Remediation of Marine Oil Spills and Water Pollution using Low Molecular Weight Organo-Gelators
Varsha Bhardwaj*and Amar Ballabh*
Department of Chemistry, Faculty of Science, The Maharaja Sayajirao University of Baroda, Vadodara, India
Submission: July 1, 2022; Published: July 11, 2022
*Corresponding author: Varsha Bhardwaj and Amar Ballabh, Department of Chemistry, Faculty of Science, The Maharaja Sayajirao University of Baroda, Vadodara, Gujarat, India.
How to cite this article: Varsha B, Amar B. Remediation of Marine Oil Spills and Water Pollution using Low Molecular Weight Organo-Gelators. Recent Adv Petrochem Sci. 2022; 7(3): 555714. DOI: 10.19080/RAPSCI.2022.07.555714
Abstract
Low molecular weight organo-gelators (LMOGs) can be employed for efficient removal of oil, toxic dyes, metal ions as well as organic pollutants from water bodies. An effective self-assembly assisted by non-covalent interactions holds the key to formation of a three-dimensional network capable of immobilizing the oil phase. The authors here present a personal perspective on the basic understanding and credibility of such gelation systems.
Keywords: Organo-gelators; Oil spills; Water pollution; Biocompatibility; Futuristic aspects; Organo gel
Abbreviations: LMOGs: Low Molecular Weight Organo-Gelators, PSOG: Phase Selective Organo-Gelation
Introduction
Molecular gels are self-assembled systems where the solute molecules (gelators) interact and combine via various noncovalent interactions such as H-bonding, van der Waals forces, π-π bonds, hydrophobic interactions, charge transfer, etc. and form one dimensional fibers, which on aggregation, form 3D assemblies which can trap a solvent, forming a gel. Gels are visco-elastic materials [1-3] which display flow characteristic on application of strain. And as the saying goes, if you wish to identify if a given substance is a gel or not, well, “If it looks like Jell-O, it’s a gel…” [4] Gels can defy gravity and an inverted vial containing a gel will likely resist its ability to flow. The underlying principle seems very simple, yet it gets complex as we dig in deeper. Since it depends on a variety of factors whether a particular molecule will congeal a particular solvent or not, most of the gelators have been discovered by serendipity and not by design. The mechanism at molecular level is not even fully understood till today. As was rightly quoted by D. J. Lloyd, “the colloid condition, the gel, is easier to recognize than to define” [5].
Gels are broadly classified based on solvents gelled by them; hydrogels, where the solvent being gelled is water and organo gels, where organic solvents are involved. The gelator molecules can belong to various classes like polymers, multi-component systems, small molecules, peptides and many more! They are employed for various high-tech applications in molecular sensing, energy storage, catalysis, tissue engineering, drug delivery, templating, cosmetics, dyes, adhesives, thickening agents, and many more [6]. They also have a great potential to be used as smart materials as they are highly versatile systems which can respond to light, heat, ultrasound waves, pH, etc. They inevitably stand out because of their indispensability, biocompatibility, and programmability.
LMOGs for Tackling Oil Spills
Marine oil spills are the result of natural or accidental discharge of crude oil and petrochemicals in the water bodies. In April 2010, the Deepwater Horizon oil rig suffered an explosion in the Gulf of Mexico and leaked millions of barrels of crude oil in the ocean. This, however, was not the first report of marine oil spill. According to statistics, more than a thousand oil spills are reported every year. While they are not necessarily as dreadful as the Deepwater Horizon, they still cause long-term damage and pose serious threat to marine ecosystem. The consequences can be disastrous, which contribute to environmental pollution, severely affected marine flora and fauna, spillage of non-renewable fossil fuels, to name a few. As far as tackling of oil spills is concerned, there are various methods available like absorption, dispersion, burning, bioremediation (use of microbes that feed upon and digest the oil), fertilizers, etc. [7-10] Among these, the efficiency of supramolecular gelation is unsurpassed, as it involves gelation of all the oil present on the water surface, which can be skimmed off in a semi-solid gel form and heated to retrieve back the oil.
The thermoreversibility and non-destructive nature of these systems offer added advantage as they facilitate recovery of oil, and at the same time, the gelator can be recycled and reused. [11-15]. Although the idea of employing LMOGs for treating oil spills has been known since the 1970s, it was only in 2001 when a solid report of amino acid based LMOG by Bhattacharya et. al. burst the bubble around the theory. Gels were reported in aliphatic hydrocarbons as well as commercial oils even in biphasic oil-water mixtures [16]. Since then, a variety of different molecules such as organic salts (multi-component systems) [17-21] amides [22,23] esters [24] polymers [25,26] urea-based moieties [27], amino acid derivatives (peptides) [28-31] cholesterol-based moieties [32,33] sugar-based moieties [34-37] etc. have been exploited for their ability to selectively gel organic solvents in presence of water (figure 1).
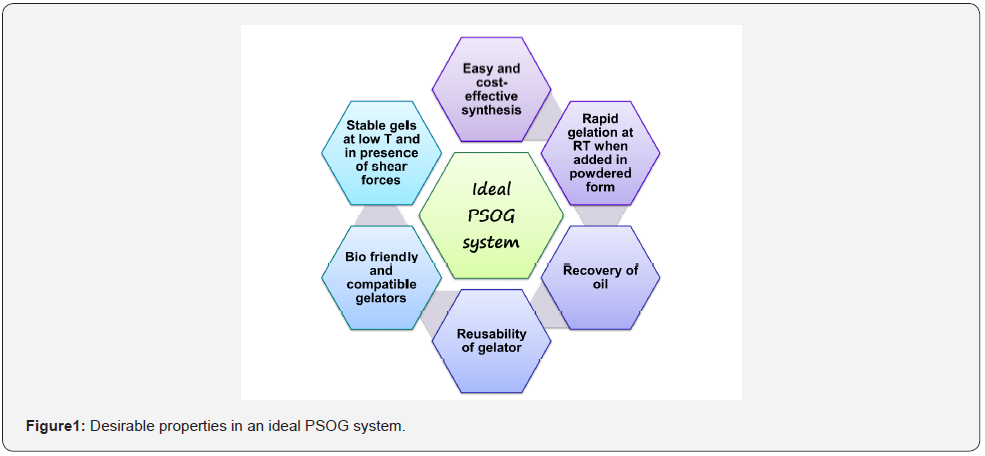
Even though, there are certain practical problems that make the use of LMOGs for phase selective organo-gelation (PSOG) ambiguous. For e.g., such systems involve a heat-cool cycle or use of a co-solvent which is ideally miscible in both phases. This allows an even dispersion of gelator molecules in dissolved form, which can be advantageous in forming a stronger gel. However, use of co-solvent causes contamination of both, organic as well the aqueous phase, and thus, can end up doing more harm than good. Hence it is ideal for a gelator to selectively congeal oil in presence of aqueous media at room temperature, preferably in powdered form. Another key point is, most of the studies reporting PSOG focus on gelation of kerosene, petrol, diesel, engine oil, etc. Crude oil-based reports are limited in literature because it remains unavailable to most of the researchers. The reason behind raising this point is, crude oils can contain natural acids and other elements which can impede gelation behavior [21], and since such systems are highly sensitive towards the constituents of both phases, we can comfortably say that systems congealing petrol, diesel and kerosene may not necessarily form gels in crude oil.
As far as designing of PSOGs is concerned, there are various reports in literature which highlight different aspects of molecules, in terms of interactions, crystal engineering, structureproperty relationship, etc. [38-43]. Nonetheless, the key to a successful design should inevitably consider all these facets. The amphiphilicity of gelator in any biphasic system governs its ability to interact with both the phases. It also regulates the formation of fibers which self-assemble via various interactions to immobilize a solvent. It is believed that hydrophobic interactions control the self-assembly in hydrogels and hydrophilic interactions control the gelation behavior in organic solvents. In presence of a biphasic system, such interactions can be disabled, and a highly amphiphilic gelator is likely to remain at the interface, interacting with both the phases, and consequently, it will fail to gel either solvent or invariably form an emulsion [44-47].
LMOGs for Controlling Water Pollution
Supramolecular gels can be very efficiently employed for sequestration of dyes dissolved in aqueous medium. The self assembled fibers of an organo gel can interact with the dye molecules present in water. This causes the dyes molecules to be absorbed by the gel, thereby eliminating them from water. In certain reports, gelation has been carried out along with the dye molecules. This can significantly decrease the concentration of pollutant dye from water, with impressive adsorption efficiencies. The same concept can be applied for efficient and selective uptake of heavy metal ions, anions, nano particles, and even certain organic pollutant species [11,48]. Many of the reported studies have also developed mechanisms of ‘switch-on/off’ gelation, where gelation is triggered or obstructed in presence of the active moieties. This can ease recovery of precious ions and contribute to tackling water pollution at large.
Conclusions and Futuristic Aspects
In summary, much progress has been made in PSOG and its extended high-tech applications. However, there are many aspects which can be developed to enhance our understanding of topics in hand and this article touches only the tip of iceberg. An ideal designing paradigm is highly favorable for getting much needed insights into the practical applications for crude oil gelation, interactions between the pollutant ions/species and gelator fibers, reusability of gelators, selectivity of gelation and pollutant uptake, etc. The design of new LMOGs is always challenging due to multiple factors such as 1 tides, 2. continuous flow of hot/cold air may bring disruption to the formation of organo gel containing fuel/crude oil 3. especially in the hostile marine environment. The design of an ideal candidate which can exhibit gelation behavior calls for a universal gelling compound in all petrochemicals, such as crude oil and commercial fuels with a high mechanical strength and sol-gel transition induced by sunrays. Moreover, a stable gel containing fuel can assist in transportation as well. These factors can contribute to the ultimate challenge of creating designer, selfprogrammable soft materials with desired applications.
Acknowledgment
The authors thank Department of Chemistry, Faculty of Science, The Maharaja Sayajirao University of Baroda, for providing infrastructural facilities.
References
- (2006) Molecular Gels. Materials with Self-Assembled Fibrillar Networks. Eds. RG Weiss, P Terech, Springer, Dordrecht, USA.
- (2018) Molecular Gels. Structure and Dynamics. Ed. RG Weiss. Royal Society of Chemistry, London.
- Dastidar P (2019) Designing Supramolecular Gelators: Challenges, Frustrations, and Hopes Gels 5(1): 15.
- Terech P, Weiss RG (1997) Low molecular mass gelators of organic liquids and the properties of their Gels Chem Rev 97(8): 3133-3159.
- Lloyd DJ (1926) Colloid Chemistry. 1. Chemical Catalog Co, New York.
- Hirst AR, Escuder B, Miravet JF, Smith DK (2008) High-tech applications of self-assembling supramolecular nanostructured gel-phase materials: From regenerative medicine to electronic devices. Angew Chem Int Ed Engl 47(42): 8002-8018.
- Schrope M (2011) Oil spill: Deep wounds. Nature 472: 152-154.
- Wang S, Liu K, Yao X, Jiang L (2015) Bioinspired surfaces with super wettability: new insight on theory, design, and applications. Chem Rev 115(16): 8230-8293.
- Buist L, McCourt J, Potter S, Ross S, Trudel K (1999) In situ burning. Pure Appl Chem 71(1): 43-65.
- Adebajo MO, Frost RL, Kloprogge JT, Carmody O, Kokot S (2003) Porous Materials for Oil Spill Cleanup: A Review of Synthesis and Absorbing Properties. J Porous Mater 10: 159-170.
- Okesola BO, Smith DK (2016) Applying low-molecular weight supramolecular gelators in an environmental setting-self- assembled gels as smart materials for pollutant removal. Chem Soc Rev 45: 4226-4251.
- Ohsedo Y (2016) Low-molecular-weight organo gelators as functional materials for oil spill remediation. Polym Adv Technol 27(6): 704-711.
- Vibhute AM, Sureshan KM (2020) How Far Are We in Combating Marine Oil Spills by Using Phase-Selective Organo gelators? Chem Sus Chem 13(20): 5343-5360.
- Kim H, Zhang G, Chung TCM, Nam C (2022) A Role for Newly Developed Sorbents for Remediating Large-Scale Oil Spills-Reviewing Recent Advances and Beyond Adv Sustain Syst 6(1): 2100211.
- Jin G, Hao YZ, Hong WZ, Jin H, Lu AS, et al. (2016) Advanced Sorbents for Oil-Spill Cleanup: Recent Advances and Future Perspectives Adv Mater 28(47): 10459-10490.
- Bhattacharya S, Krishnan GY (2001) First report of phase selective gelation of oil from oil/water mixtures. Possible implications toward containing oil spills. Chem Commun (2): 185-186.
- Trivedi DR, Ballabh A, Dastidar P (2003) An Easy to Prepare Organic Salt as a Low Molecular Mass Organic Gelator Capable of Selective Gelation of Oil from Oil / Water Mixtures. Chem Mater 15(21): 3971-3973.
- Trivedi DR, Ballabh A, Dastidar P, Ganguly, B (2004) Structure-property correlation of a new family of organo gelators based on organic salts and their selective gelation of oil from oil/water mixtures. Chem - A Eur J 10(21): 5311-5322.
- Trivedi DR, Ballabh A, Dastidar P (2005) Facile preparation and structure-property correlation of low molecular mass organic gelators derived from simple organic salts. J Mater Chem 15(26): 2606-2614.
- Ballabh A, Trivedi DR, Dastidar P (2006) New series of organo gelators derived from a combinatorial library of primary ammonium monocarboxylate salts. Chem Mater 18(16): 3795-3800.
- Yan L, Li G, Ye Z, Tian F, Zhang S (2014) Dual-responsive two-component supramolecular gels for self-healing materials and oil spill recovery. Chem Commun 50(94): 14839-14842.
- Eftekhari SB, Bagheri A, Younesi AH, Akbari A, Paige FM (2020) Supramolecular self-assembly of oleylamide into organogels and hydrogels: a simple approach in phase selective gelation of oil spills. Soft Materials 18(1): 55-66.
- Bhardwaj V, Ballabh A (2021) A series of multifunctional pivalamide based Low Molecular Mass Gelators (LMOGs) with potential applications in oil-spill remediation and toxic dye removal. Colloids Surfaces a Physico chem Eng Asp 632: 127813.
- Yu H, Liu B, Wang Y, Wang J, Hao Q (2011) Gallic ester-based phase-selective gelators. Soft Matter 7(11): 5113-5115.
- Suzuki M, Hanabusa K (2010) Polymer organogelators that make supramolecular organogels through physical cross-linking and self-assembly. Chem Soc Rev 39(2): 455-463.
- Hanabusa K, Suzuki M (2014) Development of low-molecular-weight gelators and polymer-based gelators. Polym J 46: 776-782.
- Hoshyar MP, Mohammad BZ, Rahmati A (2021) Synthesis and application of bisurea derivatives: Effect of structural differences on the gelation properties. J Environ Chem Eng 9(3): 105220.
- Konda M, Maity I, Rasale DB, Das AK (2014) A new class of phase-selective synthetic β-amino acid-based peptide gelator: From mechanistic aspects to oil spill recovery. Chempluschem 79(10): 1482-1488.
- Agarwal DS, Gogoi N, Chowdhury D, Sakhuja R (2016) Amino acid appended cholic acid-azobenzene dyad: An effective & smart phase selective gelator for aromatic solvents. RSC Adv 6(80): 76632-76641.
- Li J, Huo Y, Zeng H (2018) Combinatorial identification of a highly soluble phase-selective organogelator with high gelling capacity for crude oil gelation. J Mater Chem A 6(22): 10196-10200.
- Guchhait S, Roy S (2019) Efficient peptide based gelators for aromatic organic solvents and vegetable oils: application in phase selective gelation and dye entrapment. J Sol-Gel Sci Technol 89(3): 852-865.
- Xue M, Gao D, Liu K, Peng J, Fang Y (2009) Cholesteryl derivatives as phase-selective gelators at room temperature. Tetrahedron 65(17): 3369-3377.
- Panja A, Raza R, Ghosh K (2020) Cholesterol-Coupled Diazine-Phenol Gelator: Cyanide Sensing, Phase-Selective Gelation in Oil Spill Recovery and Dye Adsorption. Chemistry Select 5(38): 11874-11881.
- Jadhav SR, Vemula PK, Kumar R, Raghavan SR, John G (2010) Sugar-Derived Phase-Selective Molecular Gelators as Model Solidifiers for Oil Spills. Angew Chemie 49(42): 7695-7698.
- Shao ZL, Wei LL, Zhi GL, Jing CL, Xiao CZ, et al. (2013) A phase-selective supramolecular gelator as oil solidifier. Appl Mech Mater 457-458: 284-287.
- Vibhute AM, Muvvala V, Sureshan KM (2016) A Sugar-Based Gelator for Marine Oil-Spill Recovery. Angew Chem Int Ed Engl 55(27): 7782-7785.
- Soundarajan K, Mohan DT (2019) Sugar-benzohydrazide based phase selective gelators for marine oil spill recovery and removal of dye from polluted water. Carbohydr Res 481: 60-66.
- Escuder B, Miravet JF (2006) Silk-inspired low-molecular-weight organogelator. Langmuir 22(18): 7793-7797.
- Andrew RH, Ian AC, Thomas RB, Juan FM, Beatriu E, et al. (2008) Low-molecular-weight gelators: Elucidating the principles of gelation based on gelator solubility and a cooperative self-assembly model. J Am Chem Soc 130(28): 9113-9121.
- Dastidar P, Roy R, Parveen R, Sarkar K (2019) Supramolecular Synthon Approach in Designing Molecular Gels for Advanced Therapeutics. Adv Ther 2(1): 1800061.
- Aramballi JS, Oxana K, Sankarasekaran S, Samuel JB, Gearóid MÓ, et al. (2017) Supramolecular Chemistry: A Toolkit for Soft Functional Materials and Organic Particles. Chem 3(5): 764-811.
- Dastidar P (2008) Supramolecular gelling agents: can they be designed? Chem Soc Rev 37(12): 2699-2715.
- Dawn A, Shiraki T, Haraguchi S, Tamaru S, Shinkai S (2011) What Kind of “Soft Materials” Can We Design from Molecular Gels? Chem Asian J 6(2): 266-282.
- Raghavan SR, Douglas JF (2012) The conundrum of gel formation by molecular nanofibers, wormlike micelles, and filamentous proteins: Gelation without cross-links? Soft Matter 8(33): 8539-8546.
- Weiss RG (2014) The Past, Present, and Future of Molecular Gels. What Is the Status of the Field, and Where Is It Going? 136(21): 7519-7530.
- Du X, Zhou J, Shi J, Xu B (2015) Supramolecular Hydrogelators and Hydrogels: From Soft Matter to Molecular Biomaterials. Chem Rev 115(24): 13165-13307.
- Yue X, Subraveti SN, John G, Raghavan SR (2021) Phase-Selective Gelation of the Water Phase in an Oil-Water Mixture: An Approach Based on Oil-Activated Nanoparticle Assembly in Water. Langmuir 37(27): 8107-8114.
- Piepenbrock MOM, Lloyd GO, Clarke N, Steed JW (2010) Metal- and Anion-Binding Supramolecular Gels. Chem Rev 110(4): 1960-2004.






























