Wear and Corrosion Resistance of Base Oil with Nano Calcined Purified Petroleum Coke and Octadecyl Benzyl Triethanol Ammonium Bromide Ethoxylate. Part 2
AMA Omar*
Egyptian petroleum research institute, Nasr City, Cairo, Egypt.
Submission: July 15, 2022; Published: August 17, 2022
*Corresponding author: AMA Omar, Egyptian petroleum research institute, Nasr City, Cairo, Egypt.
How to cite this article: AMA Omar. Wear and Corrosion Resistance of Base Oil with Nano Calcined Purified Petroleum Coke and Octadecyl Benzyl Triethanol Ammonium Bromide Ethoxylate.Part 2. Recent Adv Petrochem Sci. 2022; 7(4): 555716. DOI: 10.19080/RAPSCI.2022.07.555716
Abstract
Egyptian micro particles petroleum coke (-300 mesh) was purified using a flotation method before being calcined at 600 degrees Celsius. These particles are dispersed in base oil at 0.1-1 wt.% in the presence of 0.00035mol/L dodecyl benzyl tritanol ammonium ethoxylate bromide with 10 units of ethylene oxide (OBT). The effects of (OBT) alone and in conjunction with calcined purified petroleum coke (99.8%) on wear, corrosion resistances of base lubricating oil were studied. The findings revealed that octadecyl benzyl tritanol ammonium bromide ethoxylated and petroleum coke have a synergistic effect as an anti-wear and corrosion inhibitor. Wear resistance improved by 90 percent, and anti-corrosion performance improved by 87 percent. These findings were investigated in terms of OBT dispersing microparticles of petroleum coke in the oil phase and increasing their adsorption on the steel phase. It found that inhibited copper surface is cleaner than blank copper. The results also confirm that mixed OBT and calcined petroleum coke have a synergistic effect. The isotherm was investigated in a manner that was very similar to the Frumkin adsorption isotherm. Equilibrium adsorption was reached in the oil process when relatively low amounts of OBT were utilized at their critical micelle concentration. The adsorbed amount did not show a continuous increase after critical micelle concentration, suggesting that adsorption did not occur after the monolayer was completed, indicating that adsorption did not occur after the monolayer was completed.
Keywords: Lubricating Oil; Additives; Petroleum Coke; Flotation; Anti-Wear; Anticorrosion
Introduction
The inclusion of dispersion to protect nanoparticles solid while also improving their disparity in oil is a popular strategy [1]. Surfactants are surface-active, amphiphilic chemicals that are introduced to a base liquid to lower surface tensions. They are made up of two parts: a polar hydrophilic head group and a polar long hydrocarbon chain that allows them to self-assemble into micelle-like structures and achieve energy balance. When the hydrophobic end of the surfactants occupies the surface of nanoparticles, their potential to cluster/aggregate is greatly reduced like sodium dodecyl sulfate, poly (vinylpyrrolidone), and cetyltrimethylammonium bromide [1,2].
Cheng and colleagues evaluated the effects of different dispersants on the particle size and size distribution of Cu nanoparticles. The extensively employed capping agents are long-chain alkyl thiols, notably O, O′-dialkyl dithiophosphate [3-5]. The size and concentration of copper nanoparticles have been discovered to have a significant impact on their tribological properties [3].
The octadecyl benzyl triethanole ammonium ethoxylate bromide with 20-unit ethylene oxide as wetting and dispersing agent combined activated Si-C-Ni composite powder with base lubricating oil in varying ratios of 1-4% by weight was studied early by the author [6]. Calcium palmitate CAP’s adsorption behavior on petroleum coke and steel improved wear resistance until it reached its CMC. Also, the effectiveness of calcium palmitate adsorbed onto steel surfaces was determined by its CMC. Furthermore, XPS research revealed that C-C bonds were the primary link between the calcium palmitate molecule and the surface of petroleum coke.
The author concluded that calcium palmitate and petroleum coke with a high ash content have a synergistic effect as anti-wear and anti-corrosion resistance of lubricating oil toward contacting surfaces, resulting in a stable coating [7]. Dibutyl 3, 5-di-t-butyl 4-hydroxy benzyl phosphate, triphenyl phosphorothioate, sulfurized octadecanoic, sulfurized docosanoic acid, triethylenetetramine zinc dialkyl dithiophosphate, and poly alkyl methacrylate are some of the most popular additives used in lubricating oil formulations [2-5]. Metal-to-metal contact is reduced by the fluid film. Lubricant selection is critical for reducing friction and material wear. Nanoparticles have a large surface area per volume; they have a lot of surface energy. To increase the properties of polymers, several fillers are widely used, such as glass and carbon fibers, kaolin, talc, and mica. Polyamide has been shown to have a higher wear resistance than other polymers when sliding against a steel counterpart [8-10].
The use of dodecyl benzyle triethanole ammonium ethoxylate bromide with 10 units of ethylene oxide OBT as a dispersion agent for purified calcined petroleum coke in base lubricating oil for anti-wear and anti-corrosion carbon steel was investigated in this study. Frumkin adsorption isotherm on the surface of petroleum coke was used to suggest a mechanism for its efficiency.
Experimental
The author synthesized the octadecyl benzyl triethanole ammonium ethoxylate bromide with 10units of ethylene oxide (OBT) as stated elsewhere [11]. According to Omar et al. [12- 14], the surface attributes of the surfactant, including surface excess and efficiency were calculated. The surfactant’s adsorption isotherm on calcined pure petroleum coke (-300 mesh size) was also examined.
Feeds
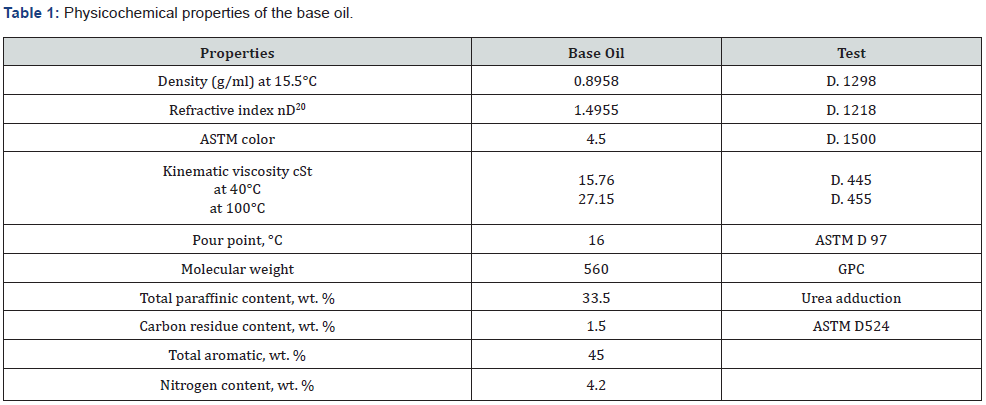
The following table lists the physicochemical properties of paraffinic base oil (Table 1) [6]. After flotation and purification, petroleum coke is separated into concentrate and tail. The author published the ideal circumstances and a comprehensive chemical analysis early [15]. In this paper, we will employ the pure concentrate fraction, which accounts for around 90% of the feed and calcined at 600 0C. The following table 2 shows the complete chemical analysis of the petroleum coke concentrate fraction after calcination.
Tests for Corrosion and Wear
A copper specimen of 30mm in length was embedded in oil contain 0.1% to 1% of dispersed coke (CPK) with and without OBT. The specimens were demoted after 24 hours, and all samples were taken out of the solution, cleaned in petroleum ether, and dried. The copper was then dried and measured after that. The following equation was used to calculate the inhibitor’s efficiency based on the weight reduction.
Percentage efficiency of inhibitor = [(W0 - W) / W0] × 100
Where W0 represents the average final weight (in mg) of copper embedded in oil, and W represents the average final weight (in mg) of copper embedded in inhibited samples [6].
Oil was mixed with different percentages of nano calcined petroleum coke CPK (0.1% to 1%) in the presence of 0.00005mol/L OBT. Wear tests were carried out with sliding speeds of 100, 200, 300, and 400rpm in wet conditions at room temperature according to ASTM G77. The sample’s carbon steel was of the cylindrical shape of 1.5mm diameter and 20mm long. During the test, the sample was passed against stainless steel ring with a different normal load (4-8Kg). Test duration varies from 1-20min for each sample. The wear of the sample was measured by its weight loss [6].
Adsorption Isotherm
Samples of calcined petroleum coke CPK (1.000g) were distributed in 100ml of OBT dissolved in petroleum ether in small vessels that could be tightly sealed with appropriate caps. The suspension present therein was continuously stirred, to achieve equilibrium. After that, the suspension was filtered, and the caked precipitate was dried. The isotherm of adsorption was calculated for various concentrations of additive OBT [6].
Results and Discussions
The inclusion of a dispersion additive to protect petroleum coke nanoparticles and improve their disparity is occurred by OBT. As we know surfactants are amphiphilic, surface-active chemicals that are used to lower surface tensions in a base liquid. They are made up of two parts: a polar hydrophilic head group and a polar long hydrocarbon chain that allows them to selfassemble into micelle-like structures to achieve energy balance. When the hydrophobic end of surfactants occupies the surface of nanoparticles, their potential to cluster/aggregate is greatly reduced. The nanoparticles, which have high surface energy, tend to congregate in colloidal systems, making uniform dispersion difficult. OBT has dispersed the cations and/or counter-anions, making them ideal candidates for overcoming the dispersibility problem. The characteristics of the petroleum coke incorporated lubricant are greatly improved after uniform dispersion is achieved.
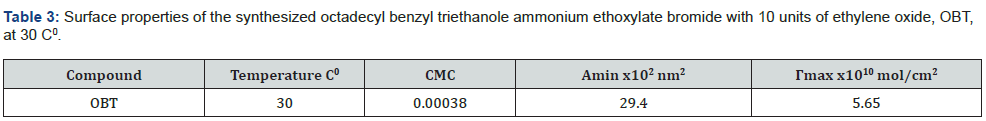
At 30 °C, the surface properties of OBT in petroleum ether 60/80 °C are determined (table 3). It has a CMC value of 0.00038 mole/L, which is owing to the tail group of the surfactant being more hydrophobic than the hydrophilic group (-triethanol ammonium ethylate). OBT has a surface excess of 5.65 x 10-10mole/ cm2 indicating that it prefers to adsorb on the surface rather than form micelles. The essential surface tension was confirmed by the surface tension reduction efficiency at CMC (18mN/m).
Adsorption Isotherms
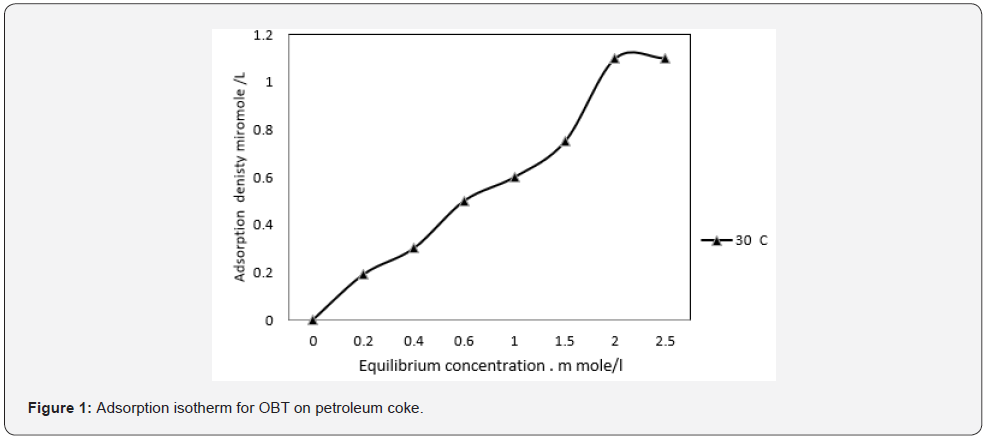
The OBT adsorption isotherms onto petroleum coke fraction are shown in (figure 1 at 30°C) Frumkin adsorption isotherm is comparable to the isotherm illustrated. Equilibrium adsorption was reached at 1.08m.mol, when relatively low amounts of OBT were utilized in the petroleum ether at their critical micelle concentration. These results are due to OBT’s preference to adsorb on oil and various phases or contact surfaces. Adsorption did not occur after the monolayer was formed, as evidenced by the continuing increase in adsorbed amount until critical micelle concentration. It is clear the adsorption on petroleum coke surface takes place by free molecule ion and reaches a maximum at its critical micelle concentration of dissolved OBT which is known as CMC. This indicates that OBT adsorption onto petroleum coke is the product of physical adsorption.
Wear and Corrosion Tests
Petroleum coke CPK was used as additives in base oil with OBT. It was found that the addition of CPK at 1wt. %, reduce wear of both antagonist surfaces. Figure 2 depicts the wear loss/ area (wear rate) of oil samples when subjected to various loads and sliding times. It is obvious that when the ratio of petroleum coke to absorbed OBT increases, the wear resistance increases. Wear resistance is increased by 90% when petroleum coke is scattered and decreases slightly with increasing time. The author suggests that the additive molecule OBT and distributed calcined petroleum coke CPK, adsorbed on the metal surface to form an adsorbed layer between contact surfaces.
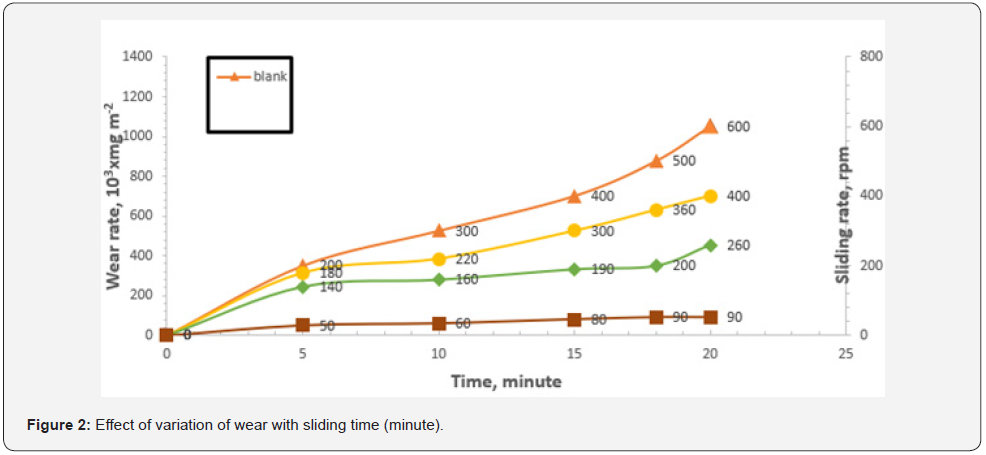
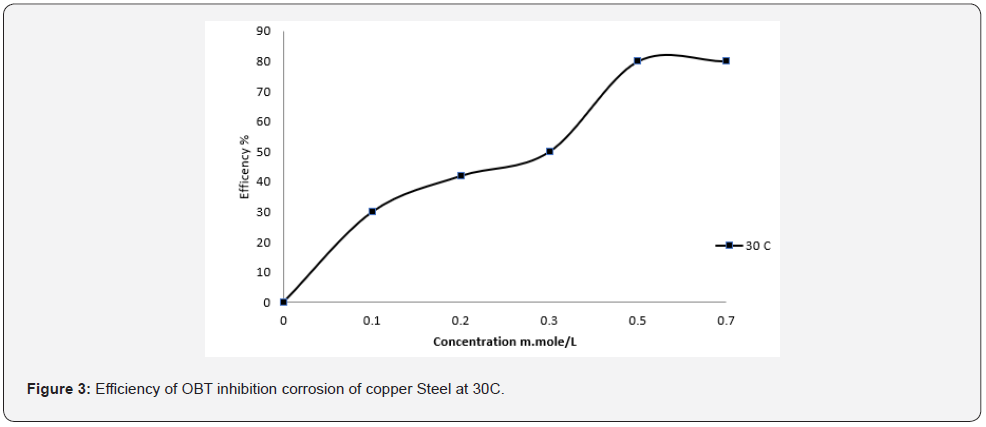
On the other hand, OBT has an inhibitory efficiency of 85 percent in oil, with a peak around its CMC, which can be attributed to OBT adsorption on metal surfaces, which forms a barrier layer as shown in figure 3. This result indicates the degree of adsorption of OBT and the rise in surface coverage till CMC, which is consistent with previous work [6, 7].
Conclusions
The tribological applications of calcined purified petroleum coke nanoparticles CPK as lubricant additives have been studied. OBT is used to prevent nanoparticles from aggregating in a base oil. OBT’s adsorption behavior on petroleum coke and steel improved wear resistance until it reached its CMC. As a result, OBT and petroleum coke CPK have a synergistic effect as anti-wear and anti-corrosion resistance of lubricating oil toward contacting surface, generating a stable layer. The increased surface area of petroleum coke concentrate PK makes it more effective in antiwear. Also, the efficiency of OBT adsorbed onto steel surfaces was proven to be dependent on its CMC.
References
- Liu W, Wang X (2008) Nano lubricants Made of Metals. In: Martin JM, Ohmae N (eds.), Nano lubricants. John Wiley & Sons: Chichester, UK, pp. 93-147.
- Parka BK, Jeong S, Kim D, Moon J, Lim S, et al. (2007) Synthesis and size control of monodisperse copper nanoparticles by polyol method. J Colloid Interface Sci 311(2): 417-424.
- Zhou J, Yang J, Zhang Z, Liu W, Xue Q (1999) Study on the structure and tribological properties of surface-modified Cu nanoparticles. Mater Res Bull 34(9): 1361-1367.
- Lisiecki I, Billoudet F, Pileni MP (1996) Control of the shape and the size of copper metallic particles. J Phys Chem 100(10): 4160-4166.
- Zhang X, Yin, H Cheng X, Hu H, Yu Q, et al. (2006) Effects of various polyoxymethylene sorbitan (tweens) and sodium dodecyl sulfate on reflux synthesis of copper nanoparticles. Mater Res Bull 41(11): 2041-2048.
- Omar AMA (2019) Wear Resistance of Lubricating Oil Contains Nano Composite Ni Si-C and Dodecyl Benzyl Triethanol Ammonium Bromide Ethoxylated with 20 Unit of Ethylene Oxide. J Miner Met Mater Eng 5: 33-37.
- Omar AMA (2021) Tribological characterization of lubricants oil with nano concentrated highly ash Egyptian petroleum coke and calcium palmitate. Maghrebian Journal of Pure and Applied Science 7(2).
- Chen L, Zhang D, Chen J, Zhou H, Wan H (2006) The use of CTAB to control the size of copper nanoparticles and the concentration of alkyl thiols on their surfaces. Mater Sci Eng 415(1-2): 156-161.
- Kukureka SN, Hooke CJ, Rao M, Liao P, Chen YK (1999) The effect of fibrereinforcement on the friction and wear af polyamide 66 under dry rolling-sliding contact. Tribol Int 32: 107-116.
- Yang EL, Hirvonen JP, Toivanen RO (1991) Effect of temperature on the transfer film formation in sliding contact of PTFE with stainless steel. Wear 146(2): 367-376.
- Osman MM, Omarv El- Sabagh (1997) Corrosion inhibition of benzyl triethanol ammonium chloride and its ethoxylate on steel in sulfuric acid solution. J Material chemistry and physics 50(3): 271-274.
- Morsi AK, Omar AMA (2009) Development of a nickel polymer composite by using friendly environmental polymeric surfactants. Material Science and Engineering 521-522: 397- 400.
- Abdel Hamid Z, Omar AMA (2003) Development of electrodeposition of nickel polymer composite by zwitterionic surfactants. J of surfactants and detergents 6: 163-166.
- Abdel Hamid Z, Omar AMA (1999) The relation between anionic/nonionic surfactant and electrodeposition of Ni-polytetrafluoroethylene polymer composite. J Anti-corrosion method and materials 46(3): 212.
- Omar AMA (1994) PhD Thesis, Ain Shams University, Faculty of Science.






























