Desulphurisation of Petroleum Coke by Solvent Extraction
Hassan Al-Haj Ibrahim*
Arab University for Science and technology Hamah, Syria
Submission: December 19, 2018; Published: January 23, 2019
*Corresponding author: Hassan Al-Haj Ibrahim, Arab University for Science and technology Hamah, Syria.
How to cite this article: Hassan Al-Haj I. Desulphurisation of Petroleum Coke by Solvent Extraction. Recent Adv Petrochem Sci. 2018; 6(2): 555686 DOI: 10.19080/RAPSCI.2019.06.555686
Abstract
Solvent extraction is the simplest approach to the desulphurization of petroleum coke. The selectivity of solvent extraction can be enhanced by coke depolymerisation using different methods such as mild hydrogenation, microwave heating, oxidation, pre-pyrolysis and the use of catalysts. In particular, pre-oxidation of petroleum coke at a moderate temperature (600K) renders the coke more amenable to the solvent extraction treatment. Extractions with oxidized samples of Syrian coke were made in a Soxhlet extractor for two hours using o-chlorophenol and pyridine as solvents. A maximum desulphurization of 76% was achieved
Keywords: Petroleum coke Desulphurisation Solvent extraction Pre-Oxidation
Introduction
Environmental considerations and the demand for high-quality low-sulphur petroleum coke call for an effective and commercial process for the desulphurization of coke. Solvent extraction would offer the simplest approach to desulphurisation if it were possible to selectively dissolve the organic sulphurcompounds present in the coke. As materials of similar chemical structure are more likely to be mutually soluble in one another, aromatic and similar compounds might be used as solvents for the extractive desulphurization of petroleum coke where the sulphur is mostly organic and thiophenes make up most of the sulphur present in the coke.
(1) Heating just below the boiling point
(2) B, B, dichloroethylether.
(3) At room temperature
Experience with coal indicates that weak organic acids such as phenols and nitrobenzene are more effective than other organic solvents [1]. These could be used to dissolve sulphides and disulphides and possibly some thiophenes as well. When coal was treated by solvent extraction about 80% of the organic sulphur was removed. It was suggested that successive sequential extraction of coal might significantly improve the desulphurization efficiency of solvent extraction [2]. The effective removal of the thiophenic sulphur in petcoke is not, however, likely with simple solvent extraction.
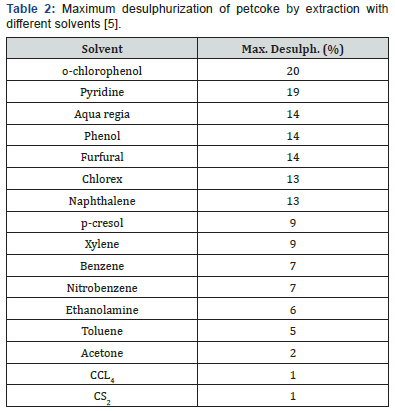
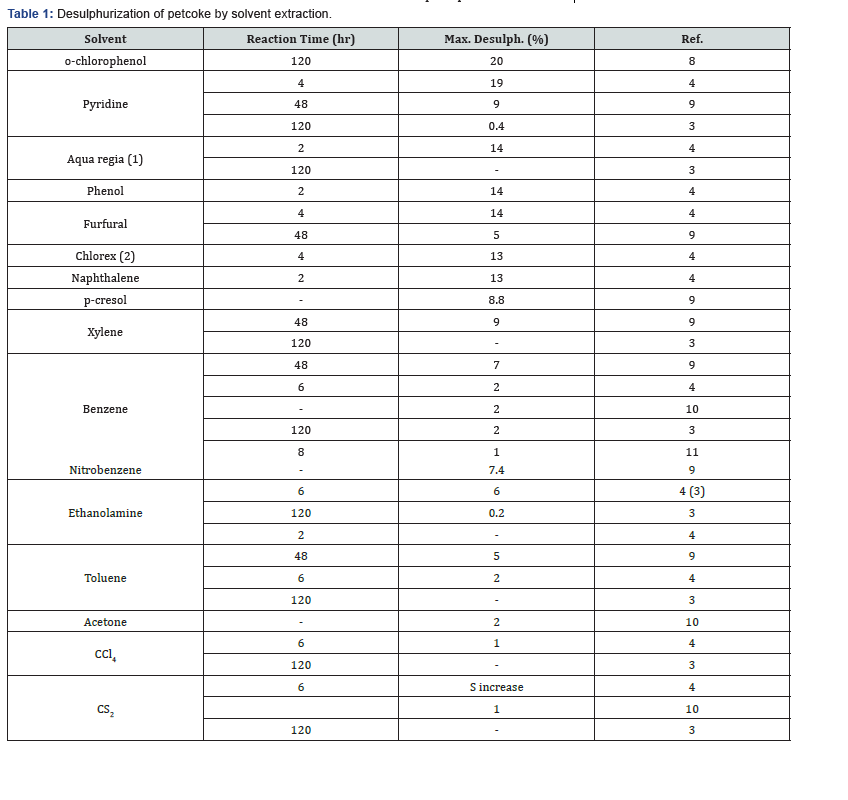
Extractions with coke using a large variety of solvents were made in a Soxhlet extractor. No sulphur removal was reported when petroleum ether [3], Dioxane [3] or hydrochloric acid [3,4] were used. Extractions made with other solvents led to somesulphur removal, but in no case was more than 20% sulphur removed when extractions were made with different single solvents [5] (Tables 1). Treatment of Syrian petcoke, produced by the delayed coking unit at the Homs Oil Refinery, by solvent extraction using different solvents such as benzene and sodium carbonate proved to be equally ineffective, with a maximum sulphur removal not exceeding 3% [6]. As can be seen from Table 2 the most efficient solvents used were o-chlorophenol and pyridine with a maximum desulphurization of 20% and 19% respectively. However, when two solvents, ferric chloride and benzene, were used consecutively in the treatment of Egyptian petroleum coke, more than 35% sulphur removal was obtained [7,8].
Ortho-chlorophenol or 2-chlorophenol C6H5ClO is a liquid at room temperature with a boiling point of 449K. It is an unpleasant water-soluble combustible liquid that is strong irritant to tissue and toxic by skin absorption, inhalation or ingestion and strong irritant to tissue (Figure 1).
Phillips & Chao [9] used o-chlorophenol as the extraction solvent. The maximum desulphurisation achieved was 20%. Their results show that the smaller the particle size of coke, the greater the amount of sulphur removed. At 430K the optimum reaction time was found to be two hours. Longer time will not further reduce the sulphur content by any significant amount,for all the soluble sulphur would have been removed by the end of the second hour.
Pyridine is a heterocyclic organic compound C5H5N. It is a colourless, flammable liquid soluble in water with a boiling point of 388K. It is a widely used polar but aprotic solvent and it has been used in the refining industry for the treatment of lubricating oils. Sabott [4] tried to use it for refluxing coke for a period of four hours. The extracted coke was filtered and washed ten times with ether. The pyridine coke mixture formed a slime which was difficult to filter and wash. His and other results are given in Table 1.
For the extraction with phenol and naphthalene, the molten solvent was mixed with coke for two hours and the mixture extracted with ether or ethyl alcohol respectively, filtered and washed with more ether or ethyl alcohol. Extractions with furfural (furfural aldehyde) formed a mixture of brownishcoloured slime that was extremely difficult to filter and wash.
The Girbotol process makes use of ethanolamine to remove H2S from gases. Treating coke with ethanolamine removed a maximum of 6% sulphur (Table 1). An extraction using toluene gave an extract with a slight yellow colour which was apparently due to some oil which had been removed from the coke. It was evident that the oil adsorbed on the coke did not contain appreciable amounts of sulphur [4].
Carbon disulphide (CS2) is a solvent for elemental sulphur. Sabott made an extraction with CS2 as a solvent for six hours and the residue washed ten times with ethyl alcohol. Upon analysis, the coke showed a sulphur content increase (from 1.27 to 1.39%). This indicates that CS2 was probably not washed completely from the coke. The CS2 extract had a faint yellowish colour that was probably due to the decomposition of the solvent [4,3]. Schafer reported about 1% sulphur removal on extracting with CS2 [10]. Mahmoud et al. [3] obtained no sulphur removal with CS2 after an extraction that was continued for five days.
Coke Depolymerisation
The above results indicate clearly that simple solvent extraction is not an effective method of desulphurisation. The ineffectiveness of solvent extraction as a desulphurization technique is directly related to the nature of sulphur compounds in coke. While free or elemental sulphur, which could be removed by solvent extraction, may be found occasionally in petroleum coke [1,11-13], most of the sulphur in coke exists as organic sulphur bound to the carbon matrix of the coke [1,14]. Effective desulphurization of petroleum coke involves the rupture of the thiophenes, which make up most of the sulphur present in the coke. The selectivity of solvent extraction can therefore be enhanced only if the coke macromolecule could be cleaved. Coke depolymerisation can be effected by different methods including increasing the extraction temperature, mild hydrogenation, microwave heating [15], oxidation, pre-pyrolysis and the use of catalysts. Although the effectiveness of depolymerisationtechniques has not been fully investigated, tentative results obtained by different workers indicate that depolymerisation does in fact lead to improved selectivity of the solvent extraction process. When some depolymerisation took place as a result of increasing the extraction temperature the effectiveness of desulphurization by solvent extraction was significantly improved as reported by Phillips & Chao [8]. Their results were as follows:
These results can be explained by the fact that some coke depolymerization must have taken place as a result of the extraction temperature increase. Mild hydrogenation at temperatures below 700K and in the absence of a solvent strongly increased the benzene and pyridine yields for several coals [1]. Oxidation was variously reported to increase, decrease or not affect the solubility of coal [1].
Pre-Oxidation of Petroleum Coke
Pre-oxidation of petroleum coke at a moderate temperature (600K) renders the coke more amenable to the solvent extraction treatment. The beneficial effect of pre-oxidation is probably due to the oxidation of the sulphur bonded into the coke matrix, which would be expected to weaken the C-S bonds without at the same time destroying the hydrocarbon matrix. Partial oxidation of the hydrocarbon matrix would, however, be expected and this favours the removal of sulphur, but the key to a desulphurization process based on oxidation would be the minimization of the oxidation of the remainder of the hydrocarbon matrix, which is not always easy [1]. Sulphur removal is probably related to the resulting weight loss of the coke and its improved porosity.
The oxidation potential of organic sulphur compounds would lie roughly in the same region as amines and alcohols. These compounds are vastly more susceptible to oxidation than ethers and benzene derivatives [1]. Organic thiols may be oxidized to disulphides, which may in turn be oxidized to give finally sulphonic acids.

Thus, at every former sulphur position, which has been oxidized and hydrolyzed, an alcohol functional group would remain.
The polarity difference between the sulphur compounds and hydrocarbons found in coke may also be increased as a result of pre-oxidation. Such an approach may be based on the polarity difference between the thiophenic compounds and the hydrocarbons. Because thiophenic compounds comprise many different compounds, they represent a range of solvent polarities, in some cases quite similar to some of the hydrocarbons found in coke. The critical polarity difference between individual hydrocarbon and thiophenic compounds is therefore variable, and in some cases may be insufficient to allow a functional separation by solvent extraction. Polarity difference can, however, be increased by oxidizing the thiophenic sulphur to the corresponding more polar mono- and dioxides, the alkylatedaryl thiophene sulphoxide or sulphone. This in turn facilitates the separation and makes possible the extraction of sulphur after a preliminary step of oxidation [16].
During pre-oxidation, water that enters the coke during the hydraulic coke cut is removed at about 400-500K. The volatile matter, for the most part, distils off also during pre-oxidation exposing thereby the pores and typical lamellar structure of the treated coke. A certain concentration of CO due to the partial combustion of coke and volatiles soon developed.
Coke pre-oxidation at lower temperatures is to be preferred because of its expected effects on maximizing the rate of desulphurization and reducing coke consumption at the same time. The optimum temperature for pre-oxidation was found to be in the range 570-620K [17-21]. Pre-oxidizing the coke at a lower temperature (550K) did not have a beneficial effect [9], while the reaction was found to be too slow at 630K [19].
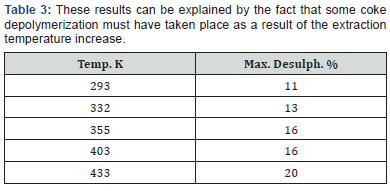
In agreement with work published by earlier workers [10,17,18,22,23] pre-oxidation of Syrian coke in the low temperature range used lead to some sulphur removal (7%) [20]. In comparison, when coal was heated to 450 ˚C in air, a maximum of 20% of the organic sulphur was removed [1]. The amount of sulphur removal is related to the weight loss regardless of temperature, but the rate of sulphur reduction is greater at lower temperatures (Table 3), a fact that indicates, according to some, a more selective SO2 formation at lower temperatures. This preferential oxidation of sulphur as compared with carbon, which takes place at lower temperatures, allows more effective desulphurization with a lower consumption of coke. Lower temperature pre-oxidation promotes higher levels of hydrodesulphurization as well.
As to be expected, pre-oxidation would be accompanied by an increase in oxygen and nitrogen and a decrease in carbon content. The coke tends to oxidize or “cure” by adsorbing oxygen at temperatures below 200 ºC. Addition of oxygen in general tends to promote cross-linking between the graphitoid structures in coke irreversibly decreasing its anisotropy [24]. The presence of oxygen atoms may also strengthen the coke structure sufficiently to make the coke less susceptible to sintering [9]. Carbon-nitrogen complexes are not likely to be formed at this low temperature range [24].
Coke porosity increases on oxidation due to the creation of micro and macroporosity. The increased microporosity mayrepresent the creation of shrinkage cracks as well as pores formed due the escape of volatile matter and other weight loss. As a result of such increase in the coke porosity, its apparent density (DA) measured by Hg pycnometer decreases. The real or true density (DR10-20) of the coke, measured by He pycnometer, also changes significantly on oxidation. With Syrian coke, the density was found to increase from 1.39g/cm3 to 1.52 [21]. This increase is slightly greater than the corresponding increase found on thermally treating the coke to a similar temperature range [25].
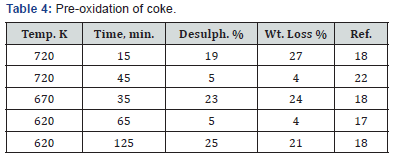
Some increase in the surface area on pre-oxidation is to be expected, particularly at low temperatures (< 700K) (Table 4). This may be due o the opening up of micropores in the coke matrix or the spreading apart of planes of largely polycyclic carbon causing the exposure of a larger surface area [9,18]. Pre-oxidation induces also many large cracks in almost all grain cokes at temperatures less than 700K [18]. When lump coke was pre-oxidized, a large number of small cracks were induced in the coke surface, but these were removed on polishing which indicates that they were very shallow in comparison with grain size [17]. In the work done on Syrian coke, even though there is no single experimental method that is a standard method or an approved method to be used for the determination of surface area, the BET method based on the adsorption of nitrogen at 77K was chosen because it is the preferred method that is normally assumed and quoted. The BET surface area for the treated Syrian coke was found to have increased from an average of 0.04 to 52m2/g [21].
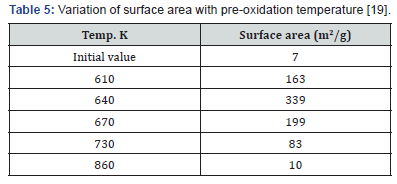
Weight loss can indeed be substantial if pre-oxidation is prolonged. Mason reported 38% weight loss after pre-oxidation at 640K for 1020 minutes [15]. The residence time used in the treatment of Syrian coke, 15 minutes, insured a much smaller weight loss, which was on average about 25% [21]. The calorific value was also reduced but only by about 14% [21]. Table 5 gives a summary of the effects of pre-oxidation on the treated coke.
There is evidence to indicate that using steam as an oxidizing agent would significantly increase sulphur removal. Also, the addition of water during the pre-oxidation treatment may accelerate oxidation especially at lower temperatures, probably by accelerating the liberation of oxidized carbon from the coke surface, thus increasing the surface area of the coke. However, using a mixture of air and steam seems to inhibit desulphurization [9,23]. Mochida et al. [18] using ground coke found this role to be small, but when lump coke was used, varying the amount of water slightly changed the desulphurization level significantly [22]..
Zannikos et al. [26] report that a combination of oxidation with solvent extraction is capable of removing up to 90% of the sulphur compounds in petroleum fractions at acceptable yields. Funakoshi and Aida used a simple solvent extraction process to remove sulphur from various hydrocarbon fuels (ranging from gasoline to straight-run bottoms). Using acetone, dimethyl formamide or other solvents, more than 90% sulphur removal could be achieved through six to eight stages of extractions with a solvent to oil ratio of 1. When an oxidation step was applied before the extraction, a higher degree of sulphur removal was obtained [27]. In similar conditions, the sulphur compounds in vacuum gas oil were removed successfully by pre-oxidation and extraction by Shiraishi & Hirai [28].
In order to investigate the effects of pre-oxidation on the efficiency of desulphurization by solvent extraction, extractions with oxidized samples of Syrian coke were made in a Soxhlet extractor for two hours using o-chlorophenol and pyridine as solvents. In the choice of the solvents and reaction time, use was made of the results summarized in Table 1.
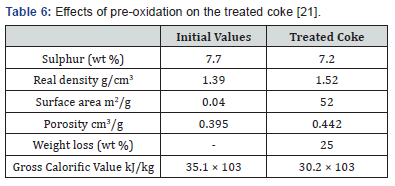
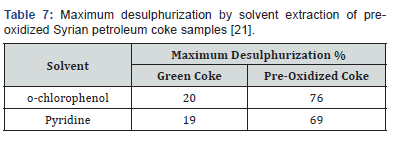
The coke samples were heated in air in an electric furnace at a controlled rate for 15 minutes at a moderate temperature (600K). The desulphurization efficiency was greatly improved when the pre-oxidized Syrian coke samples were later extracted using these two solvents. Results of the extractive desulphurization tests are given in Table 6 where a maximum desulphurization of 76% was achieved [21]. Complete elimination of the thiophenic and other sulphur compounds however was not achieved due in all probability to incomplete oxidation or to ineffective extraction where the choice of the solvent plays a very important role [21].
The fact that pre-oxidation favours the removal of sulphur is most probably related to the resulting weight loss of the cokeand its improved porosity. Treating the coke for longer periods of time would probably improve the desulphurization efficiency even further, but any advantage that is gained thereby would be offset to a certain extent by the resulting greater weight loss of the coke and its reduced calorific value (Table 7)..
Pre-pyrolysis or preheating in an inert atmosphere for several hours at temperatures of 450-670K has been shown to increase the solubility of several coals toward solvents such as pyridine and benzene, with up to a seven-fold increase of solubility in chloroform noted for extraction of a coal preheated at 600-610K for 22 hours [1].
Finally, it has been shown that the organic coal matrix can be decomposed catalytically using different catalysts such as p-toluene sulphonic acid and boron tri-fluoride-phenol. The coals treated were depolymerised to give molecules of an average molecular weight of 300-1100 soluble in the typical coal extraction solvents pyridine and benzene/ethanol [1].
Conclusion
Pre-oxidation of coke at moderate temperatures for a short period of time renders the coke more amenable to treatment by solvent extraction. Extracting pre-oxidized coke samples improved significantly the desulphurization efficiency and a maximum desulphurization of 76% was achieved using o-chlorophenol as solvent. Complete elimination of the thiophenic and other sulphur compounds however was not achieved due in all probability to incomplete oxidation or to ineffective extraction where the choice of the solvent plays a very important role.
References
- Meyers RA (1977) Coal desulphurization, Marcel Dekker, New York, USA.
- Das A, Sharma DK (2001) Organic desulfurization of Assam coal and its sulfur-rich lithotypes by sequential solvent extraction to obtain cleaner fuel, Energy Sources, Part A: Recovery, Utilization, and Environmental Effects 23(8): 687-697.
- Mahmoud BH, Ayad S, Ezz SY (1968) Desulphurization of petroleum coke. Fuel 47(6): 455-462.
- Sabott FK (1952) A study of methods of removing sulfur from petroleum coke, Quarterly of the Colorado school of mines, 47( 3): 1-22.
- Al-Haj-Ibrahim H, Morsi BI (1992) Desulfurization of petroleum coke: a review. Industrial and Engineering Chemistry Research 31(8): 1835-1840.
- Al-Haj-Ibrahim H (1995) Desulphurization of petroleum coke, Proceedings of the First Egyptian Syrian Conference on Chemical Engineering, Suez, Egypt, 2-5 Sept p. 207-212.
- Aly IHM, Magdy YH, Barakat NAM (2003) Desulfurization of Egyptian petroleum coke, Developments in Chemical Engineering and Mineral Processing 11( 3-4): 395-406.
- Phillips CR, Chao KS (1977) Desulphurization of Athabasca petroleum coke by (a) chem. oxidation and (b) solvent extraction. Fuel 56(1): 70-72.
- Parmar BS, Tollefson EL (1977) Desulphurization of oil sands coke, Can J of chem Eng 55(2): 185-191.
- Shafer WC (1952) Removal of sulfur from petroleum coke by pyrolysis. Quarterly of the Colorado school of mines 47(3): 27-37.
- Al-ansary AF, Ezz SY (1973) Desulphurization of petroleum coke by butagas. Fuel 52(1): 66-70.
- George ZM, Schneider LG (1982) Sodium hydroxide-assisted desulphurization of petroleum fluid coke. Fuel 61(12): 1260-1266.
- El-kaddah N, Ezz SY (1973) Thermal desulphurization of ultra high sulphur petroleum coke. Fuel 52(2): 128-129.
- Hussein MK, El-tawil SZ, Rabah MA (1976) Desulphurization of high-sulfur Egyptian petroleum coke. J Inst Fuel, pp. 139-143.
- Park M, Kang T (2002) A study on the desulfurization of petroleum cokes by microwave heating. Clean technology 8(1): 11-17.
- Skov SK, Dolbear GE (2006) Synergistic extractive desulfurization processes. In: C. S. Hsu CS, Robinson PR (eds), Practical advances in petroleum processing, Springer, pp. 373-379.
- Mochida I, Marutsuka T, Furuno T, Korai Y, Fujitsu H, et al. (1987) Hydrodesulfurization of petroleum cokes. High Temps-High Pressures 19(5): 545-553.
- Mochida I, Marutsuka T, Korai Y, Fujitsu H (1987) Enhanced hydrodesulphurization of cokes deposited on iron ore by air gasification. Fuel 66(1): 70-73.
- Mason RB (1959) Hydrodesulphurization of coke. Ind Eng Chem 51(9): 1027-1030.
- Hsu HL, Hardin EE, Grindstaff LI (1982) Calcining and desulphurizing petroleum coke, Br Patent, Syria.
- Al-Haj Ibrahim H (2011) Upgrading of Syrian petroleum coke by pre-oxidation. Periodica Polytechnica, Chemical Engineering 55(1): 21-25.
- Mochida I, Furuno T, Fujitsu H, Oyama T, Fujimoto K, et al. (1988) Hydrodesulphurization of needle coke in atmospheric hydrogen flow. Fuel 67: 678-681.
- El-tawil SZ, Morsi BM (1979) Thermal treatment of Egyptian petroleum coke in an atmosphere of oxygen. J Mines Met Fuels 27(6): 185-191.
- Heintz EA (1996) The characterization of petroleum coke. Carbon 34(6): 699-709.
- Al-Haj Ibrahim H, Ali MM (2005) Effect of the removal of sulphur and volatile matter on the true density of petroleum coke. Periodica Polytechnica Ser Chem Eng 49(1): 19-24.
- Zannikos F, Lois E, Stournas S (1995) Desulfurization of petroleum fractions by oxidation and solvent extraction. Fuel Proc Technol 42(1): 35-45.
- Funakoshi I, Aida T (1993) European Patent 0565324A1, 1993, and US Patent 5(753): 102.
- Shiraishi Y, Hirai T (2004) Desulfurization of vacuum gas oil based on chemical oxidation followed by liquid-liquid extraction. Energy & Fuels 18(1): 37-40.






























