppm) in the soils. The worldwide Pb concentration for surface soil averages 32 mg/kg (ppm) and it ranges from 10 to 67 mg/kg (ppm) implying that the levels at Davis & Shirtliff were significantly above this limit.
The average Pb level in the soil samples collected from Chief’s camp (B) and Railways Lower (C) were 255.50 ± 91.20 and 211.00 ± 8.26 ppm respectively and they were skewing towards the upper limit of normal range as described by Gardea-Torresdey and colleagues but above the range reported by Pendias & Pendias. The soil samples from Sinai site had the lowest level of Pb at 59.92 ± 8.42 ppm which was skewed towards the lower limit of normal range of Pb in soils. Soil sediments in the wastewater channels may enrich with pollutants present in wastewater with time. Increased Pb content in soil recovered from the open wastewater channels is a health hazard to workers who regularly clean up the channels especially when they are ignorant about the need to maximize safety measures. Lead has been associated with multiple organ problems and cancers. The soil samples collected from Chief’s camp (B), Railways Lower (C), and Davis & Shirtliff (E) sampling sites had relatively higher Hg levels, but which were within the limits set by China and US EPA for agricultural soils.
The average concentration of Cr and Ni in the soil samples from the study area which ranged between 21.37 ± 9.87 to 81.17 ± 3.80 and 11.70 ± 0.44 to 29.87 ± 1.90 ppm respectively were below the allowable limits recommended by China and US EPA but above the limits recommended by WHO for agricultural soils. It is important to note that even soils that are contaminated with low levels of heavy metals can contribute to bioaccumulation of such elements with time in organisms that are in higher trophic levels in a food chain. Pollution of the environment with traces of heavy metals from anthropogenic sources should not therefore be ignored [74-76].
The mean concentration of heavy metals was higher in soils than in wastewater samples. This was in line with a previous report by Khan and his colleagues which explained that contaminated wastewater can lead to a build-up of heavy metals in soils. Inter-elemental analysis of the metals showed several strong and positive correlations. This suggested that, these metals were from the same source, most likely the industries whose wastes were draining into the open channels in the study area. This explanation was in line with previous studies carried out in Nigeria and Pakistan. The significant correlation coefficients between pairs of metals in samples of wastewater and soils may be a pointer of a common source of heavy metal pollution in the study area, most likely anthropogenic activities.
This study showed that wastewater and soils samples from open waste channels in Nairobi industrial area contained heavy metals. Of the metals studied, the mean concentration of Ni, Cr and Pb were relatively higher than those of Tl, Hg, and Cd in the samples analyzed. The levels of Hg, Cr, Cd and Ni in wastewater samples were within the allowable limits set by WHO, WB, Kenya, China, and India. The mean level of Hg in wastewater was <0.0001ppm and this was a public health concern in the study area, based on the US EPA allowable limit of Hg in wastewater that is set at 0.00003 ppm. The level of Tl in wastewater samples was below the limit set by US EPA and this was commendable. The mean concentration of Pb in wastewater was above the allowable limits set by WHO, WB, US EPA and Kenya in 5 out of 8 sampling sites, hence becoming a public health concern in the study area.
The levels of Pb, Hg, Cr, Cd, and Ni in open drainage channels soil samples were above the limits set by WHO for agricultural and gardening soils. The mean concentration of heavy metals was relatively higher in soil than in wastewater samples at each sampling site. This was evidence of a build-up of toxic metals in the soils found in open waste channels. There was adequate evidence of clogging of the wastewater channels with mud and overgrown vegetation hence facilitating overflow and spread of contaminated wastewater and soils from the channels to residential areas nearby during the rainy seasons. The presence of domestic pigs scavenging from the open channels suggested a likely pathway through which the metallic contaminants could eventually find their way into humans. Therefore, there is need to formulate and adopt policies, strict rules among others that would translate to excellent wastewater management and treatment infrastructure hence minimizing environmental pollution and its associated health hazards as well as avail adequate reclaimed water for urban agricultural activities.
Frequent inspections and unclogging of the open waste channels should be carried out to enhance faster flow and to minimize possible spread of heavy metal contaminated wastewater to the densely populated informal settlements/ villages that neighbor Nairobi industrial area. Residents living nearby should be made aware of the health hazards that could emanate from exposure to untreated wastewater through public education and awareness campaigns. We the authors recommend the determination of heavy metals in pork available in the study area in order to provide possible evidence of bioaccumulation of these metal contaminants in human food.
Arsenic
Environmental Occurrence, Industrial Production and Use
Arsenic is a ubiquitous element that is detected at low concentrations in virtually all environmental matrices. The major inorganic forms of arsenic include the trivalent arsenite and the pentavalent arsenate. The organic forms are the methylated metabolites - monomethylarsonic acid (MMA), dimethylarsinic acid (DMA) and trimethylarsine oxide. Environmental pollution by arsenic occurs as a result of natural phenomena such as volcanic eruptions and soil erosion, and anthropogenic activities. Several arsenic-containing compounds are produced industrially and have been used to manufacture products with agricultural applications such as insecticides, herbicides, fungicides, algicides, sheep dips, wood preservatives, and dyestuffs. They have also been used in veterinary medicine for the eradication of tapeworms in sheep and cattle [77-85].
Arsenic compounds have also been used in the medical field for at least a century in the treatment of syphilis, yaws, amoebic dysentery, and trypanosomaiasis. Arsenic-based drugs are still used in treating certain tropical diseases such as African sleeping sickness and amoebic dysentery, and in veterinary medicine to treat parasitic diseases, including filariasis in dogs and black head in turkeys and chickens. Recently, arsenic trioxide has been approved by the Food and Drug Administration as an anticancer agent in the treatment of acute promeylocytic leukemia. Its therapeutic action has been attributed to the induction of programmed cell death (apoptosis) in leukemia cells.
Potential for Human Exposure
It is estimated that several million people are exposed to arsenic chronically throughout the world, especially in countries like Bangladesh, India, Chile, Uruguay, Mexico, and Taiwan, where the ground water is contaminated with high concentrations of arsenic. Exposure to arsenic occurs via the oral route (ingestion), inhalation, dermal contact, and the parenteral route to some extent. Arsenic concentrations in air range from 1 to 3 ng/m3 in remote locations (away from human releases), and from 20 to 100 ng/m3 in cities. Its water concentration is usually less than 10µg/L, although higher levels can occur near natural mineral deposits or mining sites. Its concentration in various foods ranges from 20 to 140 ng/kg. Natural levels of arsenic in soil usually range from 1 to 40 mg/kg, but pesticide application or waste disposal can produce much higher values.
Diet, for most individuals, is the largest source of exposure, with an average intake of about 50 µg per day. Intake from air, water and soil are usually much smaller, but exposure from these media may become significant in areas of arsenic contamination.
Workers who produce or use arsenic compounds in such occupations as vineyards, ceramics, glassmaking, smelting, refining of metallic ores, pesticide manufacturing and application, wood preservation, semiconductor manufacturing can be exposed to substantially higher levels of arsenic. Arsenic has also been identified at 781 sites of the 1,300 hazardous waste sites that have been proposed by the U.S. EPA for inclusion on the national priority list. Human exposure at these sites may occur by a variety of pathways, including inhalation of dusts in air, ingestion of contaminated water or soil, or through the food chain.
Contamination with high levels of arsenic is of concern because arsenic can cause a number of human health effects. Several epidemiological studies have reported a strong association between arsenic exposure and increased risks of both carcinogenic and systemic health effects. Interest in the toxicity of arsenic has been heightened by recent reports of large populations in West Bengal, Bangladesh, Thailand, Inner Mongolia, Taiwan, China, Mexico, Argentina, Chile, Finland and Hungary that have been exposed to high concentrations of arsenic in their drinking water and are displaying various clinic-pathological conditions including cardiovascular and peripheral vascular disease, developmental anomalies, neurologic and neurobehavioral disorders, diabetes, hearing loss, portal fibrosis, hematologic disorders (anemia, leukopenia and eosinophilia) and carcinoma.
Arsenic exposure affects virtually all organ systems including the cardiovascular, dermatologic, nervous, hepatobiliary, renal, gastro-intestinal, and respiratory systems. Research has also pointed to significantly higher standardized mortality rates for cancers of the bladder, kidney, skin, and liver in many areas of arsenic pollution. The severity of adverse health effects is related to the chemical form of arsenic and is also time- and dosedependent. Although the evidence of carcinogenicity of arsenic in humans seems strong, the mechanism by which it produces tumors in humans is not completely understood.
Molecular Mechanisms of Toxicity and Carcinogenicity
Analyzing the toxic effects of arsenic is complicated because the toxicity is highly influenced by its oxidation state and solubility, as well as many other intrinsic and extrinsic factors. Several studies have indicated that the toxicity of arsenic depends on the exposure dose, frequency and duration, the biological species, age, and gender, as well as on individual susceptibilities, genetic and nutritional factors. Most cases of human toxicity from arsenic have been associated with exposure to inorganic arsenic. Inorganic trivalent arsenite (AsIII) is 2-10 times more toxic than pentavalent arsenate (AsV). By binding to thiol or sulfhydryl groups on proteins, As (III) can inactivate over 200 enzymes. This is the likely mechanism responsible for arsenic’s widespread effects on different organ systems. As (V) can replace phosphate, which is involved in many biochemical pathways.
One of the mechanisms by which arsenic exerts its toxic effect is through impairment of cellular respiration by the inhibition of various mitochondrial enzymes, and the uncoupling of oxidative phosphorylation. Most toxicity of arsenic results from its ability to interact with sulfhydryl groups of proteins and enzymes, and to substitute phosphorous in a variety of biochemical reactions. Arsenic in vitro reacts with protein sulfhydryl groups to inactivate enzymes, such as dihydrolipoyl dehydrogenase and thiolase, thereby producing inhibited oxidation of pyruvate and betaoxidation of fatty acids. The major metabolic pathway for inorganic arsenic in humans is methylation. Arsenic trioxide is methylated to two major metabolites via a non-enzymatic process to monomethylarsonic acid (MMA), which is further methylated enzymatically to dimethyl arsenic acid (DMA) before excretion in the urine. It was previously thought that this methylation process is a pathway of arsenic detoxification, however, recent studies have pointed out that some methylated metabolites may be more toxic than arsenite if they contain trivalent forms of arsenic.
Tests for genotoxicity have indicated that arsenic compounds inhibit DNA repair, and induce chromosomal aberrations, sisterchromatid exchanges, and micronuclei formation in both human and rodent cells in culture and in cells of exposed humans. Reversion assays with Salmonella typhimurium fail to detect mutations that are induced by arsenic compounds. Although arsenic compounds are generally perceived as weak mutagens in bacterial and animal cells, they exhibit clastogenic properties in many cell types in vivo and in vitro. In the absence of animal models, in vitro cell transformation studies have become a useful means of obtaining information on the carcinogenic mechanisms of arsenic toxicity. Arsenic and arsenical compounds are cytotoxic and induce morphological transformations of Syrian hamster embryo (SHE) cells as well as mouse C3H10T1/2 cells and BALB/3T3 cells.
Based on the comet assay, it has been reported that arsenic trioxide induces DNA damage in human lymphophytes and also in mice leukocytes. Arsenic compounds have also been shown to induce gene amplification, arrest cells in mitosis, inhibit DNA repair, and induce expression of the c-fos gene and the oxidative stress protein heme oxygenase in mammalian cells. They have been implicated as promoters and comutagens for a variety of toxic agents. Recent studies in our laboratory have demonstrated that arsenic trioxide is cytotoxic and able to transcriptionally induce a significant number of stress genes and related proteins in human liver carcinoma cells.
Epidemiological investigations have indicated that long-term arsenic exposure results in the promotion of carcinogenesis. Several hypotheses have been proposed to describe the mechanism of arsenic-induced carcinogenesis. Zhao et al. reported that arsenic may act as a carcinogen by inducing DNA hypomethylation, which in turn facilitates aberrant gene expression. Additionally, it was found that arsenic is a potent stimulator of extracellular signalregulated protein kinase Erk1 and AP-1 transactivational activity, and an efficient inducer of c-fos and c-jun gene expression. Induction of c-jun and c-fos by arsenic is associated with activation of JNK. However, the role of JNK activation by arsenite in cell transformation or tumor promotion is unclear.
In another study, Trouba et al. concluded that long-term exposure to high levels of arsenic might make cells more susceptible to mitogenic stimulation and that alterations in mitogenic signaling proteins might contribute to the carcinogenic action of arsenic. Collectively, several recent studies have demonstrated that arsenic can interfere with cell signaling pathways (e.g., the p53 signaling pathway) that are frequently implicated in the promotion and progression of a variety of tumor types in experimental animal models, and of some human tumors. However, the specific alterations in signal transduction pathways or the actual targets that contribute to the development of arsenicinduced tumors in humans following chronic consumption of arsenic remains uncertain.
Recent clinical trials have found that arsenic trioxide has therapeutic value in the treatment of acute promyelocytic leukemia, and there is interest in exploring its effectiveness in the treatment of a variety of other cancers. In acute promyelocytic leukemia, the specific molecular event critical to the formation of malignant cells is known. A study by Puccetti et al. found that forced overexpression of BCR-ABL susceptibility in human lymphoblasts cells resulted in greatly enhanced sensitivity to arsenic-induced apoptosis. They also concluded that arsenic trioxide is a tumor specific agent capable of inducing apoptosis selectively in acute promyelocytic leukemia cells. Several recent studies have shown that arsenic can induce apoptosis through alterations in other cell signaling pathways. In addition to acute peomyelocytic leukemia, arsenic is thought to have therapeutic potential for myeloma. In summary, numerous cancer chemotherapy studies in cell cultures and in patients with acute promyelocytic leukemia demonstrate that arsenic trioxide administration can lead to cell-cycle arrest and apoptosis in malignant cells.
Previous studies have also examined p53 gene expression and mutation in tumors obtained from subjects with a history of arsenic ingestion participates in many cellular functions, cellcycle control, DNA repair, differentiation, genomic plasticity and programmed cell death. Additional support for the hypothesis that arsenic can modulate gene expression has been provided by several different studies. Collectively, these studies provide further evidence that various forms of arsenic can alter gene expression and that such changes could contribute substantially to the toxic and carcinogenic actions of arsenic treatment in human populations.
Several in vitro studies in our laboratory have demonstrated that arsenic modulates DNA synthesis, gene and protein expression, genotoxicity, mitosis and/or apoptotic mechanisms in various cell lines including keratinocytes, melanocytes, dendritic cells, dermal fibroblasts, microvascular endothelial cells, monocytes, and T-cells, colon cancer cells, lung cancer cells, human leukemia cells, Jurkat-T lymphocytes, and human liver carcinoma cells.We have also shown that oxidative stress plays a key role in arsenic induced cytotoxicity, a process that is modulated by pro- and/ or anti-oxidants such as ascorbic acid and n-acetyl cysteine. We have further demonstrated that the toxicity of arsenic depends on its chemical form, the inorganic form being more toxic than the organic one.
Various hypotheses have been proposed to explain the carcinogenicity of inorganic arsenic. Nevertheless, the molecular mechanisms by which this arsenical induces cancer are still poorly understood. Results of previous studies have indicated that inorganic arsenic does not act through classic genotoxic and mutagenic mechanisms, but rather may be a tumor promoter that modifies signal transduction pathways involved in cell growth and proliferation. Although much progress has been recently made in the area of arsenic’s possible mode(s) of carcinogenic action, a scientific consensus has not yet been reached. A recent review discusses nine different possible modes of action of arsenic carcinogenesis: induced chromosomal abnormalities, oxidative stress, altered DNA repair, altered DNA methylation patterns, altered growth factors, enhanced cell proliferation, promotion/ progression, suppression of p53, and gene amplification.
Presently, three modes (chromosomal abnormality, oxidative stress, and altered growth factors) of arsenic carcinogenesis have shown a degree of positive evidence, both in experimental systems (animal and human cells) and in human tissues. The remaining possible modes of carcinogenic action (progression of carcinogenesis, altered DNA repair, p53 suppression, altered DNA methylation patterns and gene amplification) do not have as much evidence, particularly from in vivo studies with laboratory animals, in vitro studies with cultured human cells, or human data from case or population studies. Thus, the mode-of-action studies suggest that arsenic might be acting as a cocarcinogen, a promoter, or a progressor of carcinogenesis.
Environmental Impacts of Heavy Metals
The presence of heavy metals in the environment leads to a number of adverse impacts. Such impacts affect all spheres of the environment, that is, hydrosphere, lithosphere, biosphere and atmosphere. Until the impacts are dealt with, health and mortality problems break out, as well as the disturbance of food chains.
Effect of Heavy Metals Contamination
Heavy metals contamination is becoming a serious issue of concern around the world as it has gained momentum due to the increase in the use and processing of heavy metals during various activities to meet the needs of the rapidly growing population. Soil, water and air are the major environmental compartments which are affected by heavy metals pollution.
Effect on Soil
Emissions from activities and sources such as industrial activities, mine tailings, disposal of high metal wastes, leaded gasoline and paints, land application of fertilizers, animal manures, sewage sludge, pesticides, wastewater irrigation, coal combustion residues and spillage of petrochemicals lead to soil contamination by heavy metals. Soils have been noted to be the major sinks for heavy metals released into the environment by aforementioned anthropogenic activities. Most heavy metals do not undergo microbial or chemical degradation because they are nondegradable, and consequently their total concentrations last for a long time after being released to the environment.
The presence of heavy metals in soils is a serious issue due to their residence in food chains, thus destroying the entire ecosystem. As much as organic pollutants can be biodegradable, their biodegradation rate, however, is decreased by the presence of heavy metals in the environment, and this in turn doubles the environmental pollution, that is, organic pollutants and heavy metals thus present. There are various ways through which heavy metals present risks to humans, animals, plants and ecosystems as a whole. Such ways include direct ingestion, absorption by plants, food chains, consumption of contaminated water and alteration of soil pH, porosity, color and its natural chemistry which in turn impact on the soil quality.
Effects on Water
Although there are many sources of water contamination, industrialization and urbanization are two of the culprits for the increased level of heavy metal water contamination. Heavy metals are transported by runoff from industries, municipalities and urban areas. Most of these metals end up accumulating in the soil and sediments of water bodies. Heavy metals can be found in traces in water sources and still be very toxic and impose serious health problems to humans and other ecosystems. This is because the toxicity level of a metal depends on factors such as the organisms which are exposed to it, its nature, its biological role and the period at which the organisms are exposed to the metal. Food chains and food webs symbolize the relationships amongst organisms. Therefore, the contamination of water by heavy metals actually affects all organisms. Humans, an example of organisms feeding at the highest level, are more prone to serious health problems because the concentrations of heavy metals increase in the food chain.
Effects on Air
Industrialisation and urbanisation, due to rapid world population growth, have recently made air pollution a major environmental problem around the world. The air pollution was reported to have been accelerated by dust and particulate matters (PMs) particularly fine particles such as PM2.5 and PM10 which are released through natural and anthropogenic processes. Natural processes which release particulate matter into air include dust storms, soil erosion, volcanic eruptions, and rock weathering, while anthropogenic activities are more industrial, and transportation related. Particulate matters are important and require special attention as they can lead to serious health problems such as skin and eyes irritation, respiratory infections, premature mortality, and cardiovascular diseases. These pollutants also cause deterioration of infrastructure, corrosion, formation of acid rain, eutrophication, and haze. Amongst others, heavy metals such as group 1 metals (Cu, Cd, Pb), group 2 metals (Cr, Mn, Ni, V and Zn) and group 3 metals (Na, K, Ca, Ti, Al, Mg, Fe) originate from industrial areas, traffic, and natural sources, respectively
Mechanisms of Remediating Heavy Metals
Treatment processes for acid mine water typically generate high-density sludge that is heterogeneous due to variety of metals, metalloids, and anionic components, and this makes it difficult to dispose the sludge [19]. Recent research have therefore focused on the recovery of chemical species from acid mine drainage (AMD) and secondary sludge. This is aimed at recovering valuable resources and also enabling easier and safer disposal of the treated sludge, hence reducing their environmental footprints. Disposal of metal ladened waste to landfills and waste retention ponds/ heaps lead to secondary pollution of surface and subsurface water resources. It may also lead to soil contamination, hence affecting their productivity. In order to protect human health, plants, animals, soil and all the compartments of the environment, proper and careful attention should be given to remediation technologies of heavy metals. Most physical and chemical heavy metal remediation technologies require handling of large amounts of sludge, destroy surrounding ecosystems and are very expensive.
Precipitation
A variety of alkaline chemical reagents have been used over the years for neutralization of acid mine drainage (AMD) in order to increase the pH and consequently precipitate and recover the metals. The most common alkaline reagents used for sequential recovery of minerals resources from AMD are limestone (CaCO3), caustic soda (NaOH), soda ash (Na2CO3), quicklime (CaO), slaked lime (Ca(OH)2) and magnesium hydroxide (Mg(OH)2). Some processes have recovered metals at varying pH regimes and synthesized commercially valuable materials such as pigments and magnetite. Some minerals are recovered and sold to metallurgical industries, hence off-setting the treatment costs Table (1).
Adsorption
Adsorption occurs when an adsorbate adheres to the surface of an adsorbent. Due to reversibility and desorption capabilities, adsorption is regarded as the most effective and economically viable option for the removal of metals from aqueous solution. Although efficient, adsorption is not effective with very concentrated solution as the adsorbent easily gets saturated with the adsorbate. It is only feasible for very dilute solutions, is labor intensive because it requires frequent regeneration, and it is not selective in terms of metal attenuation. Adsorption is therefore not applied in a large scale of metal remediation.
Ion Exchange
Ion exchange is the exchange of ions between two or more electrolyte solutions. It can also refer to the exchange of ions on a solid substrate to soil solution. High cation exchange capacity clay and resins are commonly used for the uptake of metals from aqueous solutions. However, this method requires high labor and is limited to a certain concentration of metals in the solution. This system also operates under specific temperature and pH. Natural and synthetic clays, zeolites and synthetic resins have been used for removal and attenuation of metals from wastewater.
Biosorption
Biosorption refers to the removal of pollutants from water systems using biological materials, and it entails the absorption, adsorption, ion exchange, surface complexation and precipitation. Biosorbents have an advantage of accessibility, efficiency and capacity. This process is readily and easily available. Regeneration is easy, hence making it very favorable. However, when the concentration of the feed solution is very high, the process easily reaches a breakthrough, thus limiting further pollutant removal.
1.1. Membrane Technologies
The use of membrane technologies for the recovery of acid mine drainage is very effective for water that has high concentration of pollutants. It uses the concentration gradients phenomenon or the opposite which is reverse osmosis. There are different types of membranes that are used for mine water treatment including: ultrafiltration, nano-filtration, reverse osmosis, microfiltration, and particle filtration.
Case Study of South Africa Acid Mine Drainage
South Africa is well endowed by mineral reserves, and this has triggered its immense dependence on mineral resources for gross domestic product and economy. However, the legacy of coal and gold mining has left in its wake serious environmental problems. The major problem is acid mine drainage. Acid mine drainage (AMD) is formed from the hydro-geochemical weathering of sulphide-bearing rocks (pyrite, arsenopyrite and marcasite) in contact with water and oxygen. This reaction is also catalyzed by iron (Fe) and sulphur-oxidising microorganisms. In a nutshell, the formation of AMD can be summarized as follows:
The oxidation of sulphide to sulphate solubilises the ferrous iron (Fe(II)), which is subsequently oxidised to ferric iron (Fe(III)):
Either these reactions can occur spontaneously or can be catalyzed by microorganisms (sulphur- and iron-oxidizing bacteria) that derive energy from the oxidation reaction. The ferric cations produced can also oxidize additional pyrite into ferrous ions:
The net effect of these reactions is to produce H+ and maintain the solubility of the ferric iron. Because of the high acidity and elevated concentration of toxic and hazardous metals, AMD has been a prime issue of environmental concern that has globally raised public concern. The discharge of metalliferous drainage from mining activities has rendered the environment unfit to foster life. Pragmatic approaches need to be developed to counter this mining legacy that is perpetually degrading the environment and its precious resources. Research and piloted studies have indicated that active and passive approaches can be successfully adopted to treat acid mine drainage and remove potentially toxic chemical species. The presence of Al, Fe, Mn and sulphates is a prime concern in addition to the trace of Cu, Ni, Pb and Zn. Metalloids of as and earth alkali metal (Ca and Mg) are also present in significant levels. Several studies have shown the feasibility of treating acid mine drainage to acceptable levels as prescribed by different water quality guidelines, but the resultant sludge has been an issue of public concern due to its heterogeneous and complex nature loaded with metal species.
Based on that evidence, research studies have been firmly embedded on the recovery of valuable minerals from AMD. There are several mechanisms used for the recovery of chemical components from AMD including: precipitation, adsorption, biosorption, and ion exchange, desalination, and membrane filtration. Out of those techniques, precipitation has been the promising technology due to the ability to handle large volumes of water with very little dosage. Adsorption and ion exchange have a challenge of poor efficiency at elevated concentrations and quick rate of saturation. Membrane technologies have the problem of generating brine that creates another environmental liability. Desalination has a problem of producing salts that have impurities, hence making them unsuitable for utilization. Freeze desalination has been a promising technology, but it has never been tried on a large scale.
Impacts of Heavy Metals in South Africa
South Africa’s geology is rich in coal and mineral reserves which contain key metals such as gold, platinum, and copper. The significant volume of mineral and coal reserves has made mining serve as a backbone in the development and growth of the country’s economy. This is evident from the massive number of mines found around the country. However, mining has been noted to cause inimical impacts to human health, organisms, and environment as a whole, with water resources being the most common victim of pollution. However, the above -mentioned concentrations depend on the pH of the AMD-concentrations decrease when pH increases. When exposed to such conditions, mortality and diseases are most likely to occur in organisms, as well as other health. In addition, AMD destroys ecosystems of organisms and also negatively impacts on the economy of the country. Heavy metals in active and abandoned mines in South Africa have impacted both surface and underground water.
Legal Requirements of Water Quality
The National Environmental Management Act (NEMA) 108 of 1998, stipulates that everyone has the right to live in an environment which is safe and unlikely to pose any deleterious effects to their health. The legislative requirements for industrial effluents are primarily governed by the Department of Water Affairs DWS Water Quality Guidelines. This purpose requires that any person who uses water for industrial purposes purify or otherwise treat such water in accordance with requirements of DWA.
Deleterious Effects of Acid Mine Drainage on Terrestrial and Aquatic Ecosystems
The introduction of effluents from mining activities into receiving streams can severely impact aquatic ecosystems through habitat destruction and impairment of water quality. This will eventually lead to a reduction in biodiversity of a given aquatic ecosystem and its ability to sustain life. The severity and extent of damage depends on a variety of factors including the frequency of influx, volume and chemistry of the drainage and the buffering capacity of the receiving stream.
Acidity
When metals in AMD are hydrolyzed, they lower the pH of the water making it unsuitable for aquatic organisms to thrive. AMD is highly acidic (pH 2-4), and this promotes the dissolution of toxic metals. Those toxic species exert hazardous effects on terrestrial and aquatic organisms. Also, if the water is highly acidic, only acidophile microorganisms will thrive on such water with the rest of aquatic organisms migrating to other regions which are conducive to their survival. Many streams contaminated with AMD are largely devoid of life for a long way downstream. To some aquatic organisms, if the pH range falls below the tolerance range, probability of death is very high due to respiratory and osmoregulation failure. Acidic conditions are dominated by H+ which is absorbed and pumps out Na from the body which is important in regulating body fluids.
Toxic Chemical Species
Exposure of aquatic and terrestrial organisms to potentially toxic metals and metalloids can have devastating impacts to living organisms. Toxic chemical species present in AMD have been reported to be toxic to aquatic and terrestrial organisms. They are associated with numerous diseases including cancer. Some of these chemical species may accumulate and be biomagnified in living organisms, hence threatening the life of higher trophic organisms such as birds. Lead causes blood disorders, kidney damage, miscarriages and reproductive disorders and is linked to various cancers. The exposure of living organisms to toxic chemical species in AMD can also lead to nausea, diarrhea, liver and kidney damage, dermatitis, internal haemorrhage and respiratory problems. Epidemiological studies have shown a significant increase in the risk of lung, bladder, skin, liver, and other cancers on exposure to these chemical species. The effects of Al, Fe, Mn, Cu, Mg and Zn on the health of living organisms are summarized in Table.
Why are Traces of Heavy Metals in Drinking Water at All?
Heavy metals enter the natural water cycle via emissions during combustion processes with rainwater, it also happens in over fertilization in agriculture via leachate and the rainwater becomes contaminated when it falls. In each country a governing body will determine which contaminants are permitted and to what limit. Waterworks control water to the point where it is fed into the public grid. There, drinking water converts into tap water and evades the supervision of the waterworks in the stretch of pipe to your tap where contamination can happen. A well-known example of this is high lead concentrations in tap water, which are due to outdated supply lines or house pipes made from lead. These pipes have been banned as components, yet cases of households with lead water are still known.
How does lead from a pipe actually get into drinking water? Lead is known to be non-water soluble as a metal. The chemical process called substance migration is the cause of metals entering water. The surfaces of two substances on a molecular or atomic level interact. Not every metal behaves the same when migrating. The less a metal releases traces of its substance to liquids, the more food-grade it is and therefore usable. For example, Copper and stainless steel are a higher food grade as they do not release traces when in contact with water. The limit values protect us from acute symptoms of poisoning. However, long-term health consequences have not been considered with the permanent consumption of the smallest quantities of heavy metals. They can accumulate in the body over years and have a harmful effect on metabolism. They can disrupt natural cell functions or cause hormone failures. There are many illnesses associated with heavy metal stress, Cancer, Multiple Sclerosis, Parkinson’s, and Alzheimer’s and even depression. In principle, heavy metals, like all toxins, affect the kidneys, liver and pancreas - organs that primarily process foreign bodies and harmful substances [86-90].
Today, water works and utilities use technologically advanced filter systems in drinking water treatment. The prescribed limit values can be largely met with the cleaning processes used. However, zero tolerance solutions are not to be expected from this field with the usual methods in the medium and long term. In industrial applications, wherever pure water is of great importance reverse osmosis is used for drinking water extraction and purification. Reverse osmosis plants discharge particles and substances from the water via semipermeable membranes and artificially generated osmotic pressure. The final result is pure H2O. So why do water suppliers not use this procedure to remove all pollutants from the water? The answer is obvious: reverse osmosis works so well that it also removes substances that are valuable to the body. In the case of drinking water, essential minerals.
However, waterworks - as well as mineral water bottlers - are prohibited by law from re-enriching their products retrospectively with what they previously withdrew from them. If natural mineral water is enriched with ingredients that are not already present in the natural spring water, it may only be marketed as table water.
Most chemical sensor applications have been based on a broad background of measurement principles and chemical reactivity developed through research in analytical and other branches of chemistry. Many fundamental ideas, devices, and materials have been adapted from other sciences and technologies (Murray et al., 1989):
• Inexpensive optical fibers from the communications industry have been applied in spectroscopically based directreading sensors and near-field microscopy (Betzig et al, 1991).
• Lithographic patterning technology widely used in the manufacture of modern microelectronics has been exploited to fabricate miniaturized electro-chemical devices such as interdigitated array electrodes, microelectrodes, and chemically sensitive field-effect transistors and to form patterned electrodes on surface acoustic wave (SAW) devices. 6 (Kepley et al., 1992; Ricco and Martin, 1992; Martin et al., 1990).
• Materials research has resulted in advanced piezoelectric materials that are employed as micro-positioners; these materials have enabled new forms of microscopy, like scanning electrochemical, scanning tunnelling, and atomic force microscopy. The availability of these micropositioners is also critical for chemical sensing on an extremely small dimensional scale (Snyder and White, 1992).
• Ultrasensitive light detection using charge coupled devices, which were developed for astronomy, is under active consideration for detection of laser-induced fluorescence from extremely small populations of molecules.
Research in the above areas involves applying new technologies to analytical chemistry and chemical sensor research. It is intrinsically multi-disciplinary, with contributions from analytical chemists, materials scientists, electrical engineers, and professionals in other fields. At the initial stages of research, the interest is generally focused on exploring and proving the principles by which, a new technology can be applied to measure a chemical substance. Open access to specialized equipment and facilities, such as those required for lithographic patterning, can be crucially important to foster interest and progress as applications to specific practical analytical and chemical sensing measurements start to appear. As previously mentioned, the most important materials-related opportunities to improve directreading chemical sensors involve the choice of materials employed to elicit stable selectivity of interaction with the target analyte.
The most frequent materials limitation for chemical sensors probably relates to the chemistry required to fashion an adequately selective response to the target analyte. Considerable potential exists to enhance the selectivity of direct-reading chemical sensors by the use of novel materials. One strategy to address this is the development of miniaturized high-speed separationsbased sensors. These have the potential for avoiding difficulties in molecular selectivity but present major challenges in improving detector sensitivity. Miniaturized total analytical systems are a relatively new area of research in analytical chemistry, but their development could greatly supplement the capabilities of existing direct reading sensors. The numerous materials issues in designing and fabricating low-cost, miniaturized separationsbased analytical systems include:
• Coatings and films with improved properties for enhanced sensor performance (e.g., chemical selectivity, chromatographic efficiency, stability under electric field gradients, electrocatalysis efficiency);
• Materials that enhance detector sensitivity and increase performance range (fiber optics)
Research efforts directed at determining which chemical sensing technologies are practical and should be developed for high-volume home and personal wellness applications are expected to have particularly high payback. New materials can lead to improvements in the selectivity of direct chemical sensors. The development of fast, miniaturized chromatographic and capillary electrophoresis systems with detectors that are sensitive to chemical structure is important for both general chemical sensing and for the more specific case of environmental monitoring. In the latter case, the requirement to monitor a given analyte over a wide range of concentrations and in a variety of environments places particularly stringent requirements on chemical sensor sensitivity and selectivity. The need to meet and possibly redefine regulatory requirements for monitoring toxins is also an important driver in the development of environmental chemical sensors. Mass-producible sensor formats are particularly important for occupational environmental monitoring in view of the need for low-cost compliance with regulatory requirements.
These materials requirements are very similar to some identified previously as being important in improving the selectivity of other direct-reading chemical sensors. It should be noted that selectivity is specific to a particular compound or class of compounds. Little potential for dual-use applications (or other secondary applications) is anticipated for coatings developed for the detection of chemical warfare agents. Nonetheless, the general lessons learned in developing chemically selective materials (understanding the role of electrical and chemical forces on surface and interfacial phenomena, molecular characterization of ion-specific membranes and modified surfaces with catalytic or enzymatic properties, etc.) can be broadly applicable and should be of help in designing materials to meet particular functional requirements.
The possibility exists of leveraging generic miniaturization techniques, including materials and processing technologies developed for mass market applications, in order to further develop compact, lightweight hand-held sensor systems for chemical weapons detection. Miniaturization techniques of particular interest include methods relating to supporting electronics and protective packaging.
Sensors & Devices for Heavy Metals in Water
A great challenge in the area of heavy metal trace detection is the development of electrochemical techniques and devices which are user-friendly, robust, and selective, with low detection limits and allowing fast analyses. This review presents the major contribution of the French scientific academic community in the field of electrochemical sensors and electro analytical methods within the last 20 years. From the well-known polarography to the up-to-date generation of functionalized interfaces, the different strategies dedicated to analytical performances improvement are exposed: stripping voltammetry, solid mercury-free electrode, ion selective sensor, and carbon-based materials, chemically modified electrodes, nano-structured surfaces. The paper particularly emphasizes their advantages and limits facing the last Water Frame Directive devoted to the Environmental Quality Standards for heavy metals. Recent trends on trace metal speciation as well as on automatic “online” monitoring devices are also evoked.
Like many other micropollutants such as drugs or cosmetics and their by-products, pesticides and industrial or household chemicals, heavy metals represent a growing environmental and health problem. They may be considered as a major source of ecological issues due to their wide overspread in natural media. Although naturally produced throughout biogeochemical processes, heavy metals occurrence in the environment mainly originates from human activities: air emissions from coal-burning plants, smelters, and waste incinerators, process wastes from mining, industrial and urban runoff all participate to their wide spreading. Once released to the environment, these metals can remain for decades or centuries since they are not biodegradable. Depending on the contamination pathway, they appear at detectable levels in food resources such as vegetables, grains or fruits, and fish or shellfish throughout bioaccumulation all along the trophic chain, thus contaminating the final consumer human being. Another contamination way is direct intoxication from domestic environment, for instance lead traces in household plumbing and old house paints.
Once penetrated inside human organism by ingestion (drinking or eating), inhalation or skin contact, heavy metals may be responsible for nausea, vomiting, diarrhea or allergic reactions for short term or low-level exposure. They can also cause severe diseases in the case of long term or chronic high-level exposure, such as reduced growth and development, cancers, organs or nervous system damages and even there are over 50 elements that are classified as heavy metals, including transition metals, some metalloids, lanthanides and actinides. Among them 17 are considered to be both very toxic and relatively accessible. Lead (Pb), mercury (Hg), arsenic (As), and cadmium (Cd) are generally considered as leader elements in human poisoning even at trace level. The general population is mainly exposed to all these metals from air, drinking water and food, fish being a major source of mercury exposure.
Moreover, smokers are highly exposed to cadmium. Some other heavy metals, including copper (Cu), zinc (Zn), nickel (Ni), cobalt (Co), selenium (Se), and bismuth (Bi) are known to play a vital role in physiological concentrations but can also be toxic in larger doses. Depending on the metal properties, the toxicity target may be different: kidneys or liver are damaged by Cu, Hg, and Pb ions while these latter two also attack central nervous system. Thus, toxicity levels are related to the nature of the metal but also to its biological and biogeochemical roles, both being strongly dependent on its speciation, i.e., the different available physico-chemical forms, namely particulate (size>1μm), colloidal (1nm-1μm) and dissolved (≤1nm) species. These latter include free metal ions, simple inorganic complexes and complexes bearing anthropogenic and natural organic ligands. Hence speciation information on heavy metals of concern appears to be data of particular relevance.
In 2000, a new European directive particularly pointed out four heavy metals (Hg, Cd, Pb, and Ni) and has established their maximal authorized as well as annual average concentration values in surface waters. As a consequence, environmental monitoring of heavy metals is of critical importance for both ecological assessments and public health preservation. In answer there is an urgent need for in-situ, real-time, and highly sensitive sensors in order to multiply control points dedicated to early warning pollution alert. Heavy metals trace detection is mainly performed using spectroscopic techniques: atomic absorption spectroscopy, inductively coupled plasma mass spectroscopy (ICP-MS), X-ray fluorescence and neutron activation analysis are the most commonly used. Their main advantages are their versatility since they are suitable for a large panel of elements, their sensitivity and their limit of detection (LOD) in the femtomolar range.
However, they suffer from several major drawbacks: expensive materials are required and qualified operators are needed to perform the multi-step sample preparation and complex analytical procedures, which are unsuitable for onsite and on time measurements necessary to prevent transient phenomena monitoring. Finally, only total metal concentration can be determined, and speciation data can be reached only by associating supplementary extraction and separation techniques such as chromatography to the spectroscopic detection. These additional steps significantly increase the risk of contamination of the sample and some modifications of the speciation may occur during sample storage or handling.
On the contrary electrochemistry represents an interesting alternative due to its numerous advantages. Electrochemical devices are mostly user-friendly since they require simple procedures. They are also reagent less, low cost, and well-suited for miniaturization and automatic in situ measurements with minimal sample changes. Thus, contamination by reagents or losses by adsorption on containers are drastically decreased. Electrochemical systems also allow quite fast analyses with experimental data obtained mostly in real time or in a few minutes. Hence, on-line monitoring of water samples becomes possible, providing dynamic data of relevance for biogeochemical survey. Nevertheless, specific developments are still required for such applications, particularly to improve sensitivity, LODs and automation
In this way, a large number of electrochemical techniques with different imposed potential or current modulations have been developed such as differential pulse voltammetry (DPV), square wave voltammetry (SWV) or Stripping Chronopotentiometry (SCP). Electrochemical sensors also allow high temporal resolution measurements to be obtained when associated to flow injection analysis (FIA) or flow electrochemical analysis cells, thus providing continuous in situ measurements. Another analytical performance of high relevance with respect to heavy metals detection concerns selectivity. In complex media, the signal of the analytical target often experiences interferences due to the presence of other species (sometimes other heavy metals). To solve this problem, several surface functionalization strategies have been developed for many years to improve sensors selectivity.
Many pioneering researchers coming from several countries have initiated and intensified works dealing with electrochemical techniques for heavy metals detection and assay in natural media. This is mainly due to the voluntary policy lead since 1998 and the Aarhus protocol in which France contracted to limit its release of Pb, Cd, and Hg at a lower level than that recorded in 1990. This goal was reached before the protocol came into effect in 2003, but the situation is still worrying: over the 2007-2009 period, 25 heavy metals have been detected in more than 10% of the analyses performed in French rivers), whereas the contamination of mussels and oysters, which constitute a good indicator of coastal water pollution, remained stable over the last 3 decades. This review provides a survey of French groups’ contribution to the development of electrochemical sensors and methods aiming at heavy metals detection. The paper particularly emphasizes the multidisciplinary of French scientific investigations through the description of the electrochemical techniques and the evaluation of the corresponding analytical performances.
Polarography
Polarography has been certainly the most studied and commonly used electrochemical technique throughout the 20th century since the pioneering work of Heyrovsky in 1922. This is undoubtedly the consequence of the particular properties of the mercury electrode: continuous renewal of the active surface area, wide cathodic potential window due to the high overpotential corresponding to hydrogen evolution, control of the hydrodynamic conditions by means of mercury drop. These characteristics make polarography a very powerful electrochemical technique for the study of inorganic, organic, organometallic, or biological compounds, not only from a theoretical point of view but also for analytical applications. In this frame the assay of heavy metals has been the subject of numerous papers due to the large inclination of mercury to form amalgams with major metal compounds.
For concentrations higher than 10-5 M linear sweep voltammetry (LSV) on a dropping mercury electrode (DME) or on a static mercury drop electrode (SMDE) generated at the end of a glass capillary is well-suited. For lower concentrations, the faradic current becomes smaller, and the double-layer charging current is not negligible anymore. Pulse techniques, i.e., normal pulse (NPV), differential pulse (DPV) and square wave (SWV) voltammetries have been favored to partially suppress the background current and thus improve the LOD. In the case of trace metals detection these potential pulse programs have been associated with anodic (ASV) or cathodic (CSV) stripping voltammetries on a hanging mercury drop electrode (HMDE) inside which the analyte is preconcentrated by constant potential electrolysis prior to analysis. The resulting methods, i.e., LSASV, DPASV and SWASV and their combination, allow LODs down to 10-12 M to be reached (Bard and Faulkner, 2001). For instance, Superville et al. assembled an automatic anodic stripping analysis system with a SMDE to undertake a real-time routine analysis of the dynamic behavior of trace metals (Zn, Pb, Cd) in rivers, pond and seawater. Furthermore, a CSV was included to estimate simultaneously the concentration of dissolved oxygen and reduced sulfur species. Magnier et al. perfected a procedure to assay lead and zinc by ASV and copper by CSV in certified reference freshwater and in the French Deûle river, Cu analysis requiring the complexation with 8-hydroxyquinoline [91,92].
The need for determination of very low concentrations has favored the development of specific electrochemical techniques with new potential perturbation modes providing high resolution and/or improved sensitivity. In this way Zlatev et al. particularly emphasized the advantages of differential alternative pulses voltammetry (DAPV) on HMDE to analyse mixtures of species exhibiting very close half-wave potentials (like Pb2+ and Tl+ or Co2+ and Ni2+) or species couples with high concentration ratios for which the analysis by DPV is hampered by complete peaks overlapping. DAPV takes advantage of the high-resolution power of the second order voltametric techniques (as radiofrequency polarography) combined with the high sensitivity and instrumental simplicity of DPV or SWV. DAPV principle is based on the superimposition on the main electrode potential E of a pair of single successive rectangular pulses characterized by small, equal amplitudes (RT/nF) and durations (from 1 to 100 ms) but opposite polarities.
The overall current recorded at this potential E corresponds to the deviation of the average of the corresponding cathodic and anodic amperometric responses. Thus, the resulting currentpotential curve exhibits the typical shape of a first-order peak derivative passing three times through zero (for potential ranges corresponding to residual and diffusion-limited currents and for the electrode potential equal to the half-wave potential) with peak amplitudes proportional to the electroactive species concentration. The resolution power of DAPV was highlighted through the analysis of a solution containing Pb2+, Tl+, In3+, and Cd2+. The sensitivity and the LOD (54 nM) were found to be similar to those obtained using classical DPV but with species having half-wave potentials difference in the range from 28 to 50 mV and concentration ratios from 1:1 up to 80:1 without any preliminary preparation of the sample.
Mercury Film
Despite very good analytical performances in terms of sensitivity and stability of the response vs. time, the low vapor pressure and the high toxicity of mercury encouraged extensive research on polarographic methods involving reduced amounts of mercury. One way consists in thin film mercury electrodes (TFME) electrodeposited on solid state materials like glassy carbon (GC). The group of Riso used SCP with low constant current for the quantification of Fe(III) in estuarine and coastal filtered waters. The procedure was proved to be highly sensitive, but analysis required several pre-treatment steps, i.e., filtration of the sample and complexation with solochrom violet. They also succeeded in detecting ultra-trace (70 pM) Sb(III) in seawater by using the same electrochemical method. The application of a double electrolysis potential during the pre-concentration step allowed the analysis to be independent from the Cu level. For the determination of Se(IV), insufficient reproducibility and sensitivity of Hg film was observed by. This problem was overcome by incorporating Cu(II) ions during the plating procedure on GC electrode surface. The metallic Cu dissolved in the Hg film was found to play an important role in peak current enhancement. A LOD 0.8 nM was reached by SWCSV and the relative standard deviation was 5.2% (n = 5) for 1 μM Se(IV).
A potentiometric stripping method has been proposed by Riso et al. for the simultaneous measurement of Cu, Pb, and Cd in ocean waters. The mercury coating was electrodeposited in situ on a GC rotating electrode at the beginning of each analysis by applying a potential step at -1.1 V/SCE for 10 min. Then an electrolysisstripping cycle was carried out. Metals concentrations were compared with a reference standard solution containing all three metals. The obtained LODs were 0.7 nM, 15 pM and 9 pM for Cu, Pb, and Cd, respectively. However, it has to be noticed that the total duration of the analysis was quite long, about 75 min.
In order to approach solid mercury-free electrodes, Munteanu et al. worked on the electrodeposition of a mercury monolayer by constant potential electrolysis with increasing electrolysis time.
An exceptional sensitivity for Pb2+ assay was obtained when the mercury monolayer-on-carbon electrode was used with fast (v > 1 kV/s) ASV. This result was revealed to be due to the ionization of Pb atoms in the mercury layer, which catalyzes the oxidation of atomic hydrogen adsorbed on the Hg layer. A remarkable LOD of 80 fM was recorded on a cylinder electrode with a 1 s preconcentration time.
Mercury Screen Printed Electrode (HgSPE)
Potin-Gautier’s group has developed an alternative strategy based on mercury micro arrays screen printed electrodes (SPE). This micro system allowed mass transport of the analytes to be enhanced compared to macro systems. This device was successfully tested for Cd2+ detection in synthetic and river samples, providing a LOD of 11.6 nM using SWASV. More recently SCP was implemented as the second stage of the electrochemical technique “Absence of Gradients and Nernstian Equilibrium Stripping” (AGNES-SCP) for the determination of free metal concentration, namely Zn2+, Cd2+, and Pb2+, with both an HMDE and a SPE. The results showed higher sensitivities and lower LOD and LOQ (in the nanomolar range) with SPE, which was linked to a higher product of mercury volume times the gain (AGNES parameter). Finally, SPE was modified by a microwell for the assay of labile Cd2+, thus reducing the sample volume down to 200μl. A LOD of 2 nM was reached using SWASV with only a 60 s pre-concentration step. Unfortunately, the performances of this electrode were pH-dependent out of the pH range 4-7. Nevertheless, all these mercury-based techniques are promised to disappear in a few years since no mercury will be authorized from 2015.
Bismuth
Bismuth film electrode (BiFE) is often considered to be a good alternative to Hg electrode and has been extensively used for electroanalysis. More “environmentally friendly” and less toxical than Hg, Bi is considered to be a safe material, as it is a noncarcinogenic element (except for foetus and embryo). However, in high doses, it presents similar toxicity to other heavy metals apart from these effects are much more reversible. The main advantage of Bi with respect to trace analysis is its capability to form binary or multi-component alloys with numerous other heavy metals. It also has the particularity of being the most diamagnetic metal, thus avoiding conductance problems. Another interest of Bi compared to Hg is its insensitivity to dissolved oxygen, thus making unnecessary any deaeration step.
Most of the time, the Bi film is plated before analysis onto the electrode by potentiostatic reduction of a Bi(III) solution, although code position during trace metal reduction has also been reported. BiFE performances compare favorably with Hg electrodes, affording high sensitivity and well-defined stripping signals. For instance, Guo et al. compared the anodic stripping voltametric response of HgFE and BiFE obtained in a 10 mM Zn(II) solution. The BiFE presented a well-defined and higher stripping peak compared to HgFE. Its sensitivity was found to be twice as high as that obtained on HgFE. This electrode was then used to detect zinc contained in milk vetch used in traditional Chinese medicine. The response was linear in the range from 0.5 to 3 μM and a LOD of 10 nM was reported. Nevertheless, it has to be noticed that the cathodic limit of the potential window is higher than on Hg.
In 2005, Legeai et al. proposed an interesting alternative to the classical Pt or GC substrates by electrodepositing Bi film onto Cu since the adherence of the film was found to be better in this latter case (Legeai et al., 2005). By using DPASV as the detection method, this BiFE exhibited very good performances toward Cd2+ assay in acidified tap water with a linear concentration range between 10-8 and 10-6 M. It was also successfully tested for the simultaneous determination of Cd2+, Zn2+ and Pb2+ ions at 10-5 M. Good accuracy (<5%) was confirmed by comparison of the electrochemical data with those obtained by ICP-MS and mercury-drop electrode on aquatic plant extracts. These results were later generalized to Ni2+ analysis, by using adsorptive stripping voltammetry and dimethylglyoxime as the complexing agent (Legeai et al., 2006).
However, BiFEs suffer a major drawback since the formation of Bi hydroxides, which occurs at pH higher than 4.3, makes the analytical results irreproducible due to film surface changes. To overcome this problem, Legeai et al. used a Nafion-coated Cu substrate for Bi electrodeposition and observed that the Bi particles which were incorporated into the membrane did not experience any hydroxylation reaction (Legeai and Vittori, 2006). This system was successfully used for Cd2+ and Pb2+ detection and exhibited good stability (only 10% decrease after 23 days).
In a very close approach, Urbanova et al. succeeded in increasing the active surface area of the electrode by depositing Bi onto a polystyrene spheres template (Urbanova et al., 2010). After polystyrene dissolution by toluene washing, the resulting ordered porous film exhibited an original three-dimensional structure which significantly enhanced the analytical performances. These latter were found to be strongly correlated to the Bi film thickness. By using the thickest, porous film (2.2 μm thickness) and DPASV method, linear concentration ranges between 178 nM and 1.16 μM for Cd2+ ions and between 96.5 and 627 nM for Pb2+ ions were obtained. The LODs were 5.3 nM and 6.3 nM respectively, with a pre-concentration time of 300 s. The standard deviations were lower than 4% in both cases.
Finally, the best performances of Bi electrodes were reported by Lu et al. for trace metal vapors detection (Lu et al., 2010). In this case, Bi powder was simply mixed with carbon ink and the resulting Bi SPE was covered with a 0.1 mm thickness hydrogel layer by immersion in an agarose solution. This latter acted both as solid electrolyte and preconcentration agent, allowing Pb vapor concentrations between 48 and 386 pM to be determined with a LOD around 4.8 pM. Similar results were obtained with Zn and Cd.
Carbon Electrode Materials
Nowadays, particular attention is paid to more ecofriendly solid electrodes. GC or graphite felts are the easiest carbonbased material to obtain (structured with sp2 carbon atoms). Alternatively, Boron doped diamond (BDD) or diamond like carbon (DLC) with sp3 carbon atoms structure are very interesting for their specific properties (McCreery, 2008).
Graphite
Graphite felts are porous electrodes made of 10 μm diameter carbon fibers. They generally present a high specific surface area (around 1200 cm2.g-1) and a high void volume (around 90%) which enhances hydrodynamics, thus improving the LOD of the resulting sensor. Feier et al. exploited these particular properties to elaborate a flow sensor for Zn2+ trace analysis (Feier et al., 2012). A LOD of 500 nM was reached by LSASV with a 5 min duration cathodic preconcentration step. Nevertheless, the selectivity toward Zn2+ detection was strongly affected by several interfering species such as Fe3+, Cu2+, Co2+, and Ni2+. Nasraoui et al. elaborated a Pb sensor by using a similar flow cell and the same electrochemical method (Nasraoui et al., 2009). A LOD as low as 1 nM was obtained for an overall analysis time of 11 min.
In order to increase the sensitivity, Khadro et al., elaborated a SPE involving active surface carbon deposit (1 mm diameter) for the assay of Hg2+ and Ni2+ (Khadro et al., 2009). Using DPASV as mutual detecting mode, the sensitivity was respectively 10 and 20 times higher than those obtained with a traditional batch electrochemical cell including a three times higher diameter working electrode. Consequently, the LODs decreased to 0.15 and 2.56 nM for Hg2+ and Ni2+, respectively. Despite satisfactory performances, the main drawback concerned electrode fouling which obliged the regular regeneration of the surface between analyses and reduced the electrode life-time: Nasraoui et al. observed a decrease about 10 and 20% of the electrochemical signal after the second and third regeneration, respectively (Nasraoui et al., 2009).
Amorphous Nitrogenous Carbon Thin Film (A-Cnx)
a-CNx thin film is a carbon-based material (around 80% sp2 carbon) including nitrogen atoms. It is synthesized by a radio-frequency magnetron sputtering technique. The amount of nitrogen incorporated into the film can be controlled by the composition of the plasma (N2/Ar ratio) used for the deposition. The main property of a-CNx electrodes concerns their broad electrochemical window, which makes it particularly suitable for electrodetection of many species. This was exploited by Seck et al. for the simultaneous assay of Cd2+ and Cu2+ (Seck et al., 2012). The presence of Cu modified the peak current related to Cd2+ as compared to Cd2+ detection in Cu-free solution. Anyway the a-CNx electrode allowed the detection of 2 ppb of Cd2+ with concentration of Cu2+ up to 140 ppb.
Diamond Like Carbon
Diamond like carbon (DLC) is a carbon-based material containing a mixture of sp2 (graphite) and sp3 (diamond) carbon phases. Several methods have been developed to produce DLC films: plasma enhanced chemical vapor deposition (CVD) techniques, ion beam, filtered cathodic vacuum arcs. DLC exhibits some unique properties, such as high elastic modulus, high mechanical hardness, very low surface roughness and chemical inertness. Khadro et al. used DLC doped with boron to improve its conductivity (Khadro et al., 2011). The resulting electrode was exploited for the simultaneous assay of many heavy metals in water, namely Cd2+, Pb2+, Ni2+, and Hg2+, in concentration ranges up to 200 nM. LODs of 8.9, 4.8, 34, and 5 nM were reached, respectively.
1.2. Boron Doped Diamond
Boron doped diamond (BDD) is the most recent carbon-based material used for electroanalytical purpose. Diamond films can be deposited using CVD systems involving activation of gases by either microwave plasma or a hot filament. Traditionally electrical conductivity of diamond films is obtained through doping with boron (p-type behavior). The advantages of such material are manifold compared to previous carbon-based ones: BDD has an extremely high chemical stability, presents a wide potential window in aqueous media (-1.35 to +2.3 V/NHE at 0.1 mA cm-2 in 0.5 M H2SO4) and generates a low background current. Moreover, it is extremely resistant to fouling phenomena, thus making its surface state very reproducible with time. Unlike graphite felt, BDD exhibits however lower specific area. In order to increase the sensitivity two major ways have been envisaged. In the one hand, Le et al. associated a BDD electrode with a micro-electrodialyser as a pre-concentration step (Le et al., 2012).
The corresponding device allowed the assay of Pb2+ ions with a linear concentration range between 96 and 490 nM and a LOD of 19 nM. The same analytical device was used for the simultaneous detection of Zn, Cd, Pb, and Cu (El Tall et al., 2007). Quantification was possible for the first three heavy metals, but the presence of Cu caused interferences. Compared to GC, BDD electrode exhibited an enhanced sensitivity (3 or 5 times) and a longer lifetime in real samples (El Tall et al., 2007). In the other hand, Sbartai et al. developed a new electrochemical microcell micromachined by a femtosecond laser for the simultaneous detection of Cd, Ni, Pb, and Hg (Sbartai et al., 2012). Reduction of the electrode size resulted in mass transport amplification. LODs of 0.4, 6.8, 5.5, and 2.3 nM were thus obtained, respectively. Quantitative results were recorded for concentrations up to 200 nM. These performances are comparable to those obtained on DLC by Khadro et al. for Pb2+ and more accurate for Hg2+, Cd2+, and Ni2+ (Khadro et al., 2011).
However, a non-linear calibration curve for Hg was obtained in the former paper, which can be explained by the presence of Clions in the electrolyte solution, leading to Hg2Cl2 formation at the electrode surface.New carbon materials like a-CNx, DLC or BDD which exhibit high chemical stability and offer a wide potential window hold significant promises for electronalytical applications. Moreover, there is a wealth of opportunities for nanoscale electrochemical devices based on carbon materials. Nevertheless, interceptions between Cations go through either the development of specific calibrations or the chemical modification of the electrode.
Ion Selective Electrodes
Ion-selective electrodes (ISEs) are potentiometric sensors that involve a selective membrane which minimizes matrix interferences (Bobacka et al., 2008). The response of these sensors is based on an equilibrium state complexation reaction between the analyte and the probe with kinetic properties strongly depending on the membrane composition. The potentiometric measurement as well as the nature of the molecular interactions generally allow LODs around 1 μM to be reached and a linear concentration range from 10-5 to 10-2 M in a pH window from 3 to 6. Innovative changes have been made in recent past years to improve these analytical performances. In this context several functionalized materials have been used, including glass, liquid or polymer membranes. In the latter case an ionophore is generally used as the sensing platform. In this way, Yaftian et al. first synthesized phosphorylated calix arene coated on a graphite electrode for the assay of Pb traces (Yaftian et al., 2006).
This sensor exhibited a particularly quite long lifetime (up to 8 weeks) and a relatively short response time (17 s). This latter was significantly shortened to 7 s by using hexahomotrioxacalix arene as the ionophore, due to fast exchange kinetics complexationdecomplexation processes (Yaftian et al., 2007). Furthermore, the concentration range covered four decades (between 10-8 and 10-4 M). The specificity of this ISE toward Pb2+ was successfully tested in synthetic solution in the presence of 22 interfering species. Nevertheless, no measurement was done in real sample water. In both cases, the electrode was also used in potentiometry for the titration of Pb2+ solution using a standard EDTA solution.
Glassy materials represent a good alternative, particularly in micro-sensor fabrication. Beyond the advantages of all solid-state devices, vitreous materials are well-suited for the production of homogeneous thin layers which allow potentiometric measurements to be done despite their poor conductivity. Thereby, several types of solid-state membranes have been synthesized like the widely used chalcogenide one, in which the conduction over the membrane is provided by halides or metallic ions. Chalcogenide glasses exhibit better chemical durability in acidic media, and in many cases, afford better selectivity and reproducibility than arene ionophores. Several French research teams have investigated such ISEs toward Cu(II) determination. Cali et al. used chalcogenide glassy-crystalline Cu-As-S alloys (Cali et al., 2002). The resulting sensor exhibited a very short response time (1-3 s) and a LOD close to 10-6 M with a linear concentration range between 10-6 and 10-2 M. These results are available within a pH range from 2 to 6.
The resulting sensor exhibited a very short response time (1-3 s) and a LOD close to 10-6 M with a linear concentration range between 10-6 and 10-2 M. These results are available within a pH range from 2 to 6. Similar analytical performances were obtained by Essi et al., with a Cu-Ag-S thin film for the assay of Cu(II) & Ag(I) ions (Essi and Pradel, 2011). They were satisfactorily compared with those obtained by ICP-MS in real samples. Furthermore the specificity of the sensor was not damaged by the simultaneous presence of Ca2+, Mg2+, Pb2+, Cd2+, and Zn2+ (Essi and Pradel, 2011). Mear et al. investigated a thin film of Ge28Se60Sb12 chalcogenide glass including Cu in order to quantify Cu(II) ions (Mear et al., 2005). A linear range was found between 10-5 and 10-3 M with a LOD of 3 μM. The concentration range was enlarged between 10-6 and 10-2 M for Cd(II) ions assay by coupling the membrane electrode with a pre-concentration module.
Chemically Modified Electrodes
From the analytical point of view, the wide and increasing success of chemically modified electrodes (CME) may be explained by the offered possibility to purposely design the surface of conventional electrodes. By combining the intrinsic properties of the modifier and a given electrochemical reaction, CMEs exhibit significantly improved response compared to unmodified electrodes (Murray et al., 1987; Gilmartin and Hart, 1995; Cox et al., 1996). For heavy metals trace detection, the modification plays a critical role especially during the preconcentration step, by favoring selective and enhanced accumulation, thus leading to higher sensitivity and lower LODs (Arrigan, 1994). During the detection step, the modification also often favors electron transfer kinetics. The modifier may be a mineral such as silica or clay, a polymer, an inorganic or organic compound or a metal nanoparticle-based material. Depending on its nature, the modifier is associated to the electrode by adsorption, covalent binding, coating or even dispersion into a conductive matrix.
Minerals
Minerals such as silica-based materials, clays and zeolithes, are of particular interest for ion exchange voltammetry (IEV) (Wang, 1989). Basically, they act as an ion selective film inside which the analytical target is preconcentrated at open-circuit potential by an exchange process. In a second step, the analyte incorporated within the ion-exchanger film is detected by using an anodic stripping technique (Walcarius, 1998).
In France, the research on silica-modified electrodes with respect to heavy metals assay is mainly represented by the group of Walcarius. In 1997 this group published the first report dealing with silica-modified carbon paste electrode (SMCPE) devoted to electroanalysis, with Cu(II) as the analytical target (Walcarius and Bessiere, 1997). By using a 10 min preconcentration step and SWASV, a LOD of 2 nM was reached. This SMCPE exhibited a good reproducibility since up to 30 detection procedures were performed over a period of a week without any noticeable loss of sensitivity. However, this system suffered the classical drawback of CPE, namely a gradual dissolution process. In this particular example, another severe drawback was the necessary use of an ammonia medium in order to ensure Cu(II) accumulation via its interaction with surface silanolate groups. This pioneering work has been later extended to various silica-based materials and the influence of interfering species has been studied (Walcarius et al., 1999a).
Clearly, this SMCPE failed at high ionic strength since important cations concentration resulted in competition for the ion-exchange sites. A very similar study has been reported with Hg(II) as the analytical target (Walcarius et al., 2000). Using the same procedure and materials, Hg(II) has been found to suffer no influence of ionic strength and no particular interference with other metallic species, even Ag(I). This result has been explained by considering the formation of soluble Hg(II) hydroxides in the experimental conditions used.
To overcome the drawback of adding a complexing agent in order to ensure metal accumulation, the group of Walcarius has developed several organically modified silica CPEs. Aminopropyl groups (Walcarius et al., 1999b; Etienne et al., 2001) or a carnosine dipeptide (Sayen et al., 2003) have been co-condensed with silane precursors to afford the desired functionalized silica materials. Cu(II) was detected with similar LODs to those reported in the former study (Walcarius and Bessiere, 1997) without adding anything to the accumulation medium. The aminopropyl-grafted silica CPE was successfully tested for Cu(II) detection in laboratory tap water (Etienne et al., 2001). Cu(II) suffered important competition from Co(II), Ag(I), and Hg(II) for the binding sites of the carnosine-modified silica CPE, thus limiting the practical usefulness of this sensor in real media (Sayen et al., 2003).
An interesting alternative to CPE has been proposed later by using silica films coated onto Au (Walcarius and Sibottier, 2005) or GC (Etienne et al., 2007; Sanchez and Walcarius, 2010) electrodes. Whereas these films have been prepared most of the time by a classical surfactant-templated synthesis (Etienne et al., 2007; Sanchez and Walcarius, 2010) an original electrochemically induced sol-gel deposition has been also reported (Walcarius and Sibottier, 2005). All these films were functionalized either by thiol or amine groups. Cu2+ (Walcarius and Sibottier, 2005), Ag+ (Etienne et al., 2007) and Hg(II) (Sanchez and Walcarius, 2010) were the analytical targets, and the LODs obtained were 40, 6, and 24 nM respectively.
From a more general point of view, all these studies on silica-based modifiers proved that the key feature with respect to analytical performances, and particularly sensitivity, is the analyte diffusion inside the porous structure of silica, thus making porosity a more predominant factor than the amount of surface silanol groups (Walcarius et al., 1998). Thus, mesoporous silicabased materials, which exhibit well-defined three-dimensional structures, appear to be much more adapted for heavy metal preconcentration than amorphous ones (Walcarius et al., 2003), whatever they are functionalized (Ganesan and Walcarius, 2008) or not (Walcarius et al., 1998). When functionalized, the amount of organic groups is also of importance, its effect on the analyte preconcentration passing by a maximum, since too much organic groups lead to a decrease in pore size which hampers the accessibility of the binding sites (Etienne et al., 2007; Ganesan and Walcarius, 2008; Sanchez and Walcarius, 2010).
Clays may also be used to perform IEV. These minerals exhibit relatively large specific surface areas and ion-exchange properties associated to an ability to sorb and intercalate many compounds. In a very close approach to what has been reported for silicabased material, their surface may be functionalized by organic groups (Navratilova and Kula, 2003), affording the possibility to tune charge selectivity of clays in IEV (Tonle et al., 2004).
In France, the group of Walcarius explored the potentialities of clays functionalized by organic groups and mixed with CPE toward Hg(II) (Tonle et al., 2003, 2005) and Pb(II) (Tonle et al., 2011) determination. With respect to Hg(II), the comparison of thiol-functionalized vs. amine-functionalized clays showed that the former one exhibited a better LOD (68 and 87 nM, respectively, using DPASV with 10 min accumulation), in accordance with the greater affinity of Hg for sulfur groups (Tonle et al., 2005). Thiolfunctionalized clays have been also coated as thin films onto GC (Tchinda et al., 2007, 2009). The LOD has been greatly improved since a 6 nM value was reached using DPASV with only 3 min accumulation, the linear range being from 50 to 800 nM (Tchinda et al., 2007). It has to be noticed that increasing the accumulation time allowed another linear range to be observed from 4 to 20 nM together with a remarkable LOD of 0.5 nM (Tchinda et al., 2007).
Macrocyclic Compounds
Macrocyclic compounds have received much attention in the last two decades due to their particular three-dimensional shape which provides a suitable cavity for selective complexation of heavy metals (Kolthoff, 1979). French groups particularly focused their research on cyclams (Goubert-Renaudin et al., 2009a,b; Nasraoui et al., 2010a,b), crown-ethers (Parat et al., 2006; Betelu et al., 2007) and calixarenes (Dernane et al., 2013; Rouis et al., 2013).
With respect to these latter compounds, Dernane et al. proposed to detect Cd2+ thanks to a p-tert-butylcalix[8]arene membrane deposited onto Au electrode (Dernane et al., 2013). In this work the metal cation coordination was favored by the presence of the four ketone groups on the calixarene, thus allowing a 0.1 μM LOD to be reached. However, the results seemed a little bit cautious since detection was performed simply by CV without further preconcentration step. Moreover, the variation of peak current density as a function of the logarithm of metal concentration was proposed as a calibration plot, which makes non-sense with respect to classical equations of CV.
Rouis et al. have built an impedancemetric sensor dedicated to Hg2+ detection by immobilizing a β-ketoimine calix[4]arene derivative in a conducting poly(p-phenylene vinylene) membrane deposited onto indium-tin oxide (ITO) electrodes (Rouis et al., 2013). The main originality of this work was the study of light excitation effect of the β-ketoimine calix[4]arene while optimizing detection parameters. Charge transfer processes at the electrode/ electrolyte interface were found to be improved under light excitation, thus providing an enhancement of the sensitivity toward Hg2+. The best results were obtained under blue light emission, providing a charge transfer resistance Rct = 4.47 kΩ and a linear range from 0.1 nM to 0.5 μM.
Parat et al. reported the use of a thin-film mercury-coated screen-printed carbon electrode covered by a crown-ether based membrane for the determination of Cd and Pb (Parat et al., 2006). The crown-ether membrane aimed at protecting the active surface from interferences during the analysis. The size of crownethers cavity has been proved to impact the performances of the electrode. The best results have been obtained for the smallest crown-ether, because its cavity size fitted well to metal ions radii, although Pb was a bit favored. LODs of 6 nM and 8 nM for Cd2+ and Pb2+, respectively, have been reached by using LSASV with 2 min preconcentration. Tests were successfully conducted on river water and soil solution extracts. This system was then applied in a later work to the semi-continuous monitoring of Cd and Pb in tap water by FIA and afforded reproducible results for 42 h (Betelu et al., 2007).
In the first work dedicated to Cu(II) detection, the functionalization aimed at stabilizing cyclams on silica in order to improve the sensor lifetime. However, increasing the number of functionalization’s per cyclam lead to an increase in cycle rigidity, which has been correlated to lower performances with respect to Cu(II) uptake (Goubert-Renaudin et al., 2009a). The modified CPE afforded good stability, with a 7% relative standard deviation for 45 measurements and was successfully tested on tap water. In a second work, dealing with Pb(II) determination, the aim of the functionalization was to improve the analyte preconcentration step by favoring complexation (Goubert-Renaudin et al., 2009b).
Thus, the inverse trend was observed, i.e., the more functionalized the cyclam, the better the performance. The best system allowed LODs down to 2.7 nM to be reached. Among potential interfering species, only Hg(II), Cd(II), and Cu(II) gave rise to significant loss of signal, mainly because of competition for the binding sites. The group of Geneste also used such kind of cyclams to functionalize graphite felt electrodes (Nasraoui et al., 2010a,b). Accumulation of Pb(II) at open-circuit potential by flowing the aqueous solution to analyze, followed by LSASV allowed a 25 nM LOD to be reached. However, this system exhibited relatively poor stability since a 20% decrease of the response was reported after regeneration.
Polymers
Electroactive surface modification by means of polymer deposition or electrodeposition represents a broad research field, leading to numerous papers every year. With respect to trace metals analysis, polymer films allow the immobilization on the electrode of a large number of ligands which may complex metal ions to be accumulated (Trojanowicz, 2003; El Kaoutit, 2012; Li et al., 2012). The polymers used for surface modification may be natural or prepared purposely by chemical synthesis. However, with respect to French groups’ research activities, no work was found dealing with natural polymers dedicated to trace metal detection. The only papers found in the literature were about polysaccharides (Crini, 2005) and chitosan (Vieira et al., 2011) and concerned heavy metals removal.
Concerning chemically synthesized polymers, the group Moutet has developed two different groups of substituted polypyrrole derivatives for trace metal determination. The first one is based on “poly(pyrrole-EDTA like)” and takes advantage of the well-known complexing properties of EDTA to improve metal preconcentration (Heitzmann et al., 2007; Buica et al., 2009a,b). These studies were devoted to the assay of Cu(II), Pb(II), Cd(II), and Hg(II). In the first two ones, the selectivity of the modified electrode has been tuned by varying the accumulation time and the pre-structuration of the polymer. The film thickness was also proved to influence selectivity. However, the electrode was insensitive to Cd(II) whatever the conditions adopted. To overcome this problem, Heitzmann et al. have chosen an imprinted polymer strategy: the polymer was electrodeposited in the presence of Cd(II) ions which were then removed from the metallopolymer film (Heitzmann et al., 2007).
The resulting functionalized electrode was thus able to detect Cd(II) as well as the other three metal cations. By introducing 4 pyrrole fragments on the same EDTA skeleton instead of only two in the former studies, an enhanced stability and a better controlled dimensionality was conferred to the polymer, thus making the sensor response independent on the film thickness (Buica et al., 2009a). The global complexing capability of the polymer was also greatly improved by the presence of 2 amine and 4 amide coordinating groups per monomer unit. The second group of polymers developed by the group of Moutet is based on poly(pyrrole-malonic acid) (Heitzmann et al., 2005; Pereira et al., 2011). Here, the analyte complexation occurred with the anionic form of malonic acid, which is known to easily coordinate various metal ions. This sensor was also tested with Cu(II), Pb(II), Cd(II), and Hg(II). It allowed LODs around 10-10 M to be reached for each metal cation, and exhibited good stability since the same current response was obtained for 2 assays at a 3 weeks interval using the same electrode stored without any particular precaution.
Another kind of polymer based on styrene units gave rise to one report by Moutet (Rivas et al., 2006). Styrene was copolymerized with acetamide acrylic acid or itaconic acid. These latter are hydrophilic whereas styrene is hydrophobic, thus providing interesting mixed properties to the resulting copolymers. The incorporated carboxilate groups were used for the accumulation and detection of Pb2+ but the system exhibited poor performances since the response was linear only in the narrow range from 10-5 to 10-3 M. Bessbousse et al. proposed a more sophisticated system based on a nanoporous β-poly(vinylidene fluoride) (β-PVDF) membrane (Bessbousse et al., 2011). The nanopores were further functionalized by track-etched poly(acrylic acid) (PAA), and thin, porous Au films were sputtered on each side of the membrane. This very sophisticated electrode has been proved to detect Pb2+ but no very clear analytical results were provided.
Polythiophene also gave rise to one report (Zejli et al., 2007). The polymer was electrodeposited on a Pt electrode by cyclic voltammety and used to detect Ag (I) by DPASV taking advantage of the inductive effect of the C-S dipole of thiophene units. However, the linear range was found to be very narrow, from 0.65 to 9.3 μM.
A contribution from the group of Noguet has also to be noticed (Yasri et al., 2011). In this original work, a graphite electrode coated by a 3,4 poly (ethylenedioxythiophene): poly(styrene sulfonate)- [PEDOT: poly (styrene sulfonate)] copolymer was used to detect Pb2+ by chronoamperometry at -0.35 V. The linearity range was from 2 to 100 nM and the LOD was 0.19 nM for 30 s accumulation at -0.65 V. The system exhibited good stability since only a slight decrease was noticed after 11 days. It was also successfully tested for the determination of Pb2+ in different vegetables extracts.
In order to improve the detection, limit the group of Chevalet exploited the high-resolution power of a multipulse electroanalytical method, namely multiple square wave voltammetry (MSWV) (Fatouros et al., 1986; Krulic et al., 1990). MSWV is based on the superimposition of several pairs of opposite pulses of constant amplitude on each step of a staircase waveform. However, MSWV differs from previously described DAPV by the electrode response: instead of the double cathodic and anodic current recorded in the latter case, MSWV response results in the integration of the successive currents. The MSWV-DD (DD for double differential) technique was used in combination with a Nafion-coated electrode for the determination of trace species like methylmercury (Moretto et al., 1999) and Fe (Ugo et al., 2001). The perfluorinated cation exchanger Nafion was used to preconcentrate the analyte and was simply deposited on a GC disk by droplet evaporation. The detection capabilities of this polymer-coated electrode combined to the sensitive MSWV-DD method allowed a calculated LOD down to 45 pM to be reached for methylmercury (Moretto et al., 1999).
Nano-Scaled Materials
During the last two decades, nano-scaled materials have aroused a great interest with respect to analytical applications due to their specific physico-chemical properties (Murray, 2008). Improvements resulting from those nanomaterials for electroanalysis are manifold: enhanced diffusion of electroactive species, higher effective surface area of nanoparticles (NPs), electrocatalytic and conductive properties, improved selectivity and higher signal-to-noise ratio. With respect to trace metal analysis, gold nanoparticles (AuNPs) are the most commonly used material (Lin et al., 2011; Liu et al., 2011). They can be obtained either by chemical or electrochemical ways.The French contribution to this topic is very recent, and the corresponding works all considered the structuration of the nanoparticle-based modified electrode to be a key feature with respect to analytical performances.
Ottakam Thotiyl et al. designed a multilayer arrangement of citrate capped AuNPs immobilized by a thiol group onto an Au electrode for the detection of As (III) (Ottakam Thotiyl et al., 2012). The anionic AuNPs were deposited layer-by-layer alternatively with cationic polyelectrolyte to afford a layered nanocomposite film. As(III) was detected by its electrocatalytic oxidation using DPV without any accumulation step, and a LOD of 0.48 μM was reported. This study evidenced a strong correlation between the amperometric response and the number of layers of the nanocomposite film.
Our group has developed a Hg(II) sensor based on GC electrode functionalized by electrodeposited AuNPs and has studied the influence of the electrodeposition method on the analytical performances (Hezard et al., 2012a,b). Namely, cyclic voltammetry, chronoamperometry and potentiostatic doublepulse were used. It was shown that both the electrodeposition mode and the charge used for the Au precursor reduction had a dramatic influence on the size and density of AuNPs. These latter two parameters were strongly correlated to the analytical performances: the best results were obtained for dense deposits of small NPs (Hezard et al., 2012b). By using a 5 min accumulation time and SWASV, a LOD of 0.4 nM was reached.
An important development in the frame of nano-scaled materials concerns single (SWCNTs) or multi-walled carbon nanotubes (MWCNTs) since their discovery in 1991 (Ijima, 1991). Their unique structure offers very interesting properties such as high specific surface area, high chemical stability, good electrical conductivity and adsorption capacity, which give rise to wide applications in electronics, composite materials, energy storage, and of course sensors (Fam et al., 2011). With respect to this latter domain the association of various CNT functionalization procedures, including organic (polymers, proteins…) or inorganic (metal nanoparticles, metal oxides…) modifiers and several transducing modes allowed the elaboration and development of chemical, gas, humidity, biomedical or environmental sensors which have been the subject of several reviews (Jacobs et al., 2010; Vashist et al., 2011; Gao et al., 2012).
Extensive international research have been made to assay heavy metals by using unmodified (Yue et al., 2012) or CNTs modified either with cysteine (Morton et al., 2009), thiacalixarene (Wang et al., 2012), Sb nanoparticles (Ashrafi et al., 2014) or with imprinted polymeric nanobeads (Rajabi et al., 2013), most of the time associated with ASV. They allowed Pb, Hg, Cu, Cd, or Zn to be detected down to 3 nM and in linear concentration ranges up to 7 μM. Surprisingly, to the best of our knowledge, no French research team has exploited CNTs for heavy metals detection yet. Interest has mainly focused on micro and supercapacitors (Ghimbeu et al., 2011), batteries (Carn et al., 2013) and energy storage devices (Sathiya et al., 2011) or biofuel cells (Both Engel et al., 2013; De Poulpiquet et al., 2013). Concerning sensors works essentially dealt with gaz sensors (Bondavalli, 2010) and above all enzymatic (Singh et al., 2013), RNA (Tran et al., 2013) and DNA (Zhang et al., 2011)-based electrochemical biosensors.
Biosensors
In general terms, a biosensor is defined as an analytical tool associating a bioactive compound (mono- or multi-enzyme system, antibody, and microorganism) which can specifically recognize species of interest and a transducing element (Frew and Hill, 1987). Thus, it may be viewed as a particular kind of CME in which the modifier is a biological element. Enzymes are known as good modifiers with respect to heavy metals since these latter often strongly inhibit enzymatic reactions (Dennison and Turner, 1995).
The group of Chovelon reported several works about conductometric biosensors based on Chlorella vulgaris, a microalgae the cell walls of which bear enzymes such as alkaline phosphatases and esterases (Chouteau et al., 2004, 2005). In both papers, the microorganism was immobilized onto interdigitated Pt electrodes using bovine serum albumin and glutaraldehyde as a crosslinker. In the first work, Cd2+ was detected as low as 8.9 nM but at least one hour was needed to obtain such a result. The second study (Chouteau et al., 2005) provided several improvements since both Cd2+ and Zn2+ were detected with an 89 nM and 0.15 μM LOD, respectively, but with a significantly lower exposure time (ca. 30 min). It has to be noticed that bioassays were found to reach a similar LOD to the biosensor but only after 4 h exposure time, illustrating the wise enzyme immobilization strategy used. Finally, an originality of this latter work was that the biosensor was also capable of detecting pesticides, taking advantage of the fact that esterases were selectively inhibited by these organic compounds whereas alkaline phosphatases were not affected.
Tekaya et al. have also reported a conductometric biosensor based on alkaline phosphatase from the cyanobacterium Arthrospira platensis (Tekaya et al., 2013) deposited on the ceramic part of interdigitated Au electrodes. In this case, inhibition measurements were performed after 24 h incubation, allowing LOD down to 10-20 M to be reached for both analytical targets, namely Cd2+ and Hg2+. Despite this very appreciable LOD, no information was provided about sensor lifetime. This is mainly due to the fact that the authors considered their study as a proof of concept aiming at providing a global response to the presence of heavy metals.
A last conductometric biosensor was reported 2 years ago by Soldatkin et al. (2012). This sensor consisted in a three-enzyme system prepared by mixing invertase, mutarotase and glucose oxidase with bovine serum albumin. Although a bit complicated, the sensor exhibited good sensitivities toward Hg2+ and Ag+ without experiencing any interference from organic compounds. After a relatively short incubation time (ca. 10-20 min), the LOD were found to be 25 and 100 nM for Hg2+ and Ag+, respectively. It has to be noticed that this biosensor was very selective since Cd2+, Zn2+, Ni2+, Pb2+, Cu2+, and Co2+ did not affect the enzymes activities.Interestingly, the authors have investigated a reactivation procedure: dipping the biosensor into EDTA or cysteine solution for 45 min allowed a significant reactivation of the enzymes together with the identification of the inhibitor metal, since EDTA reactivated Ag+ inhibition only whereas cysteine was selective to Hg2+ inhibition.
The group of Cosnier proposed an amperometric sensor toward Hg(II) based on the same three enzymes which were entrapped in a clay matrix deposited on Pt electrode (Mohammadi et al., 2005). The electrochemical response was provided by the oxidation of the enzymatically produced hydrogen peroxide. After 20 min incubation, Hg(II) was detected in the range 10 nM to 1 μM. It has to be noticed that several Hg species have been tested, such as methylmercury or phenylmercury, all of them leading to invertase inhibition. Depending of the analytical target, the biosensor recovery by using cysteine was more or less effective, but never complete. The system was quite selective to Hg(II) since all metal cations tested induced no interferences except Ag+.
Finally, the group of Burstein has reported 20 years ago a bi-enzymatic system coupled to an oxygen sensor (a Clark electrode) which offered an original solution to the enzyme recovery issue (Gayet et al., 1993). In this work, several enzyme combinations were tested but the most interesting one was that involving L-lactate oxidase and L-lactate dehydrogenase. Actually, the former enzyme was immobilized on the membrane of the oxygen sensor whereas the latter one remained in solution. Since only L-lactate dehydrogenase was affected by the presence of heavy metal cations, the sensor regeneration simply consisted in a renewal of the solution containing this very cheap enzyme. Unfortunately, the interest of this system was limited due to the relatively poor LOD obtained for the different analytical targets.
The Particular Issue of Trace Metal Speciation
In addition to the monitoring of total heavy metals concentrations in the environment, speciation analysis provides very useful complementary informations. However, speciation analysis often implies the determination of very low concentrations of minor species, typically in the range of nM to pM or even lower. Hence electrochemistry and especially anodic stripping techniques are particularly well adapted to such kind of analysis.
For instance, Bourgeault et al. studied dissolved Cu speciation and bioavailability in freshwaters by using DPASV, diffusive gradient in thin films and ISE, and compared their results to those obtained by modeling (Bourgeault et al., 2013). It was found that Cu accumulation in aquatic mosses was better correlated to the weakly complexed Cu species measured using DPASV than to free Cu concentration measured using an ISE, thus highlighting the contribution of electrochemical techniques to speciation studies. In a close approach combining DPASV and conductometry, Terbouche et al. examined the complexation of Zn(II) and Cd(II) by new humic acids from Algeria, these latter class of natural polymers being known to have an influence on heavy metals biogeochemical cycle and bioavailability (Terbouche et al., 2011). Based on the strong complexing capacities evidenced in this work, the authors suggested their humic acids to be used for metal uptake.
A last speciation study combining electrochemical techniques was provided very recently by Rotureau (2014). Here the already discussed “AGNES” technique was used for the determination of free metal in solution while SCP at scanned deposition potential (SSCP) allowed dynamic speciation information to be reached. This latter technique consists in performing classical SCP with varying deposition potentials, and then plotting the transition time as a function of the deposition potential (van Leeuven and Town, 2002). Rotureau’s work dealt with Cd and Pb speciation dynamics in clay minerals, and it was shown that the interaction of Cd with clays may be described as a chemically homogeneous, labile system over a wide pH range whereas strong pH effects were evidenced in the case of Pb.
SSCP has also been used by Parat et al. to study Cd speciation parameters in the presence of several ligands, namely nitrilotriacetic acid (NTA) and 2,6-pyridinedicarboxylic acid (PDCA) (Parat et al., 2011b). By using a Hg film SPE and a 60 s deposition time, a LOD of 2.2 nM was reached and some information upon speciation extracted. In particular, it was demonstrated that Cd-NTA complex is less labile than Cd-PDCA. It has to be noticed that classical SCP has been used by the group of Riso for the determination of Se(IV) and total Se in seawater on a HgFE (Riso et al., 2004). However in this case, speciation information was reached by tuning the experimental parameters of water pretreatment, the electrochemical ones remaining nearly the same. Thus, Se(IV) concentration was determined by performing SCP on untreated water, whereas total Se was reached by analyzing UV-digested water.
Among all anodic stripping methods, pseudopolarography is particularly well-suited to provide useful information on metal speciation in model solutions and in natural water. This electrochemical technique allows thermodynamic and kinetic properties of the different forms of metal ions to be reached. The current-potential curves obtained, called pseudopolarograms, depict the influence of the metal deposition potential on the peak height, the peak potential or the half-peak width of the subsequent anodic voltammogram. The shifts in peak potentials as well as the changes in the slope of the curve indicate a modification in the reaction kinetics or reversibility. These evolutions make it possible to discriminate between different fractions of metals, including free or labile species and electroinactive metal complexes, and to detect the presence of complexing ligands at low concentration. Compared to classical linear or pulsed voltammetry, pseudopolarography allows the speciation of trace metals at natural concentration level (generally less than nM). Pizeta et al. have taken benefit of this properties to detect Pb(II) in sediment and interstitial water with an iridium solid microelectrode covered by a thin mercury film (Pizeta et al., 2005).
The electrode sensitivity varied between 4 and 20 nA /μM and its LOD was about 30 nM. The amperometric response was repeatable enough, avoiding memory effect and electrode surface blocking. Particularly the results highlighted the existence of strong complexes in sediment and in non-filtered interstitial water with high pH. More recently this procedure was successfully applied for the study of the interactions of natural (NOM) or dissolved (DOM) organic matter with Cu and Zn (Louis et al., 2008; Nicolau et al., 2008) in seawater sample. The unpolluted marine water containing DOM about 1 mg L-1, measurements were possible in the latter case after concentrating DOM in large water volumes by nanofiltration and reverse-osmosis. Anyway, the results gave comprehensive elements on the role of marine DOM in metals speciation: the dependence of pseudopolarograms with pH clearly indicated the differences of DOM sites for Cu and Zn complexation regarding their stability and acidity constants. The main drawback is the time-consuming of the method which requires a set of anodic stripping voltammograms to be recorded for each pseudopolarogram.
To end this section dedicated to heavy metal speciation, let’s talk about some works which are certainly not the most original ones in terms of electroanalytical methods, but present particularly interesting automated systems. Zaouak et al. recently developed a system for Cd semicontinuous monitoring which allowed the determination of total and bioavailable Cd (Zaouak et al., 2010a,b). The analyses were performed using a Hg film deposited on a SPE, this latter electrode being integrated in a fully automated device allowing all the operations needed for a complete set of measurements. Thus, a measurement cell, a storage cell and a plating cell for the electrode renewal were comprised in the whole device, together with a hydraulic conveyor which allowed the three-electrode system to move from one cell to another. This automated device was tested on tap water by recording measurements every 160 min for 18.5 h and exhibited a LOD of 1.78 nM. Total and bioavailable Cd were obtained by adjusting the pH value of the treated water.
Another example of automated electroanalytical device was reported by Hezard et al. for Fe(III) and Fe(II) quantification in acid mining drainage water (Hezard et al., 2009). In this particular case, the redox species were concentrated enough to allow direct determination using an unmodified Pt electrode. Moreover, the presence of sulfide anion S2 - in acid drainage water induced an important negative shift in potential for O2 reduction, resulting in a well-separated reduction wave for Fe(III). Thus, both Fe(III) and Fe(II) were determined using a single potential scan without deaerating the solution. An originality of this work was that both sample renewal and hydrodynamic control were achieved by a fluidic system based on gravity instead of a pump
Sensing Heavy Metals Using Mesoporous-Based Optical Chemical Sensors
Heavy metal pollution is one of the more serious environmental problems; therefore, there is a constant demand for the development of new analytical tools for its monitoring. An optical chemical sensor represents a good alternative to classical instrumental methods. The mesoporous materials used in optical chemical sensors’ fabrications have properties such as high porosity, exceptional adsorption capacity, tuneable 3D shape, geometry, and morphology, which enable improved limit of detection, response time, and selectivity properties of optical sensors. In this review, we firstly present the properties of mesoporous materials, provide a brief description of sensing mechanisms, and briefly discuss the importance of continuous monitoring. Recent advances in those mesoporous silica-based optical sensors used for heavy metal detection have been reported and their advantages and limitations also discussed. This review covers publications that have appeared since 2008.
The monitoring of heavy metals within the environment, drinking water, food, and biological fluids has become essential due to the raising of environmental awareness and increasingly stringent regulations for pollution control. Heavy metals, by definition, are metals with densities of >5 g.cm-3. They are released into the environment mainly by industrial activities. In small quantities, certain heavy metals such as iron, copper, manganese, and zinc are nutritionally essential for a healthier life. However, heavy metals such as Hg, As, Pb, and Cd are highly toxic and carcinogenic, even at the trace level. The toxicity and bioaccumulative properties of most heavy metals make its control a top-priority environmental task.
Standard methods for heavy metal determination cover a wide range of laboratory-based techniques such as atomic absorption spectroscopy (AAS), inductively coupled plasmaatomic emission spectroscopy (ICP-AES), inductively coupled plasma-mass spectrometry (ICP-MS), and X-ray fluorescence spectroscopy (XFS). Although these methods are sensitive and selective they require time-intensive sample pretreatment and expensive analytical instrumentation, as well as highly qualified staff. On the other hand, optical chemical sensors (OCSs) have great potential for detecting multiple heavy metals on-site. OCSs are a group of chemical sensors in which electromagnetic (EM) radiation is used to generate an analytical signal in a transduction element. The interaction of this radiation with the sample causes the change of a particular optical parameter that can be related to the concentration of the analyte. OCSs usually rely on the principle of an immobilised indicator (organic dye) that changes its optical properties (absorption, transmission, emission, lifetime, etc.) on binding the analyte. When designing an OCS with the desired sensor characteristics, it is not only the selection of a suitable indicator, solid support (matrix), and immobilisation technique that is important but also the morphology and suitable functionalisation of such materials. Over the years, different optical chemical sensors have been introduced based on silica mesoporous materials for the determination of mercury, copper, zinc, and other heavy metal ions.
The purpose of the presented review was to provide a general overview on the latest studies relating to mesoporous silica-based optical chemical sensors for heavy metals’ determination. The papers in question mainly focused on the receptor part of the OCS and not on the development of the whole sensor system including the transducer and signal processing unit. The syntheses, properties, and other applications of mesoporous materials are already described in detail elsewhere.
Mesoporous Material
Over the past decade, mesoporous materials due to their highly porous natures combined with low absorption and emission within the visible spectra have been shown to be excellent candidates for OCSs. Mesoporous materials are a class of nanostructures with well-defined mesoscale (2-50 nm diameters) pores, surface areas up to 1000 m2/g, and large pore volumes (~1.0 mL/g). In general, these ordered mesostructured materials are formed from solution by the coassembly and crosslinking of network-forming inorganic species (typically oxides) in the presence of structure-directing agents (SDAs). The SDAs are typically surfactants or block copolymers that self-organise into mesoscale (2-50 nm) structures, according to the solution’s composition and the used processing conditions. Mesoporous inorganic materials can have various mesophase structures, for example: 2D-hexagonal (space group p6mm), biscontinuous cubic (space groups Ia-3d, Pn-3m, and Im-3m), cage-type cubic (space groups Pm-3n, Fm-3m, Im-3m, Fd-3m, etc.), cage-type hexagonal (space group P63/mmc), lamellar (L, space group p2), and others (space groups P42/mnm, P4/mmm, c2mm, Pmmm, etc.). The structures of the mesoporous materials are highly dependent on the geometries of the surfactants, including the sizes and charging of the head groups, the length and saturation of the hydrophobic tail, and its molecular shape.
In general, high concentrations of dye molecules often lead to significant florescent self-quenching resulting from intermolecular collisions, since all the molecules are completely free within the solution. Moreover, it is known that the packaging of dye molecules within a solid base will also cause selfquenching. MPS materials have abundant pore channels and surface silanol groups; therefore, dye molecules can be highly dispersed throughout the pore channels of MPSs and fixed at different locations of the pore’s surface by reaction with silanol groups, which means that the mobility and rotation of the dye is restricted to a fixed area. Therefore, the dye molecules are densely located in MPSs, and the generally observed self-quenching in the dye solution with high concentration can be reduced considerably. It has been reported that the fluorescence of the dye inside the MPS particles does not quench, although its concentration is ≈230 times higher than the maximum nonquenching concentration of the free dye in the solution.
The high concentrations of the dye immobilised in MPS improves the signal-to-noise ratio and can also affect the sensor’s sensitivity and detection limit (LOD). Furthermore, the ability to control the pore size, tailor the composition of the inorganic framework and internal pore-surface or channel, can affect or improve the sensor’s selectivity, since limited accessibility can help to shield the dyes from interferences. Moreover, it has also been shown that the 3D shapes and geometries of mesopores have a significant effect on LOD and response time (3D compared to 2D). This can be related to the fact that 3D morphologies and cage functionalities are expected to transport analyte efficiently using much more direct and easier diffusion to network sites. An additional benefit of mesoporous materials is also that they can be prepared in various morphological forms such as thinfilms, nanoparticles, and monoliths. The exceptional adsorption capacities of mesoporous materials may serve as in-situ preconcentrators for analyte, thus improving the LOD of a MPS-based sensor.
The interesting fields of usage regarding MPS materials for optical sensing have been reviewed a few times over recent years. In 2008, Melde et al. reviewed how mesoporous silicas had been applied to sensing optical and electrochemical changes in relative humidity, changes in pH, metal cations, toxic industrial compounds, volatile organic compounds, small molecular ions, nitroenergetic compounds, and biologically relevant molecules. A tutorial review published by Han et al. in 2009 reported on the development of silica-based organic/inorganic hybrid nanomaterials for use within biological and environmental applications, in which the chromogenic and fluorogenic probes can selectively detect and separate specific anions and neutral organic guests, as well as toxic metal ions. Recently, Jung et al. reviewed the preparing of a variety of silica nanotubes by self-assembled organogels and the recent development of silica-based organicinorganic hybrid nanomaterials for use as chemosensors for environmental studies, as well as within biological applications. Tran-Thi et al. noticed that sol-gel porous materials with tailored nanostructured cavities were being increasingly used with regard to their potential as sensitive matrices or layers of chemical sensors for the determination of gaseous and ionic analytes.
Sensing Mechanisms
The more commonly applied methods for the optical sensing of heavy metals using mesoporous materials are those based on light absorption or light emission. Absorption or colorimetric sensing is accomplished using an indicator that changes its colour upon binding the analyte; this change is not only spectroscopically determined but can also be observed visibly. In light-emission methods, the analyte concentration is determined by the change in the emission properties of a luminophore after being excited by a defined electromagnetic wavelength. Fluorescence typically occurs from aromatic molecules due to the rigid conjugated structure and the high rigid density of π electrons. Compared to the absorption-based methods, molecular emissions (fluorescence, phosphorescence, and, generally speaking, luminescence) are particularly important because of their extreme sensitivities and good specificities.
The sensitivity of the luminescence method is about 1000 times greater than that of most spectrophotometric methods. In addition, lower LOD for the desired analytes can be achieved. Measuring the emission intensity is also the most popular because the instrumentation needed is very simple and cheap. Nevertheless, measuring light-emission intensity has some disadvantages compared to emission lifetime measurements, in which the sample is excited only by a pulse of EM rather than via continuous illumination, which is the case with intensitybased methods. The precisions and accuracies of luminescence intensity-based schemes are greatly affected by fluctuations in the light-source’s intensity, detector sensitivity, inner filter effects, indicator concentration (bleaching and leaching), sample turbidity, and sensing layer thickness. One method of reducing the problems associated with intensity detection principles is the use of ratiometric measurements. This technique employs dual emission or dual excitation indicators or mixtures of two luminophores, exhibiting separated spectral areas with different behaviour. For example, the ratio of two fluorescent peaks is used instead of the absolute intensity of one peak. The sensors therefore typically contain a reference dye; the advantage of this approach is that factors such as excitation source fluctuations and sensor concentration will not affect the ratio between the fluorescence intensities of the indicator and reference dye.
When a fluorescent indicator is used for sensing heavy metals, the complexation of the metal ions with the indicator results in either enhanced fluorescence (chelation-enhanced fluorescence(CHEF)) or in decreased fluorescence (chelationenhanced quenching(CHEQ)). These mechanisms usually involve electron transfer (ET) and charge transfer (CT). Accordingly, these categories include photoinduced electron transfer (PET) and photoinduced charge transfer (PCT), also called intramolecular charge transfer. The PET mechanism is the more widely accepted and belongs to the group of turn-on fluorescent sensors, which fluoresce only in the presence of analytes. Sensors based on the PET mechanism often use a rational combination of a triple component system, namely, the “fluorophore-spacer-ionophore” format. The receptor contains a high-energy nonbonding electron pair (e.g., nitrogen or sulphur atoms), which can transfer an electron to an excited fluorophore group and result in fluorescence quenching. However, when the electron pair is coordinated by a metal ion, the electron transfer will be prevented, and the fluorescence is switched on.
Continuous Monitoring
The concentrations of trace metals within natural waters vary considerably as functions over time, depending on the discharger sources, seasons, types of urban activities, and so forth. The monitoring of dissolved heavy metals such as copper, lead, and cadmium over four-day periods within coastal waters showed that potentially most toxic forms of metals may vary in concentrations over a time scale of less than one hour. These data confirm the poor ecological relevance of the average conventional sampling protocol and the need for continuous monitoring.
Conventionally, ions have been determined by making use of so-called indicator dyes that undergo a binding reaction with ions. The ion-binding reactions with indicators are reversible in principle. In practice, however, most complexation reactions with heavy metals are irreversible. The indicator is essentially saturated with metal ions and any further increase in metal ion concentration produces little if any change in the observed signal. The decomplexation procedure is used for sensor regeneration/ reusage, which needs the appropriate stripping agent. EDTA and ClO stripping agents are used mostly. In the best case the reusage cycle can be repeated up to 6 times.
Specific Sensors for Heavy Metals’ Ions
The determination of toxic heavy metal cations by mesoporous material sensors/probes is usually based on the incorporation of appropriate dye molecules within selected mesoporous materials, where either absorbance or fluorescence is used as an optical detection method. Since 2008, researchers have mostly developed mesoporous materials for sensing mercury (Hg2+) and copper (Cu2+). Materials for sensing other cations have also been proposed (zinc, led, cobalt, chromium, etc.).
Mercury Sensing
The determination of mercury ions has been a subject of investigation by different research groups. Most of the sensing materials use fluorescence as a detection principle, whilst only a few use absorption. In regard to Hg2+ sensing material, SBA-15 is mostly used. Other mesoporous materials such as UVM-7, HOM- 9, Au-HMS core-shell MPS nanoparticles, and MCM-41 have also been studied. The more often used is covalent bonding of the indicator dye to the mesoporous material but immobilisation via H-bonds or ionic bonds is also possible.
In 2008, Ros-Liz et al. reported on the fabrication of dualfunction hybrid material for the simultaneous determination and removal (adsorbent) of Hg2+ ions within acetonitrile/water (1:1) solutions. A mesoporous 3D material such as UVM-7 was used as an inorganic support. The sensing principle is based on a chemodosimeter approach. In this case, a chromofluorogenic squarine dye is first “switched-off” (colorless and nonfluorescent) by reacting to -SH groups attached to a silica framework. The addition of sensing material to the solution containing Hg2+ ions result in a rapid and dramatic colour change of the solution from colourless to deep blue (new absorption band at 642 nm), due to the dye released when Hg2+ reacts with -SH groups in sensing materials.
After a two-minute reaction, the solid is collected by filtration and the absorbance of the resulting solution measured. Inorganic support can be partially regenerated by sample washing with concentrated HCl, which quantitatively removes the loaded mercury, and the material can therefore be used for several cycles. The apparent LOD of the probe is 4.9×10-7 M (0.1 ppm). Interestingly, the authors did not try to reduce the LOD by measuring the fluorescence. In addition, the leaching of the dye from the inorganic support raises the question of such a system regarding its practical application. A different approach was used by El-Safty et al. Where solid mesoporous cubic Pm3n discs were used for the simultaneous naked-eye detection and removal of mercury ions within aquatic samples. This method was based on a design of disc-like (HOM-9) sensors by the immobilisation of two different organic groups, however, the first an organic moiety for changing the silica surface polarity and the second a tetraphenylporphine tetrasulphonic acid (TPPS) probe for Hg2+ ions that showed prominent colour-change when in contact with the analyte.The sensing assay exhibited of 1 min and LOD of 5.9×10-9 M at pH 9. The reversibility of the disc-like sensor allowed for the retention of its functionality (sensitivity and fast) after multiple regeneration/reuse cycles using ClO4- as the decomplexation agent.
After multiple regeneration/reuse cycles (≥6) there was a kinetic hindrance, as the prolonged to 2 min but the sensitivity stayed at up to 92% and the disc was fully reversible. In 2008, and 2010 Kim et al. synthesised an Hg2+ sensitive acyclic dye which was immobilised on the surface of MPS. The sensing material was a light yellow solid and resulted in a colour change from light yellow to red within 10 s in the presence of Hg2+. The removal of Hg2+ (regeneration) was carried out by the addition of EDTA. A linear response was observed within the concentration range 1-10×10-6 M with an LOD of ~1×10-6 M. The mesoporous silicaimmobilised acyclical dye recognised the Hg2+ with a high degree of selectivity from amongst other metal ions within the aqueous solution.
Inorganic-organic hybrid fluorescence-based SBA-15 mesoporous materials have been reported over past years. In 2008, Zhou et al. reported a fluorescent sensor, R6-SBA-15, for the determination of Hg2+ within acetonitrile/water (7:3) solution by the covalent bonding of an organic fluorescent molecule Rhodamine 6G (R6G) within the channel of mesoporous silica. In 2010, they published another article involving the same indicator dye for Hg2+ determination within dimethylformamide/water (1:1) solution that was assembled into SBA-15 (RBSN/SBA-15) through intermolecular hydrogen bonding, instead of covalent bonding. Both SBA-15-based sensors resulted in a slight pink powder that quantitatively determine Hg2+ at the 10-9 M (ppb) level. However, it would be interesting to know and compare the two described R6G-based sensors, since the dyes were immobilised by two different approaches.
Namely, it was shown that covalent bonding can significantly prolong the sensor. Moreover, from the practical point of view it would be advisable to perform the measurements in water. Wu et al. fabricated a Rhodamine- (R6G-) based SBA-15 sensor that can be used to detect Hg2+ ions in water. However, the sensor’s LOD (1×10-8 M) compared to previously developed sensors was poorer. Fluorescent detection of Hg2+ ions was also proposed using pyrene-based fluorescent dye and the dansylamine derivate (DS), being covalently grafted onto SBA- 15. All the sensors showed good sensitivities and selectivities for Hg2+. Dong et al. have prepared a Rhodamine group modified SBA- 15 nanocomposite for the determination of Hg2+ ions in MeCNH2O solution (9:1 v/v).
Recently, Zhang et al. prepared a worm-like pore-structured mesoporous silica-based (HMS) sensor (Au-HMS-sensor). In this case, gold was used as a connector to prepare Au-HMS and determination was possible through Rhodamine B derivate, covalently grafted on Au-HMS. This sensor exhibited “turn-on” fluorescence enhancement and showed good selectivity for Hg2+ over other metal ions. LOD of 7×10-8 M concentration was reached within 100 s. The Au-HMS-sensor was successively regenerated by treatment with tetrapropylammonium hydroxide solution.
A different approach for detecting Hg2+ was used by Guo et al. and Zhang et al. who developed core-shell mesostructured silica as solid support, functionalised with pyrene. The LODs for both sensors were 1.7×10-8 M and 8.5×10-7 M, respectively, whereas the linear working concentration range was between 10-8 and 10-4 M in both cases. In comparison with the covalently grafted pyrene-SBA-15 sensor the LOD of the core-shell-based system is 50 times lower. MCM-41 mesoporous materials have also been used for preparation of the Hg2+ sensor. However, compared to other mesoporous materials, it seems that MCM-41 is not the best material for the fabrication of Hg2+ sensors, since both of the two sensors have rather high LODs.
In view of water legislation, the LODs of the majority of the mentioned sensors are still far from the “0.05 μg/L (2.5 × 10- 10 M)” target. The sensors developed by Zhou et al. and Song et al. have the lowest LODs and can detect the maximum allowed contaminant level of 1 μg/L (4.98 × 10-9 M) set by EU legislation for drinking water. Only a few papers have shown the practical applicabilities of the proposed sensors by evaluating those using real samples. Most of them lack water compatibility and need to be used in organic or aqueous organic solvents. Nevertheless, most Hg2+ sensors have demonstrated high selectivity towards other competing metal cations, showing that mesoporous structures may exhibit high selectivity potential, which is also an important sensor characteristic. Furthermore, the response time is also an important sensor characteristic, which has been overlooked by many authors.
Copper Sensing
Copper(II) ions have been the subject of continuous control, as copper is commonly used throughout industry and is therefore a widespread pollutant. However, it is also an essential trace element that plays important roles in a variety of fundamental physiological processes within living organisms. Two sensors are based on fluorescence quenching and two are based on colour change (absorption).
In 2010, Meng et al. reported on an inorganic-organic silica material, prepared by covalent immobilisation of the 1.8-naphthtlimide-based receptor (P2) within the channels of mesoporous silica material SBA-15 (SBA-P2). SBA-P2 exhibited a Cu2+ specific fluorescence-quenching response in ethanol/ water (3:7) solution with an LOD of 1.6×10-9 M. The sensor was highly selective towards Cu2+ ions over the interfering ionic species. Furthermore, the SBA-P2 material was applied for the fluorescence imaging of zebrafish organisms and the subsequent addition of Cu2+ ions resulted in SBA-P2 emission quenching. Presumably, being the first report on detecting Cu2+ ions in vivo using a functionalised nanomaterial, these results suggest that MPS is potentially useful for studying the toxicity or bioactivity of Cu2+ within living organisms. However, experiments regarding regeneration and should be done additionally in order to further characterise the sensor characteristics. Recently, El-Safty et al. constructed a Cu(II) ion sensor based on immobilised dithizone (DZ) in 2D hexagonal MCM-41 and 3D cubic Fd3m HOM-11 mesoporous silica microscopic monoliths. The reflectance spectra of this sensor exhibited a blue shift as a result of the binding of Cu2+ ions with the DZ. 3D shape and the geometries of the mesoporous materials significantly affected the ion diffusion and affinity of the metal-ligand binding, thus affecting the sensor’s characteristics.
On the other hand, Lu et al.used monodispersed mesoporous silica nanospheres modified by anthracene derivative (SGAAn) and fabricated a fluorescent sensor for the determination of Cu2+ metal ions in ethanol/water (3:7) solution. Determination of Cu2+ ions was possible through fluorescent quenching of the modified spheres in a few seconds within a concentration range from 5 × 10-8 to 10-4 M of Cu2+ , with the LOD being 2×10-8 M. The recovery of the sensor was repeatedly studied over 4 cycles by the use of EDTA as the recovery agent. Liu et al. designed an absorptionbased sensor for Cu2+ by using an indicator 4-(2-pyridylazo) rescinol (PAR) immobilised on functionalised hexagonal mesoporous silica (HMS).
Determination of Cu2+ ions was possible under strong acidic conditions (pH 12) through colour change from yellow to red of the modified spheres in 60 seconds within a concentration range from 6.3×10-7 M to 6.3×10-6 M of Cu2+, with LOD being 1.3×10-8 M. With the addition of EDTA as a regenerating agent, the sensor is reusable and can be used up to 6 times. The authors also showed a potential for developing sensors for other ions, such as Fe3+, Cd2+, Ni2+, Zn2+, Pb2+, Co2+, and Hg2+, using this sensor design. All of the reported sensors satisfied the quality of the Cu2+ parameter for water intended for human consumption (2 mg/L or 3.1×10-5 M) as set by the EU directive, WHO, and EPA.
Nano Sensors
What they are…What they do?!
The term nanosensor is not clearly defined. Most definitions refer to a sensing device with at least one of its dimensions being smaller than 100 nm and for the purpose of collecting information on the nanoscale and transferring it into data for analysis. Nanotechnology deals with physical or chemical properties of matter at the nanoscale, which can be different from their bulk properties. Nanosensors can take advantage of these phenomena. Important characteristics and quality parameters of nanosensors can therefore be improved over the case of classically modeled sensors with merely reduced sensing parts and/or the transducer.
Therefore, nanosensors are not necessarily reduced in size to the nanoscale, but could be larger devices that make use of the unique properties of nanomaterials to detect and measure events at the nanoscale. For instance, in noble metals such as silver or gold, nanostructures of smaller size than the de Broglie wavelength for electrons lead to an intense absorption in the visible/near-UV region that is absent in the spectrum of the bulk material. Nanosensors have been developed for the detection of gases, chemical and biochemical variables, as well as physical variables and the detection electromagnetic radiation.
Nanosensor Fabrication
Nanosensors can be prepared by using different methods. Three common methods are top-down lithography, bottom-up fabrication (such as for instance controlled lateral epitaxial growth and atomic layer deposition), and self-assembled nanostructures (usually done with biomolecules, e.g., liposomes, that combine in such a way that the biochemical detection of an analyte is converted into an electrical signal).
Nanosensors Based On Nanoparticles And Nanoclusters
Nanoparticles, primarily noble metal ones, have outstanding size-dependent optical properties that have been used to build optical nanosensors. The spectrum of a phenomenon called the localized surface Plasmon resonance (LSPR) depends on the size, shape and material of the nanoparticle itself as well as the particle’s environment. The high sensitivity of LSPR sensors can approach the single-molecule limit of detection for large biomolecules. Apart from metal nanoparticles, optical nanosensors based on fluorescence measurements have been built with semiconductor quantum dots and other optical sensors have been developed with nanoscale probes that contain dyes whose fluorescence is quenched in the presence of the analyte to be determined; nanoparticle films have been used for gas sensors; magnetic nanoparticles bound to biorecognitive molecules (i.e. DNA, enzymes, etc.) have been used to enrich the analyte to be detected.
For example, researchers have developed an enzyme biomarker test based on gold nanoparticles that can detect enzyme markers of disease known as proteases in humans, animals and food products. This nanosensor indicates when proteases are present through a visible color-change reaction. Nanosensors based on nanowires, nanofibers and carbon nanotubes.
Most sensors based on carbon nanotubes (CNTs) are field effect transistors (FET) because, although CNTs are robust and inert structures, their electrical properties are extremely sensitive to the effects of charge transfer and chemical doping by various molecules. The functionalization of CNTs is important for making them selective to the target analyte, different types of sensors are based on molecular recognition interactions between functionalism CNT and target analytes. For instance, researchers have developed flexible hydrogen sensors using single-walled carbon nanotubes decorated with palladium nanoparticles.
Nanowires and nanofibers have also been used to build chemiresistive sensors for the diagnosis of diseases. They have been used to maximize gas sensor responses in exhaled breath analysis for the detection of volatile organic compounds (which are biomarkers for various diseases; for example, acetone, hydrogen sulfide, ammonia, and toluene can be used as biomarkers for evaluating diabetes, halitosis, kidney malfunction, and lung cancer, respectively). In one example, porous tin oxide (SnO2) nanofibers have been demonstrated to detect acetone at levels of around 0.1 ppm, which is eight times lower than the required gassensing level for diagnosing diabetes.
Nanosensors Based on Graphene
Another carbon nanomaterial, functionalized graphene, holds exceptional promise for biological and chemical sensors. Already, researchers have shown that the distinctive 2D structure of graphene oxide (GO), combined with its superpermeability to water molecules, leads to sensing devices with an unprecedented speed (“Ultrafast graphene sensor monitors your breath while you speak”).Scientists have found that chemical vapors change the noise spectra of graphene transistors, allowing them to perform selective gas sensing for many vapors with a single device made of pristine grapheme, no functionalization of the graphene surface required (“Selective gas sensing with pristine graphene”).
Researchers also have begun to work with graphene foams, threedimensional structures of interconnected graphene sheets with extremely high conductivity. These structures are very promising as gas sensors (“Graphene foam detects explosives, emissions better than today’s gas sensors”) and as biosensors to detect diseases (see for instance: “Nanotechnology biosensor to detect biomarkers for Parkinson’s disease”).
Nanosensors Based on Bulk Nanostructured Materials
Whereas several properties of nanoparticles are useful for applications in nanosensors, their catalytic behavior is one of the most important with regard to electrochemical sensing devices. For instance, platinum nanoparticles supported on materials such as porous carbon or noble metals such as gold are reported to be relevant in the design of gas diffusion electrodes. Another property, their high surface area, makes nanoparticles suitable for immobilizing molecules, polymers or biomaterial coatings that allow the generation of composite materials with tunable surface properties. For example, modifying metal nanoparticles with predesigned receptor units and assembling them on surfaces could lead to new electrochemical sensors with tailored specificities. Simple and highly selective electro analytical procedures can also be achieved by proper functionalization of nanoparticles. Finally, stable nanoparticles can substitute amplifying labels of limited stability, such as enzymes or liposomes, with equivalent or improved sensitivities.
Nanosensors Based on Metal-Organic Frameworks (MOFs)
Metal-organic frameworks (MOFs) are organic-inorganic hybrid crystalline porous materials that consist of a regular array of positively charged metal ions surrounded by organic ‘linker’ molecules. The metal ions form nodes that bind the arms of the linkers together to form a repeating, cage-like structure. Due to this hollow structure, MOFs have an extraordinarily large internal surface area, which makes them ideal materials for gas sensing. By making the MOF from different metal atoms and organic linkers, researchers can create materials that selectively absorb specific gases into tailor-made pockets within the structure. One example is a thin-film a tailor-made MOF, coated onto an electrode that forms an electronic sensor that could detect traces of sulfur dioxide gas.
Other Examples of Nanosensor Applications
A simple single nanoscale sensor and light spectroscopy (fluorescence) can be used for sensing the difference in potential across a biological membrane such as a cell wall. This non-contact, optical voltage nanosensor is based on a Forster resonance energy transfer (FRET) sensor on a DNA origami. A graphene nanosensor enables real-time monitoring of insulin at physiologically relevant concentrations - down to a concentration of approximately 35 pM. This device employs an aptamer-based graphene field-effect transistor (GFET) nanosensor that can rapidly respond to external stimuli such as the binding between surface-immobilized aptamer molecules with insulin. This results in significant changes of the electrostatic charge characteristics in the close proximity of the graphene surface.
A gel-based sensor technology can help medical physicists and oncologists effectively plan radiotherapy in the clinic, reduce accidental overexposures, and reduce radiation-induced toxicity. In contrast to traditional inflexible sensors, these plasmonic nanosensor gels indicate exposure levels as radiation delivered to the hydrogel increases the intensity of color developed in the gel. By interfacing passive, wireless graphene nanosensors onto biomaterials via silk bioresorption, researchers have developed graphene nanosensor tattoos on teeth in order to monitor harmful oral bacteria. A great example of nanosensor technology is the ability of chips with molecular probes to detect intracellular biological parameters, i.e. from within a living cell.
Sensors for Intelligent Processing of Structural Polymeric Composites
Structural polymeric composites are critically important to sustaining U.S. aerospace and defense superiority. The potential market for these materials, by the end of the century, is projected to be 90,000 metric tons, a sixfold increase in tonnage since 1986, with a tenfold increase in worldwide employment to 200,000 people (AIA, 1991). Improvements in the manufacture of products that use these materials will help in the realization of such projections.
A polymeric composite typically has two primary microstructural components: a polymer material and reinforcing fibers. Two different generic types of polymers are used: thermosetting (the traditional choice) and thermoplastic (the more recent choice). Thermosetting polymers are initially lowviscosity liquids that can flow into a mold or around fibers. During the course of processing, thermosets react to increase molecular weight and viscosity, eventually becoming highly cross-linked, insoluble, infusible materials. On the other hand, thermoplastics are fully polymerized materials that melt and flow upon application of heat. Thermoplastics are processed well above their glass transition temperatures or melting points (if the material is semicrystalline) to reduce the melt viscosity and allow flow and to promote adhesion to the fibers.
The fibers in the composite are the component that provides the desired high stiffness and strength properties that make the material useful for structural applications. The polymer matrix protects the fiber, serves as a medium to transfer load between the fibers, and stabilizes the fiber when subjected to compression loading.
A composite structure is typically made by stacking layers of pre-impregnated (“prepreg”) material in prescribed directions on a tool form, compressing the stack, and then curing or consolidating the composite. In the case of high-performance thermosetting resins, the curing process requires the application of heat and pressure. For small-lot production, this curing step is usually accomplished in a large pressure vessel called an autoclave. Alternative processes, such as resin transfer molding, are preferred for higher-volume mass production.
Consolidation of a thermoplastic resin does not require autoclave but does require the application of energy, such as heat. In addition, new composites are being developed that consist of blends of thermosets and thermoplastics; in these materials, curing and consolidation occur simultaneously and interactively to produce complex microstructures possessing functionally gradient material properties that can be tailored to optimize specific strength, processability, etc. Intelligent processing can improve the resultant properties of composite components. The discussion that follows examines the technologies required to fully implement intelligent processing with emphasis on the most critical sensor requirements [93,94].
Thermosetting Polymer Matrix Composites
At the present time, most of the structural polymeric composites used in the aerospace industry consist of a high strength/high stiffness fiber (e.g., graphite, boron, or aramid) embedded in a thermosetting organic-resin matrix binder (e.g., epoxy, bismaleimide, polyimide, polyester, or cyanate ester). These resins are complex chemical formulations with batch-to-batch chemical variations. Until recently, the conventional processing strategy for thermoset polymer composites was based on priori models. These models cannot account for variations in material chemistry, part geometry, autoclave sizes and heating patterns, and tolling materials. As a consequence, they assume “one size fits all” and are developed for the worst-case processing conditions. Hence these models specify a safe (i.e., protracted) cure cycle.
In contrast, intelligent processing senses the state of the material during cure. The development of these models requires the definition of events that denote the changes in material state, e.g., flow, deformation, growth, etc. A processing system is also required that is empowered (through a model or set of conditionaction relations) to adjust the process parameters in response to changes in the materials state to achieve desired end-use properties. The first steps in developing an intelligent processing system are the mapping of these desired end-use properties to the actual material parameters that can be sensed and the processing variables that can be controlled. Some properties (such as residual stresses; void volume; and fiber location, volume, and orientation) can be inferred from engineered relationships with parameters that can be sensed in situ, while other properties (such as part strength and modulus) depend upon other engineering relationships to determine properties that cannot now be measured in situ.
Identifying “need-to-be-sensed” parameters can be accomplished through a review of available models of the process or by developing a mapping of properties to parameters and to variables. In the next step, a process engineer must assess available sensor technologies that can provide the necessary information either directly or indirectly. To accomplish this assessment, the framework developed can be used as a guide to list and detail the necessary parameters for evaluating and comparing different sensor technologies. With these descriptors, the process engineer can describe the range, tolerance, and limits of the sensing requirements and identify one or more candidate sensor technologies, and their respective capability and constraints.
Performance estimates and benefits of intelligent manufacturing can only be validated in a true manufacturing environment. One example is provided by the U.S. Air Force for an application that produces polymer composite replacement parts for the A-10 aircraft. In this case, sheet metal aluminum parts for the leading edge of the aircraft wing are being replaced with a hybrid carbon and aramid fiber/epoxy-resin composite to improve performance and extend the useful life of the aircraft. Initially the composite replacements were cured using a conventional (i.e., a priori model) cure cycle. But in 1990, the Air Force’s Sacramento Air Logistics Center implemented an intelligent processing system that achieved full cure in 1.5 hours versus 7 hours required for the conventional approach; that is, a 70 percent reduction in cure time. The intelligent manufacturing system was originally implemented using the polymeric material temperature (as measured by a thermocouple) as the basis for estimating the degree of cure. Autoclave temperature profiles during cure are presented for both approaches along with the resultant test values of selected material properties. This data confirmed that intelligent processing could produce parts with strength values statistically equivalent to those conventionally processed (Warnock and LeClair, 1992).
Even though this initial implementation was quite successful, it was not without difficulty. For instance, the thermocouple signal was “noisy” and hence many data points were needed to clearly establish the trendline. To identify various levels of candidate improvements, the framework presented in Chapter 2 was applied to relate sensing needs (e.g., resin temperature) to the currently available sensor technology (e.g., thermocouple); the results of this comparison are summarized in Appendix D. Some of the thermocouple shortfalls can be partially ameliorated, but nonetheless, a new sensor technology was clearly needed. For example, autoclave temperature does not directly relate to the degree of resin cure (i.e., extent of the formation of cross-linked polymer bonds).
One such multiuse technology which addresses both the limitation of thermocouples and the need for more microscopic property monitoring is photon scattering fiberoptic sensing, as described in Appendix D. Photon-scattering sensor technology uses the energy and momentum distribution in the scattered photon flux to extract a wealth of useful and interpretable information regarding the physical and chemical nature of the material being processed. Suitable electromagnetic radiation scattering measurements (i.e., Rayleigh, Brillouin, and Raman scattering) can theoretically provide a direct measurement of material properties that include bulk modulus, thermal diffusivity, sound attenuation factor, sonic speed, heat capacity ratio, chemical composition, bulk viscosity, shear viscosity, and the free energy of mixing (Maguire and Talley, 1995). The scattered flux can be detected by receiving optical fiber at any given angle, and transported at the speed of light to a remote spectrometer.
Near-infrared Raman spectroscopy, using fiber optics to provide a relatively simple optical contact with the material, can allow direct determination of the degree of cure. As an example, an epoxy-amine material has a Raman spectral peak at 1,250 cm-1 resulting from the vibration of the polymer’s epoxide ring structure and another one at 2,870 cm-1 arising from the stretching of C-H bonds. During cure, the number of the C-H bonds does not change significantly, while the number of epoxide groups notably decreases. Therefore, the 2,870 cm-1 peak can be used as an internal standard to determine the degree of change in the 1,250 cm-1 peak over time (Maguire and Talley, 1995).
Sensor array useful in many intelligent processing applications. Continued research and development of multiuse sensor technology will provide crucial enabling science and understanding for addressing these intelligent processing sensor opportunities. The last section of this chapter discusses research needs and opportunities for photon-scattering sensor technology.
Thermoplastic Polymer Matrix Composites
Future systems, ranging from aircraft parts and printed circuit boards to applications that must survive in the rigors of space, will require further innovation in intelligent processing, particularly in the development of low-cost out-of-autoclave8 composite fabrication techniques and new material systems such as components made of thermoplastic matrix composites. A newly developed out-of-autoclave technique involves the use of a unique energy transduction mode known as direct electric heating that is, passing a current through the carbon reinforcing fibers (Miller and Van den Nieuwenhuizen, 1993). Electric heating of the fibers provides for a versatile temperature source capable of accommodating complex parts possessing varying curvature and laydown angles. It can produce a high strength, low void content composite. In the discussion that follows, processing needs, together with the unique hardware for part lay-up, are discussed to identify requirements for new sensor configurations.
Thermoplastic material part quality is defined by the resultant physical properties, such as ultimate strength, void content, and elastic modulus. However, in-process measurements of some of these quantities (e.g., ultimate strength) cannot be directly obtained, since their measurement requires part destruction (University of Delaware, 1989). Void content, a commonly used measure of laminate quality, can be measured by x-rays, ultrasonic pulses, or determination of local electrical or by thermal conductivity using infrared sensors. Controlling void content during the process requires measuring both temperature and pressure in real-time during processing and making appropriate adjustments. Thus, temperature must be measured; if it begins to exceed the prescribed range, the tape head pressure can be adjusted accordingly across the face of the tape head to bring the temperature back in line [95-97].
Polymer nanocomposites
Polymer Nanocomposite-Based Electrochemical Sensors and Biosensors.
Polymer nanocomposites (PNCs) have electrochemical properties as transducers which can be used for the manufacturing of electrochemical sensors and biosensors. They possess significant variations in responsiveness, synthesis, and morphology, which help in a significant level of variations in conductivity. Afar from the economic aspect of the PNC-based sensors, the improved performance on the electronic side stands apart among its peers through the basal plane ratio of the nanofillers, method of doping, kinetic properties of the electrode, biological response and environmental impact. The impact of nanofillers in PNCs plays a significant role in sensing, processing, and actuating capabilities of the electrodes of electrochemical and biosensing applications.
The “active states of PNCs” rests on three pillars: high electrical conductivity rate, large surface area and fast electron rate which leads to best electricidal sensor outcomes. PNCs helps in the material technological advancement of electrochemical sensors which have high sensitivity and selectivity, lower detection limits, reproducibility and stability. All these increased used the PNCs in electrochemical sensor research which were manufactured through chemical synthesis or polymerization methods and could be easily scaled up for various applications. The electrochemical sensors along with the immunosensors and biosensors are becoming the norm of the day. Detections limits and sensing technologies are improved consistently due to developments happening in the unique properties of PNCs especially conductivity and electrochemical activity. The interactive fillers facilitate ion diffusion that impacts the sensing applications through intercalation into the PNC matrices leading to better stability of active electron transfer sites and detection limits. These active fillers help in reducing the layer thickness in PNC leading to ultrathin electrochemical detector technology.
PNCs stand as an outstanding leader with significant advantages in large specific interaction surface area, reduced dimension of fillers and efficient electron transfer rate.
Electrochemical Sensors
PNCs are widely used in the development of electrochemical sensors. The electrochemical sensors are based on three categories of PNCs. PNCs of conductive polymers and inorganic nanomaterials, PNCs of conductive polymers and Grp, and PNCs of conductive polymers and CNTs. Once interaction has occurred between the PNC-based electrochemical sensors and the target analyte, an electronic signal is detected by the transduction system.
Electrochemical sensors based on polymer nanocomposites
Polymer nanocomposites based on conductive polymers and inorganic nanomaterials
Metal and metal oxide nanoparticles have been extensively studied as electrochemical sensing materials due to such beneficial features as their small size; unique chemical, physical, and electronic properties; flexibility in fabricating novel and improved sensing devices; and good sensitivity to the ambient condition. The assimilation of nanoparticles of metals into PNC matrices set the stage for enhanced electrocatalytic electrode detection leading to multiple modern-day applications. For example, a Zinc oxide nanoparticle intercalated into polypyrrole (ZnO-PPy) PNC showed excellent Xanthine detection by through xanthine oxidase enzyme immobilization. A glassy carbon electrode (GCE) modified with ultrathin polypyrrole nanosheets decorated with Ag nanoparticles was fabricated for the detection of hydrogen peroxide (H2O2). The modified device showed high sensitivity toward the reduction of H2O2.
Similar electrochemical sensor based on polypyrrole-platinum (PPy-Pt) PNC was fabricated for the detection of H2O2. Another voltammetric sensor based on a polyaniline-gold nanoparticle (PANI-AuNP) PNC deposited on GCE was used for the detection of epinephrine (EP) and uric acid (UA). Exploiting the advantages of PNCs, two GCEs modified with PANI-TiO2 and PANI-NiCo2O4 PNCbased electrochemical sensors were developed for the detection of glucose. TiO2 nanotubes (TNTs) were intercalated into a PANITNT PNC composite for through hydrothermal method for the detection (a reported sensitivity of 11.4 μA mM-1) of glucose (a reported sensitivity of 11.4 μA mM-1) by the immobilization of glucose oxidase.
Polymer Nanocomposites Based on Conductive Polymers and Graphene
Graphene (Grp), an allotrope of carbon, has become the new material of interest and widely integrated into sensor research from the beginning of this millennium due to its unique properties of electrical conduction and 2-dimensional existence. Grp-PNCbased electrochemical sensors are used for electroanalytical detection of target molecules with high precision of selectivity and sensitivity, which showed spectacular detection limits over a wide range. An electrochemical sensor fabricated for the detection of 4-aminophenol (4-AP) using a PANI-Grp-GCE-modified device showed a detection limit of 6.5×10-8 M and sensitivity of 604.2 μAmM-1. A sensor was fabricated with a PANI-Grp-based PNC onto an ITO plate with immobilized horseradish peroxidase enzyme with a sensitivity limit of 0.15 mA ng.mL-1. A PANI-Grp-GCE-based PNC sensor for the elimination of calcium antagonist lercanidipine in pharmaceutical formulations for medical purposes showed a detection limit in the range from 5 to 125 ng.mL-1.
A sensor was fabricated with a PANI-Grp-based PNC onto an ITO plate with immobilized horseradish peroxidase enzyme with a sensitivity limit of 0.15 mA ng.mL-1. A PANI-Grp-GCE-based PNC sensor for the elimination of calcium antagonist lercanidipine in pharmaceutical formulations for medical purposes showed a detection limit in the range from 5 to 125 ng.mL-1. The same PANIGrp-GCE-based PNC sensor showed the detection of nitazoxanide compound which was an added advantage. Electrochemical sensors based on PPy-based PNC are becoming popular these days due to their specific applications through their overoxidized form polypyrrole (PPyox). Fabrication of polypyrrole-graphene (PPyox/Grp) helped in the simultaneous detection of adenine and guanine through an electrodeposition method. PPy-Grp composite was electro-polymerized with pyrrole and graphene oxide (GO), followed by electrochemical reduction of GO composite. The electrochemical sensor’s significant improvement in the sensing of adenine and guanine is due to the specific structure of the nanocomposite. The adenine and guanine showed strong π-π interactions, and cationic selectivity.
The detection of Dopamine (DA) using PNCs was the Holy Grail in neurochemical studies ad it is a prominent neurotransmitter, which plays a role in neurological disorders such as Parkinson’s disease and schizophrenia. A poly(ionic liquid)-functionalized polypyrrole-graphene oxide (PIL-PPy- GO)-based PNC electrochemical sensor was fabricated by the polymerization of 1-vinyl-3-ethylimidazolium bromide on N-vinyl imidazolium-modified PPy-GO films. The PILs helped in changing the surface charge which dispersibility of the PIL-PPy- GO composite and helped in the detection of DA. Another sensor used for the detection of DA was a PNC-based poly (3,4-ethylene dioxythiophene)-graphene oxide (PEDOT-GO) fabricated by electrodeposition showed significant sensing capabilities. A onestep electrochemical redox synthesis process of PEDOT-Grp PNC film was done using simultaneous electrodeposition of PEDOT and electrochemical reduction of GO on a GCE with high detection sensing of the ascorbic acid molecules. In this sensor PEDOTGrp thin film PNC mediated the electron transfer between AO and electrode interface resulting in significant improvement in electrocatalytic activity and sensitivity of ascorbic acid molecules. Jain et al. introduced the combination of Grp and a conducting PANI-Bi2O3 PNC, the synergic effect of which enhanced the performance of sensors used for the electrocatalytic oxidation of etodolac in pharmaceutical formulations.
In recent years, molecularly imprinted polymers (MIPs) with high selectivity, affinity, chemical stability, and easy preparation for the template molecule are a promising candidate for developing a new generation of recognition elements for sensors. A double-layered membrane-sensing interface was fabricated based on rGO-PANI nanocomposites and MIPs embedded with AuNPs for sensitive and selective detection of serotonin (5-hydroxytryptamine, 5-HT). The obtained sensor showed remarkable selectivity to serotonin against the interferences caused by ascorbic acid and other interferents with a good detection limit of 11.7 nmol.L-1.Polymer Nanocomposites Based on Conductive Polymers and Carbon Nanotubes.
PNCs based on conductive polymers helped in improving the sensing properties of the electrochemical sensors with enhanced selectivity and stability. A PPy-multiwalled carbon nanotube (MWCNT)-toluene sulfonic acid-based PNC was fabricated fr the detection of cholesterol with ITO-coated glass was the substrate for the sensor. The sensor showed high sensitivity and a fast response time of 9 s. Sodium dodecyl sulfate-doped PPyox) with carboxylic acid functionalized MWCNT-modified GCE were reported for the detection of the anticancer drug pemetrexed (PMX). The results showed that overoxidation of the PPy film conferred a negative charge density on the porous layer, which in turn enhances the adsorption of PMX. Xu et al. fabricated a carbon paste electrode (CPE) modified with a PEDOT-CNT nanocomposite. They used this electrode for the analysis of hydroquinone, DA, and nitrobenzene.
DNA Biosensors
PNCs are widely used these days in DNA biosensors. The medico biological field is growing leaping and bounds. In this era of 23 and me everything possible with DNA is bouncing through the boundaries of technology like DNA CRISPR editing, gene mapping. Biological agents for nefarious purposes and forensics. A basic DNA sensor work on a simple principle. You plant a DNA probe on the surface and this planted DNA chain hybridizes with its complementary pair. This hybridization technically called transduction can be detected optically and electrochemically. The electrochemical detection of transducers through electrochemical sensors leads us to DNA biosensors and are now extremely popular.
The schematic illustration of the most popular DNA biosensor based on polyaniline-gold nanoparticle-chitosan-graphene sheet (PANI-AuNP-Cts-GS) composite with a functional capture probe for the detection of BCR/ABL fusion gene in chronic myelogenous leukemia (CML). The capture probe used a hairpin structure and was dually labeled with a 5′-SH and a 3′-biotin. The biotin electrode probe was used for the detection of streptavidin-alkaline phosphatase (AP) enzyme which in turn cause an electrochemical signal caused by the catalytic reduction of 1-naphthyl phosphate to 1-naphthol picked up by Diffuse Pulse Voltammetry (DPV) with a detection range of 10-1000 pM. A DNA biosensor fabricated with PANI-AuNP PNC was used for the detection of Ag+. It works on the following principle: the electrochemical biosensor regenerates cysteine leading to the release of Ag+ from the cytosine to Ag+- Cytosine complex and reused again. The fabricated biosensor showed excellent selectivity with a good detection limit for silver ions.
A DNA biosensor fabricated with PANI-AuNP PNC was used for the detection of Ag+. It works on the following principle: the electrochemical biosensor regenerates cysteine leading to the release of Ag+ from the cytosine to Ag+-Cytosine complex and reused again. The fabricated biosensor showed excellent selectivity with a good detection limit for silver ions. Another DNA electrochemical biosensor was developed using polyaniline nanofibers (PANI-nf) enrapturing AuNPs making (PANI-nf- AuNP), a PNC. Gold was used as the electrode for the detection of Staphylococcus aureus DNA from the PANI-nf-AuNPs sensor, where the detection concentration varied from 150×10-12 to 1×10- 6 mol.L-1. A DNA biosensor based on the PANI-Fe3O4-CNT PNC was manufactured for sensing Neisseria gonorrhoeae through a DNA probe. The fabricated biosensor showed sensing in the range from 1×10-19 to 1×10-6 M through DPV measurements. The most recent DNA biosensor based on PANI-AuNPs PNC detected the microRNA-16 using a streptavidin-AP conjugate to biotinylated target sequences through transduction with a detection limit of 0.1 nM. DNA biosensor made with polypyrrole-polyaniline-gold (PPy- PANI-Au) PNC responded to the target DNA through transduction, noncomplementary and single- and double-base-mismatched target DNA-chains with a detection limit between 1×10-6 and 1×10-13 M. Nanotube DNA biosensor based on polypyrrole and poly(3,4-ethylenedioxythiophene) (PPy-PEDOT) PNC, which was functionalized with Ag nanoparticles sensed DNA transduction through EIS detection. The DNA chains used for detection were thiol-capped on the modified sensor.
Electrochemical Immunosensors
PNCs are superior candidates for the fabrication of electrochemical Immunosensors, where the antibodies are the probes which form ionic complexes with the corresponding antigen pair with a specific target. Electrochemical Immunosensors are becoming widely used in clinical diagnosis applications, doping or impurities or detecting biological components in the food industry and detecting the biomolecules of environmental origin and impact. The most widely reported are discussed in this section and the immunosensors based on CNT-PPy-goat IgGs showed the interaction between the goat IgGs and its antigoat IgGs, which changes the charges at the sensor surface with changes in conductance level. The response time for the anti-goat IgG was 1 min.
A label-free impedance immunosensor for human chorionic gonadotropin (hCG) detection using a PPy-PPa-hCG-modified carbon ink electrode was fabricated by the deposition of a PPy-pyrole-2-carboxylic acid copolymer. The hCG antibody was immobilized via the COOH groups of pyrrole-2-carboxylic acid, as a linker for covalent biomolecular immobilization. This immunosensor has a detection limit of the hCG antigen in the range of 100 pg mL-1 to 40 ng mL-1. The next progress was PANI-AuNP hybrid electrochemical immunosensor with the gold electrode for the detection of prostate antigen (PSA). The immunosensor showed effective immobilization of anti-PSA with excellent sensing performance (1.4 μA M-1) and detection limit (0.6 pg mL-1) through effective electron transport. For the detection of aflatoxin B1, an electrochemical immunosensor based on a Grp-CP-AuNPIL composite film was used.
The fabrication was in a five-part series mode as Grp-CP-AuNP-IL pattern. Poly (DPB), 2,5-di-(2-thienyl)-1-pyrrole-1-(p-benzoic acid) helped in the electrochemical stability as a CP. The covalent bonding through the antibody immobilization via carbonyl groups of the polymer helped in preventing the antibody loss, resulting in a detection limit of 1.0 fM. For ofloxacin detection, an immunosensor was fabricated based on a dual-amplification mechanism resulting from Au nanoclusters embedded in the pre-synthesized PPy film as the sensor platform and multienzyme antibody-functionalized gold nanorods as the label. The electrochemical response was in the range of 0.08 and 410 ng mL-1 with a low detection limit of 0.03 ng mL-1.
An electrochemical immunosensor was fabricated a PNC-based Grp-AuNP-poly-DPB-AuNP-IL for the detection of microcystin-LR through electrodeposition method on GCE. In this electrochemical sensor, the Grp-gold helped in the electron transfer of [Fe(CN)6]3-, and the poly 2,5-di-(2-thienyl)-1-pyrrole- 1-(p-benzoic acid)- gold nanoparticle (poly-DPB-AuNP) enhanced the electrical conduction and subsequent immobilization of the microcystin-LR antibody. A Grp-PANI-based PNC electrochemical sensor for the estradiol using horseradish peroxidase-graphene oxide-antibody (HRP-GO-Ab) was designed where carboxylated GO serves the antibody carrier property while the horseradish peroxidase helped in catalytic hydrogen reduction on the electrode. This estradiol immunosensor detected estradiol in tap water and milk samples, with average recoveries of 97.25% and 96.6%, respectively [91-96].
The electrochemical immunosensors with electrochemiluminescence (ECL) sensing property was achieved through Quantum dots (QDs). This was based on graphene oxide nanosheet-polyaniline nanowire-CdSe quantum dot (GO-PANICdSe) which detected human interleukin-6 (IL-6). A ZnSe QDAzure I-AuNP-PEDOT-modified Pt electrode electrochemical immunosensor helped in the detection of alpha-fetoprotein (AFP) through electrochemiluminescence (ECL) sensing (detection limit ~1.1 fg mL-1). The sensing mechanism was as follows: ZnSe QDs immobilize the antibody, the nanoAu-PEDOT facilitated the electron transfer, and Azure I did the catalytic reduction of redox dye with two active amino groups.
PANI-AuNP-modified paper working electrodes (PANI-AuNP PWEs) were fabricated for the simultaneous determination of two tumor markers, carcinoembryonic antigen (CEA) and AFP, in real human serum samples. An electrochemical immunosensor for prolactin hormone was also constructed by immobilizing the antigen onto poly (pyrrolepropionic acid) CP and carbon nanotube (pPPA/CNT) hybrids deposited onto a GCE and labeled with AP enzyme with a reported detection limit of 104 ng mL-1. Polypyrrole (PPy)-based PNC was used to manufacture a bioelectrode for the detection of human C-reactive protein antigen (Ag-αCRP). This was made possible with the inorganic nanoparticles (3-mercaptopropionic acid (MPA)-capped Pt nanoparticles. First, the Ab-αCRP was immobilized covalently through specific carboxyl groups linkages through Pt(MPA)- NPs within the polypyrrole (PPy)-based PNC film by carbodiimide coupling. The resulted electrochemical immunosensor showed excellent fine probe orientation with a detection capacity of 10 ng mL-1-10 μg mL-1.
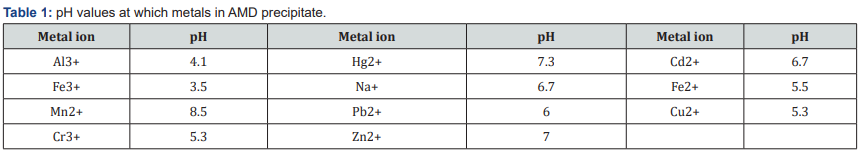
Another label-free impedimetric immunosensor based on multifunctional PNCs was based on (polypyrrole-pyrrolepropylic acid-reduced graphene oxide (PPy-PPa-rGO)) for the detection of mycotoxin aflatoxin B1. An enzyme-free electrochemical immunosensor modeled on the sandwich pattern was used for the detection of Carbohydrate Antigen72-4 (CA72-4). The sensing electrode was nano porous gold (NPG) film and asymmetric multicomponent (AMNPs) nanoparticles based on PANI-Au were used as labels. The NPG helped in the creased immobilization of Ab1 on the electrode, while the PANI-Au AMNPs impacted on the reduction capability of the electrochemical immunosensor. The doping of AuNPs and PANI films with potassium ferricyanide over a gold electrode was used to detect a carcinoembryonic antigen (CEA). This PNC-based biocompatible electrochemical immunosensor showed excellent conductivity and redox electroactivity. The detection of CEA was analyzed through electrode response of [Fe (CN)6]3- as the redox mediator.
Results and Discussion
Figure 1
Characterization
Figure 2, 3
The Future of Sensor Technology
As technology develops, the use of sensors will continue to expand into every aspect of our lives. Engineers and scientists around the world will use sensors to enhance transportation systems, medical procedures, nanotechnology, mobile devices, virtual and augmented reality, and even Artificial Intelligence (AI).
References
- Fergusson JE (1990) The Heavy Elements: Chemistry, Environmental Impact and Health Effects. Oxford: Pergamon Press, United Kingdom.
- Duffus JH (2002) Heavy metals-a meaningless term? Pure Appl Chem 74(5):793-807.
- Bradl H (2002) Heavy Metals in the Environment: Origin, Interaction and Remediation, Academic Press, 6, London.
- He ZL, Yang XE, Stoffella PJ (2005) Trace elements in agroecosystems and impacts on the environment. J Trace Elem Med Biol 19(2-3): 125-140.
- Goyer RA (2001) Toxic effects of metals. In: Klaassen CD, (eds). Cassarett and Doull’s Toxicology: The Basic Science of Poisons. McGraw-Hill Publisher, New York, pp. 811-867.
- Herawati N, Suzuki S, Hayashi K, Rivai IF, Koyoma H (2000) Cadmium, copper and zinc levels in rice and soil of Japan, Indonesia and China by soil type. Bull Env Contam Toxicol 64(1): 33-39.
- Shallari S, Schwartz C, Hasko A, Morel JL (1998) Heavy metals in soils and plants of serpentine and industrial sites of Albania. Sci Total Environ 209(2-3): 133-142.
- Nriagu JO (1989) A global assessment of natural sources of atmospheric trace metals. Nature 338: 47-49.
- Arruti A, Fernández-Olmo I, Irabien A (2010) Evaluation of the contribution of local sources to trace metals levels in urban PM2.5 and PM10 in the Cantabria region (Northern Spain) J Environ Monit 12(7): 1451-1458.
- Sträter E, Westbeld A, Klemm O (2010) Pollution in coastal fog at Alto Patache, Northern Chile. Environ Sci Pollut Res Int 17(9): 1563-1573.
- Pacyna JM (1996) Monitoring and assessment of metal contaminants in the air. In: Chang LW, Magos L, Suzuli T, editors. Toxicology of Metals, CRC Press, Boca Raton, Florida, pp. 9-28.
- WHO/FAO/IAEA. World Health Organization. Switzerland: Geneva; 1996. Trace Elements in Human Nutrition and Health.
- Kabata- Pendia A (2001) Trace Elements in Soils and Plants. CRC Press, Boca Raton, Florida.
- Hamelink JL, Landrum PF, Harold BL, William BH (1994) Bioavailability: Physical, Chemical, and Biological Interactions. CRC Press Inc, Boca Raton, Florida.
- Verkleji JAS. In: The effects of heavy metals stress on higher plants and their use as biomonitors In Plant as Bioindicators: Indicators of Heavy Metals in the Terrestrial Environment.
- Markert B (1993) VCH, New Yor, pp. 415-424.
- Stern BR (2010) Essentiality and toxicity in copper health risk assessment: overview, update and regulatory considerations. Toxicol Environ Health A 73(2): 114-127.
- Harvey LJ, McArdle HJ (2008) Biomarkers of copper status: a brief update. Br J Nutr 99(S3): S10-S13.
- (2002) Agency for Toxic Substances and Disease Registry (ATSDR) Toxicological Profile for Copper. Atlanta, GA: Centers for Disease Control.
- Tchounwou P, Newsome C, Williams J, Glass K (2008) Copper-induced cytotoxicity and transcriptional activation of stress genes in human liver carcinoma cells. Metal Ions Biol Med 10: 285-290.
- Chang LW, Magos L, Suzuki T (1996) Toxicology of Metals. CRC Press, Boca Raton, Florida, USA.
- Wang S, Shi X (2001) Molecular mechanisms of metal toxicity and carcinogenesis. Mol Cell Biochem 222(1-2): 3-9.
- Beyersmann D, Hartwig A (2008) Carcinogenic metal compounds: recent insight into molecular and cellular mechanisms. Arch Toxicol 82(8): 493-512.
- Yedjou CG, Tchounwou PB (2006) Oxidative stress in human leukemia cells (HL-60), human liver carcinoma cells (HepG2) and human Jerkat-T cells exposed to arsenic trioxide. Metal Ions Biol Med 9: 298-303.
- Yedjou GC, Tchounwou PB (2007) In vitro cytotoxic and genotoxic effects of arsenic trioxide on human leukemia cells using the MTT and alkaline single cell gel electrophoresis (comet) assays. Mol Cell Biochem 301(1-2): 123-130.
- Tchounwou PB, Centeno JA, Patlolla AK (2004) Arsenic toxicity, mutagenesis and carcinogenesis - a health risk assessment and management approach. Mol Cell Biochem 255(1-2): 47-55.
- Tchounwou PB, Ishaque A, Schneider J (2001) Cytotoxicity and transcriptional activation of stress genes in human liver carcinoma cells (HepG2) exposed to cadmium chloride. Mol Cell Biochem 222(1-2): 21-28.
- Patlolla A, Barnes C, Hackett D, Tchounwou PB (2009) Potassium dichromate-induced cytotoxicity, genotoxicity and oxidative stress in human liver carcinoma (HepG2) cells. Int J Environ Res Public Health 6(2): 643-653.
- Patlolla A, Barnes C, Yedjou C, Velma V, Tchounwou PB (2009) Oxidative stress, DNA damage and antioxidant enzyme activity induced by hexavalent chromium in Sprague Dawley rats. Environ Toxicol 24(1): 66-73.
- Yedjou GC, Tchounwou PB (2008) N-acetylcysteine affords protection against lead-induced cytotoxicity and oxidative stress in human liver carcinoma (HepG2) cells. Intl J Environ Res Public Health 4(2): 132-137.
- Tchounwou PB, Yedjou CG, Foxx D, Ishaque A, Shen E (2004) Lead-induced cytotoxicity and transcriptional activation of stress genes in human liver carcinoma cells (HepG2) Mol Cell Biochem 255(1-2): 161-170.
- Sutton DJ, Tchounwou PB (2007) Mercury induces the externalization of phosphatidylserine in human proximal tubule (HK-2) cells. Intl J Environ Res Public Health 4(2): 138-144.
- Sutton D, Tchounwou PB, Ninashvili N, Shen E (2002) Mercury induces cytotoxicity, and transcriptionally activates stress genes in human liver carcinoma cells. Intl J Mol Sci 3(9): 965-984.
- (2000) Agency for Toxic Substances and Disease Registry (ATSDR) Toxicological Profile for Arsenic TP-92/09. Center for Disease Control, Atlanta, Georgia.
- Tchounwou PB, Wilson B, Ishaque A (1999) Important considerations in the development of public health advisories for arsenic and arsenic-containing compounds in drinking water. Rev Environ Health 14(4): 211-229.
- Centeno JA, Tchounwou PB, Patlolla AK, Mullick FG, Murakat L, et al. (2005) Environmental pathology and health effects of arsenic poisoning: a critical review. (In: Naidu R, Smith E, Smith J, Bhattacharya P, eds). Managing Arsenic in the Environment: From Soil to Human Health, CSIRO Publishing Corp, Adelaide, Australia.
- Rousselot P, Laboume S, Marolleau JP, Larghero T, Noguera ML, et al. (1999) Arsenic trioxide and melarsoprol induce apoptosis in plasma cell lines and in plasma cells from myeloma patients. Cancer Res 59(5): 1041-1048.
- (1978) National Research Council Canada (NRCC) Effects of Arsenic in the Environment. National Research Council of Canada, Ontario, Canada, pp. 1-349.
- Morton WE, Dunnette DA (1994) Health effects of environmental arsenic. (In: Nriagu JO, eds). Arsenic in the Environment Part II: Human Health and Ecosystem Effects. John Wiley & Sons, Inc, New York, pp. 17-34.
- (2001) National Research Council. Arsenic in Drinking Water.
- Tchounwou PB, Centeno JA (2008) Toxicologic pathology. (In: Gad SC, eds). Handbook of Pre-Clinical Development. John Wiley & Sons New York, pp. 551-580.
- Tchounwou PB, Patlolla AK, Centeno JA (2003) Carcinogenic and systemic health effects associated with arsenic exposure-a critical review. Toxicol Pathol 31(6): 575-588.
- Tchounwou PB, Wilson BA, Abdelgnani AA, Ishaque AB, Patlolla AK (2002) Differential cytotoxicity and gene expression in human liver carcinoma (HepG2) cells exposed to arsenic trioxide and monosodium acid methanearsonate (MSMA) Intl J Mol Sci 3(11): 1117-1132.
- Yedjou GC, Moore P, Tchounwou PB (2006) Dose and time dependent response of human leukemia (HL-60) cells to arsenic trioxide. Intl J Environ Res Public Health 3(2):136-140.
- Chappell W, Beck B, Brown K, North D, Thornton I, et al. (1997) Inorganic arsenic: A need and an opportunity to improve risk assessment. Environ Health Perspect 105(10): 1060-1067.
- Centeno JA, Gray MA, Mullick FG, Tchounwou PB, Tseng C (2005) Arsenic in drinking water and health issues. In: Moore TA, Black A, Centeno JA, Harding JS, Trumm DA, editors. Metal Contaminants in New Zealand. Resolutionz Press, New Zealand, pp. 195-219.
- Abernathy CO, Liu YP, Longfellow D, Aposhian HV, Beck B, et al. (1999) Arsenic: health effects, mechanisms of actions and research issues. Environ Health Perspect 107(7): 593-597.
- Hughes MF (2002) Arsenic toxicity and potential mechanisms of action. Toxicol Lett 133(1): 1-16.
- Wang Z, Rossman TG (1996) In: The Toxicology of Metals. Cheng LW, eds. CRC Press, Boca Raton, Florida, 1: 221-243.
- Belton JC, Benson NC, Hanna ML, Taylor RT (1985) Growth inhibition and cytotoxic effects of three arsenic compounds on cultured Chinese hamster ovary cells. J Environ Sci Health 20(1): 37-72.
- Li JH, Rossman TC (1989) Inhibition of DNA ligase activity by arsenite: A possible mechanism of its comutagenesis. Mol Toxicol 2(1): 1-9.
- Jha AN, Noditi M, Nilsson R, Natarajan AT (1992) Genotoxic effects of sodium arsenite on human cells. Mutat Res 284(2): 215-221.
- Hartmann A, Speit G (1994) Comparative investigations of the genotoxic effects of metals in the single cell gel assay and the sister-chromatid exchange test. Environ Mol Mutagen 23(4): 299-305.
- Patlolla A, Tchounwou PB (2005) Cytogenetic evaluation of arsenic trioxide toxicity in Sprague-Dawley rats. Mut Res - Gen Tox Environ Mutagen 587(1-2): 126-133.
- Basu A, Mahata J, Gupta S, Giri AK (2001) Genetic toxicology of a paradoxical human carcinogen, arsenic: a review. Mutat Res 488(2): 171-194.
- Landolph JR (1989) Molecular and cellular mechanisms of transformation of C3H/10T1/2C18 and diploid human fibroblasts by unique carcinogenic, non- mutagenic metal compounds. A review. Biol Trace Elem Res 21: 459-467.
- Takahashi M, Barrett JC, Tsutsui T (2002) Transformation by inorganic arsenic compounds of normal Syrian hamster embryo cells into a neoplastic state in which they become anchorage-independent and cause tumors in newborn hamsters. Int J Cancer 99(5): 629-634.
- Anderson D, Yu TW, Phillips BJ, Schemezer P (1994) The effect of various antioxidants and other modifying agents on oxygen-radical-generated DNA damage in human lymphocytes in the Comet assay. Mutation Res 307(1): 261-271.
- Saleha Banu B, Danadevi K, Kaiser Jamil, Ahuja YR, Visweswara Rao K, et al. (2001) In vivo genotoxic effect of arsenic trioxide in mice using comet assay. Toxicol 162(3): 171-177.
- Hartmann A, Peit G (1994) Comparative investigations of the genotoxic effects of metals in the single cell gel assay and the sister chromatid exchange test. Environ Mol Mutagen 23(4): 299-305.
- Barrett JC, Lamb PW, Wang TC, Lee TC (1989) Mechanisms of arsenic-induced cell transformation. Biol Trace Ele Res 21: 421-429.
- Tchounwou PB, Yedjou CG, Dorsey WC (2003) Arsenic trioxide - induced transcriptional activation and expression of stress genes in human liver carcinoma cells (HepG2) Cell Mol Biol 49(7): 1071-1079.
- Zhao CQ, Young MR, Diwan BA, Coogan TP, Waalkes MP (1997) Association of arsenic-induced malignant transformation with DNA hypomethylation and aberrant gene expression. Proc Natl Acad Sci USA 94(20): 10907-10912.
- Liu Y, Guyton KZ, Gorospe M, Xu Q, Lee JC, et al. (1996) Differential activation of ERK, JNK/SAPK and P38/CSBP/RK map kinase family members during the cellular response to arsenite. Free Rad Biol Med 21(6): 771-781.
- Ludwig S, Hoffmeyer A, Goebeler M, Kilian K, Hafner H, et al. (1998) The stress inducer arsenite activates mitogen-activated protein kinases extracellular signal-regulated kinases 1 and 2 via a MAPK kinase 6/p38- dependent pathway. J Biol Chem 273(4): 1917-1922.
- Trouba KJ, Wauson EM, Vorce RL (2000) Sodium arsenite-induced dysregulation of proteins involved in proliferative signaling. Toxicol Appl Pharmacol 164(2): 161-170.
- Vogt BL, Rossman TG (2001) Effects of arsenite on p53, p21 and cyclin D expression in normal human fibroblasts- a possible mechanism for arsenite’s comutagenicity. Mutat Res 478(1-2): 159-168.
- Chen NY, Ma WY, Huang C, Ding M, Dong Z (2000) Activation of PKC is required for arsenite-induced signal transduction. J Environ Pathol Toxicol Oncol 19(3): 297-306.
- Porter AC, Fanger GR, Vaillancourt RR (1999) Signal tansduction pathways regulated by arsenate and arsenite. Oncogene 18(54): 7794-7802.
- Soignet SL, Frankel SR, Douer D, Tallman MS, Kantarjian H, et al. (2001) United States multicenter study of arsenic trioxide in relapsed acute promyelocytic leukemia. J Clin Oncol 19(18): 3852-3860.
- Murgo AJ (2001) Clinical trials of arsenic trioxide in hematologic and solid tumors: overview of the National Cancer Institute Cooperative Research and Development Studies. Oncologist 6(2): 22-28.
- Puccetti ES, Guller S, Orleth A, Bruggenolte N, Hoelzer D, et al. (2000) BCR-ABL mediates arsenic trioxide-induced apoptosis independently of its aberrant kinase activity. Cancer Res 60(13): 3409-3413.
- Seol JG, Park WH, Kim ES, Jung CW, Hyun JM, et al. (1999) Effect of arsenic trioxide on cell cycle arrest in head and neck cancer cell-line PCI-1. Biochem Biophys Res Commun 265(2): 400-404.
- Alemany M, Levin J (2000) The effects of arsenic trioxide on human Megakaryocytic leukemia cell lines with a comparison of its effects on other cell lineages. Leukemia Lymphoma 38(1-2): 153-163.
- Deaglio S, Canella D, Baj G, Arnulfo A, Waxman S, et al. (2001) Evidence of an immunologic mechanism behind the therapeutic effects of arsenic trioxide on myeloma cells. Leuk Res 25(3): 237-239.
- Tully DB, Collins BJ, Overstreet JD, Smith CS, Dinse GE (2000) Effects of arsenic, cadmium, chromium and lead on gene expression regulated by a battery of 13 different promoters in recombinant HepG2 cells. Toxicol Appl Pharmacol 168(2): 79-90.
- Lu T, Liu J, LeCluyse EL, Zhou YS, Cheng ML, et al. (2001) Application of cDNA microarray to the study of arsenic-induced liver diseases in the population of Guizhou, China. Toxicol Sci 59(1): 185-192.
- Harris CC (1991) Chemical and physical carcinogenesis: advances and perspectives. Cancer Res 51(18 Suppl): 5023s-5044s.
- Graham-Evans B, Colhy HHP, Yu H, Tchounwou PB (2004) Arsenic-induced genotoxic and cytotoxic effects in human keratinocytes, melanocytes, and dendritic cells. Intl J Environ Res Public Health 1(2): 83-89.
- Stevens JJ, Graham B, Walker AM, Tchounwou PB, Rogers C (2010) The effects of arsenic trioxide on DNA synthesis and genotoxicity in human colon cancer cells. Intl J Environ Res Public Health 7(5): 2018-2032.
- Walker AM, Stevens JJ, Ndebele K, Tchounwou PB (2010) Arsenic trioxide modulates DNA synthesis and apoptosis in lung carcinoma cells. Intl J Environ Res Public Health 7(5): 1996-2007.
- Yedjou CG, Tchounwou PB (2009) Modulation of p53, c-fos, RARE, cyclin A and cyclin D1 expression in human leukemia (HL-60) cells exposed to arsenic trioxide. Mol Cell Biochem 331(1-2): 207-214.
- Yedjou C, Sutton LM, Tchounwou PB (2008) Genotoxic mechanisms of arsenic trioxide effect in human Jurkat T-lymphoma cells. Metal Ions Biol Med 10: 495-499.
- Brown E, Yedjou C, Tchounwou PB (2008) Cytotoxicty and oxidative stress in human liver carcinoma cells exposed to arsenic trioxide. Metal Ions Biol Med 10: 583-587.
- Yedjou CG, Thuisseu L, Tchounwou C, Gomes M, Howard C, et al. (2009) Ascorbic acid potentiation of arsenic trioxide anticancer activity against acute promyelocytic leukemia. Arch Drug Inf 2(4): 59-65.
- Yedjou C, Rogers C, Brown E, Tchounwou P (2008) Differential effect of ascorbic acid and n-acetylcysteine on arsenic trioxide - mediated oxidative stress in human leukemia (HL-60) cells. J Biochem Mol Tox 22(2): 85-92.
- Yedjou GC, Moore P, Tchounwou PB (2006) Dose- and time-dependent response of human leukemia (HL-60) cells to arsenic tic trioxide treatment. Intl J Environ Res Public Health 3(2): 136-140.
- Miller WH, Schipper HM, Lee JS, Singer J, Waxman S (2002) Mechanisms of action of arsenic trioxide - review. Cancer Res 62(14): 3893-3903.
- Gesamp (1987) IMO/FAO/UNESCO/WMO/WHO/IAEA/UN/UNEP Joint Group of Experts on the Scientific Aspects of Marine Pollution: Report of the seventeenth session. World Health Organization, Geneva, Switzerland.
- (1988) Wilson DN Association Cadmium. Cadmium - market trends and influences; London. Cadmium 87 Proceedings of the 6th International Cadmium Conference, pp. 9-16.
- S Environmental Protection Agency (EPA) (2006).
- (1993) International Agency for Research on Cancer (IARC) Monographs - Cadmium, Lyon, France.
- Paschal DC, Burt V, Caudill SP, Gunter EW, Pirkle JL, et al. (2000) Exposure of the U.S. population aged 6 years and older to cadmium: 1988-1994. Arch Environ Contam Toxicol 38(3): 377-383.
- (2008) Agency for Toxic Substances and Disease Registry (ATSDR) Draft Toxicological Profile for Cadmium, GA, Atlanta.
- Satarug S, Baker JR, Urbenjapol S, Haswell-Elkins M, Reilly PE, et al. (2003) A global perspective on cadmium pollution and toxicity in non-occupationally exposed population. Toxicol Lett 137(1-2): 65-83.
- Davison AG, Fayers PM, Taylor AJ, Venables KM, Darbyshire J, et al. (1988) Cadmium fume inhalation and emphysema. Lancet 1(8587): 663-667.
- Mascagni P, Consonni D, Bregante G, Chiappino G, Toffoletto F (2003) Olfactory function in workers exposed to moderate airborne cadmium levels. Neurotoxicol 24(4-5): 717-724.






























