Solid Fuel Production from Straw
Hassan Al-Haj Ibrahim*
Arab University for Science and Technology, Syria
Submission: July 12, 2019; Published: August 26, 2019
*Corresponding author: Hassan Al-Haj Ibrahim, Arab University for Science and Technology, Hamah, Syria
How to cite this article: Hassan Al-Haj Ibrahim. Solid Fuel Production from Straw. Recent Adv Petrochem Sci. 2018; 6(4): 555691. DOI: 10.19080/RAPSCI.2019.06.555691
Abstract
Straws are agricultural by-products available in large quantities, and if utilized efficiently, can provide appropriate feedstocks for competitive energy production with consequent reduction of dependence on fossil fuels. Straws can be used either as a solid fuel of high calorific value and reduced pollutants emission or they may be converted into coke-like biomass or charcoal fuel. They may be used either separately or mixed with other biomass or waste products.
Keywords: Straw Size reduction Densification Torrefaction Combustion Solid fuel Pyrolysis Palletisation Van krevelen coal
Introduction
Straws are agricultural by-products available in large quantities. They include in general both grain and grass straws. Grain straws are the dry stalks of cereal plants, after the grain and chaff have been removed. Straws make up about half of the yield of cereal crops such as barley, canola, corn, oats, rice, rye, sorghum and wheat. Of these straws rice and wheat straws are abundant agricultural residues with low commercial value.
If utilized efficiently, straws can provide appropriate feedstocks for competitive energy production with consequent reduction of dependence on fossil fuels. In China alone, it is estimated that 400 million tonnes of wasted straw could be used as a substitute for 200 million tonnes of coal [1].
Straws can be used either as a solid fuel of high calorific value and reduced pollutants emission or they may be converted into coke-like biomass or charcoal fuel. They may be used either separately or mixed with other biomass or waste products
The use of Straw as a Solid Fuel
Straw fuel may be used directly in the form of bales. Raw straw, however, may have inferior properties which makes it very difficult to handle, transport, store and utilise in its original form. In particular, raw straw has high moisture content, low energy and bulk densities and irregular shape and sizes. Furthermore, the fibrous nature of raw straw makes it tenacious and difficult to reduce to small homogeneous particles. These properties of straw can have negative impacts during energy conversion such as lower combustion efficiencies and gasifier design limitations [2,3].
The raw straw has to be processed in order to facilitate efficient handling, transportation and usage of straw in commercial operations. The entire process involves securing of baled straw from agricultural fields followed by size reduction (chopping and grinding) and densification or torrefaction.
Size reduction
Is a critical process prior to densification. It is an important energy intensive unit operation essential for bioenergy conversion process and densification to reduce transportation costs [4,5]. Particle size reduction increases the total surface area, pore size of the material and the number of contact points for inter-particle bonding in the compaction process [6].
For grinding, hammer mills are normally used as they are relatively inexpensive, easy to operate and because of their ability to finely grind a greater variety of materials than any other machines and produce a wide range of particles [7,8].
Various types of size reduction equipment’s, including a multi-blade cutter, a hammer mill, a refiner, and a colloid mill, were used in the study of the pre-treatment of rice straw from the viewpoint of energy consumption. The specific energy consumptions for grinding wheat straw with the hammer mill screen sizes of 0.8 and 3.2 mm were 51.6- and 11.4-kW h t-1, respectively [9].
Grinding experiments were conducted by Adapa et al. [10] on barley, canola, oat and wheat straw using a forage chopper and a hammer mill (screen sizes of 30, 6.4, 3.2 and 1.6 mm) to determine specific energy requirements, and geometric mean particle size and distribution of ground material. Specific energyrequired by hammer mill showed a negative power correlation with hammer mill screen sizes. The highest specific energy was consumed by wheat straw (42.572.04 kWh t-1) at 1.6 mm and the lowest by canola straw (1.460.30 kWh t-1) using 30 mm hammer mill screen size. The chopper consumed highest specific energy (3.15 0.09 kWh t-1) to chop barley and lowest specific energy (1.96 0.33 kWh t-1) to chop canola straw. The particle density of canola and oat straw significantly increased with a decrease in hammer mill screen size from 30 to 1.6 mm [10].
Densification
Of biomass including straws is a viable technology for upgrading and converting biomass materials into durable highgrade briquettes or pellets with substantially superior properties when compared with standard wood pellets. Densification is carried out by a process of compression and extrusion through a die. By densification of straw, its bulk density can be increased (from an initial bulk density of 40–200 kg m-3 to a final compact density of 600–1200 kg m-3 and a bulk density of 650 kg m-3) and material waste reduced. In addition, the uniform shapes and sizes of the densified products make them easy to handle using standard handling and storage equipment and allow the straw to be transported over longer distances. The densified products can also be easily adapted not only in direct combustion in typical burners or co-firing with coal or natural gas at very high rates making use of the processing infrastructures at existing coal and gas plants, but also in gasification, pyrolysis and other biomassbased conversions [3,11,12].
The energy requirement for densification depends primarily upon the applied pressure and the method of compaction. It also depends on the moisture content of the material and its physical properties [3]. The mean values of specific compression energy can range from 5-39 MJ t-1. According to Shaw, wheat straw consumed 39.1 MJ t-1 while pre-treated wheat straw using steam explosion consumed only 7.2 MJ t-1 [13]. The compression of barley straw in a circular die required a specific energy consumption of 5–25 MJ t-1 depending on the compact density [14]. Faborode and O’Callaghan reported that chopped barley straw consumed 28–31 MJ t-1 of energy, while un-chopped material consumed 18–27 MJ t-1 [15].
In order to customise and manufacture high quality densified products, it is essential to consider the effects of the various independent variables such as applied pressure, grind size and moisture content on desirable quality parameters including density and durability [3].
Increasing the applied pressure increases the compact density. The mean densities of barley, canola, oat and wheat straw compacts increased from 907 to 988 kgm-3, 823 to 1003 kgm-3, 849 to 1011 kgm-3 and 813 to 924 kgm-3, respectively, upon application of pressure in the range of 31.6–138.9 MPa. The specific energy required for compression and extrusion of a compact significantly increased with an increase in pressure [3].
Work by different researchers indicates that the finer the grind, the higher the density and quality of compact [129]. Also, lower moisture materials produce in general denser compacts. However, there is in practice an optimum moisture content for the production of high-quality pellets, as the moisture in biomass acts as a facilitator of natural binding agents and a lubricant at the same time [3].
Rice straw pellets may be manufactured by moisturizing cut rice straw with water and extruding the moisturised straw through dies, wherein the amounts of water and extrusion pressure are controlled [16].
Solid fuel briquettes may be made from straw by a method comprising crushing and compression, where a piston-mould process may be used for compression. The controlling factors in this process which greatly affect the compressive strength of the briquettes include the size distribution of the crushed straw, the pressing temperature and die pressure. Die pressure in particular has a significant effect on the physical characteristics such as elongation and voidage of the briquettes formed and hence on their combustion characteristics as was confirmed by experimental studies in which the samples were densified under die pressure ranges of 20-140 MPa [11,12]. When samples of rice straw were densified the optimum die pressure was found to be about 80 MPa [12]. Mixing a certain percentage of a binder such as rice bran, soybean residue and sawdust (of for ex. Acacia confuse) or other biomass waste with the crushed straw was found to increase the calorific value of the briquettes and decrease the energy required for compression. The effects of the binder are related to its type and percentage ratio [17,18].
Powder mixtures of rice straw (especially Oryza sativa), bamboo or wood sawdust with calcium compounds (e.g. Ca (OH)2) may be used for the production of straw briquettes by extrusion of the mixture through rollers. The briquettes are suitable for combusting in a boiler with reduced noxious emissions, and the boiler flue gases are treated in an adsorption tower to remove NOx, SOx and dust, and the resulting gases containing CO2 are then utilised in photosynthesis for the plant growth [19].
Different straws such as rice, wheat, corn, sorghum and tobacco straws may be mixed with other combustible wastes such as wood chips and herbal residues, or crop processing residues (peanut shell, rice hull, bagasse) and the resulting mixture used for the production of fuel rods or block-shaped fuel through a process of crushing or pulverizing, oven-drying, screening, compressing, and moulding or extruding. An additive containing a clay component (aluminium silicate) may be added before extrusion moulding. The compressed fuel rods have the advantages of long combustion time, high calorific value (15,900 kJ/kg), low cost, and little pollution. They can be used for combustion directly in boilers and power generation or they may be used in the production of charcoal. The ashes generatedin the combustion of the fuel rods can be used as fertiliser [20- 23].
A 50% savings in energy was achieved when a uniaxial press was applied for the densification of straw for the production of briquettes or pellets and the bulk density lowered from 1.00 to 0.80 g/cm3 [24].
Torrefaction
Is a mild form of pyrolysis or thermal treatment in an inert atmosphere at temperatures typically ranging between 200- 320°C. It serves to improve the properties of biomass in relation to thermochemical processing techniques for energy generation such as combustion, co-combustion with coal or gasification. Torrefaction also eliminates all biological activity reducing the risk of fire and stopping biological decomposition. About 10% of the energy content in the biomass is lost as a result of the torrefaction process, but this energy of the volatiles can be used as a heating fuel for the process itself. During torrefaction, moisture and low weight organic volatile components are removed, and the biomass loses typically 20% of its mass (dry bone basis). In addition, torrefaction partly depolymerises the biopolymers (cellulose, hemicellulose and lignin) and the long polysaccharide chains, producing a hydrophobic, dry, blackened solid product as “torrefied biomass” or “bio-coal” with an increased energy density (on a mass basis) and greatly increased grindability. As a result, significantly lower energy is required to process the torrefied fuel and it no longer requires separate handling facilities when co-fired with coal in existing power stations [2].
Experiments at different torrefaction conditions in nitrogen were undertaken by Bridgeman et al. to examine optimization of the process for wheat straw. Progress of torrefaction was also followed by chemical analysis (C, H, N, O, ash), and it was seen that the characters of the straw fuel begin to resemble those of low rank coals in terms of the van Krevelen coal rank parameter. In addition, the results indicated that the volatile component of straw is both reduced and altered producing a more thermally stable product with higher calorific value. The difference between the mass and energy yield was shown to improve for the higher torrefaction temperatures investigated. The combustion behaviour of raw and torrefied fuels was studied further by differential thermal analysis. It was shown that both volatile and char combustion of the torrefied sample become more exothermic compared to the raw fuels, and that depending on the severity of the torrefaction conditions, the torrefied fuel can contain up to 96% of the original energy content on a mass basis [2].
Torrefaction of straw with pelletisation is gaining attention, because it increases its energy density, making it possible to transport it still further. This processing step also makes storage much easier, because torrefied straw pellets are hydrophobic. Because the torrefied straw pellets have superior structural,chemical and combustion properties to coal, they can replace all coal and turn a coal plant into an entirely biomass-fed power station.
Conversion of Straw into Coke-Like Biomass or Charcoal Fuel
High calorific value easy-to-ignite coke-like biomass fuel briquettes can be manufactured from rice, wheat, rape and corn straws mixed with cattle manure, sawdust and other wastes and biomass material. Different processes have been suggested for the treatment of the biomass mixture.
In one process, the biomass mixture is pulverised, fermented at 30 ºC and preheated at 200-250 ºC for 20 min to remove part of the volatile components and increase viscosity. The volatile components are collected and treated to generate fuel gas and tar, and the preheated biomass raw materials are compressed to obtain biomass fuel briquette. The obtained fuel briquette has a high density, long burning time and low ash content [25]. In another process, the biomass mixture, after pulverization and extrusion, is oven dried and carbonised at 600-800ºC for 10-11 h [26]. In a third process, the straw is pulverised, dried at 150 ºC, mixed with other raw materials, press formed at 200 ºC, and finally carbonised at 300-350 ºC [27]. In experiments conducted by Lee et al., the optimum reaction temperature range for the production of bio-oil by fast pyrolysis was found to be 410- 510ºC [28].
Alternatively, straw charcoal (with carbon content 30-35%) can be manufactured from straws and other crop and wood wastes by incomplete gasification. The straw-derived charcoal is rich in nutrients for crops and can be used as adsorbent for contaminated soil remediation [29]. Activated charcoal (typically obtained from rice straw) may also be gasified in an enclosed chamber with a limited amount of oxidant for generating fuel gas composed mainly of carbon monoxide and hydrocarbons [30]. Experimental results show that the gasification reactivity of rice straw char increases with decreasing pyrolysis temperature and increasing heating rate [31].
Straw Combustion
Straw combustion is divided into two stages. One is the emission and combustion of volatiles and the second is the combustion of fixed carbon. The existence of catalysts in the first step enhances the emission of volatiles from the straw. The action of catalysts in the second step of straw combustion may be as a carrier of oxygen to the fixed carbon [32].
In one study, the effects of different catalysts on the ignition and combustion of rice and wheat straw in air- and oxygenenriched atmospheres were investigated by thermogravimetric analysis [32].
Conventional fossil fuel combustion equipment and boilers may be used for the combustion of solid straw fuels with few modifications [17]. Straw fuels however, especially wheatstraw, have in general high contents of alkali metals, chlorine and moisture, which make these fuels difficult to burn in conventional fossil fuel combustion equipment. In order to limit the combustion problems resulting from the high alkali metals and chlorine contents, the straw may be used on small speciallydesigned grate-fired boilers that use relatively low superheater temperatures (440–520 C). However, the grate-fired boilers are relatively expensive and have a lower power efficiency compared to pulverised coal-fired boilers [33], and special boilers may have to be designed for the combustion of straw fuels in certain cases [34,35]. In one design, a natural circulation, bottom supported bi-drum configuration was chosen to ensure compact design [36].
Of the alkali metals in straw, potassium is the principal constituent due to its role as a macronutrient in plant nutrition. The high contents of potassium and chlorine are very undesirable in power plant fuels because they result in increased problems with corrosion [37]. During rice straw pyrolysis, more than 90% of alkali in the straw is water-soluble and contribute to the alkali release [38].
In grate firing, higher ash content adversely affects the combustion stability [39]. The use of straw fuels as a boiler fuel may also be limited because of slag formation and the rapid formation of fouling ash deposits on boiler heat transfer surfaces, which result in high boiler operating costs [40-42]. The limited furnace volume and high flue-gas exit temperatures of most biomass boilers promote slag or deposits from straw fuels. Slagging and deposition may reduce heat transfer and power plant efficiency and necessitate the design of more tolerant heat exchange equipment. It was suggested by Yomogida et al. [43] that decreasing the furnace exit gas temperature so that the ash solidifies prior to the first crossflow heat exchanger may reduce fouling severity and the severity and tenacity of the deposits [43].
In order to reduce the ash content, and chlorine and nitrogen content, and increase the calorific value of rice straw, cut rice straw is added to different acid solutions and heated in a reactor filled with activated carbon to 25-100 ºC for 1-24 h, filtered and dried. The acids that can be used include hydrofluoric acid, acetic acid or a mixed solution of acetic and anhydrous citric acids. The treated rice straw can be mixed with an adhesive selected from starch, paraffin wax, molasses, vegetable oil, vegetable fatty acid and lignin sulphate, extruded for 3-30 min to form a solid fuel of calorific value greater than 4,300 kcal/kg, an apparent density larger than 500 kg/m3, ash content less than 0.7 and chlorine and nitrogen contents less than 0.05% and 0.03% respectively [44].
To increase the combustion efficiency of straw briquettes in power plants and industrial boilers, the addition of antislagging additives composed of magnesium carbonate, aluminium oxide and calcium carbonate has been suggested which would inhibit alkali volatilization and solve slagging problems that may occurduring combustion [34,45]. It may also be possible to inhibit agglomeration of straws on burning or gasification by using a swirling-flow air distribution apparatus for forming swirling flow at the bottom of the fluidised bed [46].
Extensive ash slagging and undesirable fireside fouling normally associated with straw fuels can be overcome by fuel leaching [47]. Preliminary research shows that key inorganic elements that are responsible for ash fouling can be easily removed from the fuel by leaching with water [40]. Results of elemental composition and ash fusibility show that simple leaching of potassium, chlorine and other inorganic constituents from straw with water can result in significant improvements in its ignition and combustion characteristics consistent with the loss of alkali and other elements from the fuel [48]. Pilotscale combustion and full-scale boiler experiments reveal a remarkable change in the fouling tendency of leached rice straw, which would make it technically suitable as boiler fuel under normal operating conditions [47,49,50]. Pilot combustion tests showed lower rates of fouling when burning leached rice straw compared to unleached fresh material.
Furthermore, extensive agglomeration may also be avoided as a result of leaching. Bench-scale combustion in an atmospheric fluidised bed combustor confirms that the extensive bed agglomeration that occurs with combustion of untreated rice straw does not take place with leached rice straw at combustion temperatures up to 900ºC [48,49]. Pyrolysis rates obtained by dynamic thermogravimetric analysis were found to be slower for leached materials, probably as a result of a reduction in catalytic effects due to alkali metal salts.
Experimental evidence indicates that leaching of the alkali elements leads to the increase of the ash fusion temperature and the reduction of the amount of volatile ash [51]. Results obtained by Dayton et al. clearly indicate that leaching biomass effectively reduces or eliminates the release of alkali metal vapours during combustion. Leached and unleached samples of rice straw, wheat straw, switchgrass, wood fuel, sugarcane bagasse and banagrass (Pennisetum purpureum) were tested in bench-scale combustion studies using an alumina-tube flow reactor housed in a variable temperature furnace and coupled to a molecular beam mass spectrometer system. Total relative amounts of HCl(g), SO2(g), NaCl(g), KCl(g), and other inorganic constituents were compared for leached and unleached samples [52].
In addition, blending leached rice straw into a wood-based fuel blend does not increase but rather leads to a decline in ash deposit formation rate. The mechanism for a decline in deposition rate is not fully understood but may be related to the increased loading rate of ash in the furnace and possibly higher erosion rates of deposits that do form [49,53].
When naturally leached rice straw blended with wood and other fuels was fired in three different types of existing boilers: a stoker-fired travelling grate, a circulating fluidised bed, and a suspension fired unit, no adverse effects due to slagging orfouling were observed and no bed agglomeration occurred in the fluidised bed unit, but straw firing increased the ammonia injection rate that controlled NOx emissions which increased due to the higher nitrogen content of the straw [47,50].
Leaching occurs naturally in the field (field or natural leaching) when the straw fuel is exposed to rainfall, or it can be conducted under controlled conditions in industrial applications or at the power plant site (on-site leaching) [40,54].
Field leaching of potassium, chlorine and total ash has been reported for rice and barley straws [93,190]. Observations on three rice farms in California indicated that potassium, chlorine and total ash are leached from rice straw by rainfall regardless of rice variety, grain harvest method, straw arrangement, or stubble length. Leaching of sulphur by natural precipitation, however, was not clearly established (Table 1) [55].
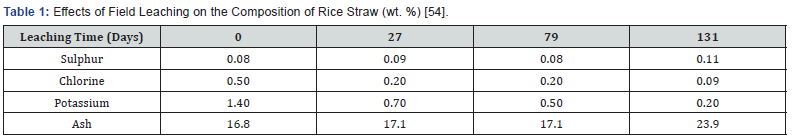
Field leaching is economically attractive because it requires no material handling or waste water disposal or treatment. However, it is only an option in areas with sufficient precipitation. Collection of straw can then be undertaken only after the last rain is received and after allowing for a period of drying time depending on the amount of cumulative rainfall received during the winter months. The timing of straw collection in spring will have to take account also of the planning of field preparation activities for the next crop.
A pre-treatment process to remove potassium and chlorine from straws may be based on pyrolysis followed by char leaching [35,37]. The straw is pyrolysed at moderate temperatures at which the potassium is retained in the char. Potassium and residual chlorine are then extracted from the residual char by water. To evaluate this pretreatment process, wheat straw chars were experimentally investigated in the laboratory. The laboratory experiments showed that three fractions ofpotassium in the straw reacted differently: 35–58% of the char potassium was dissolved very fast, followed by a secondary slow potassium release that was strongly influenced by particle sizes, water temperature, char type and water KCl content. The residual 5–10% of the char potassium remains in the char and could not be removed with pure water.
The combustion of straws may introduce a number of pollutants into the atmosphere. Table 2 shows some of the pollutants expected during the combustion of straws and other agricultural residues [55]. The unburned pollutants include CO, HC, tar, PAH (polychlorinated aromatic hydrocarbons), CxHy and char particles. These pollutants are usually due to poor combustion which is a result of low combustion temperature, insufficient mixing of fuel with combustion air and too short residence time of the combustible gases in the combustion zone [56].

Volatile organic compounds, including polycyclic aromatic hydrocarbons, may also be emitted by the combustion of straw. In a study by Keshtkar and Ashbaugh, particle size distribution in the smoke emission of rice straw was examined. Total polycyclic aromatic hydrocarbons emissions from rice straw burns were 18.6 mg per kg of fuel. The less volatile 5- and 6-ring compounds were predominately on smaller particles where they condensed in the early stages of combustion while the more volatile 3- and 4-ring compounds formed on larger particles as the smoke cooled [57]. Christian et al. found in comprehensive laboratory measurements of biomass-burning emissions thatAcetol (hydroxyacetone) was a major, previously unobserved emission from burning rice straw (21-34 g/kg) [58].
Experimental evidence indicates that nearly all reactive nitrogen (forming NO) is released from the straw fuel after the introduction of the fuel into the hot fluidised bed [59].
Particulate matter emitted from wind tunnel simulations of rice, wheat and barley straws burning was collected and analyzed for major elements and water-soluble species. Primary constituents of the particulate matter were C, K, Cl and S. Carbon accounted for roughly 50% of the straw fuel PM. Particulatematter from rice straw in the size range <10 μm aerodynamic diameter (PM10) had the highest concentration of K (24%) and Cl (17%) and barley straw PM10 contained the highest S (4%). K:C ratios of 0.20 and 7.5 were computed for particulate matter in the size class <10 μm and <2.5 μm respectively. Analysis of water soluble species indicated that ionic forms of K, Cl, and S made up the majority of these elements from all straw fuels [60].
Significant steroid modulating activities were found in the combustion soot of rice straw. GC/MS results show that polycyclic aromatic compounds and their derivatives, substituted phenolic compounds and their derivatives, aromatic carbonyl compounds and higher molecular weight ketones may be the main steroid disrupters in the soot [61].
Fluidised bed combustion is ideally suited to burn solid straw fuels since uniform mixing ensures efficient combustion, even at temperatures as low as 600 to 650ºC.
The principal problem in fluidised bed combustion of straw fuel appears to be the large increase in inorganic material that can severely affect the bed operation [62]. Ash content, which may cause extensive slagging, deposition and bed agglomeration during fluidised bed combustion, remains one of the major problems associated with the combustion of straws and other herbaceous materials. Large-sized ash particles may act as the necks in the formation of agglomerates and small-sized ash particles may contribute to bed defluidization [63].
Examination of straw particles indicate that a narrow continuous surface zone borders virtually all straw particles where high concentrations of alkali metals such as potassium and sodium and other elements are found. It was shown, in a study in which a scanning electron microscope coupled with energy-dispersive spectroscopy is used, that such alkali metals melt as a result of high combustion temperatures and coat the surface of ash particles making them sticky with a tendency to adhere to the surface of bed particles, thus contributing to bed defluidization [63,64].
It was found by examining bed materials from fluidised bed combustion of wood and rice straw fuel blends that melting of locally accumulated dust or aerosol mixture of ash particles and mullite led to the accumulation of liquid droplets, preferentially on surfaces and contact areas between bed particles sheltered from turbulence and mechanical interaction, and these formed thin discontinuous films of adhesive cement by filling of irregularities on individual and partially agglomerated bed particle surfaces. As a result of this film formation the bed particles were locally enlarged and this led ultimately to bed agglomeration. Large straw ash particles appear to have mostly been passively incorporated into the adhesive melt without melting or reaction [64].
Straw melts are often highly polymerised. The polymerised nature of rice and wheat straw melts and their low melting pointsrender these straws less attractive as fuels for many biomassfuelled power plants. However, in the highly polymerised rice and wheat straw slag the alkali metals occur as networkmodifying cations and are strongly retained in the polymerised network. Rice straw ash melt is dominated by a relatively open polymerised network that will easily accommodate the large Na+ and K+ ions. It was tentatively established by Thy et al. that about 70% of potassium in rice ash may be retained in the slag. It was further concluded by this study that the use of straw fuels, compared to wood fuels, may significantly reduce the alkali loss from high temperature molten slag [65].
Conclusion
It is manifest that straws, whether used as a solid fuel or after conversion to a charcoal fuel, are a valuable and renewable source of energy that may not be neglected in this age which is characterized by energy scarcity and rising fossil fuel prices.
References
- L Hongtao, RP Karen, X Youmin, G Ju’e (2010) Comprehensive evaluation of effects of straw-based electricity generation: A Chinese case. Energy Policy 38: 6153-6160.
- T G Bridgeman, J M Jones, I Shield, P T Williams (2008) Torrefaction of Reed Canary Grass. Wheat Straw and Willow.
- P Adapa, L Tabil, G Schoenau (2009) Compaction characteristics of barley, canola, oat and wheat straw. Biosystems engineering 104: 335-344.
- V S P Bitra, A R Womac, N Chevanan, P I Miu, C Igathinathane, (2009) Direct mechanical energy measured of hammer mill comminution of switchgrass, wheat straw, and corn stover and analysis of their particle size distributions. Powder Technol 193: 32-45.
- J Soucek, I Hanzlikova, P Hulta (2003) A fine disintegration of plants suitable for composite biofuels production. Res Agr Eng 49(1): 7-11.
- Z Drzymala (1993) Industrial briquetting - fundamentals and Methods. Studies in mechanical engineering, 13, Warszawa, PWN-Polish Scientific Publishers.
- P Lopo P (2002) The right grinding solution for you: roll, horizontal or Vertical, Feed Management 53(3): 203-206.
- R L Scholten, R R McEllhiney (1985) The effects of prebreaking in hammer mill particle size reduction. American Society of Agricultural Engineers, St. Joseph, MI, pp. 85-3542.
- F Talebnia, D Karakashev, I Angelidaki (2010) Production of bioethanol from wheat straw: An overview on pretreatment, hydrolysis and fermentation. Bioresource Technology 101: 4744-4753.
- P Adapa, L Tabil, G Schoenau (2010) Grinding performance and physical properties of non-treated and steam exploded barley, canola, oat and wheat straw. Biomass and Bioenergy.
- C K W Ndiema, P N Manga, C R Ruttoh (2002) Influence of die pressure on relaxation characteristics of briquetted biomass. Energy Conversion and Management 43(16): 2157-2161.
- C K W Ndiema, P N Manga, C R Ruttoh (2002) Densification characteristics of rice straw briquettes. Journal of the Institute of Energy 75(502): 11-13.
- M D Shaw M D (2008) Feedstock and process variables influencing biomass densification. MSc Dissertation, University of Saskatchewan, Saskatoon, SK, Canada.
- MJO ’Dogherty, J A Wheeler (1984) Compression of straw to high densities in closed cylindrical dies. Journal of Agricultural Engineering Research 29(1): 61-72.
- M O Faborode, J R O’Callaghan (1987) Optimizing the compression/briquetting of fibrous agricultural materials. Journal of Agricultural Engineering Research 38: 245-262.
- H Sato (2010) Rice straw pellets and livestock feed containing the pellets and apple pomace. Japan Patent No. JP 2010130906.
- C Chou, S Lin, W Lu (2009) Preparation and characterization of solid biomass fuel made from rice straw and rice bran. Fuel Processing Technology 90(7-8): 980-987.
- C Chou, S Lin, C Peng, W Lu (2009) The optimum conditions for preparing solid fuel briquette of rice straw by a piston-mold process using the Taguchi method. Fuel Processing Technology 90 (7-8): 1041-1046.
- S Otake, A Taya, N Ishii, E Ochi, S Sugita (1998) Solid fuel production and energy conversion for pollution control. Japan Patent No. JP 10001685.
- Zhongxi Kang (2010) Method for manufacturing compressed fuel rods. China Patent No. CN 101676370.
- B Zhang, B Li, F Fan, S Yang (2007) Method for producing rod-shaped compact straw fuel. China Patent No. CN 101139538.
- Wenjian Chen (2010) Environmentally-friendly solid fuel and its preparation from crop waste. China Patent No. CN 101798541.
- Wen Chien Chen (2010) Method of transforming a crop waste into a solid fuel and a solid fuel made by the method. USA Patent No. US 20100199550.
- A K Jain, V R Sharma, B S Pathak (1986) A note on the changes in the bulk density of crop residues due to the application of uniaxial pressure. Agricultural Wastes 16(2): 89-95.
- Wenlin Zheng (2010) Method for manufacturing coke-like biomass fuel briquette. China Patent No. CN 101629115.
- Xuping Shen (2010) Method for manufacturing solid fuel from cattle manure, straw, and sawdust. China Patent No. CN 101629114.
- L Zhang, G Wang (2004) Method for preparation of solid fuel briquets from straw. China Patent No. CN 1699525.
- K Lee, B Kang, Y Park, J Kim (2005) Influence of Reaction Temperature, Pretreatment, and a Char Removal System on the Production of Bio-oil from Rice Straw by Fast Pyrolysis, Using a Fluidized Bed. Energy & Fuels 19(5): 2179-2184.
- Q Zhang, J Zhou, S Yi, B Song (2008) Simultaneous manufacture of charcoal, fuel gas, and pyroligneous liquors by gasification of biomass and wood waste. China Patent No. CN 101186833.
- Hanyuan Tang (2007) Furnace for generating coal gas from charcoal, China Patent No. CN 101037619.
- Y Huang, X Yin, C Wu, C Wang, J Xie, Z (2009) Study on CO2 gasification reactivity of rice straw chars. Ranliao Huaxue Xuebao 37(3): 289-295.
- Y Zhaosheng, M Xiaoqian, L Ao (2009) Thermogravimetric analysis of rice and wheat straw catalytic combustion in air- and oxygen-enriched atmospheres, Energy Conversion and Management 50: 561-566.
- S H Enestam, M K Fabritius, S K Hulkkonen, J T Roppanen (2003) Control of ash related operational problems in BFB combustion of biofuels and waste. Proceedings of the International Conference on Fluidized Bed Combustion pp. 541-546.
- T R Miles, T R Miles Jr, L L Baxter, R W Bryers, B M Jenkins, et al. (1996) Boiler deposits from firing biomass fuels. Biomass and Bioenergy 10(2-3): 125-138.
- Ayhan Demirbas (2003) Demineralization of Agricultural Residues by Water Leaching. Energy Sources 25(7): 679-687.
- P A Jensen, B Sander, K Dam-Johansen (2001) Removal of K and Cl by leaching of straw char. Biomass and Bioenergy 20: 447-457.
- S Rajaram, K T U Malliah (1990) Development of fluidized bed boilers for biomass and other waste fuels. Proceedings of the Intersociety Energy Conversion Engineering Conference 5: 113-118.
- C Yu, Z Luo, M Fang, K Cen (2004) The experimental research of alkali transformation during rice straw pyrolysis. World Renewable Energy Congress VIII: Linking the World with Renewable Energy, Denver, CO, United States, 3: 11-15.
- X Lu, Y Yu, P Lin, Y Luo, J Duan (2009) Grate firing characteristics of typical biomass fuels. Dongli Gongcheng 29(3): 282-286.
- R R Bakker, B M Jenkins (1996) Feasibility of fuel leaching to reduce ash fouling in biomass combustion systems. Biomass for Energy and the Environment, Proceedings of the European Bioenergy Conference, Copenhagen, 2: 980-985.
- B Sander (1997) Properties of Danish biofuels and the requirements for power production. Biomass and Bioenergy 12(30): 177-183.
- F Frandsen, H P Nielsen, P A Jensen, L A Hansen, H Livbjerg H (1999) Deposit and corrosion in straw and coal-straw co-fired utility boilers. In: Gupta (Edt.), Impact of mineral impurities in solid fuel combustion, New York, Kulwer Academic/Plenum Publishers, USA, pp. 272-283.
- D E Yomogida, B M Jenkins, B R Hartsough, V M Tiangco (1999) Model performance of a biomass-fueled power station with variable furnace exit gas temperature for fouling control. Proceedings of the Biomass Conference of the Americas, Oakland, Calif., 2: 1409-1416.
- D G Oh (2010) Method for manufacturing solid fuel from rice straw. Korea Patent No. KR 2010022948.
- Y Tian, L Zhao, H Meng, Z Yao, L Sun, (2010) Antislagging additive for biomass solid fuel briquet and its preparation method, China Patent No. CN 101775324.
- S Yuan, Y Yuan, C Peng (2009) Swirling-flow air distribution apparatus for biomass fluidized bed. China Patent No. CN 101571288.
- R R Bakker, B M Jenkins, R B Williams, W Carlson, J Duffy (1997) Boiler performance and furnace deposition during a full scale test with leached biomass. Making a Business from Biomass in Energy, Environment, Chemicals, Fibers and Materials, Proceedings of the Biomass Conference of the Americas, Montreal, 1: 497-510.
- R R Bakker, B M Jenkins, R B Williams, D Pfaff (1999) Fluidized bed combustion of leached rice straw. Proceedings of the Biomass Conference of the Americas, Oakland, Calif., 2: 1425-1431.
- R R Bakker, B M Jenkins, R B Williams (2002) Fluidized Bed Combustion of Leached Rice Straw. Energy & Fuels 16(2): 356-365.
- B M Jenkins, R B Williams, R R Bakker, S Blunk, D E Yomogida (1999) Combustion of leached rice straw for power generation. Proceedings of the Biomass Conference of the Americas, Oakland, Calif., 2: 1357-1363.
- B M Jenkins, R R Bakker, L L Baxter, J H Gilmer, J B Wei (1997) Combustion characteristics of leached biomass. Developments in Thermochemical Biomass Conversion 2: 1316-1330.
- D C Dayton, B M Jenkins, S Q Turn, R R Bakker, R B Williams, (1999) Release of Inorganic Constituents from Leached Biomass during Thermal Conversion. Energy & Fuels 13(4): 860-870.
- A Ghosh, B C Bhattacharyya (1999) Biomethanation of white rotted and brown rotted rice straw. Bioprocess Engineering 20(4): 297-302.
- R R Bakker, B M Jenkins (2003) Feasibility of collecting naturally leached rice straw for thermal conversion. Biomass and Bioenergy 25: 597-614.
- JE Hustad, T Nussbaumer (1997) Overview of biomass combustion, developments in thermochemical biomass conversion, In: DGB Boocock, AV Bridgwater (Edt.), Developments in thermochemical biomass conversion. Chapman and Hall, London, pp. 1229-1246.
- S Q Turn, B M Jenkins, J C Chow, L C Pritchett, D Campbell (1997) Elemental characterization of particulate matter emitted from biomass burning: wind tunnel derived source profiles for herbaceous and wood fuels. Journal of Geophysical Research, [Atmospheres] 102 (D3): 3683-3699.
- J Werther, M Saenger, EU Hartge, T Ogada, Z Siagi (2000) Combustion of agricultural residues, Prog Energy Combust Sci 26: 1-27.
- H Keshtkar, L L Ashbaugh (2007) Size distribution of polycyclic aromatic hydrocarbon particulate emission factors from agricultural burning. Atmospheric Environment 41(13): 2729-2739.
- T J Christian, B Kleiss, R J Yokelson, R Holzinger, P J Crutzen (2003) Comprehensive laboratory measurements of biomass-burning emissions: 1. Emissions from Indonesian, African, and other fuels. Journal of Geophysical Research 108 (D23): ACH 3/1-ACH 3/13.
- J Konttinen, M Hupa, S Kallio, F Winter, J Samuelsson (2005) NO formation tendency characterization for biomass fuels. Proceedings of the International Conference on Fluidized Bed Combustion, p. 225-235.
- W Z Wu, J X Wang, G F Zhao, L You (2002) The emission soot of biomass fuels combustion as a source of endocrine disrupters. Journal of Environmental Science and Health, Part A: Toxic/Hazardous Substances & Environmental Engineering 37(4): 579-600.
- P Thy, B M Jenkins, R B Williams, C E Lesher (2004) Slag formation and potassium volatilization from rice straw blended wood fuel. Preprints of Symposia - American Chemical Society, Division of Fuel Chemistry 49(1): 89-92.
- H Liu, Y Feng, S Wu, D Liu (2009) The role of ash particles in the bed agglomeration during the fluidized bed combustion of rice straw. Bioresource Technology 100(24): 6505-6513.
- P Thy, B M Jenkins, R B Williams, C E Lesher, R R Bakker (2010) Bed agglomeration in fluidized combustor fueled by wood and rice straw blends. Fuel Processing Technology 91(11): 1464-1485.
- P Thy, C E Lesher, B M Jenkins (2000) Experimental determination of high-temperature elemental losses from biomass slag. Fuel 79(6): 693-700.






























